Abstract
Existing studies primarily explored chronic obstructive pulmonary disease (COPD) in smokers, whereas the clinical characteristics and the disease course of passive or nonsmokers have been rarely described. In the present study, patients hospitalized and diagnosed as acute exacerbation of COPD (AECOPD) were recruited and followed up until being discharged. Clinical and laboratory indicators were ascertained and delved into. A total of 100 patients were covered, namely, 52 active smokers, 34 passive smokers, and 14 nonsmokers. As revealed from the results here, passive or nonsmokers developed less severe dyspnea (patients with modified Medical Research Council scale (mMRC) <2, 0.0% vs. 8.8% vs. 14.3%,
Introduction
Chronic obstructive pulmonary disease (COPD) refers to one of the most prevalent chronic airway diseases characterized by persistent airflow limitation and respiratory symptoms; it acts as the fourth leading cause of death worldwide, of which China might contribute to nearly one-third of these deaths. 1,2
It has been generally known that cigarette smoking is the critical risk factor for COPD. However, over the past few years, some studies have revealed that other factors (e.g. passive exposure to cigarette smoke, occupational exposures, and indoor biomass exposure) could elevate the risk of COPD and account for nearly one-third of COPD morbidity, especially in China. 1,3,4
Though COPD in nonsmokers has contributed a substantial proportion of COPD morbidity and mortality, its clinical characteristics, disease course, and therapeutic response have been rarely discussed. 5,6 Some studies reported that COPD patients in nonsmokers developed fewer symptoms, lower levels of inflammatory biomarkers, less impairment in airflow limitation and gas exchange, and a lower prevalence of emphysema, in contrast to former or current smokers. 3,7 However, other studies suggested that COPD patients in nonsmokers exhibited more lung fibrosis and similar clinical characteristics as compared with smokers. 5 Moreover, many of the mentioned studies primarily elucidated clinical and tomographic features of stable COPD with biomass exposure history, whereas the characteristics, disease course, and therapeutic response of acute exacerbation of COPD (AECOPD) in passive or nonsmokers have not been described.
Hence, this pilot study aimed to elucidate whether there were differences in clinical and radiographical characteristics of AECOPD of active, passive, and nonsmokers. Furthermore, the disease course after standard treatment of these AECOPD patients was also assessed to provide more insights into therapy strategy regulation of different AECOPD patients.
Methods
Study population
In the present observational study, adult patients with previously clinically diagnosed COPD and acute exacerbation of symptoms were prospectively screened out and employed at affiliated Haian Hospital of Nantong University and Shanghai Jiao Tong University School of Medicine affiliated Ruijin Hospital from January 2018 to December 2018. The present study was conducted following the Declaration of Helsinki, as approved by the Institutional Review Board of affiliated Haian Hospital of Nantong University and Shanghai Jiao Tong University School of Medicine affiliated Ruijin Hospital. All adult participants provided written informed consent for participating in the present study.
Including criteria were (1) patients clinically diagnosed as AECOPD upon admission and (2) lung function having been ascertained before the present exacerbation when patients were in stable disease, with postbronchodilator ratio of forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) less than 70%, complying with diagnosis criteria of Global Initiative for COPD (GOLD). 1
Excluding criteria were patients with acute asthma exacerbation, active tuberculosis, bronchiectasis, bronchiolitis obliterans, generalized bronchiolitis, or an unstable cardiac condition in 4 months. Besides, patients with previously clinically diagnosed asthma or positive bronchodilator reversibility test before the present exacerbation were excluded as well.
Patients covered in the present study were split into three groups: COPD in active smokers, passive smokers, and nonsmokers. Active smokers were defined as former or current smokers with at least 5 pack-year of tobacco exposure. Passive smokers were patients exposed to long-term environmental tobacco smoke (at least 5 years of 40 hours per week at home or workplace) but not active smokers. The other patients were classified as nonsmokers.
Data collection
At the recruitment, all patients were assessed by a medical interview, a physical examination, laboratory tests as well as computed tomography scan. Cough severity was ascertained with day-time cough symptom scoring system, 8 grading cough symptoms from 0 to 3, with 0 denoting no cough. Dyspnea severity was ascertained with the modified Medical Research Council scale (mMRC). Whether patients had phlegm or wheezing was also evidenced. In 24 hours before hospital discharge, all the symptoms were reassessed.
Spirometry was performed following the instruction of the American Thoracic Society/European Respiratory Society when patients were in stable disease. 9 Indicators were ascertained, covering FEV1, FEV1 percentage of predicted value (FEV1%), FEV1/FVC, the ratio of residual volume and total lung capacity, diffusing capacity of the lung for carbon monoxide (DLCO) as well as the ratio of DLCO and alveolar volume.
Arterial oxygenation index (calculated as oxygen partial pressure (mmHg) divided by the fraction of inspired oxygen) and carbon dioxide partial pressure (PaCO2) were ascertained by arterial blood gas analysis upon admission and in 24 hours before patients were discharged. The relative improvement of oxygenation index or PaCO2 was calculated as changed values divided by baseline values and multiplied by 100%. Tomographic characteristics, for example, bullae number, bronchial thickening (with or without), and pulmonary fibrosis severity (none, scattered (mild), periphery localized (intermediate), diffused (severe)), were assessed by two different radiologists. By discussion among radiologists and researchers, discrepancies were addressed.
Medications (e.g. antibiotics, inhaled corticosteroid (ICS) and long-acting β2 agonists (LABA) or long-acting antimuscarinic antagonists (LAMA), short-acting β2 agonists (SABA), and systemic corticosteroid) were recorded. Furthermore, noninvasive ventilation time and hospital time were ascertained.
Statistical analysis
The sample size was calculated with G*Power, version 3.1.9.2, with Change of mMRC score: baseline PaCO2, oxygenation index, and mMRC score; Variation of cough severity: baseline cough severity and mMRC score; Change of oxygenation index: baseline oxygenation index, corticosteroid dose, noninvasive ventilation (NIV) time, and antibiotic time; Change of PaCO2: baseline PaCO2, corticosteroid dose, NIV time, and antibiotic time; Hospital time: baseline mMRC score, cough severity, oxygenation index as well as PaCO2.
All statistical analyses were conducted with IBM SPSS, version 20.0 (IBM Corp., Armonk, New York, USA), and a
Results
Demographic characteristics of COPD in active, passive, and nonsmokers
During the study, a total of 129 patients were screened out, and 100 patients were finally covered (detailed flowchart in Figure 1), namely, 52 active smokers, 34 passive smokers, and 14 nonsmokers. All of the active smokers were male, while only about half of passive or nonsmokers were male, complying with an existing study suggesting that cigarette smoking was remarkably common in Chinese men. 3 COPD patients in passive smokers achieved higher body mass index (BMI) than active or nonsmokers.

Flowchart of the study.
Nonsmokers with COPD largely involved more farmers and fewer workers, as compared with active or passive smokers. Biomass exposure and previous pulmonary diseases (e.g. tuberculosis and recurrent pulmonary infection during the early years) were more common in passive or nonsmokers as compared with active smokers.
Among all three groups, other demographic characteristics (e.g. age, educational status, and occupational exposure) were balanced. More details regarding demographic characteristics are listed in Table 1.
Demographic characteristic of subjects.a
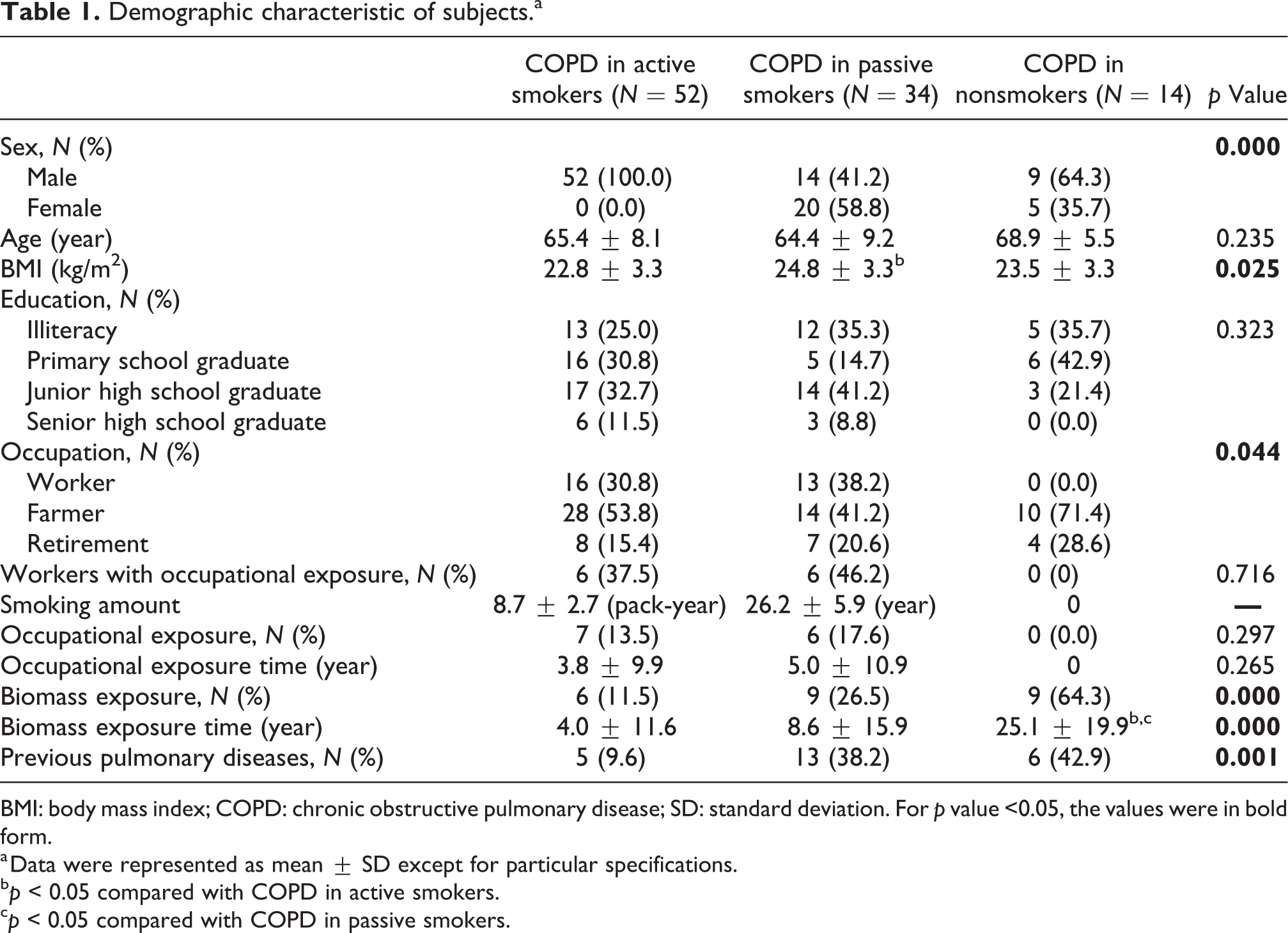
BMI: body mass index; COPD: chronic obstructive pulmonary disease; SD: standard deviation. For
a Data were represented as mean ± SD except for particular specifications.
b
c
Passive or nonsmokers with AECOPD had milder disease severity than active smokers
The GOLD severity of over 50% active smokers reached GOLD 3 or 4, while only 20% passive or nonsmokers were that severe when patients exhibited stable status. For mMRC score upon admission, none of the active smokers was less than 2, whereas 8.8% passive smokers and 14.3% nonsmokers reached below the level (Figure 2(a)). Nevertheless, other symptoms (e.g. cough, phlegm, and wheezing) were similar among the three groups (Figure 2(b) and Table 2).

Proportion of patients with different (a) dyspnea and (b) cough severity, and (c) oxygenation index or (d) carbon dioxide partial pressure (PaCO2), pre- and posttreatment. *
Clinical, radiological, and pulmonary functional characteristic of subjects.a
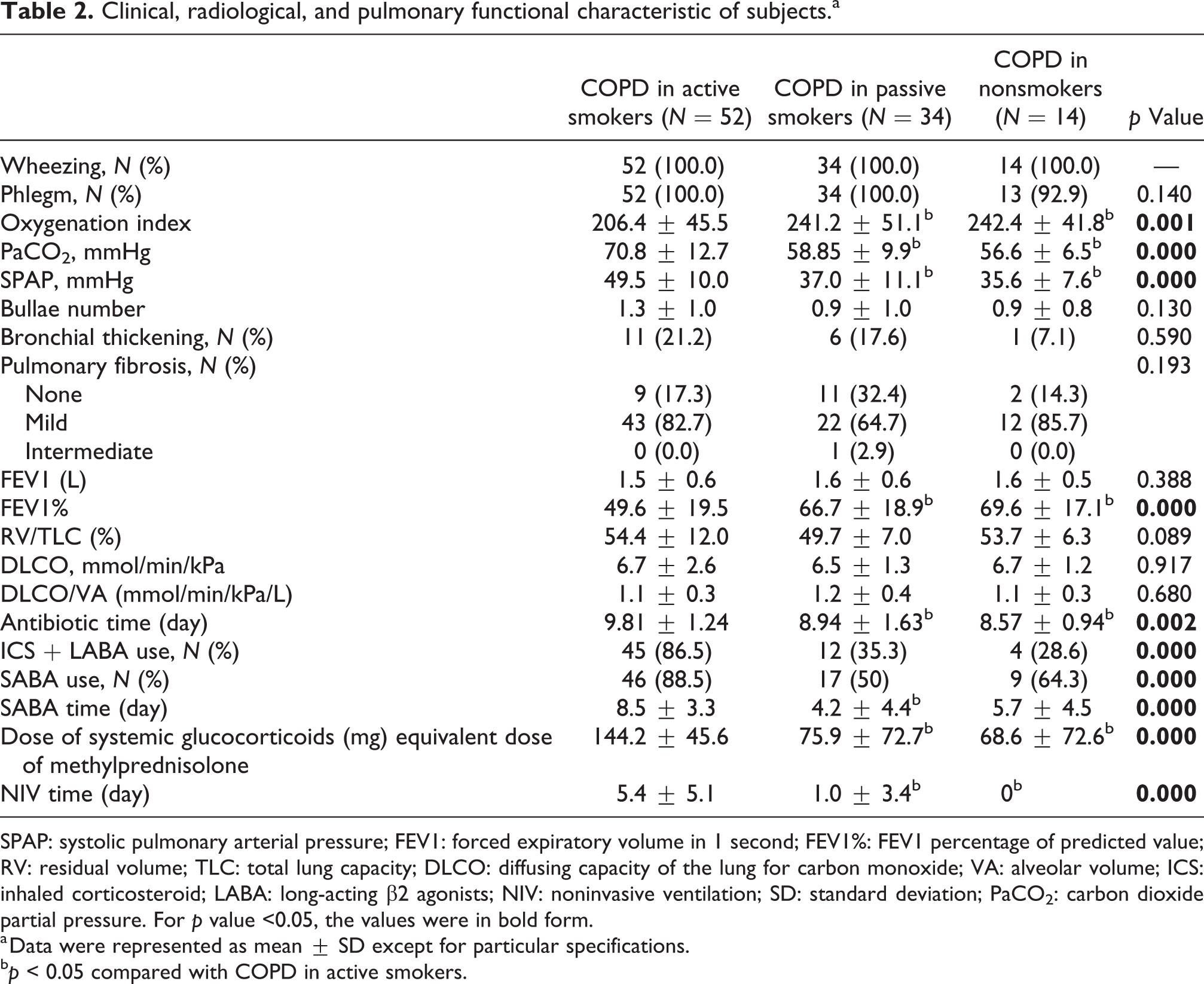
SPAP: systolic pulmonary arterial pressure; FEV1: forced expiratory volume in 1 second; FEV1%: FEV1 percentage of predicted value; RV: residual volume; TLC: total lung capacity; DLCO: diffusing capacity of the lung for carbon monoxide; VA: alveolar volume; ICS: inhaled corticosteroid; LABA: long-acting β2 agonists; NIV: noninvasive ventilation; SD: standard deviation; PaCO2: carbon dioxide partial pressure. For
a Data were represented as mean ± SD except for particular specifications.
b
To assess the severity of these patients more objectively, oxygenation index, PaCO2, and SPAP were also ascertained, and it was suggested that passive and nonsmokers achieved higher oxygenation index (206.4 ± 45.5 vs. 241.2 ± 51.1 vs. 242.4 ± 41.8 mmHg,
Furthermore, tomographic characteristics might exhibit difference as well. A trend was identified that active smokers tended to develop more severe emphysema, passive smokers milder pulmonary fibrosis and emphysema, and nonsmokers milder emphysema and bronchial thickening (Table 2).
Passive or nonsmokers with AECOPD might have better prognosis after treatment than active smokers
Regardless of smoking status, most patients could have mitigation of symptoms and the improvement of laboratory indicators after undergoing standard COPD therapy (Figure 2 and Table 3). However, different groups might exhibit different intensity of therapy and exact change of indicators. Though all the patients received LAMA as bronchodilator therapy as chronic therapy, and patients with different smoking status were administrated with similar types of antibiotics as acute therapy, active smokers were more likely to undergo ICS + LABA as chronic therapy and SABA as acute therapy. Moreover, the treatment courses of antibiotics, SABA, and NIV as acute therapies were also longer for active smokers. Furthermore, active smokers were also administrated with more systemic corticosteroids as acute therapy (more details in Table 2).
Changes of clinical and laboratory indicators.a

mMRC: modified Medical Research Council scale; PaCO2: carbon dioxide partial pressure; NIV: noninvasive ventilation; OR: odds ratio; CI: confidential interval. For
a Data were represented as mean ± SD except for particular specifications.
b Data were represented as OR (95% CI,
c
Though active smokers had been treated more aggressively, their improvement of clinical and laboratory indicators might not be the optimal (Figure 2 and Table 3). Improvement of mMRC score, indicating dyspnea symptom mitigation, was similar among the three groups (percentage of patients with mMRC improvement, 65.4% vs. 47.1% vs. 71.4%,
On the contrary, improvement of PaCO2 was more significant in active smokers (absolute amelioration: −12.6 ± 8.8 vs. −5.4 ± 5.4 vs. −4.6 ± 3.1 mmHg,
Since male patients were more common in active smokers, sensitivity analysis with only male patients was conducted, and similar results could be achieved. For male patients, dyspnea amelioration was also similar among the three groups (percentage of patients with mMRC amelioration, 65.4% vs. 35.7% vs. 66.7%,
To specifically evidence that smoking status affected disease course of AECOPD after treatment, effects of confounders and statistically different baseline characteristics were excluded by analysis of covariance for continuous variables and logistic regression for ordinal variables. Minimal regulation sets were identified according to directed acyclic graphs (DAGs). 10
After regulation, nonsmokers achieved the most noticeable amelioration of dyspnea and mitigation of cough symptoms, while passive smokers achieved the best hypoxemia amelioration, whereas nonsmokers had similar hypoxemia amelioration with passive smokers (more details in Table 3).
Discussion
Cigarette smoking, especially active smoking, has been long considered the most critical risk factor for COPD, whereas recent studies have reported other factors contributing to a large proportion of COPD. 1,4,11 –15 This study showed that nearly half of the COPD patients were not active smokers, and most of them were long-term exposed to environmental tobacco or biomass, thereby reflecting the necessity of banning cigarette smoking in public or working places, as well as the significance of the promotion of more clean fuels than wood, coal, crop residuals, and other biomass fuels in China.
Existing studies reported that COPD in nonsmokers exhibited different pathological, clinical, and tomographic characteristics as compared with smokers. 5,16 –20 One earlier study in Mexico reported that COPD patients in smokers had lower age, BMI, FEV1%, and FEV1/FVC in comparison with COPD patients with biomass exposure, complying with the present study. 17 Similar results were also achieved in another study in China, suggesting that lower BMI, FEV1%, and FEV1/FVC could be identified in smokers, whereas slight difference of age was found, and cough and phlegm symptoms were more common in smokers. 19 Later studies demonstrated that nonsmoking-related COPD patients exhibited milder symptoms and better lung function, 3,7 whereas these studies primarily focused on COPD with biomass or occupational exposure, and rare studies investigated the characteristics of COPD in passive smokers. Zubair et al. reported that more than half of COPD patients exposed to passive smoke were GOLD 1 or 2, complying with our study, whereas a direct comparison of active, passive, and nonsmokers was not drawn.
Furthermore, our study revealed that COPD in passive smokers had similar disease severity with nonsmokers, whereas it adversely impacted more female patients. Unlike the existing studies, 16,21,22 significant differences in radiologic changes were not identified between smokers and nonsmokers, probably because radiologic indicators used here were less sensitive to reveal the difference. Nevertheless, active smokers were found to have more bullae, though without statistical significance, supporting the conclusion from existing studies that COPD in smokers was emphysema predominant.
As recommended in GOLD guideline, 1 LAMA, LABA, ICS, antibiotics, systemic corticosteroids, and noninvasive ventilation acted as appropriate according to the severity of the patients here. Moreover, as expected, regardless of smoking status, all the patients gained enhancement of clinical or laboratory indices with these therapies, and treatment intensity of active smokers was higher as compared with passive or nonsmokers, which made sense since active smokers had more severe disease. However, despite higher treatment intensity, the disease amelioration of active smokers was not the most prominent, except for PaCO2 change, probably due to longer treatment course of noninvasive ventilation. Furthermore, for more severe patients with type 2 respiratory failure, amelioration of dyspnea was more common in nonsmokers as compared with passive or active smokers, revealing that lesser cigarette exposure might lead to a better therapeutic response.
To exclude the effect of baseline severity on the disease course of AECOPD patients, some outcome indicators were regulated. After regulation, amelioration of active smokers was the worst in terms of almost all the outcome indicators, with amelioration of dyspnea and cough symptoms the most conspicuous for nonsmokers and increase of oxygenation index for passive smokers, complying with a recent study reporting nonsmoker status as a critical predictor of transition from poor health status toward recovery. 23 Moreover, since the amelioration of hypoxemia was easier to achieve for passive smokers, oxygen support of lower concentration might be sufficient for these patients.
There were some inevitable limitations in the present study. First, this was an observational study, and the number of patients was limited, restricting the extrapolation of the results to larger population. Second, though some confounding factors were considered and regulated via statistical methods, the heterogeneity of different groups still might influence the results. Besides, patients covered here were more severe than the majority of AECOPD patients who could be treated in the community, which demonstrates that the conclusion from the results might only be valid for a minority of AECOPD patients. Lastly, all the patients of the present study came from the southeast region of China, and so the findings might not apply to other areas of the world and other healthcare systems where COPD phenotypes and COPD treatment strategies might differ.
In brief, the present study verified that AECOPD patients in active smokers had more severe symptoms, hypoxemia, CO2 retention, and pulmonary function impairment than passive or nonsmokers. Moreover, based on the regulated values, this study found that the disease course of AECOPD was correlated with smoking exposure status; nonsmokers were found with the best symptom mitigation, and passive smokers were found with relatively better hypoxemia amelioration. However, given the limitations in the present study, further research with a larger sample size and fewer confounders should be conducted to confirm the conclusion. Furthermore, the effect of early smoking quitting and a more accurate amount of smoking exposure that leads to different disease course or therapeutic response of AECOPD patients should also be explored in subsequent research.
Footnotes
Authors’ note
XL and ZW contributed equally to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Shanghai Key Discipline for Respiratory Diseases (2017ZZ02014).
