Abstract
Keywords
Introduction
According to the data provided by Statistics Poland, in 2019, 17.6% of women and 19.5% of men aged >15 years were obese. 1 The Eurostat European Health Interview Survey reported a similar prevalence of obesity showing that 17% of adults living in the European Union were obese. 2 Available obesity forecasts are not optimistic and indicate that its prevalence will increase, reaching maximum levels between 2026 and 2054.3,4
Obesity impacts multiple facets of an individual’s well-being including physical, psychological and social outcomes.5,6 This chronic disease not only elevates the risk of numerous cardiovascular complications but also contributes to the development of sleep-related breathing disorders and hypoventilation.7–9 Bariatric surgery is often the most effective treatment option for obese patients who have previously unsuccessfully attempted lifestyle modifications such as diet, exercise and pharmacotherapy. Between 2008 and 2018, 6.5 million people worldwide underwent surgical interventions for obesity, with the number of procedures per inhabitant varying across different countries. 10 In 2019, the Comprehensive Specialist Care in Bariatrics (KOS-BAR) pilot program was initiated to provide complex medical care for patients undergoing bariatric surgery. Patients enrolled at 15 centres met the inclusion criteria of age (≥18 years), diagnosis of obesity due to excess calorie intake, and having a body mass index (BMI) ≥ 40 kg/m2 or 35 – 40 kg/m2, where surgically induced weight loss is expected to improve obesity-related comorbidities. 11
OSA increases morbidity and mortality from respiratory complications after bariatric surgery. Patients with obesity are at higher risk for complications like sputum retention, atelectasis, bronchopulmonary infections, and pulmonary embolism, which can significantly impact postoperative recovery. 12 The highest risk of complications among OSA patients who underwent bariatric surgery is driven by more than twice increased risk of cardiac complications. 13 Patients with obesity hypoventilation syndrome may exhibit an even higher risk of cardiopulmonary complications and longer length of hospital stay, compared to patients with OSA alone.14,15
Awareness of sleep-related breathing disorders is increasing among specialists who qualify patients for bariatric surgery. Screening, sleep studies and appropriate treatment, including positive airway pressure, are gradually becoming standard of care.16,17 Obesity hypoventilation syndrome is characterized by the coexistence of obesity, sleep-disordered breathing, and daytime hypercapnia in the absence of other causes of hypoventilation. The lack of consistency in the definitions of OHS across the literature hinders reliable comparisons of study findings.18–22 Regardless of the criteria used, researchers consistently suggest that the incidence of OHS among bariatric surgery patients is strongly underdiagnosed. 23 In addition to precise diagnostic criteria, it is essential to assess the stages of hypoventilation in obesity.24,25
This study aimed to analyse the prevalence of OHS and hypoventilation staging in patients qualified for bariatric surgery under the KOS-BAR program in the Department of Pulmonology, Jagiellonian University Medical College.
Methods
Study group
It was a retrospective, single-centre analysis of the medical records of 134 patients enrolled in the KOS-BAR program. The patients were hospitalized between January 2022 and December 2023 at the Department conducting the study. The study protocol received approval from the Ethics Committee.
Data collection
We evaluated every patient during their hospital stay through a comprehensive medical history, physical examination and anthropometric measurements (weight, height and BMI). Diagnostic procedures included polysomnography (PSG), transcutaneous CO2 measurement (percutaneous capnometry or tcPCO2), chest x-ray, spirometry and arterial blood gas analysis.
All participants underwent full-night diagnostic PSG with transcutaneous CO2 measurement in a hospital setting. The monitored parameters included electroencephalography (EEG) derived from F3, F4, C3, C4, O1, and O2 electrodes, electrooculogram, electrocardiogram; and mentalis muscle electromyography recorded with surface electrodes. Airflow was measured using a thermistor and a nasal pressure transducer, while snoring was recorded via a microphone attached to the neck skin. Respiratory movements were evaluated with inductance thoracic and abdominal bands. Blood oxygen saturation (SpO2) was monitored using finger pulse oximetry. Throughout the night, patients were videotaped with an infrared camera and data were recorded on the Alice 6 Diagnostic Sleep System (Philips North America Corporation, Andover, MA, USA). Respiratory events were scored according to the American Academy of Sleep Medicine guidelines. Polysomnography outcomes included total sleep time, sleep efficiency, sleep architecture, the apnea-hypopnea index (AHI), mean and minimal saturation, total sleep time with SpO2 < 90% (T90), oxygen desaturation index (ODI), body position during sleep and hypoxic burden. Hypoxic burden was defined as a sum of individual areas under the oxygen desaturation curve. 26
Additionally, we measured transcutaneous carbon dioxide pressure (tcPCO2) as a surrogate for arterial blood gas samples using continuous, noninvasive percutaneous capnometry with an ear sensor (Monitor TCM5 Basic, Radiometer, Copenhagen, Denmark). The data we collected was recorded with AQURE TC ResQ Software (Radiometer, Copenhagen, Denmark). Arterial blood samples for critical parameter assessment were obtained from every subject during wakefulness, after 15 min in an upright sitting position, at rest; and analysed using a point-of-care device (ABL90 FLEX PLUS, Radiometer, Copenhagen, Denmark).
We measured pH, arterial carbon dioxide partial pressure (PaCO2), arterial oxygen partial pressure (PaO2), and serum bicarbonate concentration (HCO3-).
To evaluate the severity of comorbidities and the long-term mortality risk, we used the Charlson Comorbidity Index (CCI) to analyse the available data for hospitalized patients. 27 This weighted index predicts 10-year survival in patients with multiple comorbidities and is expressed as a score starting at 0. The higher the score, the higher the predicted mortality risk. The variables analysed included age and clinical conditions, each assigned a score based on the associated risk.
Obesity-associated hypoventilation – definition and classification
According to The European Respiratory Society (ERS) Task Force such parameters as hypercapnia during sleep, hypercapnia during wakefulness, and serum bicarbonate level are used to differentiate the following five stages of obesity-associated hypoventilation
28
: • stage 0 – patients who are at risk of developing hypoventilation with a BMI >30 kg/m2, coexisting OSA and no hypercapnia; • stage I – obesity-associated sleep hypoventilation – patients with a BMI >30 kg/m2, coexisting OSA or hypoventilation during sleep with intermittent hypercapnia during sleep and serum bicarbonate level <27 mmol/l during wakefulness; • stage II – obesity-associated sleep hypoventilation – patients with a BMI >30 kg/m2, coexisting OSA or hypoventilation during sleep with intermittent hypercapnia during sleep, but with serum bicarbonate level ⩾27 mmol/l during wakefulness; • stage III - obesity hypoventilation – patients with a BMI >30 kg/m2, with coexisting OSA or hypoventilation during sleep, and sustained hypercapnia during wakefulness; • stage IV – obesity hypoventilation syndrome – coexisting obesity (BMI >30 kg/m2), daytime hypercapnia (arterial carbon dioxide tension ⩾45 mmHg) and sleep-disordered breathing/hypoventilation, cardiometabolic comorbidities (subject to excluding other disorders that may cause alveolar hypoventilation).
Intermittent hypercapnia during sleep is confirmed if the American Academy of Sleep Medicine Scoring Manual rules are met: 1. arterial partial pressure of carbon dioxide or its surrogate ⩾55 mmHg for at least 10 min, or 2. arterial partial pressure of carbon dioxide or its surrogate increases for at least 10 min by 10 mmHg to minimum ⩾50 mmHg (in comparison to an awake baseline value).
29
Statistical analysis
Continuous and categorical variables were expressed as medians (interquartile ranges (IQR)) and as counts (percentage), respectively. The comparison of categorical variables was performed using the chi-square test or Fischer’s exact test, while continuous variables were compared using the Mann-Whitney U test. A p-value <0.05 was considered statistically significant. This was a complete case analysis. All statistical analyses were performed using R software version 3.6.0 (R Project).
Results
Study group
Baseline characteristics.

BMI: body mass index; CCI: Charlson comorbidity index; COPD: chronic obstructive pulmonary disease; IQR: interquartile range; n: number; NAFLD: non-alcoholic fatty liver disease.
Fisher’s exact test was used for variables with the number of cases in any subgroup <5.
The patients in our study group were referred to the two main types of surgery: laparoscopic sleeve gastrectomy (LSG) and laparoscopic Roux-en-Y gastric bypass (LRYGB). Of the 134 patients, the majority underwent LSG (n = 114, 85.1%), while LRYGB was performed in nine patients (6.7%). The remaining 11 patients (8.2%), had not undergone surgery at the time of data analysis for the following reasons: they either dropped out of the program or were still awaiting surgery.
Sleep-related respiratory disorders
Results of polysomnography, capnometry and blood gas analysis.

AHI: apnea hypopnea index; HCO3−: serum bicarbonate concentration; IQR: interquartile range; n: number; ODI: oxygen desaturation index; OSA: obstructive sleep apnea; PaCO2: arterial carbon dioxide partial pressure; PaO2: arterial oxygen partial pressure; SP90: percentage of sleep time with oxygen saturation below 90%; SpO2: blood oxygen saturation measured via pulse oximetry; tcPCO2: transcutaneous carbon dioxide gas pressure.
Fisher’s exact test was used for variables with the number of cases in any subgroup <5.
Arterial blood gas analysis was unavailable in 7 patients.
Factors associated with hypoventilation
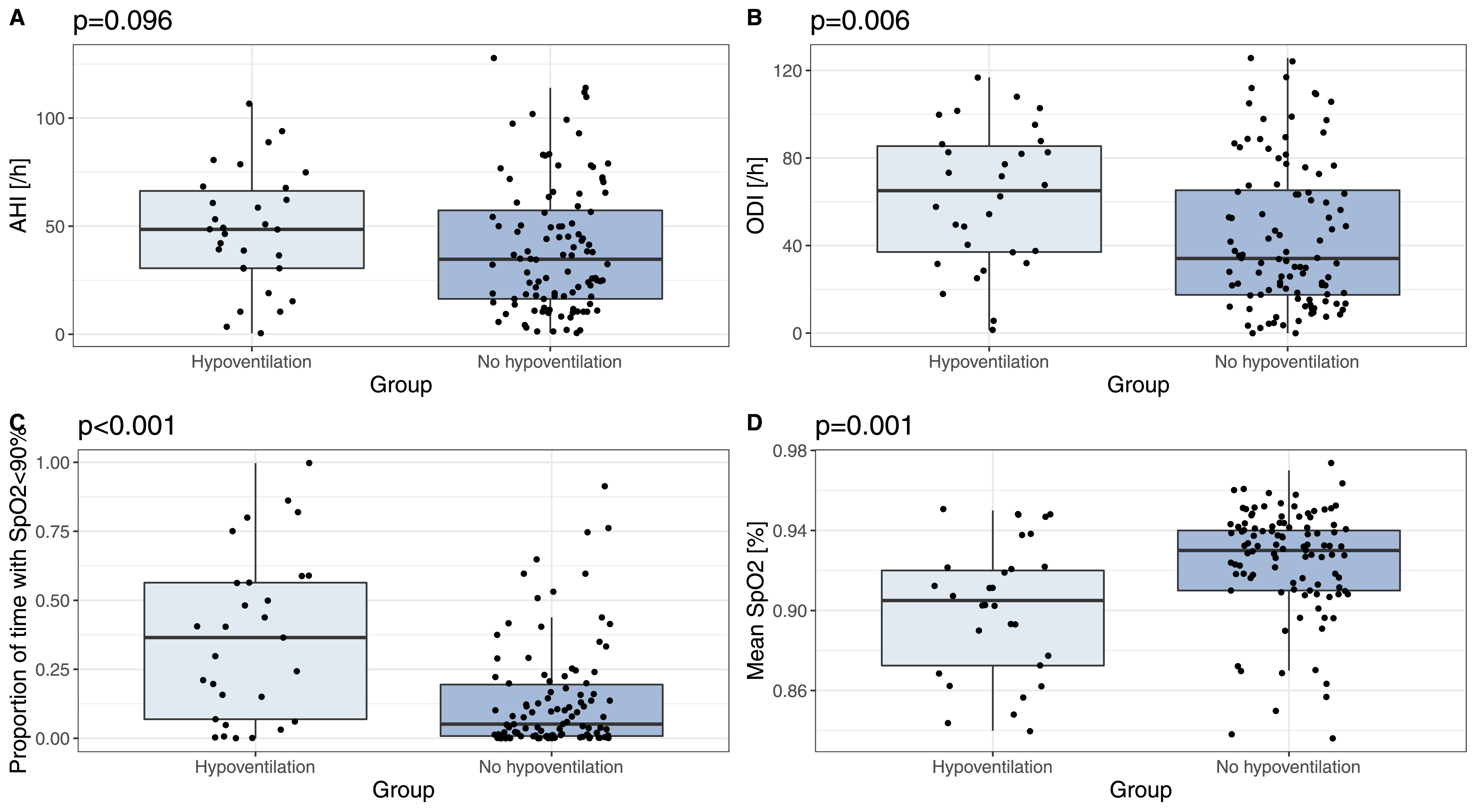
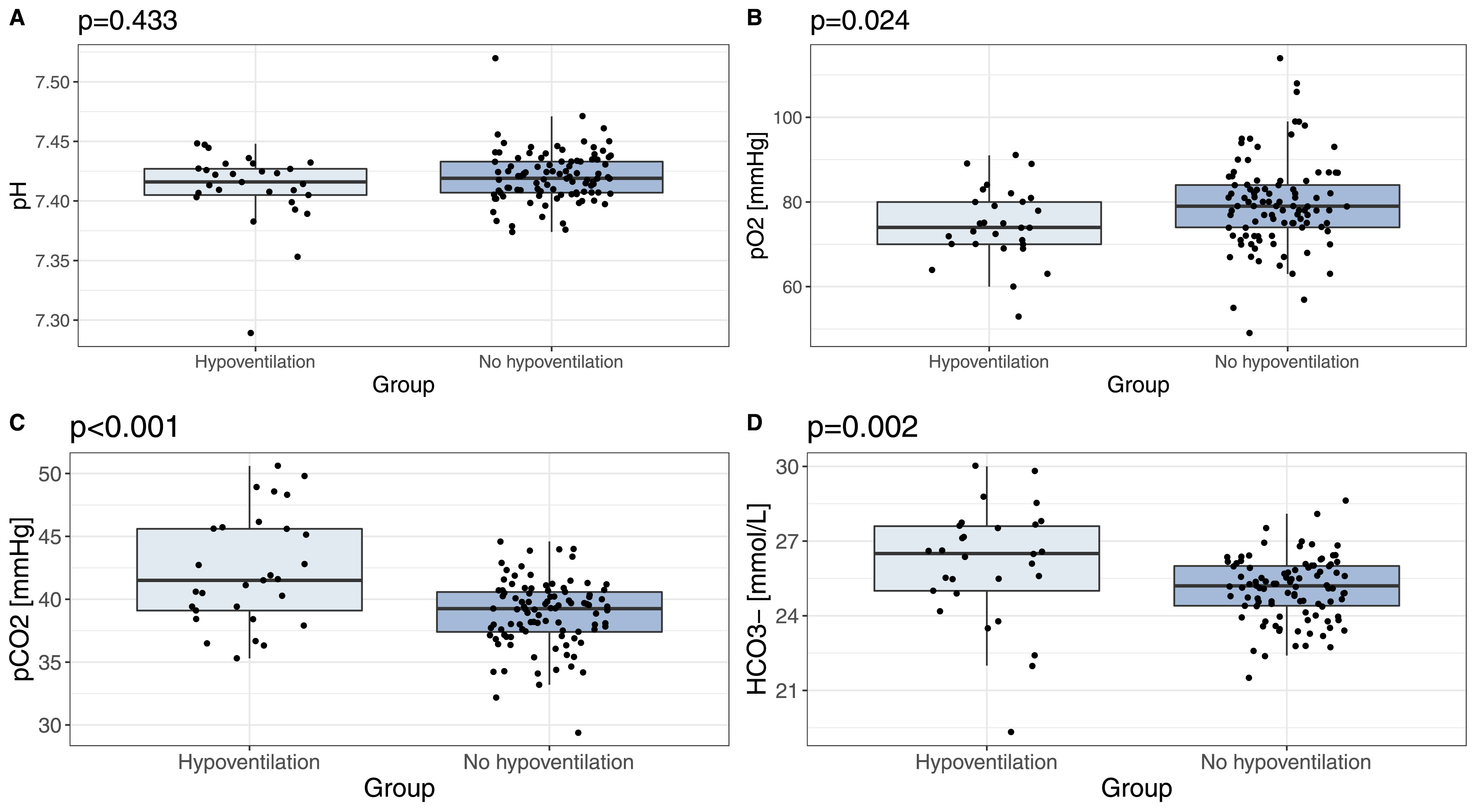
Patients with hypoventilation exhibited higher CCI scores compared to the remaining patients (1.0 vs 0.0, p = 0.022) and were more likely to be diagnosed with severe OSA (80.0% vs 52.9%, p = 0.008; Figure 1). Polysomnography revealed lower mean SpO2 (90% vs 93%, p = 0.001), higher ODI (65.1/h vs 34.1/h, p = 0.006) and a greater proportion of the sleep time with SpO2<90% (36% vs 5%, p < 0.001) (Figure 2). Arterial blood gas analysis showed higher PaCO2 (41.5 vs 39.3, p < 0.001), lower PaO2 (74.0 vs 79.0, p = 0.024) and higher HCO3- (26.5 vs 25.2, p = 0.002) levels in the hypoventilation group (Figure 3). Patients with hypoventilation more frequently exhibited increased HCO3− levels (37.9% vs 4.1%, p < 0.001). Increased HCO3− were found in 9/10 (90.0%) patients with hypoventilation stages III or IV and in 10/11 (90.9%) patients with hypoventilation stages II–IV. Daytime hypercapnia was exclusively present in patients with hypoventilation. Proportion of obstructive sleep apnea (OSA) severity in hypoventilation and non- hypoventilation groups. Comparison of polysomnographic characteristics between the groups: (A) According to AHI - apnea hypopnea index, (B) according to ODI - oxygen desaturation index, (C) according to proportion of sleep time with oxygen saturation below 90%, (D) according to mean SpO2- blood oxygen saturation measured via pulse oximetry. Comparison of ABG results between the groups: (A) Comparison of pH, (B) comparison of partial pressure of oxygen in arterial blood, (C) comparison of partial pressure of carbon dioxide in arterial blood, (D) comparison of serum bicarbonate level.
Discussion
This retrospective study, including more than 130 patients scheduled for bariatric surgery, demonstrates that the majority of these patients suffer from OSA, with one-quarter meeting the hypoventilation criteria. 28 Application of hypoventilation staging indicates that the majority of these patients are classified into stages I and IV. As hypoventilation is very common in the bariatric surgery group, we concluded that tcPCO2 non-invasive measurement should become a standard component of preoperative assessment for these patients. Further studies are necessary to better elucidate the impact of each stage of hypoventilation on perioperative outcomes.
OSA and OHS are significant risk factors for respiratory perioperative complications, including difficulties with bag-mask ventilation, difficult intubation, prolonged mechanical ventilation, pneumonia and death. 30 These increased perioperative risks can be mitigated by appropriate interventions, primarily positive airway pressure. On the one hand, the high prevalence of OSA in patients undergoing bariatric surgery is well documented, and clinicians involved in the surgical treatment of obesity generally recognize the necessity of preoperative screening for sleep-associated breathing disorders. 31 On the other, the current guidelines do not address the issue of routine assessment of hypercapnia while a reliable diagnosis of OHS requires comprehensive preoperative assessment including not only polygraphy or polysomnography but also capnometry and arterial blood gas analysis.
In the KOS-BAR program, a very thorough preoperative evaluation of the respiratory system was conducted, incorporating all of the above-mentioned tests. Based on our studies, we showed that among patients qualified for bariatric surgery, hypoventilation is frequent (22.4%) although notably lower than those reported in two large-cohort studies by Chindamporn et al. (68.5%) and Tran et al. (68.4%).21,32 This may be attributed to the more restrictive diagnostic criteria used in our study. In both aforementioned cohorts, hypoventilation was defined as a sleep study–based awake EtCO2 ≥ 45 mmHg. In contrast, we used the AASM definition of intermittent hypercapnia understood as arterial carbon dioxide partial pressure or its surrogate ⩾55 mmHg for at least 10 min or increase in arterial carbon dioxide partial pressure or its surrogate by 10 mmHg to minimum ⩾50 mmHg in comparison to the baseline during daytime for at least 10 min. Moreover, unlike in the aforementioned studies, we used non-invasive CO2 monitoring using tcPCO2 instead of etCO2. Even though AASM recommends both these methods as surrogates for PaCO2, evidence suggests that percutaneous measurement may be more precise compared to end-tidal CO2 measurement. 33
Our study contributes to the current literature by showing that obesity-associated sleep hypoventilation (stage I) is the most common form of obesity-associated hypoventilation in this population, followed by obesity hypoventilation syndrome (stage IV), obesity hypoventilation (stage III), and obesity-associated sleep hypoventilation (stage II). Previous studies either focused only on patients in stage II (Chindamporn et al.) or stages II-IV (Tran et al.). We propose that further studies should focus on the correlation between the stages of obesity-associated hypoventilation and the risk of perioperative complications, as well as examine the patients’ response to positive airway pressure (PAP) therapy in terms of perioperative risk reduction.
The number of sleep studies appears to be insufficient, especially given the growing need for bariatric surgery. It may be logistically impossible to perform polygraphy or polysomnography with carbon dioxide monitoring in each patient qualified for surgical obesity treatment. Consequently, it is crucial to optimize the allocation of these limited resources by reliably identifying patients at higher risk for hypoventilation and referring them for broader preoperative evaluation. In our study, the only clinical factor associated with increased risk of hypoventilation identified was a slightly elevated CCI score (1 vs 0). Previously mentioned studies indicated that higher age, male sex, diabetes and hypertension were associated with an increased risk of hypoventilation. We made similar observations in our study but comparisons were statistically insignificant, possibly due to the relatively small study sample. Interestingly, while BMI has been identified as a risk factor for hypoventilation in other studies, our analysis shows no difference in BMI between the patients with and without hypoventilation (43.6 vs 43.5 kg/m2). Based on the available literature and our findings, we may infer that clinical and demographic variables may be insufficient for reliably selecting patients who require broader preoperative assessment.
Arterial blood gas analysis is another relatively simple test allowing to assess the risk of severe OSA and OHS. First, it serves as one of the diagnostic criteria for obesity hypoventilation syndrome (stage IV). Second, it enables the measurement of bicarbonate level, which is considered a good predictor for ruling out obesity hypoventilation syndrome in patients with OSA and low to moderate risk of OHS.34,35 Our findings indicate that two-thirds of patients qualified for bariatric surgery diagnosed with hypoventilation, exhibited normal bicarbonate levels. This is similar to the Chindamporn et al. cohort, in which only 38.3% of patients with hypoventilation met the criterion of HCO3- ≥ 27 mmol/L. This observation is mainly driven by a high proportion of patients with stage I hypoventilation, who, by definition, had normal bicarbonate levels. Clinically, it underlines an urgent need for further studies evaluating the impact of each hypoventilation stage on clinical outcomes. It remains unclear whether stage I hypoventilation is associated with a higher incidence of perioperative complications. If this correlation was confirmed, the current guidelines and recommendation against relying solely on bicarbonate level in the preoperative assessment, would need to be modified.
This difference in the approach towards the sensitivity and specificity of the 27 mmol/L threshold calls for further clarification as to how the threshold was arrived at. When, in 2006, Mokhlesi et al. analysed the data collected, they did not have access to continuous CO2 measurements. 36 Their threshold was based on patients with chronic daytime hypercapnia, which occurred only in stages III and IV of obesity-associated hypoventilation. Even among their patients with diagnosed OSA, the threshold of 27 mmol/l exhibited a sensitivity of 92% and a specificity of 50% for OHS.
Finally, we investigated whether polysomnographic data is related to an increased risk of hypoventilation. Based on the previous studies, it remains unclear if the number of respiratory events (measured as AHI) is associated with a higher risk for OHS. 37 We found no substantial correlation between AHI and hypoventilation, however, among those diagnosed with hypoventilation, there were significantly more patients with severe OSA. Both Chindamporn et al. and Tran et al. reported that patients with hypoventilation had higher AHI compared to those without hypoventilation. Notably, patients enrolled in our study had more severe sleep-related breathing disturbances (median AHI 37, 6/h), compared to studies by Chindamporn et al. and Tran et al. (both 16.1/h). The reasons behind this discrepancy between the studies are yet unclear, especially given the higher BMI in the previous cohorts.
Analysis of oximetry offers additional valuable information. We found that lower mean SpO2, higher ODI and longer proportion of sleep time with SpO2<90% are distinct features of the hypoventilation group. This corroborates findings from the previously cited studies. Tran et al. calculated 1.7% (1.02; 1.01–1.02) increased odds of OHS per 1% increase in sleep time SpO2 <90%. 32 These data also confirm that patients undergoing bariatric surgery should be closely monitored for hypoxemia, particularly in the post-extubation period and, apart from PAP support, the addition of oxygen to the supporting system should be considered. Our comprehensive PSG included also the assessment of important metrics (hypoxic burden) and, despite statistically insignificant differences between the groups, we revealed that the hypoxic burden value in the hypoventilation group was twice as high. 26 This may be attributed to the relatively small group size.
To sum up, the data we obtained indicate that poor PSG oximetry results may be associated with a higher risk of hypoventilation. Furthermore, patients with low minimal saturation, high ODI and a higher proportion of time with SpO2<90% may warrant broader preoperative assessment. However, given the limitations of our study (retrospective design, a relatively small study sample), these results should be treated as hypothesis-generating and require confirmation in larger, prospective cohorts.
The present study has both strengths and limitations. Our data is based on a comprehensive sleep medicine evaluation including PSG with routine tcPCO2 monitoring and arterial blood gas analysis. Uniquely, this allowed us to examine all stages of sleep-related hypoventilation. This is the first time this type of data relating to European patients undergoing bariatric surgery is shown.
We are aware, however, that our study has limitations. Firstly, the relatively small study sample, limits the statistical impact of the study and prevents us from applying multivariable analysis. Secondly, the retrospective design of the study which carries inherent limitations and increases the risk of bias. Thirdly, the absence of data on postoperative complications which prevents us from evaluating whether clinical and polysomnographic data translate to patient outcomes. Additionally, arterial blood gas analysis was missing in seven patients, which could potentially limit our ability to diagnose hypoventilation. Finally, we did not analyse whether bariatric surgery alleviated hypoventilation. A recent meta-analysis suggested that bariatric surgery is only partially effective in treating OSA, 38 while a prospective observational study found that patients with OHS achieve lower weight loss after bariatric surgery. 19 Further research is needed to better explore these relationships and to assess the impact of bariatric surgery on weight reduction in patients across different stages of hypoventilation.
Conclusion
In this retrospective study, we demonstrated that hypoventilation is present in nearly a quarter of patients qualified for bariatric surgery, with obesity-associated sleep hypoventilation (stage I) being the most common form. We highlight the importance of recognising OSA and OHS as potential risk factors for respiratory complications during surgery. Additionally, we advocate for the inclusion of non-invasive tcPCO2 measurement as a standard component of preoperative assessment for bariatric patients. Our findings also suggest that clinical and demographic variables may be insufficient to reliably identify patients who require broader preoperative assessment.
The results are promising and warrant further validation through studies examining the impact of all stages of obesity-associated sleep hypoventilation on health outcomes and postoperative complications.
Footnotes
Acknowledgements
The authors would like to express their gratitude to the Head of Department of Pulmonology, Jagiellonian University Medical College - MD, PhD, Professor of Medicine Krzysztof Sładek for his support. Authors would also like to thank the physicians, nurses and nurse aids working in the Pulmonology Department at Jagiellonian University Medical College, Kraków, Poland. Author would like to express special appreciation for administrative support to Anna Wrzoskiewicz.
Ethical statement
Author contributions
Conceptualization: A.K.; methodology: K.P.; formal analysis: K.P.; investigation A.K., K.P., N.C.-W.; resources: A.K., K.P., N.C.-W., J.T., P.M.; writing-original draft preparation: A.K., K.P., N.C.-W., J.T.; writing- review and editing: A.K., K.P., N.C.-W., J.T.,P.M.; visualization: A.K., K.P., N.C.-W., J.T.; supervision: A.K.; project administration: A.K.. All authors have contributed substantially to the work reported. All authors edited, have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Jagiellonian University Medical College, grant number N41/DBS/001183. The APC funding source has not been established yet.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are not publicly available due to privacy restrictions. Data sharing is not applicable to this article.
