Abstract
Currently, few patients suffering from chronic obstructive pulmonary disease (COPD) who might benefit from a palliative care approach are referred to a palliative care team. Tools to identify patients eligible for a palliative care approach have been found to be difficult to apply in daily practice. Therefore, there is need for a simple and easily applicable tool to identify those patients who would benefit from referral to a palliative care team. The aim of this study was to determine if the surprise question (SQ) “Would I be surprised if this patient dies within 12 months?” in a subset of recently hospitalized COPD patients identifies those subjects. Recently hospitalized COPD patients were included, and the answer to the SQ was provided by the treating pulmonologist. The gold standards framework (GSF) prognostic indicator guidance was regarded as the gold standard test and was assessed for each patient. Sensitivity, specificity, and negative and positive predictive values were calculated to determine the accuracy of the SQ plus recent hospitalization compared to the variables of the GSF. A total of 93 patients were analyzed. In 35 patients (38%), the answer to the SQ was “not surprised”; 78 patients (84%) met ≥1 criteria of the GSF (15 (16%) did not meet any criteria). Specificity and positive predictive value for the SQ were both 100% ((78.2–100) and (87.7–100), respectively). Sensitivity was 44.9% (33.7–56.5) and negative predictive value was 25.9% (22.2–29.9). The “not surprised” group fulfilled significantly more GSF criteria. The SQ after recent hospitalization for COPD has a very high specificity compared to a standardized tool and is therefore a useful tool for the quick identification of patients who are most likely to benefit from palliative care. However, this method doesn’t identify all patients who are eligible for referral to palliative care.
Background
Chronic obstructive pulmonary disease (COPD) is a progressive disease causing about 7000 deaths in the Netherlands each year, thereby being the fourth leading cause of death. 1 Studies have shown a high symptom burden in patients with end-stage COPD, similar to patients with incurable cancer. 2,3 There is now general acknowledgment that optimal care for patients with advanced chronic organ failure such as COPD should be based on an integrated approach with integration of physical, psychosocial, as well as spiritual aspects of care. 4 However, in comparison with care for patients with malignant disease, provision of palliative care for COPD is currently limited. 5
End-of-life care preferences are rarely discussed by patients and their health care providers and referral to palliative care is often not considered. 6 –8 This might be partly due to the unpredictable course of the disease, characterized by stable periods and acute exacerbations. Moreover, a qualitative study showed that in contrast to cancer, there is a lack of understanding in patients that COPD is a life limiting disease, making a discussion about palliative care more challenging. 9
It has been suggested that hospitalization for an acute exacerbation of COPD (AECOPD) might identify patients who potentially require palliative care, due to high mortality rates and loss of quality of life. 3,10 –12 In addition, several instruments have been developed to help clinicians recognize patients who may benefit from palliative care and assess patients’ needs. An example of such an instrument is the gold standards framework (GSF) prognostic indicator guidance. 13 Other general identification tools are the supportive and Palliative Care Indicators Tool, 14 and recently the Necesidades Paliativas Centro Colaborador de la OMS - Instituto Catalán de Oncología. 15 These tools are well validated, yet complicated and difficult to use in daily practice. 13,16
The surprise question (SQ; “Would I be surprised if this patient died in the next year?”) is a simple tool to trigger the health care professional to consider referral for palliative care. 17,18 The SQ has been incorporated into several guidelines and identifications tools, 19 including the GSF. 13 These instruments warrant further exploration, especially with regard to whether the patient has unmet needs and may benefit from palliative care. However, we hypothesize that after hospitalization for COPD, the SQ can be used as a screening tool in daily practice to identify patients who are in need of palliative care. This approach might identify patients without the need of collecting clinical data or using a complex algorithm.
To test our hypothesis, we compared the SQ with a reference tool (the GSF) to identify subjects in need of palliative care in a group of patients who have recently been hospitalized for an AECOPD. Our identification tool is referred to as “hospitalization and surprise question” (HSQ).
Method
Design and study population
The study was an observational prospective study, conducted at the outpatient clinic of pulmonary medicine in the Spaarne Gasthuis, the Netherlands. The Spaarne Gasthuis is a large community-based teaching hospital with 818 beds. A palliative care team is available for consultation for both inpatients and outpatients.
Inclusion criteria were as follows: (1) known COPD; (2) hospitalization with an AECOPD between 1 January 2016 until 21 May 2017; and (3) an appointment at the outpatient clinic between 1 May and 22 June. A clinical diagnosis of COPD and AECOPD is defined according to the global initiative for chronic obstructive lung disease (GOLD) guideline 2017. 20
Patients were excluded if they missed their appointment at the outpatient clinic or if their physician felt unable to answer the SQ.
The primary outcome measures were defined as sensitivity, specificity, and negative and positive predictive value of the SQ compared to a set of indicators as stated in the GSF (Table 1). Secondary outcome measures were the prevalence of outpatients with GSF criteria and baseline clinical characteristics.
Variables based on the gold standards framework prognostic indicator guidance.

COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in 1 s; ICU: intensive care unit; MRC: medical research council; NIV: non invasive ventilation.
The GSF contains general indicators and COPD-specific indicators. Patients were assessed as “GSF1+” if ≥1 variable of the general indicators was met. Due to variation in the cutoff point for a positive score on several prognostic tools available, 15,16,21 we also conducted a stricter analysis for patients being considered GSF+ if they met ≥2 criteria of the GSF was met (GSF2+). Likewise, the cutoff for the indicator “comorbidity” differs from ≥2 or ≥1 comorbidities in some validation studies. 15,22 For this reason, we conducted the same analysis but with a cutoff point of ≥2 comorbidities for a positive score on comorbidity (GSFcom2+). For the disease-specific indicators, if ≥2 of these indicators were met, patients were assessed as “advanced disease.” “Advanced disease” is one of the general indicators of the GSF. As a consequence, patients meeting ≥2 of the specific clinical indicators were also assessed as GSF1+.
For assessment of comorbidity, we used the age-adjusted Charlson comorbidity index because it is a well validated prognostic measure for illness burden and most commonly used in contemporary clinical research. 23 Karnofsky performance status was used for assessing activities and functional performance status. 24
Data collection
First, data were obtained from the electronic patient medical records. Baseline characteristics included the following variables: (1) demographic variables, including age and sex, domestic situation (single and living together), place of living (home, residential home, and nursing home), and date of hospital admission for AECOPD; (2) COPD-related variables namely GOLD grade (I–IV, according to version 2014) 25 and smoking history; and (3) measurable variables according to the set of indicators as described in the GSF.
Additional variables were incorporated in a short questionnaire administered at the outpatient visit (Appendix 1). Finally, the patients’ treating pulmonologist was approached after the appointment at the outpatient clinic to answer the SQ. For all patients included, we used the binary response option (i.e. surprised or not surprised). If the pulmonologist answered “not surprised” to the SQ, the patient was considered SQ+. For the answer “surprised,” the patient was considered SQ−. A total of nine pulmonologists were involved in the assessment.
Statistical analysis
The statistical program SPSS version 20 was used to analyze the data. Data were summarized as mean ± SD, median (interquartile range), or count (percentage). To analyze the difference between baseline characteristics, χ 2 test for nominal variables and t-test for continuous, normally distributed variables was used. For non-normally distributed continuous and ordinal variables, Mann–Whitney U test was used. To analyze variance in answer to the SQ between the pulmonologists, Wilkinson rank sum test was used. Sensitivity, specificity, and positive and negative predictive values of the SQ tool were calculated. A two-tailed p-value of ≤0.05 was considered statistically significant.
Ethics
Ethics approval was obtained by the local ethics committee of the Spaarne Gasthuis, prior to the start of the study and informed consent was obtained from all patients prior to data collection.
Results
Patient characteristics
Of 743 patients hospitalized with an AECOPD during the period of 1 January 2016 until 30 April 2017, 109 fulfilled the inclusion criteria of having an appointment at the outpatient clinic during the period of May 1 and June 22. Median time between hospitalization and assessment was 155 days (25–535). Ten patients missed their appointment at the outpatient clinic, two patients had died and one patient was hospitalized at the time of appointment, leaving 96 evaluable patients all of whom agreed to participate. Of two patients, the involved pulmonologists could not provide an answer to the SQ because these patients were referred to them for the first time and they did not have enough information to conduct a critical assessment. In one patient, the reason for hospital admission was found to be congestive heart failure instead of COPD, and the patient was excluded from analysis. Finally, a total of 93 patients were analyzed.
Of the 93 patients, 35 (38%) were SQ+ (58 SQ−, 62%), and 78 (84%) met the standard referral index of ≥1 criteria of the GSF and were GSF1+ (15 GSF−, 16%). Sixty patients (65%) fulfilled ≥2 criteria of the GSF (GSF2+); 26 (29%) patients had ≥2 comorbidities, 10 (9%) patients had ≥3 comorbidities, with a mean Charlson comorbidity index score of 5. Eleven patients (12%) received PC at time of inclusion. An overview of all baseline characteristics is displayed in Table 2. Stratified by the answer to the SQ, SQ+ patients had a significantly lower forced expiratory volume in 1 s (FEV1), higher COPD GOLD class, and higher incidence myocardial infarction in their medical history. SQ+ patients did significantly fulfill more GSF criteria than SQ− patients. Furthermore, in the SQ+ group, there were significantly more patients receiving PC at the time of inclusion. Overall comorbidity and all other baseline characteristics, except smoking, did not differ significantly.
Baseline characteristics stratified by binary surprise question response.a
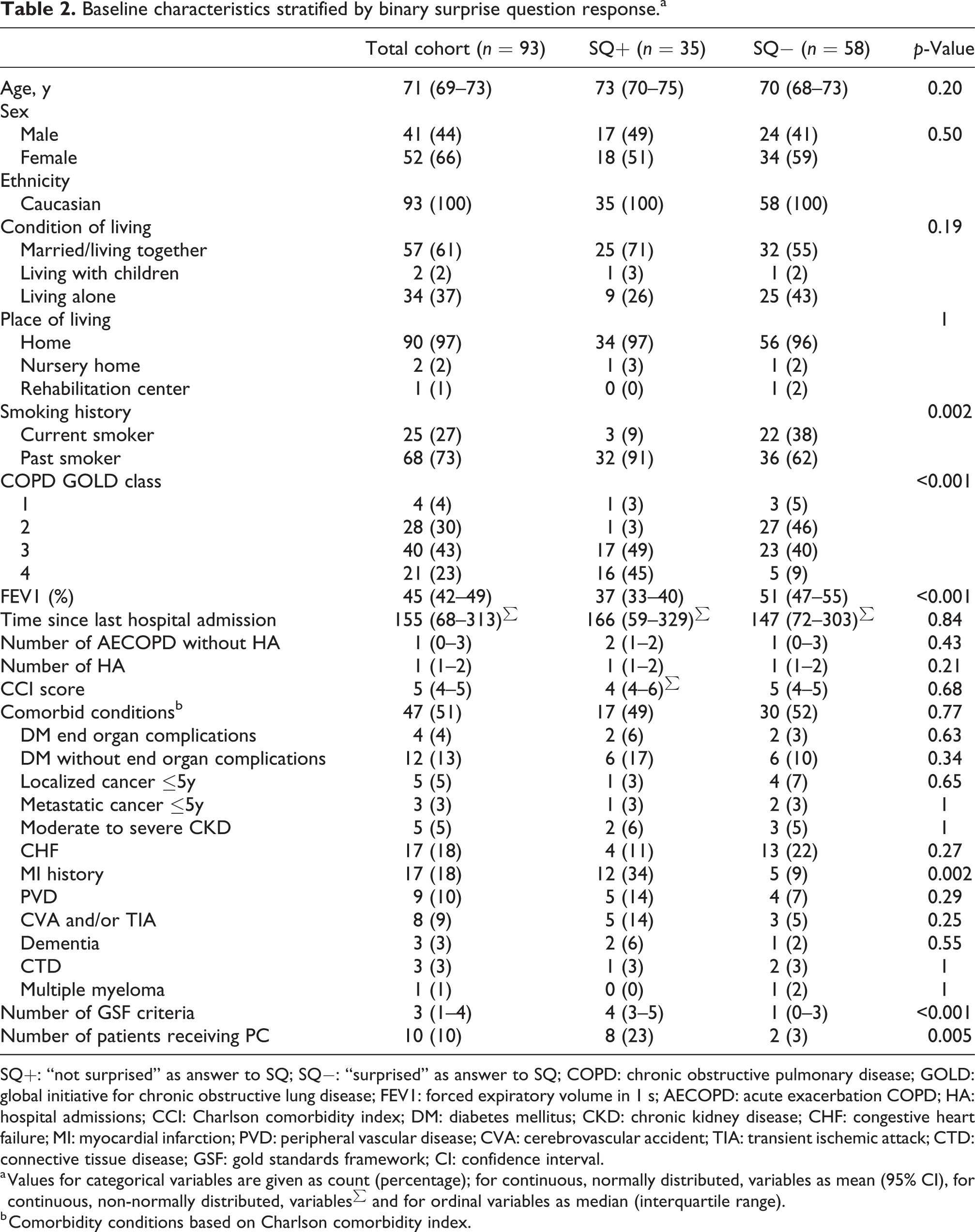
SQ+: “not surprised” as answer to SQ; SQ−: “surprised” as answer to SQ; COPD: chronic obstructive pulmonary disease; GOLD: global initiative for chronic obstructive lung disease; FEV1: forced expiratory volume in 1 s; AECOPD: acute exacerbation COPD; HA: hospital admissions; CCI: Charlson comorbidity index; DM: diabetes mellitus; CKD: chronic kidney disease; CHF: congestive heart failure; MI: myocardial infarction; PVD: peripheral vascular disease; CVA: cerebrovascular accident; TIA: transient ischemic attack; CTD: connective tissue disease; GSF: gold standards framework; CI: confidence interval.
a Values for categorical variables are given as count (percentage); for continuous, normally distributed, variables as mean (95% CI), for continuous, non-normally distributed, variables∑ and for ordinal variables as median (interquartile range).
b Comorbidity conditions based on Charlson comorbidity index.
Variance in answer to the SQ did not differ significantly between the nine pulmonologists.
Sensitivity, specificity, and positive and negative predictive values
The SQ had a high specificity and very high positive predictive value (PPV, both 100%), with low sensitivity and negative predictive value (NPV, 44.9% and 25.9%, respectively) explained by a high rate of false negatives (Table 3). Results were the same when comorbidity was scored positive if patients had ≥2 comorbidities, instead of ≥1 comorbidity. In a stricter analysis, a cutoff point of ≥2 GSF criteria was used. False positive answers to the SQ raised from 0 to 4, resulting in a slightly lower specificity and PPV (88.2%, confidence interval (CI): 71.6–96.2 and 88.6%, CI: 72.3–96.3, respectively). Due to a higher amount of true negatives, sensitivity and NPV improved, although they remained low (52.5%, CI: 39.2–65.5 and 51.7%, CI: 38.3–64.9, respectively, Table 4).
Sensitivity, specificity, positive predictive values, and negative predictive values of SQ versus ≥1 GSF indicator guidance.

GSF: gold standards framework; SQ: surprise question; SQ+: “not surprised” as answer to SQ; SQ−: “surprised” as answer to SQ; CI: confidence interval.
Sensitivity, specificity, positive predictive values, and negative predictive values of SQ versus ≥2. Criteria of GSF prognostic indicator guidance.

GSF: gold standards framework; SQ: surprise question; SQ+: “not surprised” as answer to SQ; SQ−: “surprised” as answer to SQ; CI: confidence interval.
Discussion
This study showed that all patients identified as eligible for palliative care by the use of the HSQ would also have been identified as such by the GSF prognostic indicator guidance. In other words, a positive answer to the HSQ includes nearly half of the patients that could benefit from palliative care according to the GSF prognostic indicator guidance, with no false positives. Therefore, we demonstrated that the SQ in this population, with a recent hospitalization for COPD, can be used as a simple screening tool to identify patients in need of palliative care.
Few studies focusing on the SQ have been performed, most of them in cancer patients or in patients with advanced chronic conditions in general. 15,18,26 –30 To the best of authors’ knowledge, there are only two studies concerning COPD and the SQ, 31,32 one of which compared nonvalidated prognostic indicators and the answer to the SQ. 32 Their findings are consistent with our findings, with 87% meeting at least one prognostic indicator and a PPV of a “No” answer to the SQ of 95%. A recent study in patients with diverse chronic conditions 15 compared the predictive validity of the SQ and the NECPAL tool, a content-validated tool developed by their group. In contrast to our study findings, they found that 93% of SQ+ patients were also NECPAL+ patients. However, criteria of the NECPAL tool differ from the GSF prognostic indicator guidance and need further modification. Reliability was also not tested in both of our studies, and a high inter- and intra-rater variability could be an explanation for the difference in results.
Although use of the SQ has been promoted by some, 29,33 others have highlighted concern about the implementation of the SQ into routine practice. Studies have reported confusion and discomfort among physicians using the SQ, as they preferred a more objective clinical terminology 34 and felt the SQ was too subjective to base important decisions on. 35 It must be emphasized that provision of palliative care should be based on unmet end-of-life care needs, rather than on prognostication or a well-defined timescale. The SQ simply asks clinicians whether he or she thinks the patient “is sick enough to die” and could therefore benefit from palliative care. Accordingly, the SQ has been mentioned as a general indicator of unmet needs and physical decline. 36 This is supported by the results of our study, since all patients assessed as “not surprised” by the SQ also fulfilled at least one indicator of a tool that is developed to identify patients who may have these unmet needs or show physical decline. Moreover, a recent study to the provision of PC for COPD showed that from a large primary care cohort, only 7.8% received PC during their study follow-up. 37 Our study group considered patients with more severe COPD (average FEV1 of 45%), of whom only 12% received palliative care at the time of inclusion. One of the barriers for referral to PC in COPD is the complexity of tools to identify eligible patients. 38 Therefore, simple instruments to raise the amount of referrals are required. Our study showed that the SQ combined with recent hospitalization may provide an easily applicable method to rise outpatient palliative care referrals.
Strengths and limitations
This study is based on everyday clinical practice and the outcome is easily applicable for physicians.
A limitation of this study is the fact that only COPD patients with a recent hospitalization have been included. Clinical parameters as well as the answer to the SQ may differ in patients who have not been hospitalized before, who suffer from other conditions than COPD, or when assessment takes place in a different setting (e.g. in primary care). Therefore, this study only provides information about patients with COPD who have been admitted to the hospital in the past 18 months.
Implications for further research
Since little is known about the efficacy of palliative care services in COPD, 39 further research is needed to determine patients’ and family outcomes after outpatient referral. Moreover, follow-up after referral is needed to determine if patients identified through the HSQ, experience a better quality of life after referral to a palliative care team. Further enhancement toward a comprehensive and integrated pathway for both inpatient and outpatient palliative care services is important. Finally, due to our study design with short follow-up time, we were unable to obtain mortality rates and correlate them to the answer to the SQ. This could be assessed in further research. However, the aim of this study was to investigate whether the SQ combined with hospitalization could provide an appropriate “short cut” for identifying patients in need of palliative care, rather than estimating prognosis.
Conclusion
In a subset of recently hospitalized COPD patients, the SQ presents high specificity and a high positive predictive value compared to the Gold Standard Framework Indicator Guidance and provides a quick and simple tool for identifying COPD patients who are likely to benefit from a palliative care approach. Due to a low sensitivity and a low negative predictive value, it should not be used as a stand-alone tool.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
