Abstract
Asthma and obstructive sleep apnea (OSA) are common respiratory disorders. They share characteristics such as airway obstruction, poor sleep quality, and low quality of life. They are often present as comorbidities, along with obesity, gastroesophageal reflux disease (GERD), and allergic rhinitis (AR), which impacts the disease’s control. In recent years, there has been discussion about the association between these conditions and their pathophysiological and clinical consequences, resulting in worse health outcomes, increased healthcare resource consumption, prolonged hospital stays, and increased morbidity and mortality. Some studies demonstrate that treatment with continuous positive airway pressure (CPAP) can have a beneficial effect on both pathologies. This review summarizes the existing evidence of the association between asthma and OSA at their pathophysiological, epidemiological, clinical, and therapeutic levels. It intends to raise awareness among healthcare professionals about these conditions and the need for further research.
Introduction
Asthma and obstructive sleep apnea (OSA) have distinct origins but share commonalities. Asthma is a chronic inflammatory airway disease with variable expiratory airflow limitation, while OSA involves the partial or complete collapse of the upper airways during sleep. 1 The prevalence of both conditions is rising, with asthmatic individuals having a higher likelihood of OSA. 2
There exists a bidirectional interaction between asthma and OSA, influencing each other and sharing common risk factors and comorbidities such as GERD, obesity, and AR. They adversely impact disease severity through different mechanisms in both upper and lower airways.3,4 OSA exacerbates airway inflammation in asthmatic individuals, leading to poor disease control, while asthma increases upper airway collapse, worsening OSA’s clinical course. 1
Given the significant impact of OSA on asthma, including increased inflammation, airway remodeling, and heightened exacerbations, there is a proposal to recognize OSA as a distinct asthma phenotype, 5 or even a specific treatable trait, 6 considering their correlation and phenotypical particularities. This review aims to provide an updated understanding of the correlation between OSA and asthma, exploring evidence at pathophysiological, epidemiological, clinical, and therapeutic levels.
Definition
Obstructive sleep apnea
The International Classification of Sleep Disorders, third edition, categorizes sleep-related respiratory disorders into four types, including obstructive sleep apnea (OSA), central sleep apnea syndrome, sleep-related hypoventilation disorders, and sleep-related hypoxemia disorders. OSA is identified as the most prevalent in the general population. 7 The definition of OSA has evolved over time, transitioning from its initial description in 1976 to the recent simplification of nomenclature to “obstructive sleep apnea” and its abbreviation “OSA”. 8
OSA is diagnosed based on criteria such as an apnea-hypopnea index (AHI) ≥ 15 events per hour or AHI ≥5 events per hour with accompanying symptoms like excessive daytime sleepiness. 9 It involves partial or complete upper airway obstruction during sleep, resulting in altered breathing patterns, intermittent hypoxia, and sleep fragmentation. 10 This disorder is associated with decreased quality of life and various medical comorbidities, including arterial hypertension, cardiac arrhythmias, and an increased risk of heart failure. 9
Long-term consequences of OSA include chronic inflammation, endothelial dysfunction, metabolic dysregulation, and an elevated risk of stroke, type 2 diabetes, metabolic syndrome, cancer, and neurocognitive impairments. Polysomnography is the gold standard for diagnosis, and continuous positive airway pressure (CPAP) therapy is the primary treatment, achieving AHI normalization in 90% of cases. Other therapeutic options include mandibular advancement devices, surgery, upper airway stimulation therapy, positional therapy, myofunctional therapy, and weight management in overweight patients.9,10
Asthma
Asthma is a heterogeneous disease characterized by chronic airway inflammation, featuring variable respiratory symptoms such as wheezing, shortness of breath, chest tightness, and coughing, along with fluctuating expiratory airflow limitations. The severity and duration of symptoms vary, with some patients experiencing spontaneous resolution or positive responses to medications, while others endure exacerbations and severe asthma, particularly prevalent in middle- and low-income countries where most asthma-related deaths occur. 11 This condition is influenced by a spectrum of factors, encompassing environmental elements such as air pollution and allergens, host factors like obesity and infections, and genetic components, including susceptibility genes and sex. 10
The Global Initiative for Asthma (GINA) recognizes five common phenotypes, including allergic asthma, characterized by early onset and responsiveness to inhaled corticosteroids (ICS); non-allergic asthma, exhibiting variable responses to ICS; and adult-onset asthma, often associated with non-allergic traits and potential occupational factors. Additionally, a specific asthma phenotype is linked to obesity, where patients may experience significant respiratory symptoms even without eosinophilic airway inflammation, underscoring the complex interplay between asthma and obesity and emphasizing the need for personalized management strategies. Another phenotype is described as asthma with persistent airflow limitation, associated with the gradual remodeling of the airway walls over time. 11
Approximately 3%–5% of asthmatic individuals have severe asthma, defined by the European Respiratory Society (ERS) and the American Thoracic Society (ATS) as requiring high-dose ICS plus a second controller (and/or systemic corticosteroids) to prevent or manage uncontrolled symptoms. 12 The latest Consensus Document on Severe Asthma in Adults from the Spanish Society of Pneumology and Thoracic Surgery (SEPAR) further refines this definition, characterizing uncontrolled severe asthma as poorly managed despite a year of treatment with a combination of high-dose ICS/long-acting β2-agonists (LABA) and long-acting anticholinergics (LAMA) or requiring maintenance oral glucocorticoids (lasting 6 months per year, regardless of the dose or cumulative dose exceeding 1 g of prednisone or equivalent). 13
Consequently, severe asthma is defined as necessitating multiple medications (steps 5 and 6 of the Spanish Guideline for Asthma Management (GEMA) and GINA) or remaining poorly controlled despite these treatment steps. 13 Moreover, individuals with asthma frequently suffer from reduced sleep quality due to heightened nocturnal symptoms, resulting in a higher prevalence of excessive daytime sleepiness compared to non-asthmatic individuals. 5
Epidemiology
Asthma stands as the most prevalent chronic respiratory disease, ranking as the 16th leading cause of disability, affecting over 339 million people globally and contributing to around 420,000 annual deaths worldwide.14,15 In parallel, OSA is the most common chronic sleep-related respiratory disorder, impacting nearly one billion individuals. 1 It primarily affects older men and also extends its impact to children and women, especially postmenopausal women.1,16,17
Despite being separate medical conditions, individuals with asthma exhibit a higher prevalence of OSA than non-asthmatic individuals. A meta-analysis conducted by Kong et al. revealed a 49.5% prevalence of OSA in asthmatic patients, ranging from 36.39% to 62.60%, with a 2.64 times higher risk of presenting OSA compared to non-asthmatics. 3 This has mainly been attributed to structural changes and upper airway collapsibility during sleep, potentially linked to the use of inhaled and oral corticosteroids. 2 Approximately 35.1% of OSA patients also receive an asthma diagnosis, likely due to chronic intermittent hypoxia causing inflammation and remodeling of the lower airway. 18
The risk of developing OSA escalates with asthma severity, especially in difficult-to-control cases, reaching up to 9 times higher than in non-asthmatics. This heightened risk is more prevalent in women, particularly in the older population.1,18 Asthmatic patients with OSA also tend to have a higher body mass index (BMI) compared to those without OSA. 3 In contrast, OSA associates with unfavorable outcomes in asthma exacerbations, leading to more emergency department visits, decreased quality of life, and poorer disease control. 2
Pathophysiology
Both asthma and OSA exhibit distinct yet interconnected pathophysiological mechanisms. Independent pathways contribute bidirectionally to their severity, with shared risk factors and comorbidities. 18 Proposed mechanisms for worsened asthma control in OSA patients include neuro-mechanical reflex bronchoconstriction, gastroesophageal reflux (GER), inflammation, and dyspnea induced by OSA-related cardiac dysfunction. 19 On the other hand, severe asthma, steroid treatment, and comorbidities like gastroesophageal reflux disease (GERD) can lead to pharyngeal alterations promoting OSA development. 10
OSA pathophysiology primarily involves a collapsible upper airway associated with factors such as obesity. Non-anatomical factors like reduced muscle tone, low arousal threshold, and prolonged apnea contribute to cyclic hypoxemia, intrathoracic pressure variations, sleep fragmentation, and an inflammatory cascade causing harmful effects. 20 OSA’s characteristics impact distal airways, triggering a predominantly neutrophilic response, potentially leading to tissue injury. 21 Furthermore, chronic intermittent hypoxia in OSA induces inflammation marked by type 1 T helper (Th1) activation, resulting in collagen accumulation, matrix degradation, and airflow restriction. 22
Asthma’s pathophysiology involves innate and adaptive immunity activation, leading to chronic airway inflammation, edema, mucus accumulation, and remodeling. The inflammatory mechanisms that induce these permanent structural changes are associated with a combination of the response from Th1, type 2 T helper (Th2), and type 17 T helper (Th17) cell subsets, with the Th2 response playing a major role in the development of the disease. This is also associated with predisposing genetic factors. Th2 response plays a significant role, stimulated by inhaled allergens, leading to bronchoconstriction. 23 In patients with severe asthma, increased neutrophils and elevated IL-8 levels in sputum are observed.21,24
Traditionally, asthma has been characterized by an inflammatory mechanism primarily associated with the Th2 pathway. However, an alternative pathway has been described in the presence of OSA, where both Th2 and Th1 responses are simultaneously activated, contributing to asthma heterogeneity, especially airway remodeling. 24 OSA patients, especially those with thinner reticular basement membranes, exhibit specific neutrophilic asthma phenotypes and airway remodeling, potentially resisting steroid treatment.21,24
Moreover, there are two primary asthma endotypes identified: Th2-high (eosinophilic) and Th2-low (non-eosinophilic), also described as T2 high and non-T2, respectively. The Th2-high phenotypes encompass early-onset allergic asthma, late-onset eosinophilic asthma, and aspirin-exacerbated respiratory disease (AERD). Characterized by the absence of T2-high disease markers like eosinophilia, the Th2-low endotype exhibits higher expression of Th1 and Th17 inflammatory responses. This category includes non-atopic, obesity-associated, smoking-associated, and very late-onset asthma. 25 Nevertheless, none of the described endotypes, regardless of their main inflammatory response, have incorporated OSA as a significant factor related to asthma.
Impact of obstructive sleep apnea on asthma
OSA’s major impact on asthma is systemic inflammation activation, releasing cytokines and proinflammatory mediators (TNF-a, IL-6, VEGF, pentane, 8-isoprostane, CRP, leptin, MMP-9). 18 This process renders asthmatic patients more susceptible to severe crises. 10 OSA also leads to elevated neutrophils in sputum, causing airway inflammation, remodeling, steroid resistance, and heightened asthma severity. 18 On the other hand, mechanical stress during apneas and snoring induces local inflammation in respiratory tracts, worsening asthma. Edema, uvula hypertrophy, and tissue fibrosis contribute to this effect. 26
Simultaneously, OSA patients experience hypoxia and increased vagal tone during sleep, triggering nocturnal asthma attacks, as well as an augmented laryngeal stimulation which elevates parasympathetic nerve activity, causing reflex bronchoconstriction. Additionally, OSA leads to nocturnal blood accumulation in the lungs due to negative intrathoracic pressures, further intensifying bronchial hyperreactivity. 19 Apnea episodes raise cholinergic tone, activating muscarinic receptors and contributing to bronchial constriction. Structural airway changes foster a predominantly neutrophilic asthma type, hindering treatment response.19,27
Asthma’s effect on OSA
Asthma’s impact on OSA includes systemic inflammation and the use of corticosteroids, leading to pharyngeal remodeling, adipose tissue redistribution, and upper airway myopathy. This reduces protective effects during sleep, increasing susceptibility to snoring and OSA.2,10 This association is mainly seen in cases of severe asthma and difficult-to-control asthma, which are considered high-risk factors for developing OSA. 18
Asthma is associated with elevated negative intrathoracic pressures and greater pharyngeal collapsibility during asthmatic attacks. This results in intensified nocturnal symptoms, sleep fragmentation, apnea episodes, and daytime sleepiness.10,18 Additional asthma-related factors contributing to OSA risk include inflammation’s impact on control and chemoreception, influencing upper airway permeability. Asthma’s association with atopy, along with a higher prevalence of snoring in atopic women, suggests a pathogenic role of asthma in sleep disorders.10,28
Shared inflammatory pathway
In addition to the mutual effect on systemic inflammation activation, both asthma and OSA exhibit dysregulation in L-arginine and nitric oxide (NO) metabolism (Figure 1). This imbalance involves an altered ratio between L-arginine and asymmetric dimethylarginine, leading to the inhibition and uncoupling of nitric oxide synthase—an enzyme responsible for converting L-arginine into L-citrulline and NO.2,29 This includes reduced availability of L-arginine, lower NO levels, and increased arginase activity.
29
These shared characteristics extend to both individuals with OSA and obese individuals with asthma, hinting at a potential relationship in L-arginine metabolism. However, a comprehensive investigation into the role of L-arginine metabolism in individuals with asthma and OSA is yet to be fully explored.
2
Asthma and OSA pathophysiology, mutual impact, and shared inflammatory mechanisms.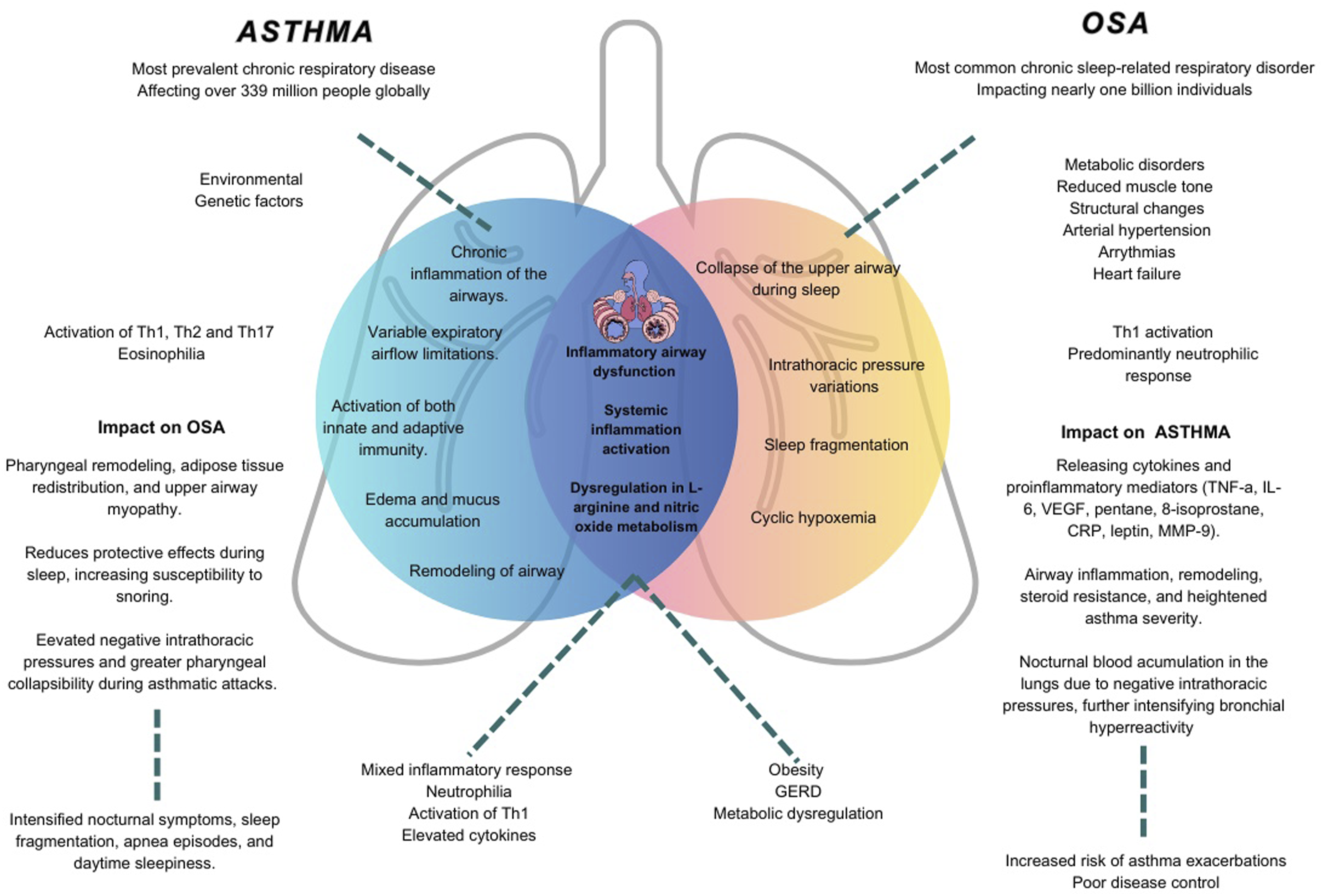
Connection between asthma, OSA, and obesity
Obesity, extending beyond a physical obstacle, profoundly impacts cellular homeostasis, interactions, and organ systems. Adipose tissue acts as a source of systemic inflammation, affecting the airway epithelium, triggering bronchial hyperreactivity, and recruiting inflammatory cells. 30
In OSA, obesity contributes fundamentally due to fat tissue accumulation around upper airways, narrowing the pharyngeal space, and causing intermittent obstruction. 9 Obesity in sleep apnea results in increased insulin resistance and leptin levels due to intermittent hypoxia, associated with obesity and metabolic syndrome. 30
Obesity significantly increases the risk of asthma by 50%, with prospective studies indicating a causal role as it precedes asthma onset, and weight gain associates with asthma development. 2 Individuals with both obesity and asthma experience greater severity of symptoms, more frequent exacerbations, and a diminished response to treatment, ultimately affecting their overall quality of life. 31
Connection between asthma, OSA, and GERD
GER involves the backward flow of gastric content into the esophagus, becoming GERD when symptomatic or causing esophageal damage. The prevalence of GERD, notably in Western Europe, North America, and South America, ranges from 20% to 40%, and 30% to 80% of people with asthma are affected.2,32 GERD is identified as a risk factor for recurrent asthma exacerbations, with an odds ratio of 4.9 (95% CI: 1.4-17.8) in a cross-sectional study, and an association with increased asthma exacerbations (rate ratio: 1.6; 95% CI: 1.3-2.0) in the Severe Asthma Research Program (SARP)-3.33,34
Two proposed mechanisms link GERD to asthma severity and exacerbations: micro aspiration of gastric contents causing airway inflammation and reflux-induced stimulation of the vagus nerve leading to bronchoconstriction. Asthma-related bronchoconstriction and hyperinflation may induce acid reflux due to pressure gradient alterations and lower esophageal sphincter (LES) abnormalities. 2 Antireflux treatment in asthma has shown symptom improvement but inconsistent effects on lung function, potentially due to varying treatment options and durations.2,32
OSA is frequently associated with GERD, with a prevalence of GERD in OSA patients of 65%, and a prevalence of OSA in GERD patients of 73%.2,35 The causal link involves increased intrathoracic pressure during apnea episodes causing LES opening and abdominal pressure increase, leading to reflux. Nocturnal GERD symptoms may also contribute to upper airway collapse.2,36 A meta-analysis by Wu et al. identified a significant association between OSA and GERD (OR: 1.75; 95% CI: 1.18-2.59), suggesting a strong connection between the two conditions. 37 Untreated OSA patients exhibit higher nocturnal reflux symptoms, and CPAP treatment reduce their prevalence, indicating potential efficacy for both diseases.38,39
Clinical implications
OSA is a significant risk factor for recurrent exacerbations in difficult-to-treat asthma, alongside factors like psychological dysfunction, respiratory infections, GER, obesity, and chronic sinus disease. However, the relationship between OSA and asthma remains unclear, posing questions about whether OSA in asthmatic patients is an independent comorbidity or an emerging asthma phenotype, given the added complexity to the inflammatory mechanisms in asthma with the development and occurrence of OSA. 5
On the other hand, since 2016, the concept of treatable traits has been proposed in the management of obstructive airway diseases, including asthma. This approach involves addressing patients in a multidimensional manner to identify and treat specific issues related to their condition. 6 A treatable trait is defined as a therapeutic target that must be identified using features such as biomarkers, genetic tests, or phenotypic characteristics. 40 Considering this framework, the correlation between OSA and asthma suggests a potential treatable trait, rather than a new phenotype, given the clinical impact between both diseases and the particular phenotypical aspects in these patients.
OSA symptoms overlap with asthma, making differentiation challenging. Higher BMI and daytime sleepiness in both conditions suggest potential coexistence, especially with frequent exacerbations or poor asthma control. 2 Notably, respiratory events during sleep, snoring included, manifest as obstructive hypopneas in asthmatic patients, potentially underestimating OSA severity. 21 Pulmonary function decline, quality of life reduction, and increased asthma exacerbation risk are linked to OSA in asthma. 41 Risk factors contributing to FEV1 decline in asthmatic patients include age, sex, smoking, exacerbations, obesity, and hypoxia.5,42
In a retrospective study conducted by Wang et al., 77 asthmatic patients diagnosed with OSA were selected. These patients were followed for over 5 years with spirometry. It was observed that asthmatic patients with OSA experienced a significantly greater decline in FEV1 per year compared to those without apnea. This decline was related to the severity of the AHI. The severity of OSA was the only independent factor associated with this decline in FEV1, even after adjusting for other confounding factors such as BMI, age, smoking, and the number of emergency department visits per year. Additionally, treatment with CPAP was observed to significantly reduce the decline in FEV1 in patients with severe OSA. 41
Similar findings were encountered in a meta-analysis conducted by Wang et al., in 2023, that investigated the effect of OSA on lung function in asthmatic patients, as well as the effect of asthma on polysomnography results in patients with OSA, yielding results that suggest an impaired lung function in asthmatic patients with OSA, especially in children, manifested by a decrease in FEV1. Although a trend towards a decreased FEV1 was also observed in adults, it did not reach statistical significance. 18
Furthermore, OSA has a negative impact on asthma symptoms, as evidenced by an increase in the asthma control test (ACT) scores and exhaled nitric oxide fraction (FeNO). 18 Moreover, individuals afflicted with both asthma and OSA encounter deleterious consequences, experiencing a 20% increase in hospitalization periods and a 25% increase in the frequency of exacerbations due to the concurrent presence of systemic inflammation. Additionally, a higher symptom burden has been identified in asthmatic patients with OSA through screening tools such as questionnaires. 43
Regarding the risk of mortality associated with these two conditions, a retrospective study in South Korea, which included 186,491 newly diagnosed asthmatic patients between 2004 and 2013, revealed a shorter median duration between asthma diagnosis and death in asthma patients with sleep disorders, emphasizing the increased risk of mortality with sleep disorders. Comprehensive management of asthma should address sleep disorders to improve long-term outcomes. 44 It is imperative to conduct screenings for OSA risk factors, notably obesity and reflux, as they play a pivotal role in averting severe asthma exacerbations.3,18,45
Treatment implications
Regarding treatment, CPAP in OSA benefits asthma-related quality of life, 46 especially in severe OSA or poorly controlled asthma, although lung function improvement is inconsistent.21,22 Long-term CPAP use shows improvements in asthma severity, ACT scores, asthma control, and a slowing of the decline in FEV1.41,47 The favorable impact of CPAP on sleep apnea has been attributed to the reduction of local and systemic inflammation and improvement in alveolar ventilation heterogeneity. 5 Short-term treatment with CPAP has been demonstrated to reduce airway hyperresponsiveness in asthma after 7 days. 46
A retrospective study identified, through questionnaires, an increase in ACT scores from 15.35 to 19.80, as well as a reduction in rescue medication use from 36% to 8% in patients using CPAP. 47 Additionally, a prospective multicenter study conducted by Serrano et al., involving 99 asthmatic patients with OSA, evaluated the effect of CPAP treatment over 6 months and found a decrease in exacerbation rate from 35.4% to 7.2%. 48 A randomized trial demonstrated reduced daytime sleepiness and improved quality of life with CPAP, emphasizing the need for further research on treatment impact. 49
Treatment options for asthma and OSA and their impact on each disorder.

Considering the bidirectional interaction between asthma and sleep apnea, screening for sleep apnea in asthmatic patients is valuable. Although polysomnography is the gold standard, practical limitations exist. A study recommends using the Stop-Bang questionnaire for early detection of sleep apnea in asthmatic patients.5,50 Physicians should diagnose and treat sleep apnea in asthma patients to delay airway remodeling and prevent lung function deterioration. 18
Conclusions
The accumulated information supports the existence of a bidirectional interaction between asthma and OSA, which is mediated by shared characteristics and risk factors that contribute to a worsening severity of both diseases. Several studies have indicated that patients with asthma have a higher risk of developing OSA. Furthermore, the presence of OSA has been observed to negatively impact the quality of life of asthmatic patients, leading to the proposition of the emergence of an asthma treatable trait.
Patients with OSA are more likely to present with non-Th2 asthma, which has a poor response to inhaled corticosteroids, the cornerstone of asthma treatment. It is crucial to continue researching to determine the percentage of symptoms in these patients that are attributable to OSA itself, as opposed to shared comorbidities such as GERD, sinusitis, and obesity.
Given the increasing incidence of sleep-disordered breathing in patients with severe asthma, the treatment of sleep apnea may improve asthma control and quality of life for these patients. Further studies are needed to determine the bidirectional impact of these conditions, but in general, physicians should screen, diagnose, and treat OSA in patients with asthma to slow down airway remodeling and prevent further deterioration of lung function.
Footnotes
Author’s note
All of the authors have approved the contents of this paper and have agreed to the journal´s submission policies.
Author contributions
All authors have read and approved the manuscript, and significantly contributed to this paper. VPM: Conception and design, literature review, manuscript writing correction, and final approval of the manuscript. CDI: Conception and design, Literature review, manuscript writing correction, and final approval of the manuscript. CS: Literature review, manuscript writing, correction, and final approval of the manuscript. FS: Literature review, manuscript writing, correction, and final approval of the manuscript. LFT: Conception and design, literature review, manuscript writing correction, and final approval of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data related to our study and materials will be available upon request by contacting the correspondence author (Liliana Fernández-Trujillo,
