Abstract
Background
People with chronic obstructive pulmonary disease (COPD) are at a higher risk of falls. This preliminary study aims to externally validate a previously developed clinical prediction model for falls in community-dwelling older adults with COPD.
Methods
This was a secondary analysis of a 12-month prospective cohort study. Older adults (≥60 years) with COPD, who reported a fall in the past year and/or had balance concerns, were tracked for 12-month future falls. Baseline predictors included 12-month history of ≥2 falls, total chronic conditions, and Timed Up and Go Dual-Task (TUG-DT) test scores. Model performance was assessed for discrimination (c-statistic), calibration (E:O, CITL, and calibration slope), and clinical value (decision curve analysis).
Results
The study included 89 participants (average age 73 ± 9 years; 83 females; FEV1%predicted = 47%). Of these, 35 (39%) reported ≥1 future fall, totaling 89 falls. The model demonstrated acceptable discrimination (c-statistic = 0.62, CI [0.51,0.72]), and calibration (E:O = 1, CITL = 0, and a calibration slope = 1). Decision curve analysis showed greater clinical value when using the prediction model compared to screening for fall history alone.
Conclusions
A 12-month history of ≥2 falls, higher total chronic conditions, and worse TUG-DT test scores, predicts falls in community-dwelling older adults with COPD. Larger studies are needed before clinical application.
Introduction
Each year in Canada, one in every three older adults living in the community will experience a fall. 1 Falls are among the leading causes of morbidity and mortality among those 60 years and older. 2 Older adults living with certain chronic conditions, such as chronic obstructive pulmonary disease (COPD), are at an even greater risk of falls, 3 with an estimated 55% increased risk of falling compared to age-matched healthy counterparts. 4 According to a systematic review of 23 studies, the pooled yearly prevalence of falls in COPD is 30%. 5 Moreover, in a population based study of over 16,000 community-dwelling older adults, COPD emerged as the strongest predictor of falls among 13 chronic conditions including heart disease, arthritis, and diabetes. 3 Taken together, this suggests that fall risk is a significant concern among older adults with COPD, highlighting the need for early detection and intervention.
A clinical prediction model is a useful tool for clinicians to screen an individual’s likelihood of falling and determine whether intervention should be initiated. An important step in clinical prediction model development, is external validation.6,7 In external validation, the performance of a clinical prediction model is evaluated in a new cohort that is separate from the cohort used from model development.6,7 In the falls literature, clinical prediction models have primarily been developed for the general older adult population, 8 with previous externally validated models achieving acceptable accuracy for generalizability (concordance (c) statistic = 0.69-0.71).9,10 Although general older adult fall prediction models apply to a wide range of individuals, disease-specific models may be preferable for populations at higher risk of falls. Previous models in other clinical populations like chronic kidney disease and hematological disorders have proposed unique prediction models for falls with potentially greater accuracy (area under the ROC curve [AUC] = 0.72-0.96),11,12 suggesting that disease-specific prediction models may be favourable. People with COPD have an increased risk of falls and may present with unique risk factors compared to general older adults, yet there are presently no externally validated clinical prediction models for falls in this population.
In our previous study, we developed a clinical prediction model for 12-month future falls among 178 individuals with COPD (mean age 72.7 ± 9.0 years). 13 Our model revealed that those reporting ≥2 falls in the previous 12 months, a higher number of chronic conditions, and worse mobility under cognitive demand (measured by slower Timed Up and Go Dual-Task (TUG-DT) test performance) were more likely to experience ≥1 future fall over 12 months. 13 Although the prediction model resulted in acceptable discrimination (c-statistic = 0.69, 95% confidence interval (CI) [0.61,0.78]), and calibration (expected to observed (E:O) ratio = 1.01, CI [0.83,1.18], calibration-in-the-large (CITL) = 0, CI [-0.37.0.35], and calibration slope = 0.93, CI [0.59,1.27]) it has yet to be externally validated. 13 Therefore, the aim of this preliminary study was to conduct an external validation of our clinical prediction model for predicting ≥1 future fall over 12 months using a sample of community-dwelling older adults with COPD.
Methods
Study design
We conducted a secondary analysis of a completed 12-month prospective cohort study aimed at identifying the optimal balance screening tests for falls in patients with COPD. 14 The study was publicly registered (ClinicalTrials.gov identifier: NTC03229473) and received ethics approval from the Hamilton Integrated Research Ethics Board (HiREB #3331 and #4335) and the Joint West Park Healthcare Centre-Toronto Grace Health Centre Research Ethics Board (JREB #17-018-WP). Study details were reported in accordance with the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) guidelines. 15
Participants
Participants were recruited from four respirology clinics located in Toronto and Hamilton, Ontario, Canada (Supplementary Material 1). Participants were included if they had a medical diagnosis of COPD according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines, 16 were aged 60 years or older, and reported a history of ≥1 fall in the previous 12 months or difficulty with walking/balance. Participants were excluded if they were unable to communicate in English, presented with cognitive impairments as measured by a score of 23 points or less on the Mini-Mental State Examination (MMSE), 17 or presented with a condition that limited their ability to safely participate in balance and mobility assessments (e.g., Parkinson’s Disease, acquired brain injury, and other neurological conditions). As recommended, to ensure the present dataset was suitable for external validation, we evaluated our sample using the first three domains of the Prediction model Risk Of Bias Assessment Tool (PROBAST): participant selection, predictors, and outcome (Supplementary Material 2).18,19
Clinical prediction model parameters
The clinical prediction model from the development dataset of 178 participants with COPD was applied to the participants in the present study for external validation using the logistic regression formula
Primary outcome
Falls were defined and prospectively tracked monthly for 12-month using the same method as the development cohort (Supplementary Material 3). 13 As per the Prevention of Falls Network Europe recommendations, 20 a fall was defined as “an unexpected event in which the participants come to rest on the ground, floor, or lower level”. Fall status was used as the primary outcome in this study and was dichotomized into ≥1 fall versus no falls.
Baseline descriptors
Baseline descriptors included age, sex, body mass index (BMI), smoking history, use of supplemental oxygen, use of a gait aid, measures of pulmonary function (FEV1% predicted, FVC% predicted, and FEV1/FVC ratio), 12-month fall history, self-reported balance concerns, total chronic conditions, total medications, COPD severity in accordance with the GOLD guidelines, 16 and exercise tolerance as measured by the six-minute walk test (6MWT). 21
Predictors
The predictors used for the analysis were measured using the same methods as the development cohort and included 12-month history of ≥2 falls, total chronic conditions, and TUG-DT test scores. 13 Fall history was collected at baseline to categorize participants into either ≥2 falls or <2 falls in the previous 12 months. Total chronic conditions were retrieved from participant clinical records and included cardiac, respiratory (including COPD), rheumatological, musculoskeletal, vascular, cancerous, gastrointestinal, neurological, and psychological conditions. The TUG-DT test is a timed performance-based test assessing mobility under cognitive demand with lower completion times indicating better mobility under cognitive demand. 22 The TUG-DT test is scored as the total time taken to go from a seated position in a chair, stand up, walk three metres forward, turn around a floor marker, walk back to the chair, and sit down, all while counting backward by threes aloud starting from 100. 22
Missing data
Variables with missing data were addressed with multiple imputation (Supplementary Material 4).6,23 As recommended in previous literature, combined estimates as well as standard errors for prognostic modelling were derived using Rubin’s rules across 20 imputed data sets.6,24 Sensitivity analysis was conducted to compare prediction model performance with and without imputed data.
Statistical analysis
Data were summarized as means and standard deviations for continuous data, median and interquartile range (IQR) for skewed data, and absolute and relative frequencies for categorical data. Prediction model performance was described as model discrimination and calibration. 18
Model discrimination relates to how well the prediction model identifies individuals with, versus without, the outcome, and was measured using the c-statistic, with scores ranging from 0.5 to 1 (higher values indicate better discrimination).6,7,25 Acceptable c-statistic thresholds are context specific, meaning there are no set thresholds or general rules for establishing acceptable predictive validity across all prediction models.18,26 Since no previous research has determined a c-statistic threshold for predicting falls in COPD, we have set a c-statistic threshold based on previous literature on fall prediction modelling in older adults of ≥0.6 as acceptable for predictive validity.8–10
Model calibration relates to the level of agreement between the predicted and observed values and was measured using the E:O ratio (ideal value of 1), CITL (ideal value of 0), and calibration slope (ideal value of 1; acceptable range = 0.8 to 1).7,25 If miscalibration was detected (deviation from ideal calibration values), model recalibration was conducted using recalibration-in-the-large (if only E:O ratio and CITL deviated from ideal) or logistic calibration (if calibration slope deviated from ideal) (Supplementary Material 5). 6 If model recalibration was conducted, model performance was re-evaluated for discrimination and calibration.
Calibration plots were used to visually evaluate model calibration performance with observed values on the y-axis and expected (predicted) values on the x-axis. 6 The calibration plots display a smoothed line representing the prediction model calibration performance, overlaying a reference line representing perfect calibration, and is interpreted by how closely the smoothed line aligns with the reference line using visual inspection.6,7
Decision curve analysis was applied to evaluate the clinical net benefit of the prediction model compared to alternative strategies with net benefit on the y-axis and threshold probability on the x-axis. 27 Net benefit represents the net total of true positives gained at a given decision threshold (the point in which the likelihood of an event is deemed actionable by a clinical decision maker), with higher net benefit indicating more true positives and fewer false positives (Supplementary Material 6). Prediction model performance was compared to alternative clinical strategies for deciding to treat, which included: treating all, treating none, and treating based on 12-month history of ≥1 fall 28 . The clinical strategy of treating based on 12-month history of ≥1 fall was included based on the World Fall Guidelines for Prevention and Management of Falls in Older Adults. 28 Model significance was set to α = 0.05, and all analyses were conducted using STATA/IC 16.1.
Results
Participant characteristics
Characteristics of community-dwelling older adult participants with COPD stratified by fall status.

Abbreviations: COPD = chronic obstructive pulmonary disease, SD = standard deviation, BMI = body mass index, 6MWT = six-minute walk test, FEV1 = forced expiratory volume in 1 second, FVC = forced vital capacity, TUG-DT = Timed Up and Go Dual-Task.
Model performance and recalibration
Model performance revealed a c-statistic of 0.62 (CI [0.51,0.72]), E:O ratio of 1.07, CITL of −0.13, and calibration slope of 0.56 (CI [-0.13,1.23]). Although the E:O ratio and CITL were relatively near the ideal values of 1 and 0 respectively, the calibration slope substantially deviated below the ideal value of 1, indicating that the predictions were too extreme, and the model was overfit.
6
Based on the calibration plot (Figure 1), the model begins to substantially deviate from ideal calibration beyond a 50% predicted likelihood of falls. Therefore, model recalibration was conducted to prevent systemically incorrect predictions and decision making.
6
The prediction model underwent logistic calibration in which the model intercept was updated to −0.261 and model slope to 0.549. Upon re-evaluation of model performance, the updated prediction model achieved acceptable calibration with an E:O ratio of 1.00, CITL of 0, and calibration slope of 1.02 (CI [-0.31,2.18]). Based on the calibration plot of the recalibrated model (Figure 2), the model is visually inline with ideal calibration. Calibration plot for the fall prediction model in community-dwelling older adults with COPD (N = 89) with the smoothed prediction model overlayed onto an ideal reference line (indicates perfect calibration). Calibration plots display the observed values on the y-axis over the expected (predicted) values on the x-axis. The spike plot at the bottom of the calibration plot shows the distribution of the observed and expected probabilities, with the observed data dichotomised into <50% below the midline and ≥50% above the midline. Calibration plot for the recalibrated fall prediction model in community-dwelling older adults with COPD (N = 89) with the smoothed prediction model overlayed onto an ideal reference line (indicates perfect calibration). Calibration plots display the observed values on the y-axis over the expected (predicted) values on the x-axis. The spike plot at the bottom of the calibration plot shows the distribution of the observed and expected probabilities, with the observed data dichotomised into <50% below the midline and ≥50% above the midline.

Decision curve analysis
Compared to treating all, treating none, and treating based on 12-month history of ≥1 fall, decision curve analysis revealed a higher net benefit using the fall prediction model when the probability threshold was between 30%–50% (Figure 3). There were no discernable differences between strategies at decision thresholds outside this range. Decision curves for treating all, treating none, treating based on 12-month history of ≥1 fall, and treating based on predicted likelihood of ≥1 fall using the prediction model derived from a sample of community-dwelling older adults with COPD (N = 89). Decision curves are plotted with net benefit on the y-axis, indicating the net true positives, over the x-axis, representing the threshold probability or the minimum likelihood of a fall that is deemed actionable by a clinical decision maker.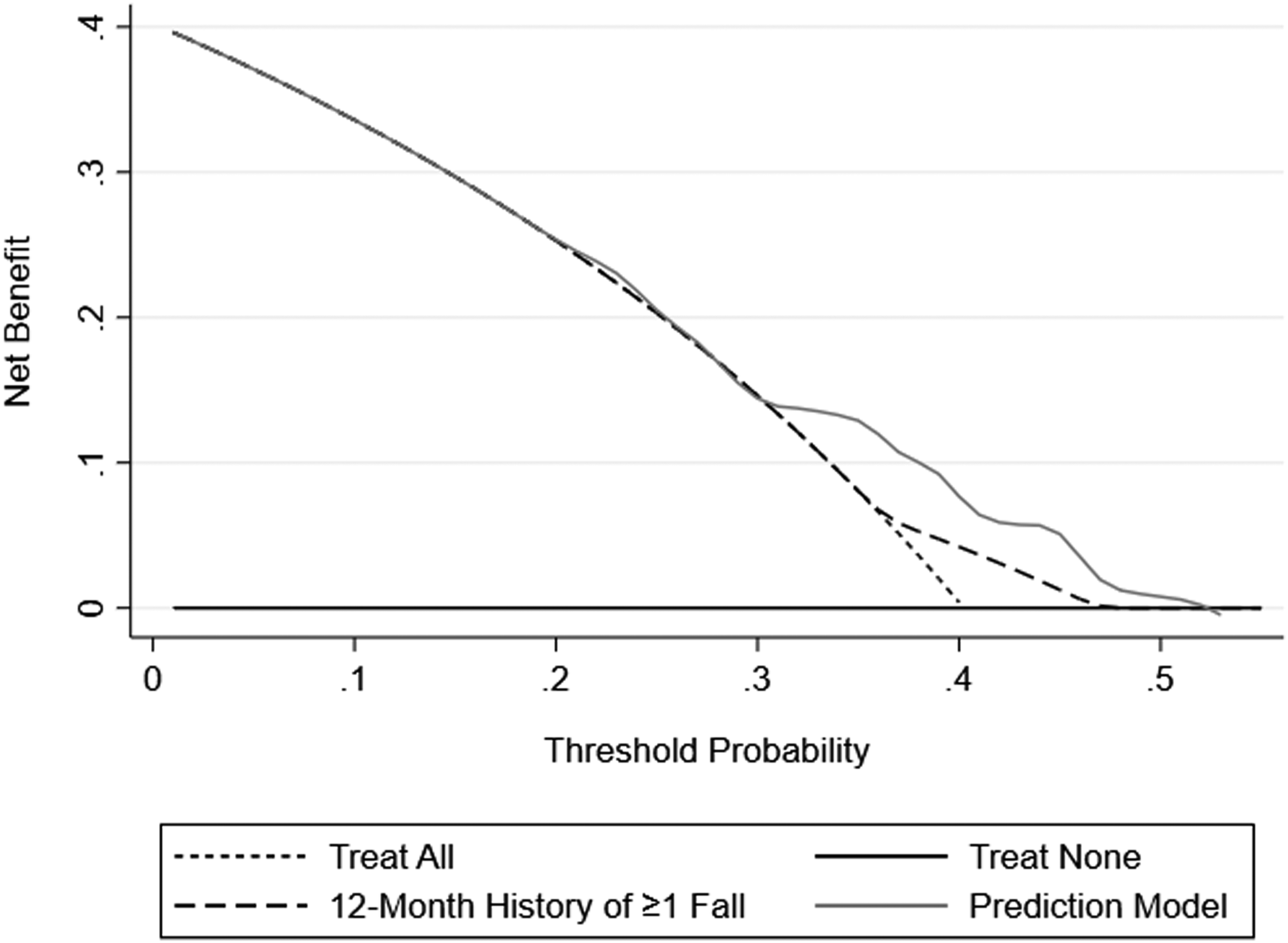
Discussion
In this preliminary study, we externally validated a clinical prediction model for falls in community-dwelling older adults with COPD. Our findings showed that the model, which includes a 12-month history of two or more falls, total chronic conditions, and TUG-DT test scores, demonstrated promising validity for predicting falls over the next 12 months. Model performance revealed acceptable discrimination and calibration after recalibration. Additionally, decision curve analysis indicated superior clinical value using the fall prediction model compared to only screening based on 12-month fall history.
Compared to model development, the externally validated prediction model observed a slightly decreased c-statistic (0.69 in model development vs 0.62 in model validation). 13 Since our model discrimination c-statistic was ≥0.6 and was relatively close to what was observed in model development, we deemed model discrimination acceptable. Like other studies in the literature,10,29,9,30 a difference in model discrimination from development to validation was expected. This discrepancy may be attributed to our initial model miscalibration, which represents the differences between contexts and settings from model development to model validation that are not captured by the predictors. 6 Model miscalibration has a direct relationship with model discrimination (i.e., lower calibration slope values typically result in lower discrimination values). 6
To address model miscalibration and reduce the risk of misclassification, model recalibration was necessary. 6 Similar to our study, van De Loo and colleagues applied model recalibration (via recalibration-in-the-large) to achieve ideal calibration values in a 12-month retrospective model validation study for predicting falls among 1,125 community-dwelling older adults (median age = 77, interquartile range [72,83]). 9 The benefit of model recalibration is that the predictors within the prediction model remain the same but the intercept and slope are modified to accommodate the changes in uncontrolled contexts and settings, like differences in geographic location, sample size, and temporal application. 6 With model recalibration, the prediction model can be applied with a setting-specific intercept and or slope without the need for re-validation since the predictors and their respective coefficients are not changed. 6 Our model performance reflects a resilient prediction model for falls in people with COPD even with a change in setting. However, due to the preliminary nature of this study, an additional external validation study with a sufficient sample size is warranted to confirm our findings.
Attaining a sufficient sample size for external validation can be challenging with a prospective cohort study design. Previous external validation studies for fall prediction among older adults employed a retrospective study design,9,10 since larger sample sizes may be more readily available. Unfortunately, this poses a problem for the interpretation of findings since it is difficult to control how predictors are measured. This was observed in the harmonization of data in the retrospective cohort study by van de Loo and colleagues, whereby the TUG test was used as a proxy for the gait speed test, while two other predictors had no data at all and were multiply imputed. 9 A future consideration is to use data from large international or national longitudinal cohort studies like the European or Canadian Longitudinal Studies on Aging.31,32 Data in these large longitudinal studies are collected prospectively, standardized (and/or harmonized) to allow comparison across countries and cohorts, and include participants with a wide range of chronic conditions.31,32
Decision curve analysis is a useful tool for clinicians to translate prediction models into clinical practice and is recommended as a component of evaluating prediction model performance.6,7 The findings of this study showed that the prediction model for 12-month future falls was superior in clinical net benefit to treating all, treating none, and treating solely based on 12-month fall history if the decision threshold was set between 30%–50% (Figure 3). For example, if a clinician is willing to intervene only when a patient has a ≥40% likelihood (or any other set threshold between 30%–50%) of falling over the next 12 months, the fall prediction model is better at detecting patients that present with either <40% or ≥40% likelihood of falling than the other strategies evaluated. A similar result was reported in older adults by van de Loo and colleagues, whereby their externally validated clinical prediction model observed a superior net benefit for predicting future falls compared to treating all, treating none, and treating based on 12-month history of ≥1 fall when the probability threshold was between 35%–60%. 9 Our near similar results with fewer predictors, offers a parsimonious model for consideration in clinical settings to better detect the need for fall prevention intervention in community-dwelling older adults with COPD.
No previous decision thresholds, to our knowledge, have been established for fall prevention intervention in COPD or in older adults. However, in a meta-analysis by Oliveira and colleagues, combining data from 23 studies on falls in people with COPD, a pooled prevalence of falls of 30% (CI [19%,42%]) was established, 5 setting the lower boundary of fall risk associated with this clinical population. At the upper range of >50% likelihood of falling, our model predictions become unstable as shown by the calibration plot in Figure 2. Deciding only to treat with a threshold >50% may be too risky, as intervention is urgent and the high likelihood of falling could result in a life-altering injury. A range of 30%–50% as a decision threshold allows clinicians to avoid treating all COPD patients and target higher risk subgroups while initiating fall prevention interventions early enough to avoid a fall. The prediction model presented in this preliminary study may thus offer a valuable clinical tool for early detection of fall risk in people with COPD. However, due to the preliminary nature of this study, the prediction model should be evaluated in a larger sample before being used in clinical practice.
Clinical prediction models used to predict the likelihood of different health outcomes, like falls, can be employed to help healthcare professionals and patients make treatment decisions to improve health outcomes, for example, whether to enroll in a fall prevention program. One potential strategy for implementing fall prediction models into clinical practice is the use of clinical decision support systems (CDSS). 33 Within CDSS, clinical prediction models can be integrated electronically to use patient information to catch key health concerns and enhance healthcare decisions. 33 The implementation of a fall prediction model using CDSS is currently being investigated in an ongoing trial by researchers in the Netherlands. 34 Although the clinical utility of our fall prediction model should be further evaluated before it is used in clinical practice, integration into CDSS is a key consideration for future research.
Our study strengths were demonstrated in our methodological approach. We complied with the Prevention of Falls Network Europe and the TRIPOD guidelines,15,20 reporting the necessary information for model comparison and model updating in future fall related studies in COPD.
Our findings, however, should be interpreted considering several limitations. First, our limited sample size likely contributed to the decreased model performance from development to validation. However, after recalibration, our model’s performance remained comparable to larger cohort studies in older adults while using fewer predictors.9,10 Additionally, our prediction model is likely unreliable for predicting falls when the predicted likelihood is outside the 30%–50% range. However, this range may be ideal, as the lower boundary of 30% reflects the annual pooled prevalence of falls in COPD, 5 and exceeding the upper boundary of 50% could indicate an imminent risk of serious falls.
Conclusion
In conclusion, these preliminary findings indicate that a clinical prediction model incorporating a 12-month history of two or more falls, total chronic conditions, and mobility under cognitive demand is externally valid for predicting 12-month falls in community-dwelling older adults with COPD. The model achieved acceptable discrimination and calibration, showing superior clinical value compared to screening for fall history alone. This prediction model could potentially support clinical decisions for fall prevention interventions. However, further research is needed to assess the model’s impact in a larger sample across various clinical settings.
Supplemental Material
Supplemental Material - Validation of a clinical prediction model for falls in community-dwelling older adults with COPD: A preliminary analysis
Supplemental Material for Validation of a clinical prediction model for falls in community-dwelling older adults with COPD: A preliminary analysis by Khang T. Nguyen, Cindy Ellerton, Joshua Wald, Natya Raghavan, Luciana G. Macedo, Dina Brooks, Roger Goldstein and Marla K. Beauchamp in Chronic Respiratory Disease
Footnotes
Acknowledgments
The authors would like to thank Dr. Roger Goldstein at West Park Healthcare Centre and Diana Hatzoglou at St Joseph’s Healthcare Hamilton for their vital support with recruitment. We also wish to acknowledge Zachary Hollingham for his help with patient assessments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Respiratory Research Network - Emerging Research Leaders Initiative.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
