Abstract

Keywords
Several devices are used for long-acting muscarinic antagonist (LAMA)/long-acting β2 agonist (LABA) inhalation therapy for chronic obstructive pulmonary disease (COPD) patients but what is the optimum device? On the basis of Bayesian network meta-analysis, all the LAMA/LABA fixed-dose combinations are at present evaluated as having similar efficacy in terms of forced expiratory volume in 1 second (FEV1). 1 However, in recent years, the association between the residual volume and the symptoms of COPD has been drawing attention. In Japan, the devices available for LAMA/LABA are dry-powder inhalers (DPIs) and soft-mist inhalers (SMIs). A previous report showed improvements in functional residual capacity and residual volume (RV) with an SMI-type LAMA/LABA over the same LABA or LAMA given as a single agent. 2 Other researchers have reported that the deposition pattern within the lungs was more peripheral for SMI than for DPI. 3
Since April 2015, among 9 patients in our hospital with COPD who sequentially used 2 kinds of LAMA/LABA devices, 8 patients (mean age: 77.5 years; range: 67–87 years; mean pack years: 84.2; range: 33.8–144; all patients were male) initially took DPI-glycopyrronium plus indacaterol for at least 8 weeks and then switched to SMI-tiotropium plus olodaterol. One patient switched from SMI to DPI. Because SMI-tiotropium plus olodaterol became commercially available 2 years after DPI-glycopyrronium plus indacaterol had become available in Japan, most patients in this study switched from DPI to SMI. All patients were using LAMA as a monotherapy before they started the combination treatment. We examined changes in lung function measures by comparing the measures taken before and after the switch to the SMI device. The first “before switch” lung function was measured on the day it was switched and the last “after switch” lung function was measured after 6 to 10 weeks. The lung function values were measured 2 to 8 hours after the last inhalation dose, and the time point of the “before switch” and “after switch” lung function measurements was almost identical for each patient. The appropriate inhalation technique for both devices was confirmed for every patient when the patient started the respective inhalations. This study was approved by the Institutional Review Board of our hospital. Changes in lung function test values were evaluated using the Wilcoxon signed-rank test (Table 1). Although no significant difference was found in forced expiratory volume in 1 second (FEV1) before and after the use of the SMI device, the vital capacity (VC) (p = 0.012) and % predicted VC (p = 0.012) differed significantly, with an average improvement of ≥200 mL in VC after the switch from the DPI to the SMI. These findings could reflect the possible superiority of tiotropium plus olodaterol over glycopyrronium plus indacaterol or of the SMI-type inhalation device or of both. In the treatment of COPD, not only FEV1 but also the increase in VC related to air trapping caused by small airway diseases should be considered. Improvement in VC leads to decreased RV, increased effective ventilation, and alleviation of rapid and superficial breathing. This is a pharmacologic effect comparable to the lung volume reduction surgery (LVRS). This could be considered a “pharmacologic LVRS effect.”
Changes of lung function parameters after switching from DPI to SMI.a
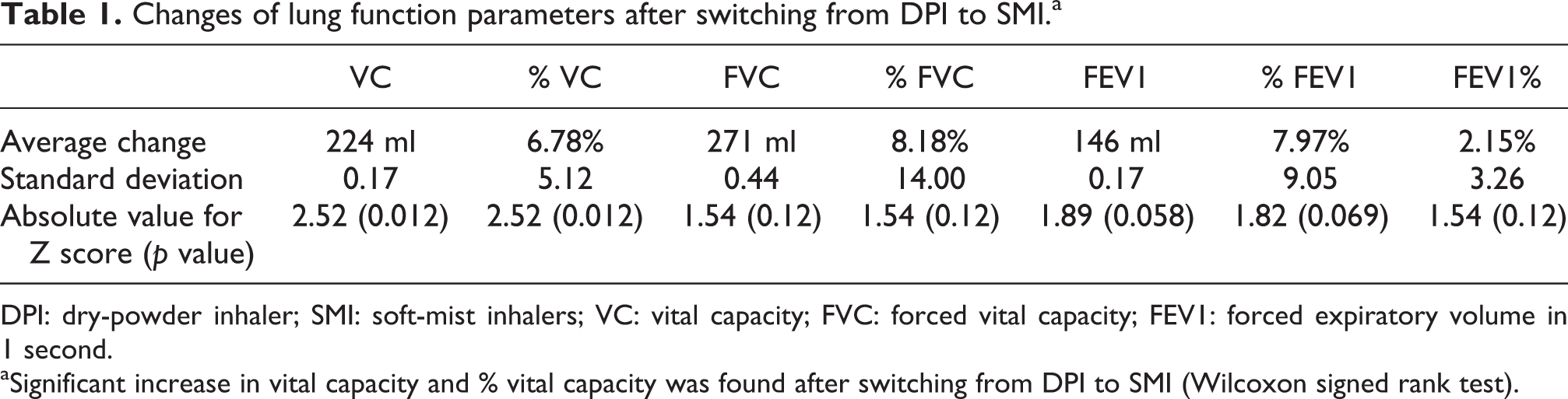
DPI: dry-powder inhaler; SMI: soft-mist inhalers; VC: vital capacity; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 second.
aSignificant increase in vital capacity and % vital capacity was found after switching from DPI to SMI (Wilcoxon signed rank test).
LAMA/LABA combination has increased the number of treatment choices, and identifying the most appropriate device for each COPD phenotype is necessary. Patients with COPD who do not show satisfactory responses to DPI-glycopyrronium plus indacaterol may benefit from switching to SMI-tiotropium plus olodaterol, especially from the viewpoint of increasing the VC. Here we have reported a retrospective evaluation of routinely treated patients. The sequence of the two treatments was not randomized. Therefore, the findings can only be indicative of what might happen in a larger population. A planned prospective investigation is required to verify this contention.
Footnotes
Authors’ note
HY analyzed the data and drafted the manuscript. HS and NHiz contributed to the design and data analyses and drafted the manuscript. NHid, NT, and HT gave advice on this article. All authors read and approved the final manuscript.
Acknowledgements
The authors thank Ms F Miyamasu, Medical English Communications Center, University of Tsukuba, for grammatical revision of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NH has received lecture fees from AstraZeneca, Astellas Pharma, and Boehringer Ingelheim.
