Abstract
Background:
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2023 report revised the combined assessment, merged the C and D groups into the E group, and revised the initial inhalation therapy recommendation.
Objectives:
This study aimed to analyze the future exacerbation and mortality of different inhalation therapies among patients with chronic obstructive pulmonary disease (COPD) in various groups based on the GOLD 2017 and GOLD 2023 reports.
Design:
This is a multicenter and retrospective study.
Methods:
Stable COPD patients from the database setup by 12 hospitals were enrolled. The patients were divided into Groups A, B, C, D, and E according to the GOLD 2017 and GOLD 2023 reports. Then, the patients were classified into long-acting muscarinic antagonist (LAMA), long-acting β2-agonist (LABA) + inhaled corticosteroid (ICS), LABA + LAMA, and LABA + LAMA + ICS subgroups. Data on exacerbation and death during 1 year of follow-up were collected.
Results:
A total of 4623 patients were classified into Group A (15.0%), Group B (37.8%), Group C (7.3%), Group D (39.9%), and Group E (47.2%). The exacerbation, frequent exacerbation, and mortality showed no differences between different inhalation therapies in Groups A and C. Patients treated with LABA + LAMA or LABA + LAMA + ICS had a lower incidence of exacerbation and frequent exacerbation than patients treated with LAMA or LABA + ICS in Groups B, D, and E. The exacerbation, frequent exacerbation, and mortality showed no differences between different inhalation therapies after combining Groups A with C.
Conclusion:
Patients in Group A should be recommended to undergo mono-LAMA, while patients in Groups B and E should be recommended treatment with LABA + LAMA, which is consistent with the GOLD 2023 report. However, it is worth considering merging Groups A and C into a single group and recommending mono-LAMA as the initial inhalation therapy.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is the most common chronic respiratory disease with high morbidity and mortality, which has become the third leading cause of death. 1 Therefore, treatment and prevention are urgently needed.
The goal of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) program is to produce recommendations for the management of COPD based on the best scientific information available and revise these annually. Until GOLD 2017, the combined assessment tool classified COPD patients into Groups A, B, C, and D based on the COPD assessment test (CAT) or modified Medical Research Council (mMRC) and exacerbation history. For patients in Group A, the initial inhalation therapy recommendation should be a short- or long-acting bronchodilator. For those in Group B, it should be a long-acting muscarinic antagonist (LAMA) or long-acting β2-agonist (LABA). Patients in Group C should receive a LAMA and those in Group D should receive a LAMA, LAMA + LABA, or LABA + inhaled corticosteroid (ICS). 2
The latest GOLD 2023 report revised the assessment tool and initial inhalation therapy recommendations, after which Groups C and D were merged into Group E. In addition, initial inhalation therapy recommendations for Group A should be a short- or long-acting bronchodilator, while Group B should receive LAMA + LABA and Group E should receive LAMA + LABA or LAMA + LABA + ICS therapy. 3 However, further studies are needed to confirm whether the latest combined assessment tool and initial inhalation therapy recommendations are more effective in reducing the risk of future exacerbation and mortality.
Therefore, the purpose of this study was to analyze the future exacerbation and mortality of different inhalation therapies among patients with COPD in various groups according to the GOLD 2017 and GOLD 2023 reports.
Patients and methods
Study participants
This was a multicenter and retrospective study. All subjects were from the outpatient COPD database (Register number: ChiCTR-POC-17010431) which included 12 comprehensive hospitals (Supplemental Table 1). The patients had been diagnosed with COPD between December 2016 and March 2022 according to the GOLD 2017 report: the ratio of forced expiratory volume in 1 s to forced vital capacity (FEV1/FVC) was <0.70 after inhaling a bronchodilator. 4 Patients with asthma, lung cancer, pneumonia, active tuberculosis, pulmonary fibrosis, and severe heart, liver, or kidney disease were excluded from this study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 5
Data collection
The baseline data including age, sex, body mass index (BMI), smoke history, biofuel exposure history, FEV1 %pred, FEV1/FVC, GOLD grades, CAT scores, mMRC scores, exacerbations, and hospitalizations in the past year and inhalation therapy regimens were collected. All patients were followed up for a duration of 1 year, during which the number of exacerbations, hospitalizations, and mortality was collected.
Study procedures
According to the GOLD 2017 report, the patients were assigned to four groups. Group A, 0 or 1 exacerbation per year and no hospitalizations, CAT scores <10 and mMRC scores of 0–1; Group B, 0 or 1 exacerbation per year and no hospitalizations, CAT scores ⩾10 or mMRC scores ⩾2; Group C, ⩾2 exacerbations or ⩾1 hospitalization per year and CAT scores <10 and mMRC scores of 0–1; Group D, ⩾2 exacerbations or ⩾1 hospitalization per year and CAT scores ⩾10 or mMRC scores ⩾2. 2 Then, each group was classified into the LAMA, LABA + ICS, LABA + LAMA, and LABA + LAMA + ICS groups based on the inhalation therapy received at their first hospital visit.
According to the GOLD 2023 report, the patients were assigned to three groups. Group A, 0 or 1 exacerbation per year and no hospitalizations, CAT scores <10 and mMRC scores of 0–1; Group B, 0 or 1 exacerbation per year and no hospitalizations, CAT scores ⩾10 or mMRC scores ⩾2; and Group E, ⩾2 exacerbations or ⩾1 hospitalization per year. 3 Then, each group was classified into the LAMA, LABA + ICS, LABA + LAMA, and LABA + LAMA + ICS groups based on the inhalation therapy they received at their first hospital visit. Patients with adjusted inhalation therapy were excluded from this study during 1 year of follow-up.
Variable definitions
Exacerbation is a COPD progression that requires antibiotics, oral corticosteroids, or hospitalization. 6 Frequent exacerbation was defined as at least two exacerbations per year. 7 A current smoker has had smoking exposure of ⩾10 pack-years, while a former smoker has had ⩾10 pack-years but has not smoked for more than 6 months. 8 Adjusted inhalation therapy was defined as changing the inhalation therapy drugs or stopping inhalation therapy drugs for more than 3 months during 1 year of follow-up. 9
Statistical analysis
Continuous variables are expressed as mean ± standard deviation or median and interquartile range (IQR). Continuous variables with a normal distribution and homogeneity of variance were analyzed with analysis of variance; otherwise, non-parametric tests were used. Multiple comparisons of differences between groups were Bonferroni adjusted. The chi-square test or Fisher’s exact test was used to analyze categorical variables. Risk factors for future exacerbation and mortality were identified and their adjusted odds ratios (aOR) and 95% confidence intervals (CI) were estimated using logistic regression analyses. Two sides and a value of p < 0.05 were considered to be statistically significant. SPSS 26.0 (IBM, Armonk, NY, USA) and Free Statistics software version 1.7.1 (Beijing, China) were used for statistical analysis of the data.
Results
The clinical characteristics of the COPD patients
A total of 4623 patients with COPD were enrolled in this study (Figure 1). The mean age was 65.7 ± 9.0 years and the majority were male (85.6%). The patients were assigned to Group A (15.0%), Group B (37.8%), Group C (7.3%), Group D (39.9%), or Group E (47.2%; Table 1).

Flow chart of this study.
The baseline clinical characteristics of the total COPD patients.

BMI, body mass index; CAT, COPD assessment test; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; GOLD, global initiative for chronic obstructive lung disease; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist; mMRC, modified medical research council.
After 1 year of follow-up, the proportion of patients suffering exacerbation, frequent exacerbation, and hospitalization were 27.7%, 12.5%, and 15.5%, respectively. The mortality was 2.2% (Table 2).
The future exacerbation and mortality in total COPD patients during 1 year of follow-up.

COPD, chronic obstructive pulmonary disease; IQR, interquartile range.
The future exacerbation and mortality of different inhalation therapies in Group A patients
There was a total of 694 patients in Group A who were assigned to LAMA (37.3%), LABA + LAMA (15.9%), LABA + ICS (16.7%), and LABA + LAMA + ICS (30.1%) groups. They were then adjusted for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year. There was no significant difference in future exacerbation, frequent exacerbation, hospitalization, and mortality among the LAMA, LABA + LAMA, LABA + ICS, and LABA + LAMA + ICS groups (Table 3 and Supplemental Table 2).
The future exacerbation and mortality of different inhalation therapies during follow-up in Groups A and B patients.
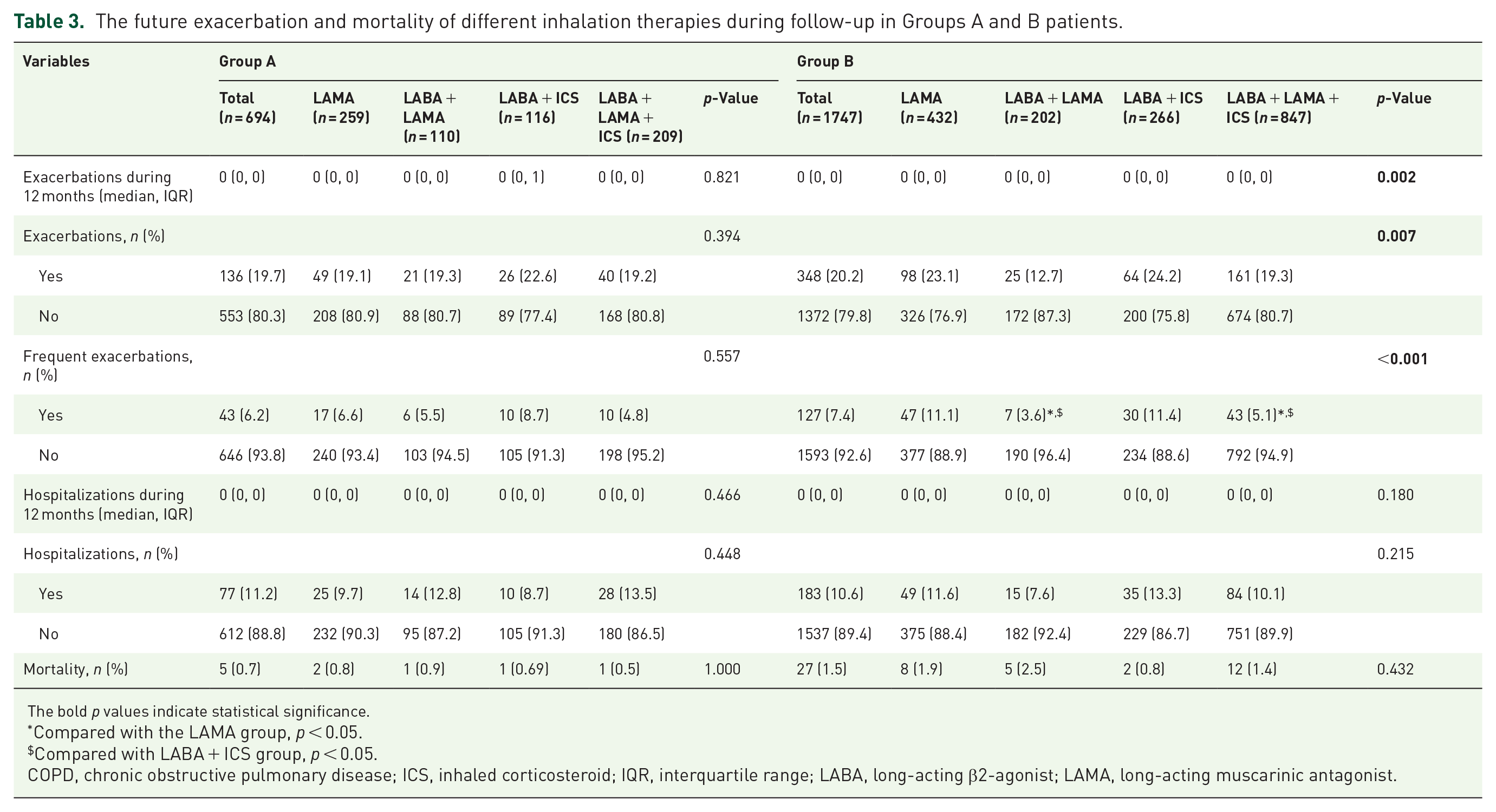
The bold p values indicate statistical significance.
Compared with the LAMA group, p < 0.05.
Compared with LABA + ICS group, p < 0.05.
COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; IQR, interquartile range; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist.
The future exacerbation and mortality of different inhalation therapies in Group B patients
A total of 1747 patients were in Group B and assigned to either the LAMA (24.7%), LABA + LAMA (11.6%), LABA + ICS (15.2%), or LABA + LAMA + ICS (48.5%) groups. After being adjusted for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year, the patients treated with LABA + LAMA or LABA + LAMA + ICS had a lower incidence of future exacerbation and frequent exacerbation than patients treated with LAMA or LABA + ICS (p < 0.05; Table 3 and Supplemental Table 3).
The future exacerbation and mortality of different inhalation therapies in Group C patients
A total of 336 patients were in Group C and assigned to the LAMA (29.5%), LABA + LAMA (14.9%), LABA + ICS (21.4%), and LABA + LAMA + ICS (34.2%) groups. After being adjusted for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year, there was no significant difference in future exacerbation, frequent exacerbation, hospitalization, and mortality among the LAMA, LABA + LAMA, LABA + ICS, and LABA + LAMA + ICS groups (Table 4 and Supplemental Table 4).
The future exacerbation and mortality of different inhalation therapies during follow-up in Groups C and D patients.

The bold p values indicate statistical significance.
Compared with the LAMA group, p < 0.05.
Compared with LABA + ICS group, p < 0.05.
COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; IQR, interquartile range; LABA, long-acting β2-Agonist; LAMA, long-acting muscarinic antagonist.
The future exacerbation and mortality of different inhalation therapies in Group D patients
A total of 1846 patients were in Group D and assigned to the LAMA (22.4%), LABA + LAMA (10.8%), LABA + ICS (11.7%), and LABA + LAMA + ICS (55.1%) groups. Afterward, patients were adjusted for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year. The patients treated with LABA + LAMA or LABA + LAMA + ICS had a lower incidence of future exacerbation and frequent exacerbation than patients treated with LAMA or LABA + ICS (p < 0.05; Table 4 and Supplemental Table 5).
The future exacerbation and mortality of different inhalation therapies in Group E patients
A total of 2182 patients in Group E and assigned to LAMA (23.5%), LABA + LAMA (11.4%), LABA + ICS (13.2%), and LABA + LAMA + ICS (51.9%) groups. After adjusting for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year. The patients treated with LABA + LAMA or LABA + LAMA + ICS had a lower incidence of future exacerbation and frequent exacerbation than patients treated with LAMA or LABA + ICS (p < 0.05; Table 5 and Supplemental Table 6).
The future exacerbation and mortality of different inhalation therapies during follow-up in Groups E and (A + C) patients.

The bold p values indicate statistical significance.
Compared with the LAMA group, p < 0.05
Compared with LABA + ICS group, p < 0.05.
COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; IQR, interquartile range; LABA, long-acting β2-Agonist; LAMA, long-acting muscarinic antagonist.
The future exacerbation and mortality of different inhalation therapies in Group (A + C) patients
A total of 1030 patients were in Group (A + C) and assigned to the LAMA (34.8%), LABA + LAMA (15.5%), LABA + ICS (18.3%), and LABA + LAMA + ICS (31.4%) groups. After being adjusted for confounding factors including age, sex, BMI, smoke history, biofuel exposure history, FEV1 %pred, CAT scores, mMRC scores, and exacerbation in the past year, there was no significant difference in future exacerbation, frequent exacerbation, hospitalization, and mortality among the LAMA, LABA + LAMA, LABA + ICS, and LABA + LAMA + ICS groups (Table 5 and Supplemental Table 7).
Discussion
Combined COPD assessment is an important tool to evaluate COPD patients in the GOLD reports and recommended initial inhalation therapy. This was different from the ‘ABCD’ groups based on the GOLD 2017 report where Groups C and D were merged into Group E to highlight the clinical relevance of exacerbation in the GOLD 2023 report. In addition, the initial inhalation therapy recommendations had changed and highlighted the important status of LABA + LAMA in Groups B and E based on the GOLD 2023 report. In fact, further studies are needed to validate the latest proposal. In this study, we are the first to analyze the future exacerbation and mortality of different inhalation therapies in various groups according to the GOLD 2017 and GOLD 2023 reports to validate whether the latest proposal is appropriate.
Although the initial inhalation therapy recommendation for patients in Group A should be a short- or a long-acting bronchodilator according to the GOLD 2023 report, mono-LAMA has in fact become the preferred initial therapy for patients in Group A among clinical practices. 10 A previous study confirmed that 97% of patients in Group A received a LAMA as the initial inhalation therapy. 11 In this study, we found that the future exacerbation and mortality showed no significant difference between the different inhalation therapies in Group A. In addition, we found that the patients treated with LABA + LAMA or LABA + LAMA + ICS had a lower incidence of exacerbation and frequent exacerbation compared to patients treated with LAMA or LABA + ICS in Groups B, D, and E. It is considered that ICS could increase the risk of pneumonia in COPD patients. Therefore, this implied that mono-LAMA should be recommended for patients in Group A, while LABA + LAMA should be recommended for patients in Groups B and E as the initial inhalation therapy. In other words, our findings support the updated GOLD 2023 report.
In fact, the number of patients in Group C was relatively small and had poor stability during follow-up. 12 In addition, we found that the future exacerbation and mortality showed no significant difference between different inhalation therapies in Group C. It is implied that mono-LAMA should be recommended for patients in Group C instead of LABA + LAMA. Considering the future exacerbation and mortality showed no significant difference between the different inhalation therapies in Group A. Therefore, we combined Groups A with C into a single group to analyze the outcomes. We found that the future exacerbation and mortality also showed no significant difference between different inhalation therapies after combining Groups A with C. Although several studies have confirmed that LABA + LAMA or LABA + LAMA + ICS were superior to LAMA or LABA + ICS in reducing future exacerbation and improving pulmonary function in COPD patients,13 –17 these studies only focused on total patients with COPD rather than determining drug effectiveness according to the GOLD groups. In addition, studies showed that the treatment with LABA + LAMA or LABA + LAMA + ICS was better than LAMA at improving symptoms and reducing the risk of exacerbation in more symptomatic COPD patients (including Groups B and D).18–19 However, our previous study found that there were no significant differences in exacerbation, hospitalization, and mortality during the follow-up among different inhalation therapies on less symptomatic COPD patients. 20 This study also confirmed that the patients in Groups A and C treated with LABA + LAMA or LABA + LAMA + ICS did not achieve better outcomes than patients treated with LAMA or LABA + ICS. Therefore, merging Groups C and D into Group E and recommending LABA + LAMA or LABA + LAMA + ICS as the initial inhalation therapy must be considered. However, merging Groups A and C into a single group (A + C) and recommending mono-LAMA as an initial inhalation therapy is also worth considering.
This study has some limitations. First, this is a retrospective study, meaning that randomized controlled trials are needed in the future. Finally, some studies have shown that the real-world prescriptions of initial inhalation therapy for COPD patients were not consistent with GOLD reports and the causes including pulmonary function, poor familiarity with recommendations, low self-efficacy, and time constraints might have an influence on the future exacerbation and mortality.20–21
Conclusion
Patients in Group A should recommended a mono-LAMA, while patients in Groups B and E should recommended a LABA + LAMA, which is consistent with the GOLD 2023 report. However, it is also worth considering merging Groups A and C into a single group and recommending a mono-LAMA as the initial inhalation therapy.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231213715 – Supplemental material for The future exacerbation and mortality of different inhalation therapies among patients with chronic obstructive pulmonary disease in various GOLD groups: a focus on the GOLD 2017 and GOLD 2023 reports
Supplemental material, sj-docx-1-tar-10.1177_17534666231213715 for The future exacerbation and mortality of different inhalation therapies among patients with chronic obstructive pulmonary disease in various GOLD groups: a focus on the GOLD 2017 and GOLD 2023 reports by Qing Song, Wei Cheng, Cong Liu, Xueshan Li, Ling Lin, Yating Peng, Yuqin Zeng, Rong Yi, Yi Liu, Xin Li, Yan Chen, Shan Cai and Ping Chen in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
