Abstract
Bronchodilation with a long-acting muscarinic antagonist (LAMA) or long-acting β2-agonist is central to the management of chronic obstructive pulmonary disease (COPD). Tiotropium, the first LAMA available for use in COPD, has been shown to be an effective bronchodilator and is generally safe and well tolerated. However, tiotropium has limitations that include a high incidence of dry mouth, slow onset of action and, in some studies, a part of the patient population did not achieve clinically significant bronchodilation. It also remains unclear whether tiotropium reduces progressive deterioration of lung function in patients with COPD. An ideal LAMA would provide clinically meaningful bronchodilation, deliver symptom relief, prevent disease progression, improve exercise tolerance and health status, prevent and treat complications and exacerbations and reduce mortality risk. A 24-h duration of action, rapid onset of action and a good safety and tolerability profile are also desirable. The once-daily LAMA, NVA237 (glycopyrronium bromide), may meet some of these characteristics. NVA237 has high selectivity for the muscarinic type-3 (M3) receptor which might potentially result in a higher therapeutic index than tiotropium, which is less selective for M3. Phase II studies showed that NVA237 once daily provides clinically significant 24-h bronchodilation with a rapid onset of action and a favourable safety and tolerability profile. Phase III studies are ongoing that will assess the long-term safety and efficacy of NVA237.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive condition characterized by airflow limitation that is not fully reversible [GOLD, 2009]. COPD was the fourth most frequent global cause of death and the 13th most common cause of disability in 2004, and is predicted to become the third most frequent cause of death and the fifth most common cause of disability by 2030 [World Health Organization, 2008]. The goals of COPD management are to relieve symptoms, prevent disease progression, improve exercise tolerance, improve health status, prevent and treat complications, prevent and treat exacerbations, and reduce mortality [GOLD, 2009]. Bronchodilators are central to the management of COPD, with regular treatment with long-acting β2-agonists (LABAs) and long-acting muscarinic antagonists (LAMAs) considered more effective and convenient than short-acting options [GOLD, 2009]. Current guidelines recommend regular use of a LABA and/or LAMA for patients with moderate, severe or very severe COPD and note that there is insufficient evidence to recommend one class over the other [GOLD, 2009].
Long-acting muscarinic antagonists
Mechanism of action
Bronchoconstriction in COPD is mediated by cholinergic mechanisms [Beeh et al. 2002]. Cholinergic nerve impulses are conducted by acetylcholine, which binds to muscarinic (M) type-1 (M1) receptors on nerve ganglions, stimulating acetylcholine release from postganglionic terminals. This acetylcholine binds to M3 receptors on airway smooth muscle and submucosal glands, stimulating bronchoconstriction and mucus production. Acetylcholine also binds to presynaptic M2 receptors triggering feedback inhibition of acetylcholine production.
Blocking M1 and M3 receptors reduces or reverses bronchoconstriction, whereas blockade of M2 receptors attenuates feedback inhibition of acetylcholine production, which may reduce the bronchodilating effects of a muscarinic antagonist [Beeh et al. 2002]. M2 receptors are also found in the heart and are believed to modulate pacemaker activity, atrioventricular conduction and contraction force [Abrams et al. 2006]. Blockade of cardiac M2 receptors may therefore result in increased heart rate. An ideal anticholinergic for use in COPD would therefore have high affinity for M1 and M3 and low affinity for M2 receptors [Beeh et al. 2002].
Tiotropium
Tiotropium was the first LAMA available for use in COPD and is delivered using the HandiHaler dry powder inhaler (Boehringer Ingelheim, Ingenheim, Germany) [Boehringer Ingelheim, 2010a] or Respimat Soft Mist inhaler (Boehringer Ingelheim) [Boehringer Ingelheim, 2010b]. Tiotropium has a duration of action of over 24 h [Lipson, 2006] and binds to M1, M2 and M3 receptors, but dissociates rapidly from M2 receptors, resulting in relatively selective M1 and M3 receptor antagonism [Lipson, 2006]. Slow dissociation from M1 and M3 receptors results in a long duration of action [Lipson, 2006].
Tiotropium has been shown to improve trough forced expiratory volume in one second (FEV1) compared with placebo [Casaburi et al. 2000], as well as reducing dyspnoea, improving health status scores, reducing COPD exacerbations and hospitalizations, improving exercise endurance and inspiratory capacity and reducing the need to start inhaled corticosteroids (ICS), LABAs and LABA/ICS combination [Troosters et al. 2010; Casaburi et al. 2005; Maltais et al. 2005; Niewoehner et al. 2005; O'Donnell et al. 2004; Celli et al. 2003; Donohue et al. 2002]. Recent data from the POET (Prevention of Exacerbations with Tiotropium in COPD) study showed that tiotropium resulted in a reduction in the rate of exacerbations (rate ratio 0.89, p < 0.02) and the risk for hospitalized exacerbations (hazard ratio 0.72, p < 0.0001), compared with salmeterol [Vogelmeier et al. 2010a]. Tiotropium has also been shown to improve dyspnoea compared with salmeterol [Donohue et al. 2002].
Although effective, tiotropium has several limitations. First, in a 6-month, randomized, placebo-controlled trial, St George’s Respiratory Questionnaire (SGRQ) total score was significantly improved with tiotropium versus placebo (4.2 versus 1.5, respectively; p < 0.01). However, approximately 50% of patients with COPD failed to achieve improvements in SGRQ scores that are considered to be clinically relevant (≥4 points) [Yorgancýoðlu et al. 2009; Brusasco et al. 2003]. In the same study, compared with placebo, transition dyspnoea index (TDI) focal score improved significantly in the tiotropium group (1.1 units, p < 0.001); although, 57% failed to achieve a change in TDI believed to be clinically meaningful (≥ 1 unit) [Brusasco et al. 2003]. In addition, the Understanding Potential Long-term Impacts on Function with Tiotropium (UPLIFT) trial indicated that tiotropium did not modify the rate of decline in lung function, compared with placebo, with both groups having similar declines in pre- and postbronchodilator FEV1 (30 ml and 42 ml per year respectively) [Tashkin et al. 2008]. However, the control group in UPLIFT included patients treated with LABAs and ICS, which may have masked the benefit of tiotropium [Miravitlles and Anzueto 2009]. In another study, treatment-naïve patients receiving tiotropium had a mean 42 ml annual decrease in FEV1, compared with 53 ml with placebo (p = 0.026) [Troosters et al. 2010], suggesting that tiotropium may reduce the decline in lung function. Second, the slow dissociation of tiotropium from M1 and M3 receptors, while advantageous in terms of the long duration of action that it confers [Barr et al. 2006; Disse et al. 1999, 1993], has the potential to prolong antimuscarinic side effects. Third, tiotropium has a slow onset of action and has been shown to increase FEV1 by less than 50 ml at 5 min after dosing on the first day of treatment, with clinically significant effects occurring within 30 min of the first dose and peak effects achieved after up to 3 h [Casaburi et al. 2000].
Tiotropium was generally well tolerated in clinical trials, although concerns were raised about cardiovascular safety, based on a meta-analysis [FDA, 2008; Singh et al. 2008]. However, no significant increase in cardiovascular events was found in the large (n = 5992) UPLIFT trial, in which serious adverse events were collected prospectively and independently adjudicated over 4 years [Tashkin et al. 2008]. The US Food and Drug Administration (FDA) concluded that the data from UPLIFT were sufficient to address the potential safety concerns raised by earlier reports [FDA, 2010].
The most frequent adverse event associated with tiotropium is dry mouth. It occurred in 5.5% of primary-care managed patients (versus 1.5% with placebo) in a recent 12-week study [Freeman et al. 2007]; whereas 16% of patients (versus 2.7% with placebo) reported dry mouth in a 1-year study [Casaburi et al. 2000]. In another study, the likelihood of dry mouth in tiotropium recipients was 4.6-fold higher than with placebo, 2.1-fold higher than with ipratropium and 4.7-fold higher than with salmeterol [Barr et al. 2006]. A pooled analysis in 17,014 patients showed that dry mouth was more frequent with tiotropium (incident rate 3.06) than placebo (1.29) [Kesten et al. 2009].
Profile of the ideal LAMA
The efficacy profile of an ideal treatment for COPD should include delivery of clinically meaningful bronchodilation, as well as symptom relief, prevention of disease progression, improvement in exercise tolerance and health status, prevention and treatment of complications and exacerbations and a reduction in mortality risk [GOLD, 2009]. Clinically meaningful bronchodilation has been defined as a difference of 100 ml in trough FEV1 [Donohue, 2005]. Definitions of clinically meaningful improvements are also available for other efficacy endpoints, including dyspnoea (≥1 point on the TDI scale [Mahler and Witek, 2005]), health-related quality of life (HRQoL; ≥4 point improvement in SGRQ [Jones et al. 1991]) and exercise endurance (54 m improvement on the 6 min walk test [ATS, 2002]). However, it is important to note that patients with COPD are highly heterogeneous, with a wide variety of clinical, genetic and pharmacological characteristics, which complicates definition and validation of the minimal clinically important differences [Kiley et al. 2005].
An ideal LAMA would also have a 24-h duration of action with once-daily dosing combined with a rapid onset of action. Once-daily dosing has the potential to improve adherence with medication in patients with COPD [Bourbeau and Bartlett, 2008] and has been associated with higher levels of persistence [Breekveldt-Postma et al. 2007]. It has also been postulated that bronchodilators with long durations of action provide a relatively consistent improvement in airway calibre over time, which contrasts with the peaks and troughs that may occur with twice daily agents or repeated use of short-acting bronchodilators [Beeh and Beier, 2010].
The ideal LAMA would also have a favourable safety and tolerability profile and be convenient and easy for patients to use. New LAMAs will be particularly welcome if they have an improved tolerability profile compared with existing agents, with a lower frequency of adverse effects (such as dry mouth) and a good cardiovascular safety profile. An ideal LAMA would also be delivered via a convenient and easy to use device that provides effective delivery of the drug to the lungs of patients with COPD.
NVA237
Characteristics
NVA237 is a dry-powder formulation of glycopyrronium bromide in development as a once-daily LAMA for the treatment of COPD. It is delivered via the Concept1 device, which was originally developed for use with indacaterol (Onbrez Breezhaler, Novartis, Horsham, UK) and has a low air-flow resistance (0.07 cmH2O½/litre/min) to ensure patients with severe lung conditions are able to use the device effectively [Pavkov, 2010]. Concept1 has been shown to deliver a consistent dose irrespective of disease severity and age, with no reported device failures in clinical trials [Pavkov, 2010].
NVA237 has a quaternary ammonium structure that differs from ipratropium and tiotropium, minimizes oral bioavailability [Ali-Melkkila et al. 1993] and has the potential to reduce systemic side effects resulting from swallowed portions of the dose. NVA237 has a high degree of selectivity for M3 receptors over M2 receptors (Novartis, data on file). As noted earlier, blockade of M1 and M3 receptors reduces or reverses bronchoconstriction, whereas blockade of M2 receptors attenuates feedback inhibition of acetylcholine production, which may reduce the bronchodilating effects of a muscarinic antagonist [Beeh et al. 2002]. In addition, agents that block M2 receptors may also result in an increase in heart rate. The selectivity of NVA237 for M3 versus M2 receptors compares favourably with that of tiotropium (selectivity ratios of 5 versus 2, respectively). NVA237 has greater kinetic selectivity for the M3 receptor versus the M2 receptor, compared with tiotropium (kinetic selectivity ratios of 9.0 versus 4.3, respectively) (Novartis, data on file). Additionally, tiotropium takes four to five times longer to equilibrate with the M3 receptor than NVA237. The more rapid equilibration of NVA237 with the M3 receptor may underlie the faster onset of action of NVA237 (4.8-fold faster) versus tiotropium [Sykes et al. 2010].
Clinical efficacy
In a 7-day, randomized, double-blind, placebo-controlled crossover study in patients with stable moderate-to-severe COPD, NVA237 once daily provided significantly greater bronchodilation than placebo, with a rapid onset of effect that was sustained throughout a 24-h period [Verkindre et al. 2010]. Mean trough FEV1 values on days 1 and 7 were significantly greater across all doses of NVA237 compared with placebo (Figure 1). NVA237 and tiotropium resulted in significant improvements in FEV1 throughout the 24-h period on day 7 compared with placebo (Figure 2(A)), with no statistically significant differences between NVA237 50 µg and 100 µg and tiotropium at any timepoint. FEV1 was significantly higher with NVA237 50 µg and 100 µg once daily than with tiotropium at 5 min and up to 2 h, with a significant difference between NVA237 100 µg and tiotropium also sustained up to 4 h (Figure 2(B)). In addition, the improvement in trough FEV1 with NVA237 50 µg was statistically superior (p = 0.0070) to 12.5 µg and numerically better than 25 µg NVA237 (p = 0.0537) on day 7. NVA237 50 µg was also statistically superior to both 25 µg (p = 0.0009) and 12.5 µg (p < 0.0001) on day 1 [Overend et al. 2010].
Trough forced expiratory volume in 1 s (FEV1) treatment contrasts (active versus placebo and NVA237 versus tiotropium; least square means, 95% confidence intervals) after (A) 7 days and (B) 1 day of treatment. (Reproduced with permission from Verkindre and colleagues [Verkindre et al. 2010]). Twenty-four-hour profile of least square means of forced expiratory volume in 1 s (FEV1) on (A) day 7 and (B) day 1 of treatment. (A) All treatments were significant (p < 0.05) versus placebo; #
p < 0.05 versus tiotropium; (B) all treatments were significant (p < 0.05) versus placebo at each post-dose time point (except NVA237 12.5 µg at 23 h 45 min); #
p < 0.05 versus tiotropium. (Reproduced with permission from Verkindre and colleagues [Verkindre et al. 2010]).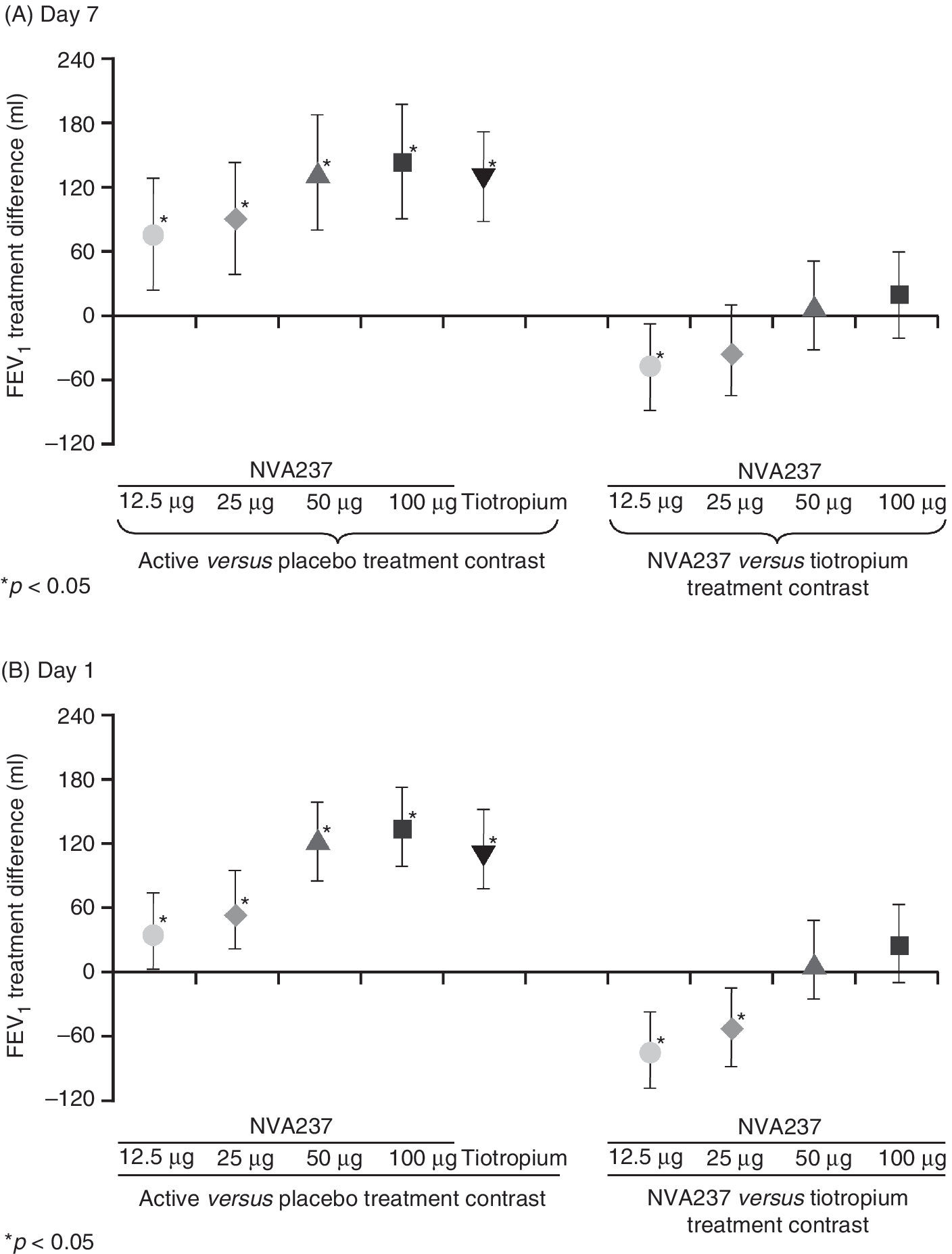

In a randomized, double-blind, placebo-controlled, two-period cross-over study, NVA237 50 µg once daily provided sustained, clinically significant 24-h bronchodilation [Fogarty et al. 2011]. On day 14, patients receiving NVA237 50 µg had significantly higher values of FEV1 area under the curve during 0–24 h (FEV1 AUC0–24h) than placebo recipients (Figure 3). In addition, trough FEV1 was significantly higher (p < 0.05) in patients receiving NVA237 than in patients receiving placebo, with a mean difference on day 14 of 154 ml that exceeded the threshold for a clinically significant effect.
Twenty-four hour bronchodilatory effect on day 14. *Only patients with forced expiratory volume in 1 s (FEV1) values in both periods for a given timepoint were included. Values are expressed as mean ± standard error. AUC, area under the curve. (Reproduced with permission of the American Thoracic Society. Copyright © 2010 American Thoracic Society [Fogarty et al. 2010]).
Clinical safety
In the 28-day study, NVA237 once daily was well tolerated and had an adverse event profile similar to that of placebo; doses studied were two to four times the dose selected for inclusion in phase III studies [Vogelmeier et al. 2010b]. The overall frequency of adverse events was similar in patients receiving NVA237 100 µg (28.3%), NVA237 200 µg (26.5%) and placebo (26.4%). Dry mouth occurred in 3.3% of patients in the NVA237 100 µg group, 7.1% of patients in the NVA237 200 µg group, and none of the placebo recipients. Thus, the incidence of dry mouth with NVA237 was low, even in patients receiving two to four times the dose currently being evaluated in phase III studies. The results compare favourably with previously published data for tiotropium 18 µg, where the frequency of dry mouth was reported to be 14.7% in a 13-week study of patients with COPD [van Noord et al. 2000]. The low incidence of dry mouth with NVA237 will be further assessed in ongoing long-term studies. The majority of adverse events in the 28-day NVA237 study were of mild severity, while severe adverse events occurred in only three patients: one in the NVA237 200 µg group (pneumonia, not considered drug related) and two in the placebo group (dyspnoea suspected to be drug related; COPD exacerbation). The 14-day and 7-day studies also showed that NVA237 was generally well tolerated at doses of up to 100 µg and was associated with low frequencies of serious and severe adverse events [Fogarty et al. 2011; Verkindre et al. 2010].
Mean QTc intervals (Fridericia’s) were similar among patients receiving NVA237 or placebo on days 1 and 28 in the 28-day study [Vogelmeier et al. 2010b] (Figure 4). Although there were statistically significant differences of up to 6 ms between the two NVA237 doses at 23 h 45 min on days 1 and 28, the differences were not considered clinically relevant. Notable changes in QTc interval from predose to maximum postdose (Fridericia’s formula; >440 ms for men and >460 ms for women) occurred in similar percentages of patients in the placebo (6.6%), NVA237 100 µg (9.8%) and NVA237 200 µg (2.0%) groups. No clinically meaningful between-group differences were noted for other electrocardiographic (ECG) variables (QT, RR, PR, QRS, and ventricular rate). Mean pulse rates were generally similar across all three treatment groups and there were no clinically meaningful differences in diastolic or systolic blood pressure between NVA237 and placebo. The 7-day study also revealed a low incidence of notable QTc interval values in patients receiving NVA237 (0–3.8%), compared with the placebo group (5.5%) [Verkindre et al. 2010]. Similarly, in the 14-day study there were no clinically relevant changes in vital signs or ECGs [Fogarty et al. 2011].
Least square mean (standard error) values for QTc interval (Fridericia’s) (safety population). *p < 0.05 versus NVA237 100 µg. (Reproduced with permission from Vogelmeier and colleagues [Vogelmeier et al. 2010b]).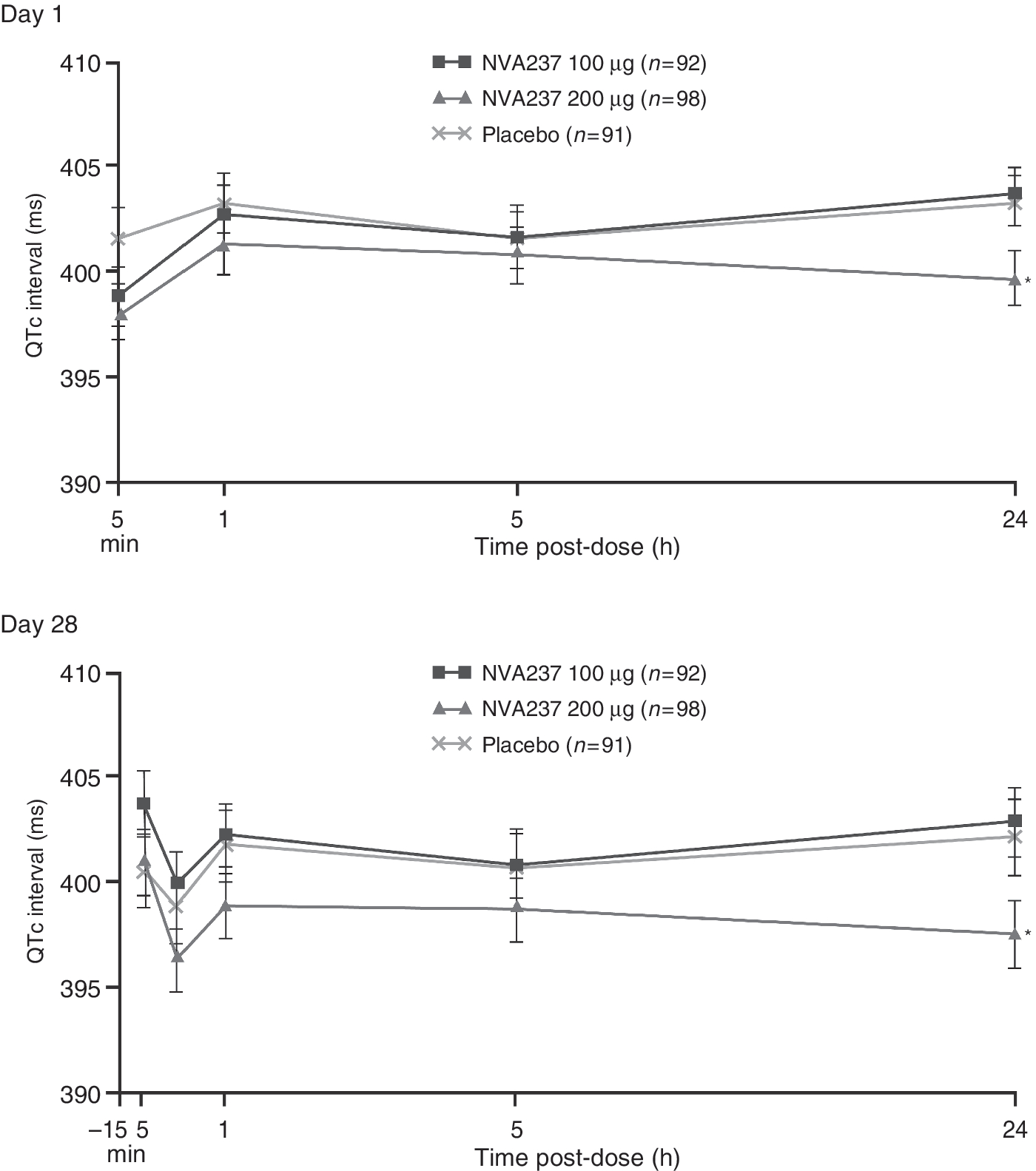
Future of NVA237
Phase III studies have been planned and are ongoing to evaluate the benefits of once-daily NVA237 in patients with moderate-to-severe COPD, including a 26-week study (GLOW1; NCT01005901) comparing NVA237 with placebo and a 52-week study (GLOW2; NCT00929110) comparing NVA237 with placebo and open-label tiotropium. Both studies will evaluate effects on trough FEV1 (primary outcome), dyspnoea, HRQoL, exacerbations and rescue medication use. In addition, a 42-day, randomized, double-blind, placebo-controlled, two-period crossover study (GLOW3; NCT01154127) will evaluate the effects of NVA237 and placebo on exercise tolerance (primary outcome) and inspiratory capacity. Other phase III studies are also planned to further profile the use of NVA237 in patients with COPD.
There may be the potential for benefits with a combination of once-daily NVA237 and a once-daily LABA such as indacaterol (Onbrez). Current guidelines recommend the use of combinations of bronchodilators, especially LABAs and LAMAs, for patients whose symptoms are not sufficiently controlled by maintenance bronchodilator monotherapy [GOLD, 2009]. The complementary mechanisms of action of LABAs (stimulation of β2 adrenergic receptors) and LAMAs (inhibition of the action of acetylcholine at muscarinic receptors) both result in smooth muscle relaxation [Verkindre et al. 2010; Johnson, 1998; Roux et al. 1998], providing the potential for additive or synergistic effects resulting in maximum bronchodilation. Combinations of LABAs and LAMAs have been shown to significantly improve lung function compared with monotherapies, as well as improving HRQoL and dyspnoea and reducing COPD exacerbation rates, with no apparent increase in safety concerns [Tashkin et al. 2009a, 2009b; Vogelmeier et al. 2008; Aaron et al. 2007; van Noord et al. 2006; Johnson, 1998].
QVA149 is a once-daily dual bronchodilator containing a fixed-dose combination of NVA237 and indacaterol and is in development for the treatment of COPD. Phase II studies have shown that once-daily QVA149 provided sustained 24-h bronchodilation that is superior to indacaterol 300 µg and 600 µg monotherapy (Figure 5) [van Noord et al. 2010]. Combining NVA237 with indacaterol (in the form of QVA149) had additive effects on bronchodilation, resulting in a mean treatment difference for trough FEV1 on day 7 of 226 ml versus placebo, 123 ml versus indacaterol 300 µg and 117 ml versus indacaterol 600 µg (all p < 0.001). This additive bronchodilatory effect of QVA149 was not associated with evidence of an additive effect on safety signals. QVA149 has been shown to be well tolerated and was not associated with significant effects on 24-h mean heart rate after 14 days of treatment [Van de Maele et al. 2010].
Trough forced expiratory volume in 1 s (FEV1) on day 7. Data are least squares mean ± SE; *p < 0.0001 versus placebo; #
p < 0.0001 versus indacaterol 300 µg and 600 µg. (Reproduced with permission from van Noord and colleagues [van Noord et al. 2010]).
Discussion
Long-acting bronchodilators are central to COPD management, with the LAMA tiotropium providing an important option [GOLD, 2009]. Although tiotropium is effective and is generally well tolerated [Casaburi et al. 2000], it has limitations, including a high incidence of dry mouth [Barr et al. 2006], a slow onset of action [Casaburi et al. 2000] and, in some studies, a part of the patient population did not achieve clinically significant bronchodilation [Yorgancýoðlu et al. 2009; Brusasco et al. 2003]. It also remains unclear whether tiotropium reduces the progressive decline in lung function typical of COPD [Troosters et al. 2010; Miravitlles and Anzueto 2009; Tashkin et al. 2008].
An ideal LAMA for use in COPD would deliver clinically meaningful bronchodilation, as well as symptom relief, prevention of disease progression, improvement in exercise tolerance and health status, prevention and treatment of complications and exacerbations and a reduction in mortality risk [Troosters et al. 2010; GOLD, 2009]. Other characteristics of an ideal LAMA would include a 24-h duration of action, rapid onset of action, and a good safety and tolerability profile.
NVA237 is a novel once-daily LAMA that may meet some of these characteristics and could have the potential to overcome some of the limitations associated with tiotropium. Additional studies, however, are needed to confirm this. The high M3 selectivity of NVA237 could be an advantage and might potentially result in a higher therapeutic index compared with tiotropium, which is less selective for M3 receptors. LAMAs with high selectivity for M3 receptors may provide effective bronchodilation that is not attenuated by blockade of M2 receptors and may be associated with a lower risk of adverse effects due to blockade of extrapulmonary M2 receptors [Abrams et al. 2006; Beeh et al. 2002].
Phase II studies have shown that NVA237 once daily provides clinically significant 24-h bronchodilation with a rapid onset of action and a favourable safety and tolerability profile [Fogarty et al. 2010; Verkindre et al. 2010; Vogelmeier et al. 2010b]. Ongoing phase III studies will determine to what extent NVA237 meets other characteristics considered desirable in a new LAMA intended for use in COPD. In addition, other studies will evaluate the potential benefits of a combination of NVA237 and the once-daily LABA indacaterol (QVA149) in patients with inadequately controlled COPD on bronchodilator monotherapy.
In conclusion, NVA237 has demonstrated clinically meaningful bronchodilator efficacy and a good safety and tolerability profile in phase II studies and is currently under investigation in phase III studies. The available data for NVA237 indicate that it has characteristics considered important in a new LAMA for use in COPD, including a fast onset of action, sustained 24-h bronchodilation with once-daily dosing and a favourable safety and tolerability profile.
Footnotes
Acknowledgements
Writing and editorial assistance was provided by Melanie Stephens (ACUMED®, London, UK) and Mark Fedele (Novartis). This assistance was funded by Novartis Pharma AG (Basel, Switzerland).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Professor Vogelmeier has given presentations at symposia sponsored by AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Janssen-Cilag, Novartis, Nycomed, Pfizer and Talecris, and was/is a member of advisory boards organized by AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Janssen-Cilag, Novartis, Nycomed, Pfizer and Talecris. Donald Banerji is an employee of Novartis Pharmaceuticals Corporation.
