Abstract
Objective
We aimed to explore the safety and diagnostic value of medical thoracoscopic lung biopsy in patients with unexplained diffuse interstitial lung disease (ILD) in a single center pilot study.
Method
We retrospectively analyzed clinical and pathological diagnostic data from 52 patients with diffuse ILD undergoing medical thoracoscopic lung biopsy.
Results
Forty-four cases of diffuse ILD were confirmed pathologically, giving a diagnostic rate of 84.6%. Among these 44 patients, 11 patients were diagnosed with cancer, including eight patients with lung adenocarcinoma, three patients with metastases; two from a gastrointestinal malignancy, and one from a granulosa cell tumor of the ovary. There were 17 cases of idiopathic interstitial pneumonia, including nine cases of usual interstitial pneumonia (UIP), four cases of non-specific interstitial pneumonia (NSIP), three cases of cryptogenic organizing pneumonia (COP), and one case of acute interstitial pneumonia (AIP). There were 12 cases of rare interstitial pneumonias, which included six cases of pulmonary alveolar proteinosis, one case each of pulmonary Langerhans cell histiocytosis (LCH) and pulmonary lymphangiomyomatosis, two cases of nodular sarcoidosis, and two cases of chronic eosinophilic pneumonia. We recorded various complications, including bleeding, infection, and pneumothorax. A total of 28 patients (53.8%) experienced at least one of the above complications, but there were no deaths associated with biopsy.
Conclusions
Medical thoracoscopic lung biopsy appears a safe and effective method for diagnosing diffuse ILD of unknown cause but further prospective studies, with larger numbers, including comparison with other established techniques are required.
Introduction
Diffuse interstitial lung disease (ILD) is relatively common in respiratory clinical practice, with patients experiencing progressive dyspnea, restrictive ventilatory dysfunction, and diffusion limitation, with or without hypoxemia and other major manifestations. Diffuse ILD manifests on high resolution computed tomography (HRCT) as streaky, netlike, honeycomb, or ground-glass lesions.1-3 ILD encompasses more than 200 conditions, it is thus important, though challenging, to achieve the correct, specific diagnosis. 4
The diagnosis of ILD requires clinical, pathological, and radiological approaches. International consensus guidelines indicate that in up to 50% of patients with indeterminate ILD, histopathological examination is required if clinical, laboratory, and HRCT results are insufficient for reliable diagnosis. 5 Therefore, we retrospectively analyzed the data from 52 patients with diffuse ILD undergoing thoracoscopic lung biopsy between January 2015 and July 2021 to illustrate the clinical diagnostic rate, safety, and treatment-directing value of rigid thoracoscopic lung biopsy.
Materials and methods
Study subjects
A retrospective analysis was conducted on data from 52, out of a total of 74, patients with indeterminate ILD. They were admitted electively to our hospital, for medical thoracoscopic lung biopsy as an original planned diagnostic procedure, between January 2015 and July 2021.
Inclusion criteria included (1) diffuse interstitial lesions in both lungs on HRCT; (2) complete clinical, imaging, and pathological data, without a conclusive diagnosis on imaging and/or bronchoscopy and/or CT-guided percutaneous lung biopsy; (3) age less than 75 years.
Exclusion criteria were (1) infectious diseases and ILD of known cause, eg connective tissue disease-related interstitial pneumonia, radiation or immune or drug-related pneumonia; (2) HRCT showing unilateral or isolated pulmonary lesions; (3) HRCT showing UIP; and (4) serious cardiovascular dysfunction, abnormal vital signs, or unstable consciousness contra-indicating surgery. Fifty-two patients, with complete data, fulfilled all inclusion criteria. The remaining 22 patients failed to complete the whole diagnostic procedure, of which 12 patients had organ failure including poor cardiopulmonary function and unstable vital signs, four patients were intolerant of surgery due to anesthetic allergy or failed to match the surgery position, and six patients did not agree for further examinations, therefor their thoracoscopy was not performed. The flow diagram of procedural was shown in Figure 1.
Technical aspects
Rigid thoracoscopic equipment and compatible biopsy forceps (Karl Storz), venous incision kits, 24-F closed drainage tubes, and single-use water-seal bottles were used.
Preoperative preparation
The purpose and importance of surgery were fully explained to all patients, and written informed consent was obtained. Preoperatively, routine blood tests, coagulation function, arterial blood gas, and blood type were analyzed, and HRCT and electrocardiography were performed. Pulmonary function testing was carried out if the patient could cooperate and had no contraindications. Blood pressure and blood glucose were controlled within the normal range before surgery, and patients with COPD received nebulized bronchodilators 1 h preoperatively. Coagulation function and platelet counts were confirmed as normal in all patients including those with chronic myelodysplasia.
Surgical technique
1. The patient was placed supine tilted forward 30°–45°, with the lesion selected for biopsy facing laterally anterior. The ipsilateral upper limb was fixed by bracket in the shape of a salutation. Standard physiological observations; electrocardiography, heart rate, pulse oxygen saturation, non-invasive blood pressure, and body temperature monitoring and intravenous catheterization were performed.
2. A sterile cover was placed over the patient, with the face unobstructed to promote spontaneous breathing. Pleural cavity puncture was performed to induce an artificial pneumothorax. Aguide wire was inserted into the puncture needle, and used for probing when approaching the thoracic cavity. When no resistance was encountered, the guide wire was considered to have reached the thoracic cavity. The catheter was then inserted along the guide wire into the thoracic cavity and fixed. Insertion was performed between the 4th–6th ribs in the midaxillary line on the target side. The puncture needle was connected to the extension tube and 1 mL of physiological saline formed a small liquid column closing the extension tube. After injecting 280–400 mL of nitrogen into the thorax, chest radiography was performed. The distance between the wall and the visceral layer was >4 cm. If lung compression was not ideal, further filtered gas was injected.
3. The midaxillary line at the 4th–6th intercostal space was the surgical site. Routine sterilization and towel draping were performed, and 2% lidocaine (10–20 mL) was administered for local anesthesia. No intraoperative vagal nerve block or intrapleural local anesthetic was required. An incision of approximately 1.0 cm was made, subcutaneous tissue and intercostal muscle were bluntly separated layer by layer, as far as the pleural cavity, and the trocar was inserted into this vertically approximately 1.0 cm. The trocar was fixed by the assistant, and the needle core was removed. The biopsy site was determined according to the lesion site on HRCT and the appearances on visceral pleural endoscopy. Biopsy forceps were used to clamp the lesion site Figure 2, and lung tissue was cut as far as possible and retracted strongly. The second and third biopsies were clamped as far as possible from the deep part of the original biopsy site. Generally, 3–6 pieces of tissue (5 × 5 mm each) were obtained and sent for histopathology. Specimens were removed from all patients using one surgical access route without displacement or incision enlargement. A closed drainage tube was placed after surgery, with a water-seal bottle connected.
Results
General characteristics
General characteristics of patients.

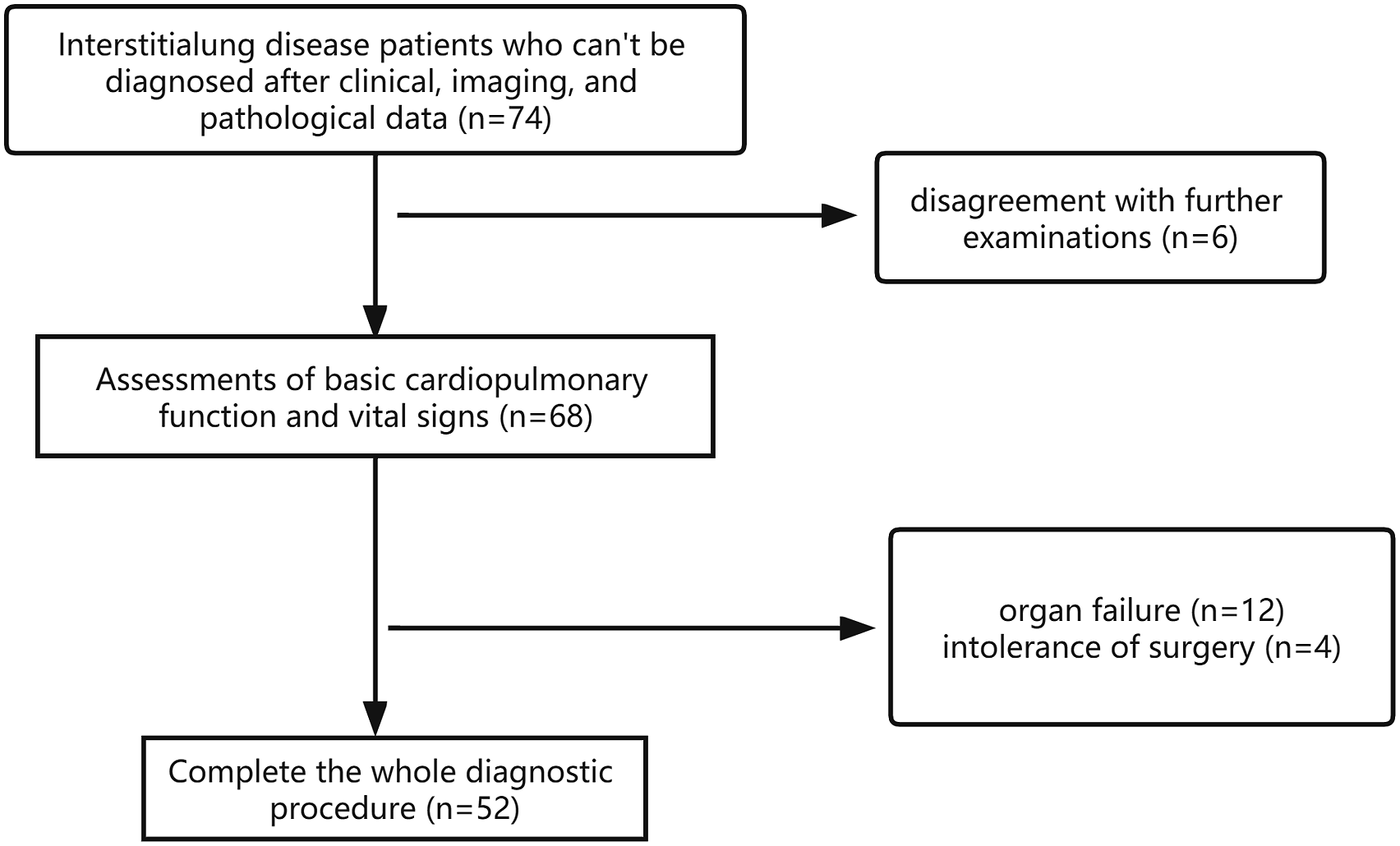
Flow diagram of procedural.

Biopsy forceps were used to clamp the lesion site.
Procedure, numbers, and biopsy sites
Surgery was completed in all cases, and patients were safely returned to the wards post-operatively. All patients underwent a smooth operation, and vital signs remained relatively stable with the exception of one patient who exhibited an intraoperative ‘pleural reaction’. This involved the development of dyspnea, chest tightness, and sweating, towards the end of surgery, with a fall in blood oxygen saturation from 98% to 92% with resolution after 5 min after dexamethasone and oxygen treatment. An example of intraoperative appearances at thoracoscopy is presented in Figure 5. Mean surgical duration was 35.5 ± 17.5 min. 16 patients (30.8%) were biopsied from the left lower lobe, and 13 patients (25%) were biopsied from the left upper lobe. The most common biopsy site was the right lower lobe in 24 cases (46.2%), with the right upper lobe biopsied in 13 cases (25%) and the right middle lobe biopsied in 5 cases (9.62%). Increased lung density of both lungs, with multiple round thin walled cystic opacities seen. Widespread bilateral scattered patchy opacification with thickened local lobules most marked peripherally in both lower lobes. Small right pneumothorax with numerous thin-walled cystic lesions in both lungs, the largest measuring approximately 1.3x1.6 cm.

Pathologically confirmed cases
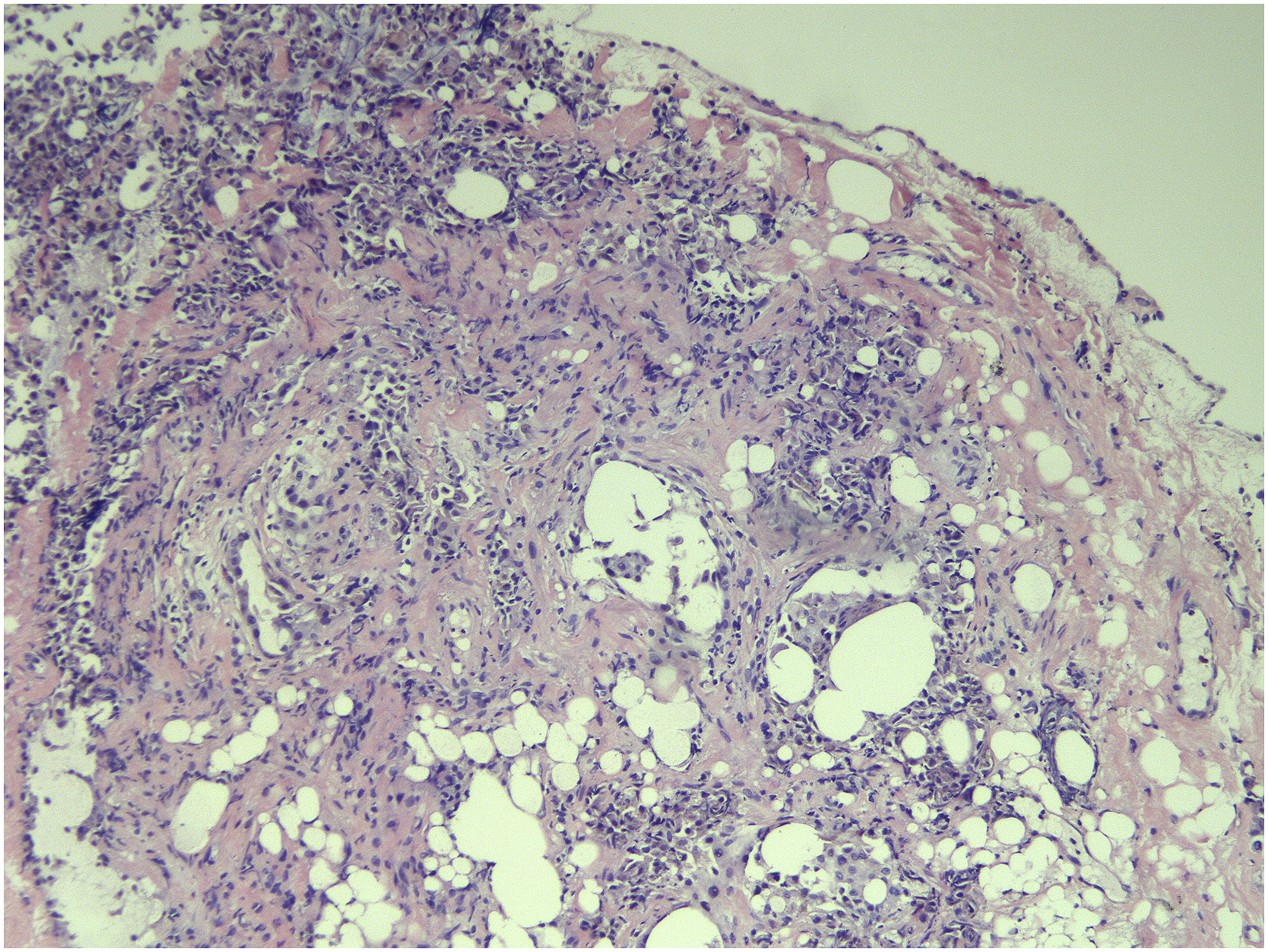
Forty-four cases of diffuse ILD were given a pathologically confirmed diagnosis: a diagnostic rate of 84.6%. Among them, 11 cases demonstrated malignancy, including 8 cases of lung adenocarcinoma, 2 cases of lung metastases from a gastrointestinal cancer, and 1 case from a granulosa cell tumor of the ovary. 17 cases of idiopathic interstitial pneumonia were noted, including 9 cases of UIP (an example is shown in Figure 6), 4 cases of NSIP, 3 cases of COP, and 1 case of AIP. 12 cases of rare interstitial pneumonias were recorded, including 6 cases of pulmonary alveolar proteinosis, 1 case of lung LCH, and 1 case of lung lymphangiomyoma. 2 cases of pulmonary sarcoidosis and 2 cases of chronic eosinophilic pneumonia were observed. Eight cases (15.4%) could not be definitively diagnosed, with pathologic examination showing chronic inflammation, fibrous tissue hyperplasia, and minor inflammatory cell infiltration, as well as other non-specific changes. At thoracoscopy, the surface of the basal segment of the right lower lobe shows pebble-like changes. The surface of the lung shows scattered sacculus-like protrusions ranging from rice grains to corn size, with scattered nodules also seen. Hematoxylin-eosin (HE) staining. ×100. Pathological changes showing lung septum thickening, fibrosis and cystic changes. In the fibrotic area, there are variable amounts of fibrosis and collagen fiber deposition in the alveolar septum, with local alveolar epithelial hyperplasia, and areas of intra-alveolar inflammatory cells. Histologic appearances are consistent with UIP.
Post-operative complications
Thirty-five patients complained of chest pain after surgery, with a Numeric Rating Scale score of 3–5, which was relieved after analgesic treatment. During surgery, there were different degrees of bleeding at the biopsy site, with the volume of blood loss ranging from 5–30 mL. Pneumothorax persisted in 17 patients (32.7%) after surgery, with an average pulmonary compression of 23.1 ± 7.3%, estimated radiologically, and the longest period of intubation required was 23 days. Postoperative infection occurred in three patients (5.8%), of which 2 patients presented with fever and one patient presented with turbid, light-yellow pleural fluid, with Klebsiella pneumoniae cultured. After appropriate antibiotics and drainage, all three patients improved and were discharged successfully after pleural extubation. No respiratory failure or acute exacerbation of ILD occurred, no respiratory support was required. Mean postoperative drainage tube duration was 6.4 ± 6.3 days. The average length of hospital stay was 7.5± 5.3 days.
Follow-up and treatment
After discharge, 28 patients attended follow-up and continuous treatment, including three patients with lung cancer, five patients with alveolar proteinosis, and 20 patients with interstitial pneumonia. Three patients died within 1 year, considered to be related to respiratory failure and multiple-organ failure caused by disease progression (malignancy and UIP). Five patients with alveolar proteinosis returned to hospital at intervals for alveolar lavage treatment, and different degrees of lesion absorption were observed on re-imaging. Of the 20 patients with interstitial pneumonia, six were treated with pirfenidone for pulmonary fibrosis; 14 were treated with a glucocorticoid combined with immunosuppressive therapy, including three who were treated with pirfenidone combined with a glucocorticoid. During follow-up, over at least 12 months, 16 patients showed improvement on HRCT while four showed no obvious change.
Discussion
Diffuse ILD represents a group of diseases with similar clinical and imaging manifestations, but differing etiologies, immunological mechanisms, pathologies and prognoses. HRCT findings in ILD, can be divided into four categories: UIP, possible UIP, uncertain UIP, and other diagnoses. International guidelines,6,7 state that a typical pattern on HRCT is sufficient to diagnose UIP in the appropriate clinical setting, excluding other possible causes of ILD. In the absence of a typical HRCT, surgical lung biopsy is required to establish a definite diagnosis of UIP.6,7 Lung biopsy should be considered in patients without definite UIP or when manifestations are inconsistent with UIP as it is important to determine ILD subtype.
This study retrospectively analyzed data from 52 patients with diffuse ILD who attended our department over a period of 5 years in whom the specific diagnosis remained undetermined. A precise diagnosis was determined by lung biopsy with a diagnostic rate of 84.6%. Various postoperative complications were documented in 28 patients, including pneumothorax, infection, and bleeding, but no acute exacerbations of ILD occurred and no respiratory support was required. There were no deaths as a result of lung biopsy. Three patients died within 1 year of surgery, all 3 deaths resulted from respiratory failure due to disease progression, despite treatment and were unrelated to surgical lung biopsy.
In this study, all 52 patients remained undiagnosed despite clinical data and HRCT findings; 19 undergoing TBLB before surgery, without reaching a clear pathological diagnosis. Previous studies have reported finding a TBLB diagnosis in only 20–30% of ILD patients, limited by size and location of the samples. 8 In addition, in UIP diagnostic sensitivity of TBLB was low at only 30%. Moreover, in NSIP and desquamative interstitial pneumonia, diagnostic specificity was lower at only 16% 9 ; likely due to small size and tissue damage in TBLB samples.10,11 In another study of 801 patients with diffuse ILD undergoing TBLB, less than one-third of the biopsy specimens were confirmatory. 12 Adequate alveolar tissue is key not only to making a definitive diagnosis, but in excluding other conditions, such as malignancy and infections.13,14
In ILD patients, if classical UIP imaging criteria do not meet American Thoracic Society/European Respiratory Society guidelines, recommend surgical lung biopsy as the gold standard,6,7 primarily because of the large specimen size and this is most often used in clinical practice.15-23 However, video-assisted thoracic surgery (VATS) requires general anesthesia and single-lung ventilation and is associated with high perioperative mortality and morbidity rates of 1.4%–4% and 5.8%–14.7%, respectively.18,19 In a study by Fisher et al., 20 the 30-days mortality after elective VATS was around 2%; results comparable with data from the United States and United Kingdom.21,23 Furthermore, mortality after non-elective surgery was much higher at 20% 23 probably related to poor baseline lung function and reduced respiratory reserve in these patients. 22 Mechanical ventilation alone may cause barometric lung injury, and ventilator-dependent atelectasis is common with single-lung ventilation, contributing to pulmonary complications.24-26
Medical thoracoscopy, unlike surgical thoracoscopic lung biopsy, requires only lidocaine for local anesthesia, without endotracheal intubation or mechanical ventilation. Patients are awake during surgery, performed through a single incision, reducing cost and tissue damage, enhancing its popularity in clinical practice. In addition, the surgical field is clearly exposed, positioning is accurate, and tissue samples can be taken from multiple locations. However, there are currently few domestic and foreign studies on medical thoracoscopic lung biopsy in patients with ILD.3,28 This may be because most lung interventional centers are equipped with semi-rigid thoracoscopes, with a small operating channel and clamps, few biopsy tissues are taken, restricting pathological diagnosis. Often, biopsies are only performed for pleural lesions. However, in our department, employing rigid thoracoscopes with larger operating channels and a wide surgical field, tissue biopsy diameter is up to 5 mm, improving the pathological diagnostic rate. Moreover, because intubation and general anesthesia are not required, unlike with VATS, it is likely that patient satisfaction is increased with reduced airway complications, length of hospital stay and costs.
Our study reported complications of lung biopsy, including bleeding, infection, and pneumothorax, with incidence rates of 15.4%, 5.8%, and 23.1%, respectively. A total of 28 patients (53.8%) had at least one complication, but no surgery-related deaths occurred, consistent with previous VATS-related studies. 17 In foreign literature,15,16 complication rates after thoracoscopic lung biopsy in patients with ILD range from 11.2% to 65.6%, with 30-days mortality rates of 0%–8.5%. Kreider et al., reported acute exacerbation of ILD, contributing to respiratory failure and death as a serious complication of VATs biopsy possibly caused by lung hyperinflation during single-lung positive pressure ventilation. 27 This complication was not seen in our study.
Our study has some limitations; Firstly, this study was a retrospective analysis. Retrospective studies are prone to selection and recall bias and cannot accurately determine the incidence of complications or relative risks. Secondly, our patients were generally in good condition, without respiratory failure or pulmonary hypertension; thus, we cannot address outcomes in the above-mentioned high-risk patients. Finally, the latest guidelines propose multidisciplinary discussion of patients with non-definite UIP, to formulate detailed diagnostic and management plans. Our study is lacking in this respect. Since ILD is a rare disease, the included patients are limited from a single center with experienced operators, we are committed to work with other regions and hospitals to obtain more data, and hope to have more comprehensive and objective results in the future.
Conclusions
Taking into account recent guidelines and the literature, we believe that medical thoracoscopic lung biopsy is an effective diagnostic modality for patients with ILD with respiratory symptoms and non-UIP manifestations on HRCT, particularly if lung function is acceptable, without pulmonary hypertension. In patients who are not elderly and do not require oxygen therapy, this procedure is a safe and effective and can achieve early diagnosis leading to more timely treatment of ILD, thereby improving prognosis. We recorded significant, not unexpected, complications; bleeding, infection and pneumothorax but no acute exacerbations of ILD, respiratory failure or deaths related to the procedure. Further larger scale, prospective, randomized trials, including a wider spectrum of patients and comparison with other established biopsy techniques, particularly cryobiopsy, are required.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
