Abstract
Background
In this study, we will derive and validate a prognostic tool to predict in-hospital death based on Chinese acute exacerbation of chronic obstructive pulmonary disease (AECOPD) patients.
Methods
Independent predictors of in-hospital death were identified by logistic regression analysis and incorporated into a clinical prediction tool.
Results
The clinical prediction model was developed with data from 1121 patients and validated with data from 245 patients. The five predictors of in-hospital death from the development cohort (Arrival ward requiring help by wheelchair or medical cart, Arterial oxygenation index, Age, Albumin and Neutrophil count) were combined to form the AAAAN Score. The AAAAN Score achieved good discrimination (AUC = 0.85, 95% CI 0.81–0.89) and calibration (Hosmer-Lemeshow chi-square value was 3.33, p = 0.65). The AAAAN Score, which underwent internal bootstrap validation, also showed excellent discrimination for mortality (AUC = 0.85, 95% CI 0.81 to 0.89) and performed more strongly than other clinical prediction tools. Patients were categorized into 3 risk groups based on the scores: low risk (0–2 points, 0.7% in-hospital mortality), intermediate risk (3–4 points, 4.1% in-hospital mortality), and high risk (5–7 points, 23.4% in-hospital mortality). Predictive performance was confirmed by external validation.
Conclusions
The AAAAN Score is a prognostic tool to predict in-hospital death in Chinese AECOPD patients.
Introduction
Chronic obstructive pulmonary disease (COPD) is a worldwide public health challenge. It is the third leading cause of death and attributable disability-adjusted life-years worldwide, especially in developing countries.1–3 COPD is also a heavy health and economic burden in China. 4 In China, the total number of people living with COPD increased by more than 60% in the past decade, from 30.9 million in 1990 to 51.5 million in 2010. 5 Recently, estimated by a national cross-sectional study, the prevalence of COPD was 8.6% among the Chinese population aged over 20 years, with nearly 100 million COPD patients in 2015. 6
Acute exacerbation of COPD (AECOPD) is a leading cause of hospitalization. 7 Among hospitalizations for AECOPD, in-hospital mortality is considerable, which ranged from 2.5% to 28%.8–11 Therefore, accurate prognostication of patients hospitalized with AECOPD is important. For patients with AECOPD requiring hospitalization, prognostic tools for predicting in-hospital death risk have been investigated. A few studies have evaluated existing severity scores derived for alternative use, such as CRB-65 (Confusion, Respiratory rate ≥30/min, Blood pressure systolic <90 mmHg or diastolic 60 < mmHg and Age ≥65 years at the time of presentation to the hospital) Score 12 and CURB-65 (Confusion, Urea >7 mmol/L, Respiratory rate >30/min, Blood pressure systolic <90 mmHg or diastolic 60 < mmHg and age >65 years at the time of presentation to the hospital) Score.13,14 On the other hand, some studies have attempted to derive new scores such as BAP-65 (elevated Blood urea nitrogen, Altered mental status, Pulse >100/min, Age >65 years),15,16 CAPS (the COPD and Asthma Physiology Score), 17 and DECAF (Dyspnoea, Eosinopenia, Consolidation, Acidaemia and atrial Fibrillation) Score. 18
However, none of the prognostic tools above are based on the Chinese population. Then, are those previous prognostic tools suit for the Chinese population, or is there a new prognostic tool more suitable for the Chinese? Therefore, in this study, we intend to derive a prognostic tool to predict in-hospital mortality in Chinese patients hospitalized with AECOPD.
Methods
Ethical approval
The study protocol was approved by the Regional Human Research Ethics Committee of the Nanjing First Hospital, Sir Run Run Hospital Affiliated to Nanjing Medical University, and Wuxi People’s Hospital Affiliated to Nanjing Medical University. Individual patient consent was waived on the condition that all patient data were de-identified before evaluation because this study was a retrospective analysis.
Participants selection
To develop the prediction model, a development cohort was conducted at Nanjing First Hospital from January 2014 to January 2017. After development and internal validation, external validation was performed with data from Sir Run Run Hospital Affiliated to Nanjing Medical University and Wuxi People’s Hospital Affiliated to Nanjing Medical University between January 2021 and November 2022. The diagnostic criteria for AECOPD were as follows: (1) a history of COPD (the patient had been clearly diagnosed with COPD, with typical clinical COPD presentations and pulmonary functions); (2) an acute worsening of respiratory symptoms such as dyspnea, cough, or sputum purulence severe enough to warrant hospital admission. 19 The inclusion criterion was COPD exacerbation requiring hospitalization. The criteria for exclusion were patients without full medical records.
Data collection
The following data were collected from the medical records: demographic details (sex, age, and smoking), preexisting medical conditions (chronic cor pulmonale, pulmonary arterial hypertension, atrial fibrillation, hypertension, diabetes mellitus, coronary artery disease, chronic liver disease, anemia, cerebrovascular disease, and cancer), admission clinical data (heart rate, respiratory rate, temperature, diastolic blood pressure, systolic blood pressure, and hypercapnic encephalopathy), arterial blood gas values (pH, arterial oxygenation index, PaCO2), laboratory investigations (chloride, sodium, potassium, urea, creatine, haemoglobin, total protein, albumin, white cell count, neutrophil count, eosinophil count, lymphocyte count, and neutrophil–lymphocyte ratio). Lastly, in our study, we introduced a new variable: arrival ward with help or independent. According to arrival ward with help or independent, patients are divided into two groups: the independent group and the dependent group. The independent group defined that patients could walk to the ward independently. Conversely, the dependent group defined that patients were so weak that they could not walk to the ward by themselves, on the contrary, they arrived at the ward requiring help by wheelchair or medical cart.
Statistical methods
Categorical variables were expressed as frequencies and percentages. The continuous variables with a normal distribution were presented as mean ± standard deviation, and those with a non-normal distribution were represented as median and interquartile range. Categorical data between the groups were compared using the Chi-square test or Fisher’s exact test as appropriate. The comparison of normally distributed continuous variables between the groups was performed using the Studentt test. Non-normal distribution of continuous variables was performed using the Mann–Whitney U-test.
To develop a predictive tool, variables univariately associated (p < 0.05) with in-hospital death were dichotomised or categorised and retained for multivariate analysis. Cut-off points were identified using the following hierarchy: a clinically relevant cut-off; visual inspection of the receiver operator characteristic (ROC) curve; or a median split. Eligible variables were regressed against in-hospital mortality using multivariate logistic regression analysis. Then, only five independent predictors were included in the final clinical prediction tool. Scores for each predictor were assigned relative to the regression coefficient. Discrimination of the prediction tool was measured by the area under the ROC curve (AUC). Calibration was assessed using the Hosmer–Lemeshow goodness-of-fit test and satisfied when the p value was >0.05.
Internal validation of the tool was assessed by calculating the AUC of 10,000 bootstrapped samples. Two dummy prognostic models were developed using: all significant independent categorical predictors of in-hospital death and a ‘full’ model containing all significant independent predictors of in-hospital death in their original form (i.e., with variables on a continuous scale where appropriate). Last, the AUCs for predicting in-hospital death between the developed clinical prediction tool and the two dummy prognostic models were compared.
The AUCs for predicting in-hospital death between the developed clinical prediction tool and the previous prediction tools (CURB-65, CRB-65, CAPS, and BAP-65) were compared. Decision curve analysis could ascertain whether the implementation of the developed prediction tool in clinical practice would be beneficial and which of the alternative tools should be chosen. In the validation cohort, we calculated the AAAAN scores of individual patients according to the developed scoring system. The outcome was a calculated predicted probability on which we built a new AUC, evaluating the discrimination of the developed scoring system. Calibration was assessed using the Hosmer–Lemeshow goodness-of-fit test. Data were analyzed using SPSS v22.0 (IBM Corporation, Armonk, NY, USA) and R software for Windows (version 3.5.0; R Core Team, 2018).
Results
Population description and comparison between survivors and those who died in-hospital.

Abbreviation: PaC02, arterial partial pressure of carbon dioxide.
Using the methods described above, the following categorical variables were entered in a multivariate logistic regression analysis: age ≥75 years; coronary artery disease; heart rate ≥100/min; diastolic blood pressure <60 mmHg; hypercapnic encephalopathy; pH < 7.3; arterial oxygenation index <300; PaCO2 > 45 mmHg; chloride <100 mmol/l; potassium ≥4.5 mmol/l; urea ≥ 8 mmol/l; haemoglobin <120 g/L; total protein <60 g/L; albumin <30 g/L; white cell count ≥10 × 109/L; neutrophil count ≥8 × 109/L; eosinophil count <0.05 × 109/L; lymphocyte count <0.9 × 109/L; neutrophil-lymphocyte ratio ≥7.45; and arrival ward requiring help by wheelchair or medical cart.
Independent predictors of in-hospital mortality according to the “categorical” multivariate regression.
Independent predictors of in-hospital mortality according to the “full” multivariate regression.

The AAAAN score.

The AAAAN Score displayed adequate discrimination between patients who survived to discharge or died in hospital (AUC = 0.85, 95% CI 0.81–0.89) and achieved good calibration (Hosmer–Lemeshow chi-square value was 3.33, p = 0.65). The area under the AAAAN Score ROC curve for predicting in-hospital mortality was 0.85 (95% CI 0.81–0.89), and the bootstrapped AUC was 0.85 (95% CI 0.81–0.89), indicating good internal validity.
Compared with the AAAAN Score (Table 4), there were no statistically significant differences between (a) the AUC for a dummy “categorical” model using all the variables in their categorical form (Table 2) (0.85 vs 0.86, p = 0.359) (Supplemental Figure S1(a)) and (b) the AUC for the “full” regression model using all variables in their original form (Table 3) (0.85 vs 0.87, p = 0.468) (Supplemental Figure S1(b)).
In our cohort, the AAAAN Score performed significantly better for the prediction of in-hospital mortality than: the CURB-65 (AUC = 0.76, AAAAN vs CURB-65, p < 0.001); the CRB-65 (AUC = 0.72, AAAAN vs CRB-65, p < 0.001); the CAPS (AUC = 0.76, AAAAN vs CAPS, p < 0.001); and the BAP-65 (AUC = 0.65, AAAAN vs BAP-65, p < 0.001), which have all been proposed as useful predictive instruments in AECOPD (Figure 1(a)). By decision curve analysis, the net benefit of applying the AAAAN Score to guide clinical decision-making was beneficial and also better than the other 4 prognostic tools (Figure 1(b)). Supplemental Table S1 showed the comparison of AUCs and cut-off values between the AAAAN Score and the other 4 prognostic tools. The highest AUC was that of the AAAAN Score (0.85), at a cut-off point of 5, with 80% sensitivity and 80% specificity. (a) Comparison of the AUCs between AAAAN Score and other prognostic tools (CURB-65, CRB-65, CAPS, and BAP-65) in the development cohort. (b) Comparison of decision curves between AAAAN Score and other prognostic tools (CURB-65, CRB-65, CAPS, and BAP-65) in the development cohort. Abbreviation: AUC = area under the receiver operator characteristic curve; AAAAN = Arrival ward requiring help by wheelchair or medical cart, Arterial oxygenation index, Age, Albumin and Neutrophil count; CURB-65 = Confusion, Urea >7 mmol/L, Respiratory rate >30/min, Blood pressure systolic <90 mmHg or diastolic 60 < mmHg and age >65 years at the time of presentation to hospital; CRB-65 = Confusion, Respiratory rate ≥30/min, Blood pressure systolic <90 mmHg or diastolic 60 < mmHg and Age ≥65 years at the time of presentation to hospital; CAPS = the COPD and Asthma Physiology Score; BAP-65 = Blood urea nitrogen, Altered mental status, Pulse >100/min, Age >65 years.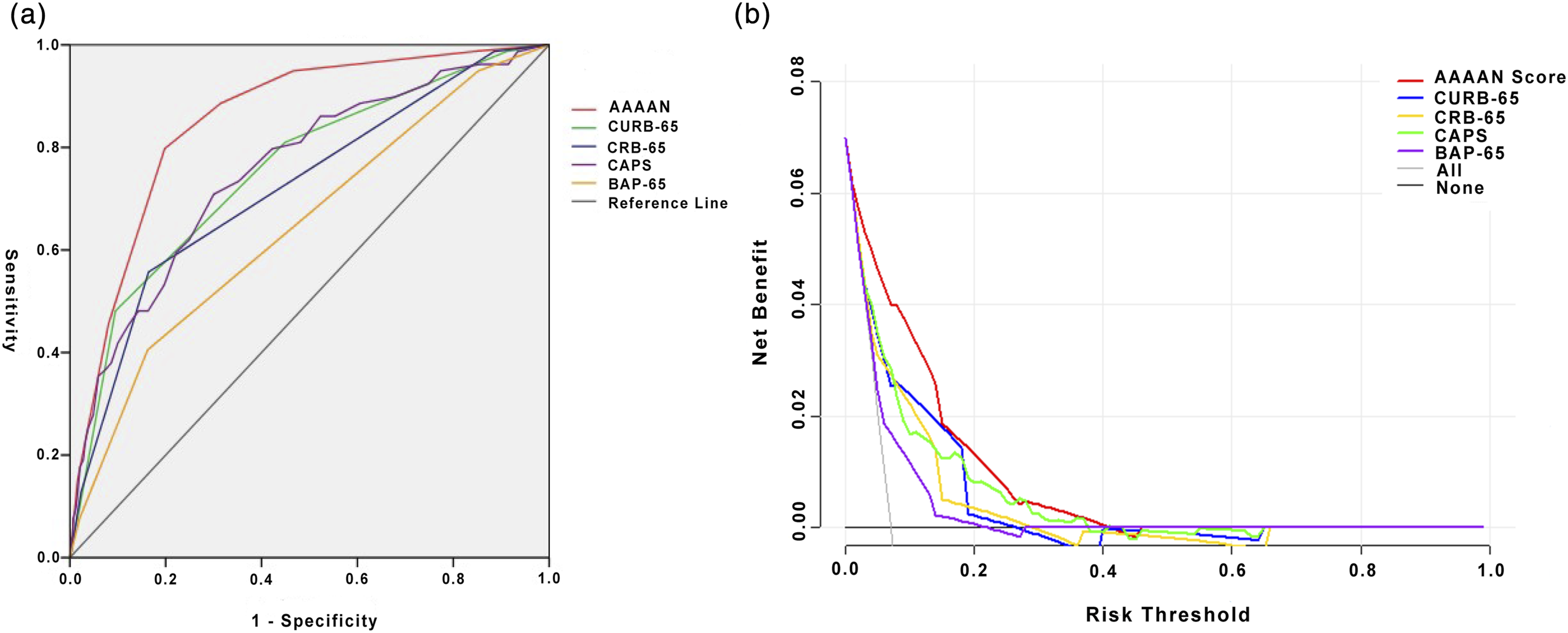
In-hospital mortality according to the AAAAN Score.

Supplemental Table S3 showed the comparison between survivors and those who died in-hospital for the validation cohort. The AAAAN Score also performed well in the validation dataset and achieved good discrimination (AUC = 0.86, 95% CI 0.78–0.93) and calibration (Hosmer-Lemeshow chi-square value was 8.76, p = 0.12). The AAAAN Score still performed significantly better for the prediction of in-hospital mortality than: the CURB-65; the CRB-65; the CAPS; and the BAP-65. (Supplemental Figure S2)
Discussion
In the present study, we identified several risk factors for in-hospital death. Most predictors of in-hospital death had been reported in previous studies, such as advanced age,8,18,20,21 low arterial oxygenation index,22,23 low serum albumin,10,18,24 and high serum urea 24 and neutrophil count. 25 However, to our knowledge, this is the first study to report that arrival ward requiring help by wheelchair or medical cart is an independently predictive factor of in-hospital death. In our study, according to the arrival ward with help or independent, patients are divided into two groups: the independent group and the dependent group. The independent group defines that patients can walk to the ward by themselves. Conversely, the dependent group defines that patients are so weak that they cannot walk to ward by themselves, while they arrive at the ward requiring help by wheelchair or medical cart. We find that patients who are so weak that they require a wheelchair or medical cart to arrive at the ward have a higher in-hospital death risk. In addition, the predictors of arrival ward requiring help by wheelchair or medical cart are similar to the mobility on level surfaces (immobile, use of wheelchair, walk with help, or independent) of Barthel Index. 26 Importantly, the information about the arrival ward with help or independently is regularly recorded in the medical records in China. Therefore, it may become an available predictor of in-hospital death for Chinese patients with AECOPD.
In this study, we compare the AAAAN Score and other prognostic tools for predicting in-hospital death risk. The AAAAN Score performs significantly better for the prediction of in-hospital death than: CRB-65, CURB-65, CAPS, and BAP-65. There have been several previous studies reporting prognostic tools (CURB-65, CAPS, and BAP-65 Scores) predicting in-hospital death for Chinese patients with AECOPD.27–29 Two studies reported that the AUCs of the CURB65 for death were 0.74.27,28 In our study, the AUC of the CURB65 for death is similar to 0.75. The AUC of BAP-65 for death was 0.67 in the previous study, 28 which is also similar to our study for 0.65. In addition, the AUC of CAPS was 0.71,29 which is 0.76 in our study.
The prediction score includes 5 variables that ranged from 0 to 7 points. Furthermore, patients are categorized into 3 risk groups based on the scores: low risk (0–2 points, 0.7% in-hospital mortality), intermediate risk (3–4 points, 4.1% in-hospital mortality), and high risk (5–7 points, 23.4% in-hospital mortality). To determine the optimal threshold value for predicting in-hospital mortality, the Youden index is used, and the best cut-off in the present model is 5 points (with a sensitivity of 80% and specificity of 80%). The in-hospital mortality is nearly one fourth in patients with scores ≥5 points.
The overall in-hospital mortality in our study was 8%. In previous studies, the in-hospital mortality of AECOPD all over the world, apart from China, ranged from 2.5% to 25%.8,18,20,30 A meta-analysis by Hoogendoorn et al. found that in-hospital mortality varied from 2.5% to 14% and the average in-hospital mortality was 7% in patients with AECOPD. 31 However, the in-hospital mortality rates were slightly lower in China, which ranged from 1.4% to 10.1%.22,23,28,32–35 Pu et al. conducted a prospective cohort study with 12831 AECOPD inpatients, and found that the in-hospital mortality rate was 1.4%. 32 Peng et al. conducted a retrospective study with 6668 patients hospitalized for AECOPD, and reported that the in-hospital mortality was 1.9%. 33 Five other studies with relatively smaller samples reported in-hospital mortality rates of 6.0%, 22 7.0%, 34 7.2%, 35 9.7%, 28 and 10.1%, 23 respectively. Therefore, the observed in-hospital mortality rate in our study is in accordance with previous studies.
There are some limitations in our study. First, it is a retrospective study from a single center, so its inherent weakness cannot be avoided. We lacked some data, such as pulmonary function tests, forced expiratory volume in the first second (FEV1), smoking status, packs per year, extended Medical Research Council Dyspnoea (eMRCD) score and frailty. In the future, prospective randomized controlled trials with large sample sizes and multi-center are warranted to validate the AAAAN Score to predict in-hospital death in Chinese patients with AECOPD. Second, complicating consolidation is common in AECOPD. However, we do not evaluate the AAAAN Score in the subgroups of patients with or without consolidation. Third, the COPD severity stage is an important characteristic for patients with AECOPD, and we lacked these data. Fourth, the DECAF score is a well-known, simple, and effective tool for predicting mortality in patients hospitalized with AECOPD. However, because we really lack data on available clinical data, for example, the eMRCD score, the comparison with the DECAF score was not included in this study.
In conclusion, this clinical prediction tool can help clinicians make a preliminary judgement on the risk of Chinese patients hospitalised with AECOPD and stratify patients into clinically relevant risk groups. High-risk patients may require early escalation of care and priority treatment.
Supplemental Material
Supplemental Material - The arrival ward requiring help by wheelchair or medical cart, arterial oxygenation index, age, albumin and neutrophil count score: Predicting in-hospital mortality in Chinese patients with acute exacerbations of chronic obstructive pulmonary disease
Supplemental Material for The arrival ward requiring help by wheelchair or medical cart, arterial oxygenation index, age, albumin and neutrophil count score: Predicting in-hospital mortality in Chinese patients with acute exacerbations of chronic obstructive pulmonary disease by Dawei Chen, Caimei Chen, Pan Zhang, Feng Zhang, Hao Zhang, Qing Sun, Jian Sun, Yan Tan, Binbin Pan and Xin Wan in Chronic Respiratory Disease
Footnotes
Author contributions
DC and XW contributed substantially to the study design, data analysis and interpretation, and the writing of the manuscript. CC, PZ, FZ, HZ, QS, JS, YT, and BP helped conduct the study, and collect and analyze the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from Xinghuo Talent Program of Nanjing First Hospital, Nanjing Health Science and Technology Development Special Fund Project (Grant No. YKK21129), and The Six-one Project of Top Talents in Jiangsu Province (Grant No. LGY2020014).
Ethics statement
The study protocol was approved by the Regional Human Research Ethics Committee of the Nanjing First Hospital, Sir Run Run Hospital Affiliated to Nanjing Medical University, and Wuxi People’s Hospital Affiliated to Nanjing Medical University. Individual patient consent was waived on condition that all patient data were de-identified before evaluation because this study was a retrospective analysis.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
