Abstract
Introduction
Lung cancer screening presents an important teachable moment to promote smoking cessation, but the most effective strategy to deliver support in this context remains to be established.
Methods
We undertook a systematic review and meta-analysis of smoking cessation interventions delivered during lung health screening, published prior to 20/07/2022 MEDLINE, PsychINFO, CENTRAL, EMBASE, CINAHL and Scopus databases. Two reviewers screened titles, and abstracts, four reviewed each full text using prespecified criteria, extracted relevant data, assessed risk of bias and confidence in findings using the GRADE criteria. The review was registered prospectively on PROSPERO (CRD42021242431).
Results
10 randomised controlled trials and three observational studies with a control group were identified. Meta-analysis of nine RCTs demonstrated that smoking cessation interventions delivered during lung screening programmes increased quit rates compared to usual care (odds ratios: 2.01, 95%: 1.49–2.72 p < 0.001). Six RCTs using intensive (≥3 behavioural counselling sessions) interventions demonstrated greater quit rates compared to usual care (OR: 2.11, 95% CI 1.53–2.90, p < 0.001). A meta-analysis of two RCTs found intensive interventions were more effective than non-intensive (OR: 2.07, 95%CI 1.26–3.40 p = 0.004), Meta-analysis of two RCTs of non-intensive interventions (≤2 behavioural counselling sessions or limited to online information audio take home materials such as pamphlets) did not show a higher quit rate than usual care (OR: 0.90, 95% CI 0.39–2.08 p = 0.80).
Discussion
Moderate quality evidence supports smoking cessation interventions delivered within a lung screening setting compared to usual care, with high-quality evidence that more intensive interventions are likely to be most effective.
Introduction
Tobacco use remains a primary driver of global morbidity, mortality, and related economic and healthcare costs, responsible for more than eight million deaths per year.1,2 Despite mortality rates declining for most cancers, lung cancer deaths have remained high, accounting for 21% of all cancer deaths in the UK. 3 Patients typically present to healthcare services at a late stage, resulting in late diagnosis and a poorer prognosis. 3 Early diagnosis through lung cancer screening programmes has achieved success in the USA and Europe. For example, the National Lung Cancer Screening trial in the US saw a 20% reduction in lung cancer mortality among their populations who underwent screening. 4 In 2019 the NHS rolled out their Targeted Lung Health Check (TLHC) pilot project with 77% of lung cancers identified being stage 1 and 2 5 and lung cancer screening has recently been recommended by the UK National Screening Committee. 6
Eligibility criteria for lung screening programmes vary slightly between countries, however participants are typically middle aged (55–75), a current or former smoker (quitting within the last 15 years), with a 30+ pack year history. Around 40–50% of people taking part still smoke, 7 and are likely to be highly nicotine dependent creating a significant barrier for cessation. 8 Attendance at screening programmes has been highlighted as a teachable moment for smoking cessation, with several randomised controlled trials (RCTs) of screening demonstrating increased quit rates among individuals taking part,8–11 however there is substantial scope to increase the quit rates observed (11–15%).
Increasing quit rates among this high-risk population has the potential to dramatically increase the impact and cost effectiveness of screening programmes, beyond their effect on early identification of lung cancers, by reducing the risk of future smoking related illness.12,13 Recent modelling estimates that 1.1 million smokers, around 15.4% of all the people in England who smoke, will meet the criteria for screening, representing a potentially huge public health benefit. 14 Identifying the most effective strategies for delivering cessation within a lung screening context is now recognised as a priority by multiple health organisations including the American Thoracic Society, NHS England, and the European Respiratory Society.15–17 Studies investigating the effects of delivering smoking cessation interventions in this context vary in terms of intervention type, frequency, and intensity of intervention. A systematic review published in 2016 concluded that lung screening programmes were optimal environments to deliver smoking cessation, however did not offer evidence on what type of smoking cessation delivery modality is most effective. 11
This area of screening and smoking cessation medicine is rapidly evolving, and despite organisations across Europe and the US recommending that cessation support be incorporated into lung screening programmes, they do not provide exact guidelines on what form this should take. We therefore conducted a systematic review and meta-analysis to determine the most effective delivery strategies for smoking cessation interventions conducted during lung health screening programmes.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) guidelines were used to complete and report this systematic review, which was registered prospectively on PROSPERO(CRD42021242431).
Inclusion criteria
1. Study type: randomized controlled trial (RCTs) and observational studies with a control group. 2. Population: current smokers enrolled in a lung health screening program, including any of the following interventions: Low Dose Computed Tomography (LDCT) scanning, thoracic radiograph, spirometry assessments, formal clinic visit with a physician specialising in respiratory health. 3. Intervention: a smoking cessation program (behavioural therapy, pharmacological, e-cigarette, motivational interviewing, group counselling, telephone counselling, online materials, and information leaflets). 4. Control: usual care or comparator as defined by the study. 5. Outcome measures: smoking quit rates, quit attempts and reductions in cigarettes smoked.
Exclusion criteria
1. Studies not published or translated into English. 2. Abstracts, conference posters, expert opinion, review articles, study protocols and student dissertations.
Search strategy
We searched MEDLINE, PsychINFO, CENTRAL, EMBASE, CINAHL and Scopus from 1950–20 July 2022. Our search strategy used exploded medical subject headings in the following: smoking OR tobacco OR vape or vaping OR e-cigarette OR cigar OR nicotine AND cessation OR cease OR stop OR quit OR intervention OR abstain OR abstinence OR relapse OR reduce OR give up AND screen OR scan OR check OR early detection OR early diagnosis OR CT) ADJACENCY2 lung OR thoracic OR pulmonary AND early detection of cancer OR tomography, emission-computed OR mass screening OR spirometry. We also reviewed reference lists of included studies.
Data extraction and quality assessment
Two researchers (PJW and NSH) reviewed all titles and abstracts for eligibility. Four independent reviewers (PJW, KEJP, SA, AP) read full text studies to decide on final inclusion and exclusion (Figure 1). Discrepancies were resolved by discussion between all co-authors. Data were extracted by PJW and AAL using a predefined standardised form and COVIDENCE software. Data recorded included study design and setting, methodology, participant baseline characteristics, type and intensity of smoking cessation intervention used, definition of control group, primary and secondary endpoints, measurement of abstinence and follow up period. Quality of the evidence including the risk of bias (ROB) was assessed by two independent reviewers (PJW and AAL) using the GRADE criteria with ROB determined using the Cochrane risk-of-bias assessment tool.18,19 A complete list of extracted data and GRADE criteria appraisal provided in online supplement. Prisma, study flow diagram.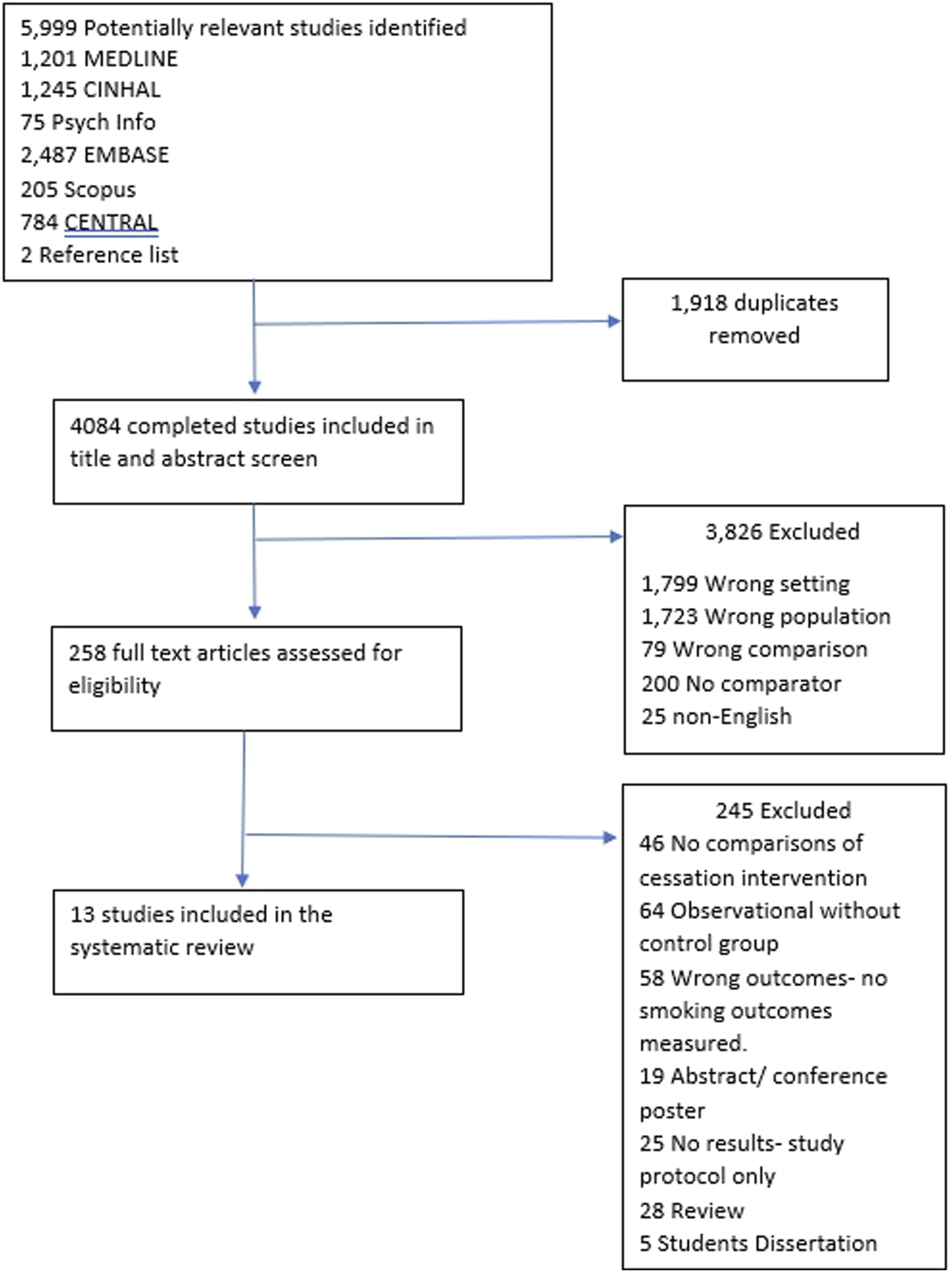
Studies were grouped by outcome assessment and intensity of the intervention. We defined intensive interventions as ≥3 behavioural counselling sessions and non-intensive interventions as those consisting of ≤2 behavioural counselling sessions or limited to online information or take home materials such as pamphlets.
Outcome measures
Primary outcomes
We considered smoking abstinence at the end of the trial study period (acknowledging that this could vary from trial to trial) as our primary outcome. Smoking abstinence could be defined as; the self-reporting of abstinence of smoking cigarettes in the past 7 days or more, following the cessation intervention, with or without biochemical verification. The measure of abstinence could include, point prevalence, prolonged or continuous and biochemical verification could include exhaled carbon monoxide (CO) or urine cotinine as set out in Piper et al.’s 2020 review for defining and measuring smoking abstinence in clinical trials. 20 To evaluate the effect of intervention intensity, we compared quit rate among studies that provided intensive interventions and non-intensive interventions, compared with control. We also assessed quit rates among studies that provided pharmacotherapy (NRT, E-cigarettes or prescription medications).
Secondary outcomes
Secondary outcomes included changes in smoking behaviour in terms of number of quit attempts and reductions in tobacco usage (daily cigarette consumption). We defined quit attempts as the number of individuals self-reporting a quit attempt post-cessation intervention and reduction in tobacco use as a reduction in the number of self-reported daily cigarettes from baseline.
We searched for information on intervention impacts in terms of long-term mortality, exacerbations of respiratory diseases and patient experience, however none of the screened or included articles measured these as endpoints.
Data analysis
A fixed effects meta-analysis was performed to estimate the pooled differences and 95% CI in quit rate and quit attempts between smoking cessation intervention groups and control group. A Mantel-Haenszel fixed effects model was used to obtain estimates for dichotomous data, expressed as odds ratios (OR). To answer our primary and secondary questions we performed the following meta-analyses on data from included RCT; quit rate between smoking cessation interventions versus usual care, quit rate between intensive smoking cessation interventions versus usual care, quit rate between intensive smoking cessation interventions + pharmacotherapy versus usual care, quit rate between non-intensive smoking cessation interventions versus usual care, quit rate between intensive + pharmacotherapy smoking cessation interventions versus non-intensive interventions, and quit attempts between smoking cessation interventions versus usual care. Heterogeneity among included studies was assessed using the I2 statistic. We contacted authors for missing primary and secondary endpoint data. The statistical analyses were performed using the Cochrane Collaboration’s Review Manager Software (RevMan V.5.2.0).
Results
Of 4084 initial studies, 3823 titles and abstracts were excluded, the main reasons for exclusion were, smoking cessation not delivered in a lung screening setting or population (wrong setting and wrong population), or no comparator group. This left 258 studies included in the full text screen, of these 13 studies were included (Figure 1). Reasons for exclusion were, no comparisons of cessation intervention, observational studies with no control group, abstracts, no smoking abstinence measures used as outcomes, review papers, study protocols and students’ dissertations. We also searched clinicaltrials.gov and the ISRCTN registry for ongoing trials in this area. We found 12 investigational trials comparing different smoking cessation interventions to control groups within a lung screening setting21–32 (Table E6: Online Supplement).
Overview of included studies
Summary of included studies.
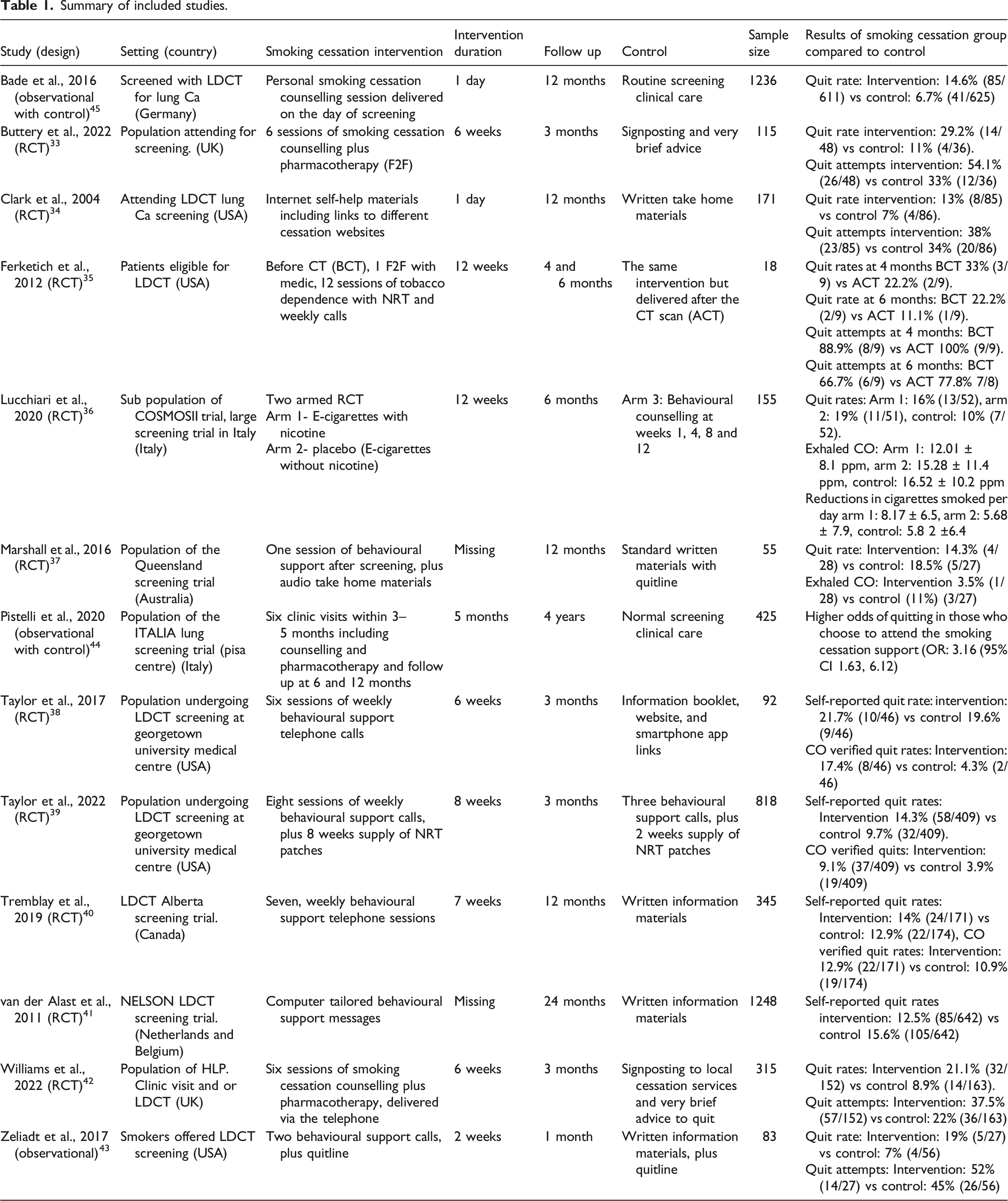
Studies investigated effectiveness of smoking cessation interventions as follows: single, in clinic session of behavioural counselling, 45 single, in clinic session of behavioural counselling and audio materials, 37 single in clinic behavioural counselling, plus NRT and subsequent weekly telephone calls, 35 internet self-help materials, 34 tailored information leaflet, 41 two behavioural support calls and Quitline, 43 multiple weekly behavioural support calls.38,40 multiple weekly behavioural support calls and pharmacotherapy,39,42 multiple weekly behavioural support, in clinic plus pharmacotherapy33,44 or e-cigarettes. 36 Timing of the intervention delivery varied across included studies from immediately on the day of the screening clinic to 5 months after screening, average length of interventions was 13.2±13.6 weeks.
A variety of comparisons were used for usual care (UC) groups e.g. signposting to local cessation clinics, written information leaflets and standard behavioural counselling, in addition to the intervention delivered before the lung health screening appointment. Of note Lucchiari et al., 36 used four sessions of behavioural counselling as their UC intervention, which is considerably more input compared to most UC interventions included.
All included studies used self-reported point prevalence smoking abstinence, either 30-days or 7-days, with five studies35,37–40 using exhaled carbon monoxide monitoring. Studies reported cessation outcomes at 1 month, 43 3 months (n = 4),33,38,39,42 4 and 6 months, 35 6 months, 36 12 months (n = 4),34,37,40,45 24 months, 41 and one at 4 years. 44 Five studies reported data on quit attempts33–35,42,43 and one study reported data on reductions in daily cigarettes smoked. 36
Description of included studies
Lucchiari et al., 36 conducted a three arm RCT, comparing the effect of behavioural counselling plus nicotine containing e-cigarettes, behavioural counselling plus nicotine free e-cigarettes (placebo) and behavioural counselling only (UC) on smoking behaviour in 155 smokers attending screening in Italy. Nine of the included studies were two-armed RCTs. In the QuLIT-1 trial Buttery et al., 33 randomised 115 smokers attending a targeted lung health check to immediate support including six sessions of behavioural counselling plus pharmacotherapy or very brief advice (VBA) to quit and signposting to local cessation services. Ferketich et al., 35 used a similar intervention (12 weeks tobacco cessation support, including pharmacotherapy), though this started a week after attending screening, rather than immediately. However, in this study the control group received the same smoking intervention delivered before the CT screening appointment (12 weeks of smoking cessation then attended screening on week 12) so it was not suitable for comparisons against “usual care”. Taylor et al., 38 randomised 92 smokers attending screening to receive six sessions of telephone behavioural counselling after participants had attended screening and received results, or a written information booklet (UC). A subsequent paper from that group randomised 818 smokers to receive intensive (8 telephone counselling sessions plus 8 weeks of NRT patch) or less intensive (3 counselling sessions, plus 2 weeks NRT patch), delivered 13 days post LDCT scan. 39 In the QuLIT-2 trial, Williams et al., 42 randomised 315 smokers attending a TLHC to receive six sessions of telephone behavioural support starting immediately, with pharmacotherapy prescriptions within 48 h of participating in the TLHC, or VBA to quit and signposting to local services (UC). A telephone intervention was also utilised by Tremblay et al., 40 who randomised 354 smokers attending LDCT screening to seven behavioural support calls delivered after participants had received screening results, or to written information leaflets only (UC).
Three RCT used low-intensity interventions; Clarke et al., 2004 34 randomised 171 smokers attending screening to receive internet materials or standard written materials delivered on the day of screening (UC). Marshall et al., 2016 37 randomised 55 smokers attending screening to receive one session of behavioural support on the day of screening with audio take home materials, or written information plus Quitline (UC). Van der Alast et al., 2012 41 randomised 1248 male smokers who were in the screening arm of the larger NELSON RCT, to receive computer tailored information leaflet or standard information leaflet after attendance to screening (UC).
The remaining three included studies used an observational design with a control group. Bade et al., 2016, 45 compared 12 months quit rates among 611 smokers attending screening who accepted the offer of one behavioural support session delivered before attendance to screening, to 625 smokers who received standard screening care. Pistelli et al., 2020, 44 explored quit rate among smokers at the Pisa centre in the ITALIA lung screening trial, screened participants were offered six smoking cessation clinic visits within 3–5 months of screening including counselling and pharmacotherapy (n = 119), those who did not accept the offer of cessation went onto routine clinical care (n = 306). Lastly, Zeliadt et al., 2017, used a waitlist design and gave smokers who recently attended a LDCT scan but had not received their results (n = 27), two telephone behavioural counselling sessions, plus Quitline and compared quit rates to recent attendees to LDCT who had received their results (n = 56).
Effect of stop smoking interventions on quit rate compared to usual care
To analyse the effectiveness of smoking cessation interventions, we performed a meta-analysis on included RCTs which compared quit rate between smoking cessation and usual care groups delivered in a screening context. Meta-analysis of eight RCTs eligible for inclusion33,34,36–40,42 (n = 1984) demonstrated that providing a smoking cessation intervention improved quit rates among current smokers enrolled in screening programmes compared with usual care (OR: 2.01, 95%: 1.49–2.72 p < 0.001), Figure 2. We included data from Arm 1 (e-cigarette with nicotine) and Arm 3 (usual care) from the Lucchiari et al.,
36
study, as this best answered the research question. Measures to assess quit rate varied between included studies, three studies used exhaled CO to confirm quits,38–40 the remaining five studies used self-reported point prevalence smoking abstinence to assess quit rate.33,34,36,37,42 Data from two RCT were excluded from the meta-analysis; Ferketich et al.,
35
as they did not use a usual care group, rather explored the effect of timing of the same intervention and van der Aalst et al.,
41
as their intervention group was too similar to their UC group (two types of information leaflet). Forest plot comparing quit rate among participants randomised to SC intervention compared with UC. Quit rates measured via self-reported (30-days or 7-days pp) or CO verification at follow up time periods ranging from 3 months to 12 months post intervention. Data from Arm 1 (E-cigarette with nicotine) and Arm 3 (UC) from Lucchiari et al.’s three armed RCT was included in this analysis. UC: usual care; SI: smoking cessation.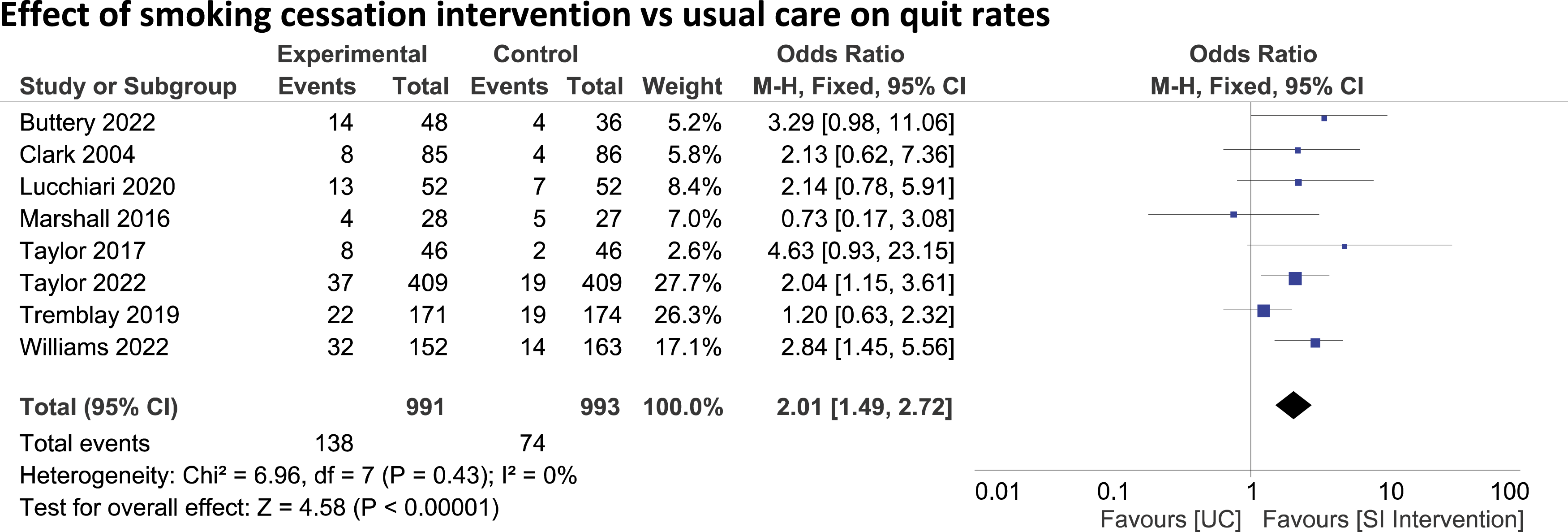
Effect of intensive cessation interventions on quit rates
Meta-analysis of the six RCTs (n = 1758)33,36,38–40 that provided an intensive smoking cessation intervention, defined as ≥3 sessions with a smoking cessation advisor, nurse, or councillor+/− pharmacotherapies, demonstrated that intensive smoking cessation interventions yield greater quit rates compared to UC (OR: 2.11, 95% CI 1.53–2.90, p < 0.001), Figure 3. We also conducted a meta-analysis among studies that provided pharmacotherapy (NRT, varenicline, e-Cigarettes), alongside behavioural counselling. Of note, only studies that provided intensive support were also the studies that provided pharmacotherapy. A meta-analysis of four RCTs33,36,39,42 (n = 1321), demonstrated that intensive smoking cessation support plus pharmacotherapy, yields higher quit rates compared to control among a screening population (OR: 2.40 95% CI 1.64–3.51, p < 0.001) (Figure 4). Forest plot comparing quit rates in studies that randomised participants to intensive SC interventions (≥3 behavioural counselling sessions compared with UC. Quit rates measured via self-reported (30-days or 7-days pp) or CO verification at follow up time periods ranging from 3 months to 12 months post intervention. Data from Arm 1 (E-cigarette with nicotine) and Arm 3 (UC) from Lucchiari et al.’s three armed RCT was included in this analysis. UC: usual care; SI: smoking cessation. Forest plot comparing quit rates in studies that randomised participants to intensive SC interventions (≥3 behavioural counselling sessions and pharmacotherapy compared with UC. Quit rates measured via self-reported (30-days or 7-days pp) or CO verification at follow up time periods ranging from 3 months to 6 months post intervention. Data from Arm 1 (E-cigarette with nicotine) and Arm 3 (UC) from Lucchiari et al.’s three armed RCT was included in this analysis. UC: usual care; SI: smoking cessation.

Effect of intensive interventions compared to non-intensive interventions on quit rate
A meta-analysis of two RCTs,36,39 (n = 922) investigating the effect of intensive interventions compared to usual care, in the case of these two studies their UC interventions would be classed at a non-intensive smoking cessation intervention itself. Results from the analysis demonstrated that intensive smoking cessation interventions compared to non-intensive smoking cessation interventions, results in higher quit rates (OR: 2.07, 95%CI 1.26–3.40 p = 0.004), Figure 5. Forest plot of self-reported quit rates in two studies that randomised participants intensive SC interventions compared with non-intensive SC interventions Quit rates measured via self-reported pp at 6 months follow up, and CO verification at 3 months follow up. Data from Arm 1 (E-cigarette with nicotine) and Arm 3 (UC) from Lucchiari et al.’s three armed RCT was included in this analysis. UC: usual care; SI: smoking cessation.
Effect of non-intensive interventions on quit rate
A meta-analysis of two RCTs34,37 (n = 226) investigating non-intensive smoking cessation interventions (≤2 behavioural counselling sessions) did not find higher quit rates compared to UC, (OR: 0.90, 95% CI 0.39–2.08 p = 0.80), Figure 6. Forest plot of self-reported quit rates in two studies that randomised participants to non-intensive SC interventions compared with UC (≤2 counselling sessions, online materials, audio self-help materials). Quit rates measured via self-reported pp at 12 months follow up. UC: usual care; SI: smoking cessation.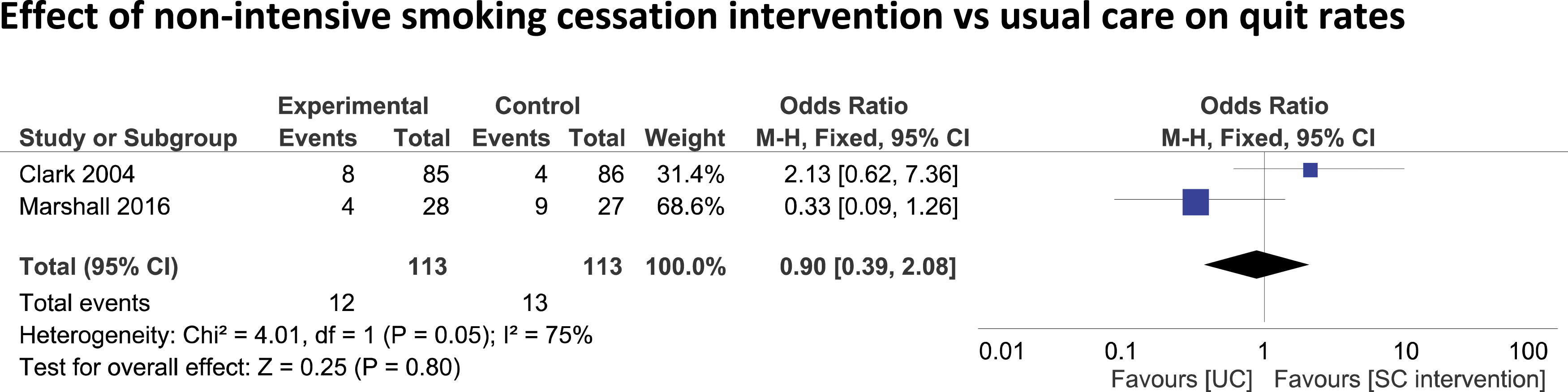
Effect of interventions on quit attempts
The impact of smoking cessation interventions on the number of participants reporting quit attempts could be compared in three RCTs.33,34,42 (n = 570), showing that smoking cessation interventions produce significantly higher quit attempts compared to UC interventions, within a screening context (OR: 1.85, 95%CI 1.28–2.66, p < 0.001) (Figure E1: Online Supplement).
Risk of bias and evidence quality
Risk of bias (ROB) and evidence quality was judged using the Cochrane risk-of-bias assessment tool.
19
Overall, ROB for each study was judged on study design and whether there was a clear or serious limitation. In the case of an unclear limitation the study was judged as having unclear ROB, if the study had a serious limitation, it was graded as high risk of bias. Four studies were assessed as having low risk of bias.33,36,39,42 We assessed four studies as having an unclear risk of bias,34,38,40,41 mainly attributed to unclear methods of blinding of outcome assessors and unclear methods of randomisation. The remaining five studies were deemed high risk of bias,35,37,43–45 attributed to the observational study design in three of the studies,43–45 with no attempt to blind outcome assessors, poor allocation concealment, selective reporting and low sample sizes seen in two of the studies37,43 (Figure 7). A more detailed description of risk of bias domains and scoring can be found on the online supplement (Table E1: Online Supplement). Risk of bias of assessment of included studies.
19

Using the GRADE criteria, 46 we found overall moderate quality evidence to support inclusion of smoking cessation interventions into lung cancer screening, attributed to strong association of results, directness of the evidence in terms of assessing our primary endpoint (quit attempts) and the precision of results (large sample size of included participants) (Table E2: Online Supplement). For intensive smoking cessation interventions, the GRADE criteria found high quality evidence supporting embedding intensive smoking cessation interventions into lung screening, compared both to usual care and to low intensity interventions, again attributed to strong association of results, directness of the evidence in terms of assessing primary endpoint (quit attempts), the precision of results (large sample size) and moderate inconsistency (heterogeneity) (Table E4: Online Supplement). In terms of non-intensive smoking cessation interventions, the GRADE criteria found low certainty evidence for non-intensive interventions, attributed to low and unclear risk of bias, large heterogeneity across included studies and the low sample sizes of Marshall et al., 2016 37 and Zeliadt et al., 2018 43 (Table E5: Online Supplement).
Discussion
This systematic review addressed the effectiveness of smoking cessation intervention strategies delivered as a component of lung health screening programmes. Overall, moderate quality evidence supports the embedding of smoking cessation interventions within screening programmes. Intensive interventions and those including pharmacotherapy support (NRT, varenicline or E-cigarettes) were supported by high quality evidence and appeared to be more effective at increasing quit rates. Where smoking cessation intervention intensity was low, the evidence supporting the recommendation of these strategies was low, suggesting a minimum intervention threshold needed to have an effect.
Given the well-established health and social harms from smoking, the value of lung cancer screening programmes will be enhanced by embedding smoking cessation interventions into them. Modelling suggests that providing behavioural cessation advice, plus pharmacotherapy can reduce the cost per quality adjusted life year (QALY) of a lung screening programme by up to 50%. 13 The economic evaluation of the Taylor et al., 2022 39 RCT comparing 8-weeks intensive intervention to 2-weeks minimal intervention, also estimated greater lifetime cost savings with cessation support. The incremental costs per QALY gained were estimated to be larger with the 8-weeks intensive counselling. 47
Results from our current review, suggest that providing more support (multiple counselling sessions and pharmacotherapy) compared with less-intensive approaches, may be more favourable for a screening population. However, there were only limited direct comparison data, leading to only a recommendation of moderate certainty. Two RCT found increases in quit rates with more intensive smoking cessation support compared with less intensive support. Lucchari et al., 2020 found that nicotine (e-cigarettes) with four sessions of behavioural counselling was more effective than behavioural support alone. 36 Taylor 2020 et al., demonstrated self-reported and CO verified quit rates were significantly higher among smokers who received eight counselling sessions plus 8 weeks of NRT patch vs three sessions plus 2-weeks supply of NRT, 39 which further supports the argument that smoking cessation interventions delivered in a screening context should provide multiple sessions and offer pharmacotherapy provision. We also note that all the intensive interventions included pharmacotherapy, so it is not possible from the available data to disentangle these two effects, although best practice is anyway for pharmacotherapy to be offered with behavioural support.
Of note, despite it being established that a combination of behavioural support combined with pharmacotherapy is likely to be the most effective modality for smoking cessation in general,48–50 many of the studies included in the review chose a less intensive approach. This may be attributed to the country the study was conducted in, in the USA pharmacotherapy prescriptions will likely have to be sourced via an individual’s health insurance plan.
The effectiveness of E-cigarettes as a stop smoking aid is set out in a recent UK NIHR Health technology assessment 51 and supported by NICE, while a living Cochrane review finds high certainty evidence that nicotine containing e-cigarette use increases quit rates compared to NRT. 52 However, this may in part be due to the typically longer duration of use of e-cigarettes compared to medicinal forms of NRT and other pharmacological agents. This probably reduces the risk of relapse to smoking, but longer-term use of e-cigarettes is itself likely to cause some health harms, albeit substantially less than those associated with smoking. Individuals should therefore be advised to quit vaping in due course if they can, though not at the expense of going back to smoking.
To date there has been one previous systematic review aimed at investigating effective smoking cessation interventions delivered during LDCT screening. Iaccarino et al., 53 conducted their review in 2019, concluding that the data was insufficient to recommend one smoking cessation intervention over another. Our current review also contains statistical pooled analyses of included studies enabling us to draw firmer conclusions from the data.
This review has certain limitations. The primary endpoint of smoking cessation was assessed at a range of time points in the studies included. The immediacy of intervention also varied as well as what was considered to be usual care. 36 This led to a high level of heterogeneity between the studies included in the meta-analysis, however this is to an extent addressed in the different analyses presented, comparing both any additional smoking cessation intervention versus usual care and more intense intervention versus less. Despite having no search limitations based on location, all included studies were conducted in high income countries, so generalisability to middle and low-income countries need to be considered. Additionally, tobacco use habits are changing and will be influenced by the social and cultural factors. As the studies included in this analysis were all conducted in Europe/USA, the extent to which these findings can be applied globally is unclear. Although not included in the meta-analysis, the findings of observational studies also support the conclusion that smoking cessation is effective in this context.
This review demonstrates that providing smoking cessation within lung health screening programmes increases quit rates. Given that smoking cessation is arguably the most successful and cost-effective intervention for reducing tobacco induced diseases, it is vital that smokers who attend screening clinics are offered support. This not only reduces risk for the individual but potentially increases the cost effectiveness of the screening intervention.13,47 The population who attend lung screening clinics will typically have a high degree of nicotine dependence and addiction, in addition to a higher risk of not only frequent respiratory exacerbations and ill health, 54 but also social isolation and loneliness.55,56 Because of that, we suggest that offering pharmacotherapy alongside behavioural counselling support, should be mandated when designing pathways to support smoking cessation within lung cancer screening. Finally, it is apparent that more research is needed within this area, particularly aimed at intensiveness and timing of the interventions.
Conclusion
Lung health screening settings are a key environment for delivering smoking cessation. All screening programmes should provide evidence-based smoking cessation alongside screening, and efforts should be made to provide sustained counselling support and provide pharmacotherapy.
Supplemental Material
Supplemental Material - Strategies to deliver smoking cessation interventions during targeted lung health screening - a systematic review and meta-analysis
Supplemental Material for Strategies to deliver smoking cessation interventions during targeted lung health screening - a systematic review and meta-analysis by Parris J Williams, Keir EJ Philip, Saeed M Alghamdi, Alexis M Perkins, Sara C Buttery, Michael I Polkey, Anthony A Laverty, and Nicholas S Hopkinson in Chronic Respiratory Disease.
Footnotes
Author’s note
Parris J Williams embedding smoking cessation into lung cancer screening increases quit rates over usual care interventions, with increased effect sizes when smoking cessation interventions are more intensive with pharmacotherapy.
Author contributions
PJW, KEJP, SMA, NSH and AMP conducted screening and data extraction, PJW and AAL conducted ROB analysis, PJW conducted pooled analyses with guidance from NSH and AAL. PJW produced the first draft to which all authors contributed. All authors have reviewed and approved the final manuscript and NSH is the guarantor.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NSH is medical director of Asthma + Lung UK and chair of Action on Smoking and Health, AAL is a trustee of Action on Smoking and Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by RM Partners West London Cancer Alliance, hosted by The Royal Marsden NHS Foundation Trust and Royal Brompton and Harefield Charities. KEJP was supported by the Imperial College Clinician Investigator Scholarship. The funders had no input into data analysis or the writing of this manuscript.
Disclosure
Nicholas Hopkinson, the manuscript’s guarantor affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
