Abstract
Background
Respiratory rate (RR) is used for the diagnosis and management of medical conditions and can predict clinical changes. Heavy workload, understaffing, and errors related to poor recording make it underutilized. Wearable devices may facilitate its use.
Methods
RR measurements using a wearable photoplethysmography-based monitor were compared with medical grade devices in complementary clinical scenarios: Study one included a comparison to a capnograph in 35 healthy volunteers; Study two included a comparison to a ventilator monitor in 18 ventilated patients; and Study three included a comparison to capnograph in 92 COVID-19 patients with active pulmonary disease. Pearson’s correlations and Bland-Altman analysis were used to assess the accuracy and agreement between the measurement techniques, including stratification for Body Mass Index (BMI) and skin tone. Statistical significance was set at p ≤ 0.05.
Results
High correlation was found in all studies (r = 0.991, 0.884, and 0.888, respectively, p < 0.001 for all). 95% LOA of ±2.3, 1.7-(−1.6), and ±3.9 with a bias of < 0.1 breaths per minute was found in Bland-Altman analysis in studies 1,2, and 3, respectively. In all, high accordance was found in all sub-groups.
Conclusions
RR measurements using the wearable monitor were highly-correlated with medical-grade devices in various clinical settings.
Trial registration
ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT03603860.
Keywords
Introduction
Respiratory rate (RR) is regarded as one of the basic measurements to assess a person’s physiological status. Within the clinical routine, trained medical staff are required to measure and record the patient’s RR as it is one of the major components of the diagnosis, management, and follow-up of several acute and chronic medical conditions. 1 Moreover, respiratory rate is known to be an early predictive vital sign of changes in the clinical status of patients.2–5 However, often it is underutilized and least recorded of all other basic vital signs,6,7 and for several reasons. Heavy workload and understaffing often do not allow proper time to complete a full 60-s measurement of manual counting, and in most cases, a less accurate 30-s or 15-s assessment multiplied by two or 4, respectively, is performed, leading to inaccuracies.7–9 Additional errors are related to the poor recording of the start and end of a breath, interruptions to counting, the motion of patients, or remembering a count. 10 Pulmonary diseases are diagnosed and monitored using devices that measure pulmonary function, which in many cases are uncomfortable and alter the breathing of the patients.11,12
Gaps and pitfalls associated with manual counting could be solved using methods that automate respiratory rate measurement. 13 There are several comfortable non-invasive methods capable of extracting respiratory-related information like thoracic bioimpedance, inductance plethysmography, or electromyography, but there is still a lack of sufficient evidence in clinical practice. 14 Thus, there is a need to validate simple methods of measuring respiratory rate to be used in diagnosing and monitoring pulmonary diseases. 15 Wearable technologies are regarded as compact devices that can be comfortably worn on a body, presenting data to users and allowing user interaction in various ways, thus providing relevant measurement capabilities while simplifying the workflow of health care providers. 16 Moreover, these devices were shown to have low false-positive alerts, early detection of patient deterioration, and could be used in multiple locations, inside and out-of-the-hospital. A relatively simple method is the optical photoplethysmography (PPG) method, often used in monitoring pulse oximetry. Several studies have shown the potential of this technique in measuring respiratory rate, yet the devices were not wearables hence with limited applicability.17–20 In this set of studies, we compared the respiratory rate measurements using a wearable PPG-based chest patch device with medical-grade devices for validation purposes. The unique feature of this specific device is its ability to collect all five basic parameters needed in clinical practice within a single device, that is blood pressure, pulse rate, blood oxygen saturation, body temperature, and respiratory rate. Moreover, as this is an optical sensor, we performed sub-analyses comparisons based on skin tone and body mass index (BMI).
Methods
Ethical consideration
This study includes data from three prospective, comparative clinical trials, approved by the Institutional Review Boards of the Tel-Aviv Sourasky Medical Center, Tel-Aviv, Israel (0032-15-TLV) and the Baruch Padeh Medical Center, Poriya, Israel (0077-18-POR and 0048-20-POR, NCT03603860). All participants were advised both orally and in writing as to the nature of the clinical study. Recruitment and signing the written informed consent forms were completed prior to their enrollment. Data collection periods were August 2018 until November 2018 for Study 1, February 2019 until February 2020 for Study 2, and March 2020 until August 2020 for Study 3.
Study outline
The PPG-based monitor was compared with accepted medical-grade devices in three clinical scenarios - healthy volunteers, post-surgery ventilated patients, and patients with acute respiratory disease.
Study 1: 35 healthy volunteers (ages 18–70 years; 18 males) were included. Manual measurements taken using a handheld capnograph (PC-900B portable capnograph and oximeter, Creative Medical, Shenzhen, China) were compared with the wearable PPG-based chest patch (Figure 1). The devices used in the studies to measure the respiratory rate. (a) Siemens Maquet Servo i Ventilator System; (b) PC-900B Handheld Capnograph and Oximeter, Creative Medical; (c) BB-613P, Biobeat Technologies Ltd.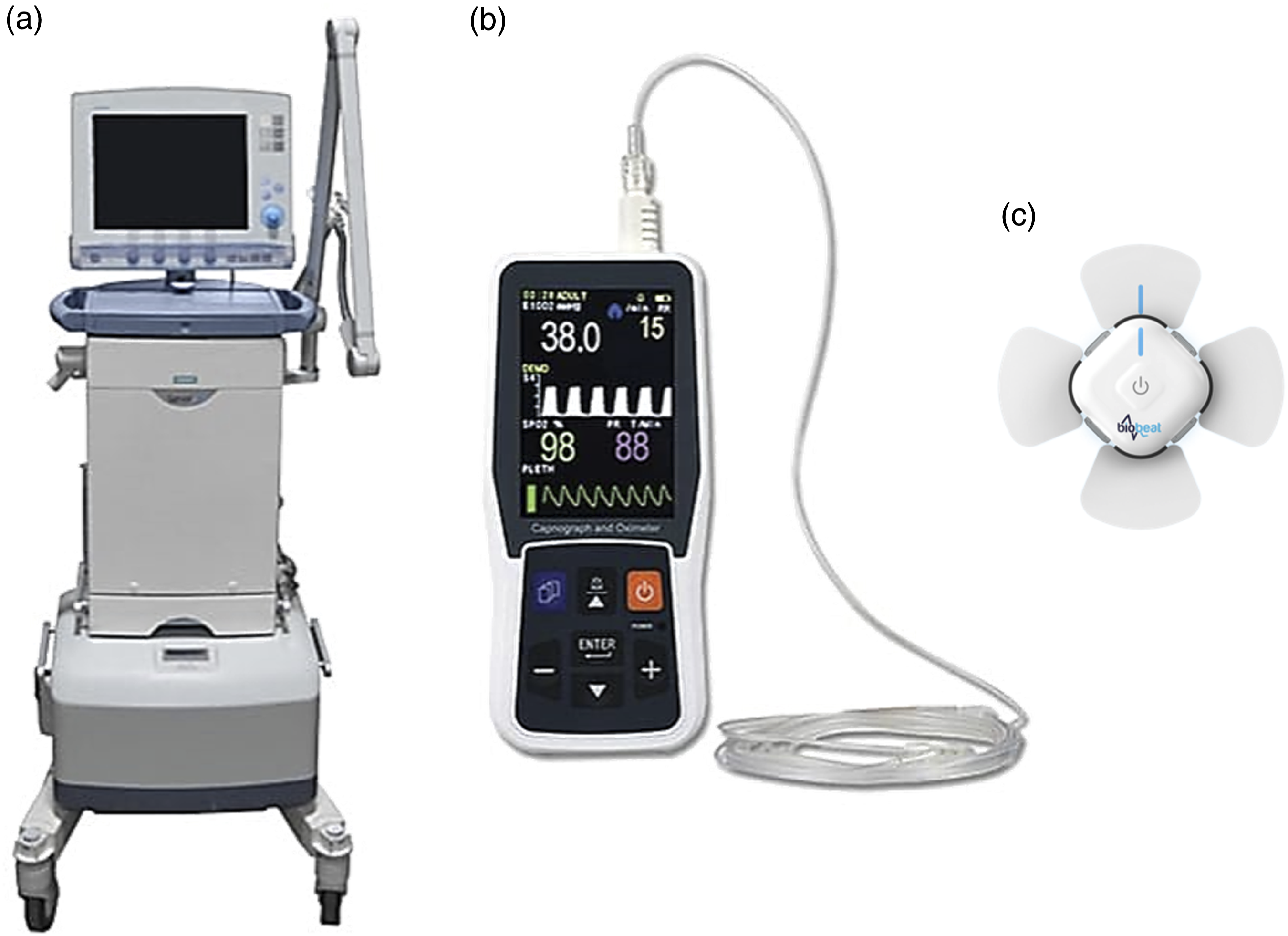
Study 2: Eighteen post-cardiac surgery patients (ages 18–81 years; 12 males) were recruited for the study. Monitoring started immediately after the surgical procedure, upon arrival to the Cardiac Surgery Intensive Care Unit (CSICU), and while patients were still ventilated. Upon arrival at the CSICU, they were connected to a Maquet Servo-i ventilator (Maquet Cardiovascular LLC, Wayne, NJ) and the wearable monitor (Figure 1). All parameters derived from the hospital monitoring systems were recorded once every minute using the medical center’s electronic medical record system and data from the wearable devices were automatically collected and recorded in the company’s cloud.
Study 3: 92 COVID-19 patients (ages 18–96 years; 52 males) were recruited for the study. Manual measurements taken using a handheld capnograph (PC-900B portable capnograph and oximeter, Creative Medical, Shenzhen, China) were compared with the wearable PPG-based chest patch.
In all studies, inclusion criteria were male and female subjects ≥18 years, exclusion criteria were pregnancy and subject younger than 18 years. In studies 1 and 2, the number of recruited patients was purposely chosen to stand regulatory demands, while in study 3, conducted within a COVID-19 isolation unit, patients were randomly recruited by the nursing staff. Only RR measurements of 4–40 breaths per minute (BPM) were included in the final analysis as this is the defined limit of measurement of the wearable devices.
The wearable device
The wearable chest patch device (BB-613P, Biobeat Technologies Ltd, Petach Tikva, Israel; Figure 1) is a wireless, wearable, non-invasive device that implements reflective PPG technology, which allows capturing unique characteristics of the PPG wave, including original wave markers. The PPG sensor provides pulse rate, respiratory rate, cuffless blood pressure, blood oxygen saturation, stroke volume, cardiac output, and more. 21 The method to monitor and track respiration rate using a PPG signal has been previously described in the literature.17–20 Briefly, during full inspiration, the increased intrathoracic pressure leads to a minute reduction in blood pressure, and hence the PPG signal becomes smaller, and when the lungs are empty, the PPG signal increases. By looking at the outer contour of the PPG wave, the respiration rate can be observed. To extract the respiration rate from the PPG wave, a filter within the respiration range is used to obtain an accurate respiration rate value.
Statistical analysis
Correlation analysis was performed using Pearson's correlation, and agreement was evaluated based on the Bland-Altman method using 95% limits of agreement (LOA).
Patients were stratified by BMI and skin tone based on the Fitzpatrick scale. 22 Analyses were performed by using IBM SPSS Statistics for Windows, version 25.0, IBM Corp (Armonk, NY). The analysis team had no access to the clinical data of the participants.
Results
No adverse events were recorded in any of the participants in all studies. Study one included 70 samples, two from each patient. 36 (52%) were from patients with normal weight (BMI <25), 24 (34%) were from patients with overweight (25 ≤ BMI <30), 10 (14%) were from patients with obesity (30 ≤ BMI), 42 (60%) were from patients with Fitzpatrick 1–3, and 28 (40%) were from patients with Fitzpatrick 4–6.
Bland-Altman analysis and correlation analysis of the three studies. Stratification based on body mass index (BMI) considered BMI <25 as normal weight, 25 ≤ BMI <30 as participants with overweight, and 30 ≤ BMI as participants with obesity. Skin tone stratification included fitzpatrick 1–3 and fitzpatrick 4–6. BPM – breathings per minute. LOA - limits of agreement. Significance was considered as p < 0.05.


Comparing respiratory rate measurements between the wearable PPG-based device and the handheld PC-900B in 35 healthy volunteers. (a) Bland-Altman analysis of all patients; (b) Pearson’s correlation curve of all patients; (c) Bland-Altman analysis of patients with normal weight (BMI <25); (d) Pearson's correlation curve of patients with normal weight (BMI <25); (e) Bland-Altman analysis of patients with overweight (25 ≤ BMI <30); (f) Pearson’s correlation curve of patients with overweight (25 ≤ BMI <30); (g) Bland-Altman analysis of patients with obesity (30 ≤ BMI); (h) Pearson's correlation curve of patients with obesity (30 ≤ BMI); (i) Bland-Altman analysis of patients with Fitzpatrick 1–3; (j) Pearson’s correlation curve of patients with Fitzpatrick 1–3; (k) Bland-Altman analysis of patients with Fitzpatrick 4–6; l) Pearson’s correlation curve of patients with Fitzpatrick 4–6.

Comparing respiratory rate measurements between the wearable PPG-based device and the Maquet Servo-i ventilator in 18 post-cardiac surgery patients. (a) Bland-Altman analysis of all patients; (b) Pearson–s correlation curve of all patients; (c) Bland-Altman analysis of patients with normal weight (BMI <25); (d) Pearson's correlation curve of patients with normal weight (BMI <25); (e) Bland-Altman analysis of patients with overweight (25 ≤ BMI <30); (f) Pearson's correlation curve of patients with overweight (25 ≤ BMI <30); (g) Bland-Altman analysis of patients with obesity (30 ≤ BMI); (h) Pearson’s correlation curve of patients with obesity (30 ≤ BMI); (i) Bland-Altman analysis of patients with Fitzpatrick 1–3; (j) Pearson’s correlation curve of patients with Fitzpatrick 1–3; (k) Bland-Altman analysis of patients with Fitzpatrick 4–6; l) Pearson’s correlation curve of patients with Fitzpatrick 4–6.

Comparing respiratory rate measurements between the wearable PPG-based device and the handheld PC-900B in 92 COVID-19 patients. (a) Bland-Altman analysis of all patients; (b) Pearson’s correlation curve of all patients; (c) Bland-Altman analysis of patients with normal weight (BMI <25); (d) Pearson's correlation curve of patients with normal weight (BMI <25); (e) Bland-Altman analysis of patients with overweight (25 ≤ BMI <30); (f) Pearson’s correlation curve of patients with overweight (25 ≤ BMI <30); (g) Bland-Altman analysis of patients with obesity (30 ≤ BMI); (h) Pearson's correlation curve of patients with obesity (30 ≤ BMI); (i) Bland-Altman analysis of patients with Fitzpatrick 1–3; (j) Pearson’s correlation curve of patients with Fitzpatrick 1–3; (k) Bland-Altman analysis of patients with Fitzpatrick 4–6; l) Pearson’s correlation curve of patients with Fitzpatrick 4–6.
Discussion
The goal of this study was to assess and validate RR measurements using a wearable wireless PPG-based sensor, compared with various medical grade devices, including capnography and measurement recorded by a ventilator. Moreover, participants were stratified based on BMI and skin tone, especially important when using a device relying on an optical measurement method. The potential of using PPG-based sensors for the measurement of RR was already shown before.17–20 In this set of studies, we have evaluated and validated the PPG-based sensor in various clinical settings, among healthy individuals and in patients suffering from COVID-19 and after cardiac surgery – two populations with known or potential lung injury. In our hands, we found high accordance between the PPG-based device and all other methods, showing its value in monitoring individuals in various clinical settings and environments. The high accordance was kept even when stratifying these populations based on BMI and skin tone, showing that the device is suitable and provides accurate and valid measurements in diverse populations.
As the merit of remote patient monitoring (RPM) becomes more evident, the need for RPM platforms that include various vital signs grows. To help reduce the workload of healthcare providers and support their workflow, RPM platforms must be able to completely replace the collection of vital signs by using, ideally, single devices that collect the various required vitals automatically. These are some of the advantages of the platform we have studied.
Moreover, recent studies showed promising results for non-contact methods to measure breathing and other related information, based on optical imaging, depth camera, or Doppler radar technologies.23–25 However, these methods are limited to fixed locations and are suitable for moderate movement conditions. This is another advantage of wearable technology as it could be used in any location. 15
A limitation of this study is the relatively small number of individuals included in each trial. Though it covers the requirements defined by the various regulators, more data from real-life settings would be important to substantiate the role of such RPM platforms in various clinical settings, both in the hospital and at home.
To conclude, we have shown that the RR measurements recorded by the wearable monitor are in high correlation with parallel measurements recorded with other medical-grade devices in various clinical settings and in a diverse population.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AE, NG, ABI, MF, MT, ADS and RM are employees of Biobeat Technologies Ltd. DN declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
