Abstract
Azithromycin was thought to prevent acute exacerbations of chronic obstructive pulmonary disease (AECOPDs) by anti-microbial and anti-inflammatory effects. However, it′s value in the treatment of critically ill patients with AECOPD before ICU admission remains unclear. Our study aimed to find whether azithromycin use prior to ICU admission leads to better clinical outcomes for those individuals. 533 critically ill patients with AECOPD from the MIMIC-IV database were included. Univariate followed multivariate logistic regression was used to select risk factors for short-term mortality. The multivariable logistic regression models were implemented to investigate the association between azithromycin use before ICU admission and short-term mortality. Lower short-term mortality was observed in the azithromycin group (p = .021), independent of differences in demographic data and other clinical outcomes (p>.05). Azithromycin use before ICU admission was proved to have a decreased short-term mortality by multivariable logistic regression (p<.05). The results remained consistent after being stratified by age, SOFA scores, pH, and cancer diagnosis. Azithromycin use prior to ICU admission was associated with lower short-term mortality for critically ill AECOPD patients.
Keywords
Background
Chronic obstructive pulmonary disease (COPD) is still the third leading cause of death and disability worldwide, with an estimated medical cost of $36 billion annually in the United States.1–4 Up to 60% of these costs are attributed to acute exacerbations of COPD (AECOPD), which could bring negative effects on health status, quality of life, and hospitalization rates.1,4–6 When AECOPD is evaluated as life-threatening based on clinical presentations, further treatment in the intensive care unit (ICU) will be necessary.1,7 However, despite advances in medical care, the mortality of these patients remains high.8,9 Therefore, establishing an appropriate treatment regimen for critically ill patients with AECOPD is a high priority.
As a kind of macrolide antibiotic, azithromycin, with potent antimicrobial and immunomodulatory effects, was proved to bring large clinical benefits to COPD patients.10–12 After repeated validation, long-term use of azithromycin has been established to prevent AECOPD for patients with COPD and was included in authoritative guidelines.1,13,14 However, it remains controversial whether azithromycin should be routinely used at the beginning of AECOPD, especially for the severe ones requiring critical care in ICU, where azithromycin is often considered to be of limited benefit.1,11,12 To address this question, we conducted this study to evaluate whether azithromycin use prior to ICU admission could provide better clinical benefits for critically ill patients with AECOPD.
Methods
Data source
All data were obtained from the Medical Information Mart for Intensive Care IV (MIMIC IV) open-source clinical database (version 1.0), which contains information for more than 50,000 patients who were admitted to the intensive care unit (ICU) of the Beth Israel Deaconess Medical Center during 2008 to 2019.15,16 One author was obliged to complete the online course and pass the online exams to gain access to the database (Xuequn, Certification number: 35,875,386) after signing a data use agreement (https://physionet.org/content/mimiciv/view-dua/1.0/). Owing to all the data in the database being anonymous, the informed consent for the individual was not required.
Cohort selection
Our inclusion criteria were as follows: (1) Patients were older than 18 years and stayed in the ICU for more than 24 hours; (2) Patients were diagnosed as AECOPD by ICD-10 code17–19 on admission; (3) To ensure that patients were admitted to ICU because of AECOPD, combined type II respiratory failure was set as an inclusion criterion. Type II respiratory failure is defined by the max partial pressure of carbon dioxide (PaCO2) > 45 mmHg (6 kPa) and the min pH < 7.35 within 12 hours before ICU admission.
20
Exclusion criteria are surgically related complications like severe trauma, burns and vital organ surgery. The primary outcome was short-term (30 days) mortality. Only the data from the first hospital and the first ICU admission was retained (Figure 1). The flowchart of patient enrollment.
Data collection and outcomes
In the present study, we extracted data of baseline parameters including age, gender, ethnicity, body mass index (BMI), sequential organ failure assessment score (SOFA), blood gas results, comorbidities, some treatments like antibiotic use, non-invasive ventilator use and invasive ventilator use, and some clinical outcomes like short-term mortality, long-term mortality, hospital duration and ICU duration. Comorbidities were erected by the Elixhauser, a table created from past retrieval codes summarized in the ICD-10 codes.17,18 Data elicitation was performed by PostgreSQL (version 10, www.postgresql.org).
Statistical analysis
SKTEST order was run in STATA (Skewness and kurtosis tests for normality), which presents a test for normality based on skewness and another based on kurtosis and then combines the two tests into an overall test statistic, to validate the variables to assess whether they fit normal distribution. Then, continuous data were presented in the tables as the mean with standard deviation (normal distribution) or median with interquartile ranges (non-normal distribution) and compared by Student t-test (normal distribution) or Wilcoxon rank-sum test (non-normal distribution). Categorical data were presented as percentages, while the significance of the differences was determined through Chi-squared test. Univariate followed stepwise back multivariate logistic regression was used to identify the risk factors for short-term death. Multivariate logistic analysis with complex models adjusted for risk factors selected above was employed to investigate the independent effects of azithromycin use before ICU admission on short-term mortality. The complex models were as follows: no adjustment (Model1), adjusted for age (Model 2), adjusted for age and SOFA scores (Model 3); adjusted for age, SOFA scores and pH (Model 4); adjusted for age, SOFA scores, pH and diagnosis of cancer (Model 5). Variance inflation factor (VIF) was taken to test collinearity between continuous variables, and VIF ≤5 was seen as non-collinearity. 21 Stata 15.1 software (StataCorp, Texas, USA) and R software (version 4.0.0, www.r-project.org) was implemented for the statistical analysis, and p < .05 was considered statistically significant.
Results
Comparisons of demographics between survivors and non-survivors.
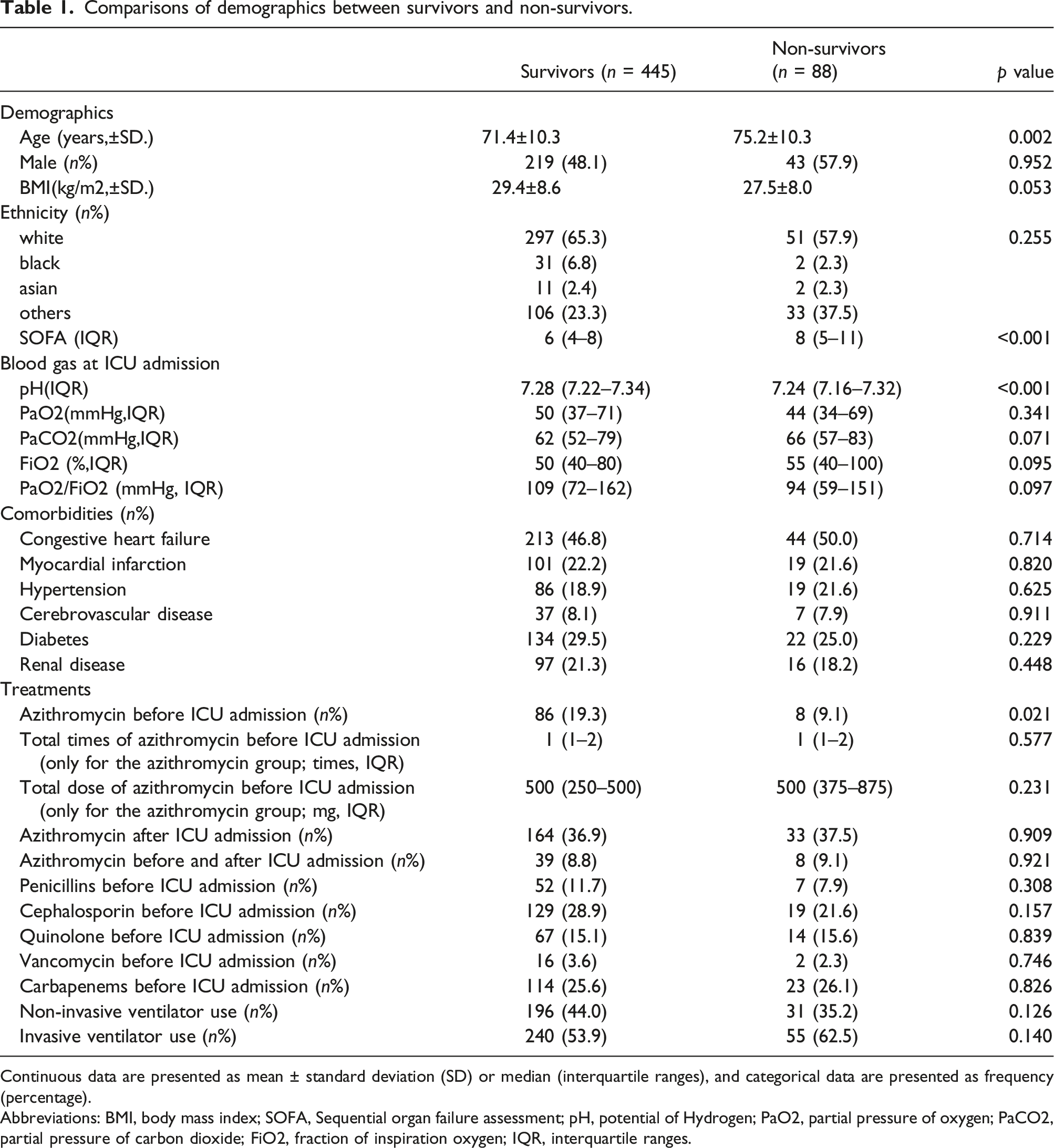
Continuous data are presented as mean ± standard deviation (SD) or median (interquartile ranges), and categorical data are presented as frequency (percentage).
Abbreviations: BMI, body mass index; SOFA, Sequential organ failure assessment; pH, potential of Hydrogen; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide; FiO2, fraction of inspiration oxygen; IQR, interquartile ranges.
Comparisons of population information and clinical outcomes between Azithromycin and Non-Azithromycin groups.
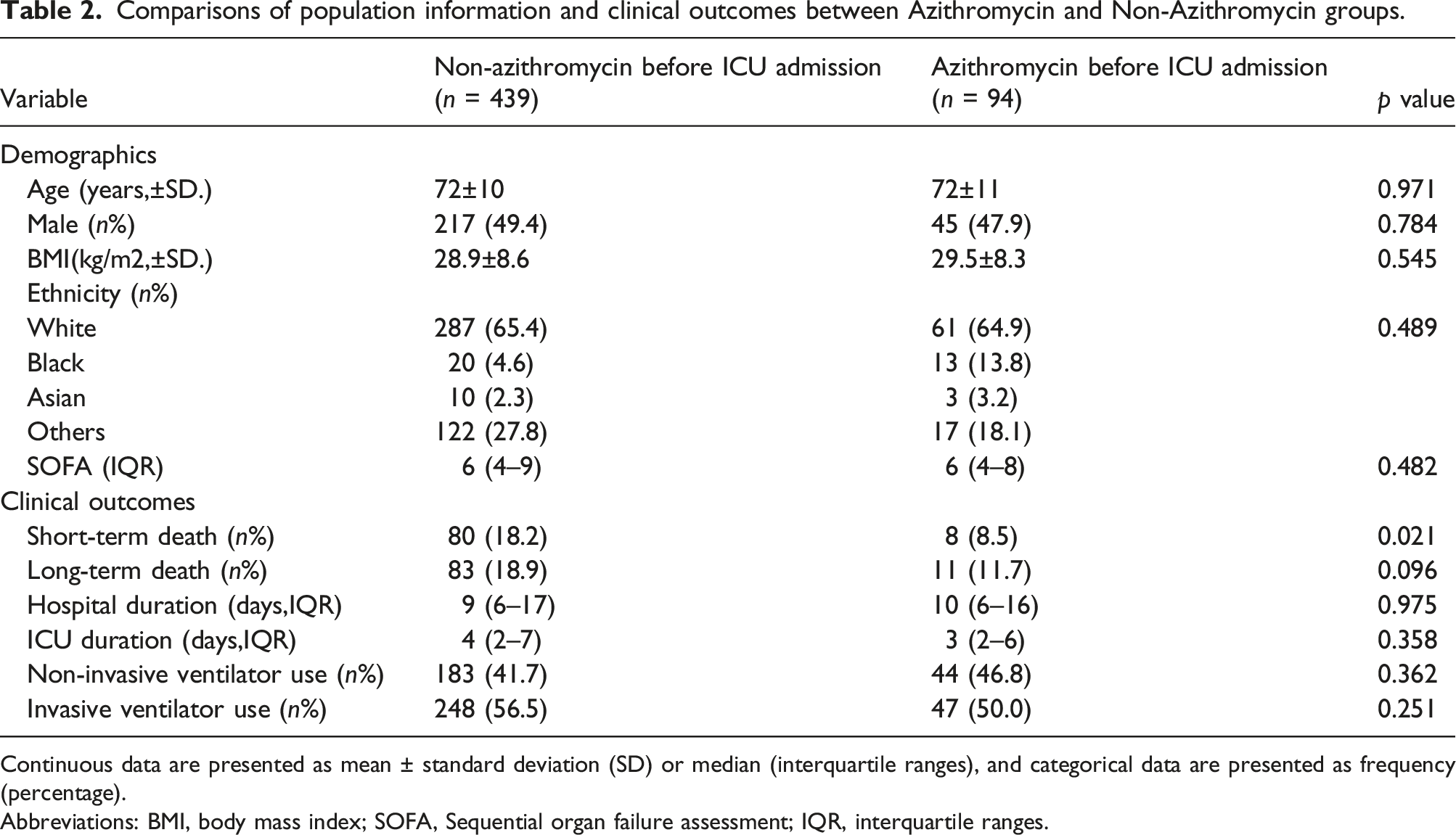
Continuous data are presented as mean ± standard deviation (SD) or median (interquartile ranges), and categorical data are presented as frequency (percentage).
Abbreviations: BMI, body mass index; SOFA, Sequential organ failure assessment; IQR, interquartile ranges.
Univariate and multivariate logistic analysis of variables and short-term mortality.

Abbreviations: OR, odd ratio; CI, confidence interval; BMI, body mass index; SOFA, Sequential organ failure assessment; pH, potential of Hydrogen; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide; FiO2, fraction of inspiration oxygen; IQR, interquartile ranges.
ORs associated with azithromycin before ICU admission and short-term mortality by multiple regression models.
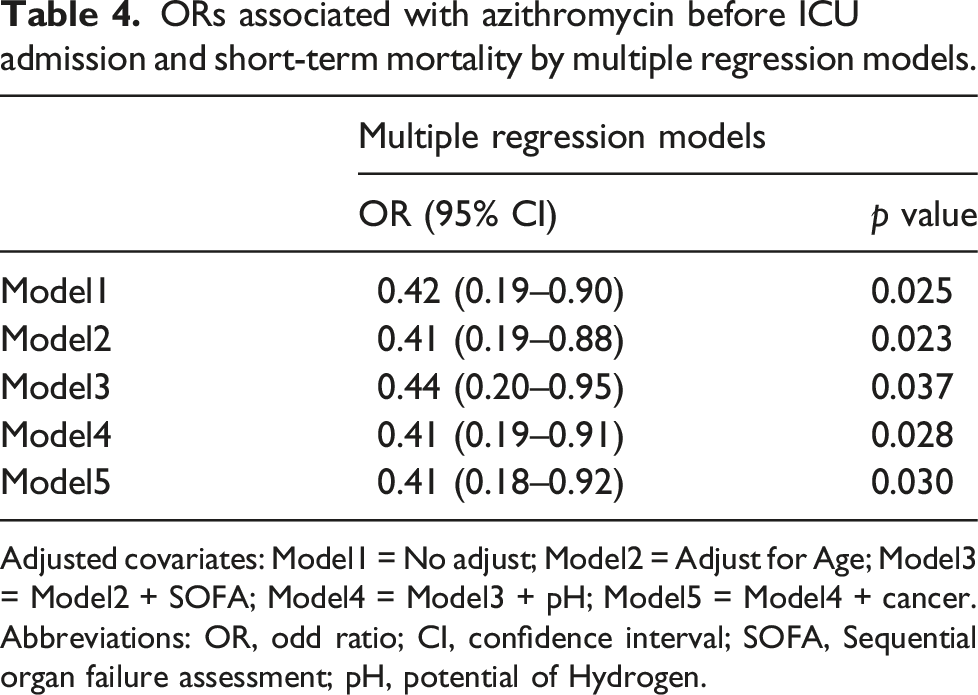
Adjusted covariates: Model1 = No adjust; Model2 = Adjust for Age; Model3 = Model2 + SOFA; Model4 = Model3 + pH; Model5 = Model4 + cancer.
Abbreviations: OR, odd ratio; CI, confidence interval; SOFA, Sequential organ failure assessment; pH, potential of Hydrogen.
Discussion
Our study showed that critically ill ACOPD patients who received azithromycin prior to ICU admission had a lower short-term mortality rate. This association was independent of the dose or times of azithromycin use and other antibiotic use before ICU admission. To our knowledge, the present study is the first time for this view to be suggested.
Despite progress in treatment, poor prognosis, including declining lung function, poorer quality of life and increased risk of mortality, is commonly seen in AECOPD patients when they are identified as life-threatening exacerbations.1,5,6 Thus, the critical care in ICU is required for these patients. A meta-analysis focusing on AECOPD patients admitted to ICU showed mortality for the collective was extremely high, reaching 17.6%–48.8% during hospitalization. 9 Another study with a similar subject gave a mortality rate as high as 27%. 23 All suggested that treatment for AECOPD patients requiring ICU admission is particularly important.
AECOPD, arousing by various infectious and noninfectious stimuli, was usually accompanied by higher inflammation status from the very beginning, making early identification and management of AECOPD meaningful in clinical practice.24,25 Research has shown that this series of inflammatory responses are not limited to lungs, but occurs throughout the body. 26 Despite the exact mechanism remains unclear, some inflammatory factors, like CRP, interleukin-6 (IL-6), interleukin-8 (IL-8), leukotriene B4 (LTB4), and tumor necrosis factor-α (TNF-α), were discovered be associated with occurrence and prognosis of AECOPD.27,28 This may be the root cause of the positive effect of corticosteroids and other anti-inflammatory drugs on the short-term recovery of symptoms and long-term clinical outcomes of AECOPD patients.1,29–31
Azithromycin, as a kind of macrolide, with both anti-microbial and anti-inflammatory effects, has repeatedly been proved to have significant effects on chronic inflammation.11–13 In the anti-microbial aspect, azithromycin has a wide spectrum, which has a strong antibacterial effect on not only Gram-positive bacteria, but also some Gram-negative bacteria, mycoplasma, chlamydia and legionella.13,32,33 In the meantime, there is a post anti-microbial effect of azithromycin. After drug withdrawal, the concentration of the drug released in the cell still exceeds the minimum inhibitory concentration of common sensitive bacteria within 5–7 days32–34 Azithromycin is often recommended for ACOPD patients with infective evidence but milder illness state and fewer comorbidities during acute exacerbation periods. For AECOPD patients evaluated as life-threatening, labeled older age, more severe state and more comorbidities, azithromycin is not the first choice and early combination with other antibiotics is not recommended.1,35,36
In addition to the coverage for common pathogenic bacteria of lung infection in AECOPD patients, azithromycin, like other macrolides, are involved in multiple ant-inflammatory activities, including suppression of some pro-inflammatory cytokines, attenuated chemotaxis and adhesion of neutrophils, hypoactive proliferation of monocytes, enhanced phagocytosis of apoptotic cells, alleviation of oxidant burst, reduction of mucous secretion, inhibition of angiogenesis, and decrease of virulence factors.10,11,37–42 Besides, azithromycin may join the response to corticosteroids by enhancing the recruitment of histone deacetylase 2, which is seen as a major contributor to the inflammatory gene repression produced by corticosteroids in patients with COPD, thus resulting in less corticosteroid use.30,31,43 Based on this anti-inflammatory effect, several studies suggested chronic azithromycin in COPD patients to prevent AECOPD.1,13,44 Numerous controlled trials implied inclusion of azithromycin in the treatment regimen may bring clinical benefits for critically ill patients with AECOPD, including reductions in hospital readmissions, improvements in time to next readmission and decrease of ICU costs.11,45,46 In a study involving more than 25,000 patients with AECOPD admitted to the ICU, macrolides initiated within 2 days of admission reduced hospital costs and 30-days ICU readmission rates. 11 Azithromycin is also used in other ICU conditions due to its antibacterial and anti-inflammatory effects. Afshar et al. 47 found that azithromycin was associated with less ICU stay days in severe sepsis patients received mechanical ventilation. Another study showed macrolide antibiotics could decreased death risk from septic shock and multiple organ dysfunctions for severe sepsis patients with ventilator-associated pneumonia. 48
There were several limitations to our study. For one thing, as a retrospective study, all our data is based on the MIMIC-IV databases. Although the sample size of our study was not small, future larger multicenter prospective studies are warranted to further validate our points. For another, in order to ensure that the extracted data were admitted to ICU because of AECOPD, we only selected patients with type II respiratory failure, which would inevitably lead to selection bias. Additionally, the use of azithromycin may simply be a surrogate marker for an unknown confounder, such as access to a primary care physician, a respirology/pulmonology specialist, and or medicare, causing confounding bias. Furthermore, the absence of data on whether patients used azithromycin before admission will lead to a decrease in persuasiveness.
Conclusion
Azithromycin use prior to ICU admission was associated with lower short-term mortality for critically ill AECOPD patients, the correlation was independent of the dose and times of azithromycin use. Therefore, we recommend the routine use of azithromycin for AECOPD patients who are admitted to the emergency department in serious conditions or whose condition changes during hospitalization and may require further treatment in ICU.
Footnotes
Authors contributions
XG: Responsible for data extraction and writing of the manuscript. HL: Responsible for data analysis and data validation. DG: Participate in data validation and proofreading of the manuscript. QL: Responsible for proofreading of the manuscript. XG and HL contributed equally to this manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MIMIC database was supported by grants from the National Institute of Biomedical Imaging and Bioengineering (NIBIB) of the National Institutes of Health (NIH) under award numbers R01-EB001659 (2003–2013) and R01-EB017205 (2014–2018).
Ethics approval and consent to participate
The project was approved by the review boards of the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center. There was no requirement of individual informed consent to extract data from the MIMIC-IV database because data from this database was publicly available and information of all patient were de-identified.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
