Abstract
There is a need for straightforward, novel diagnostic and monitoring technologies to enable the early diagnosis of COPD and its differentiation from other respiratory diseases, to establish the cause of acute exacerbations and to monitor disease progression. We sought to establish whether technologies already in development could potentially address these needs. A systematic horizon scanning review was undertaken to identify technologies in development from a wide range of commercial and non-commercial sources. Technologies were restricted to those likely to be available within 18 months, and then evaluated for degree of innovation, potential for impact, acceptability to users and likelihood of adoption by clinicians and patients with COPD. Eighty technologies were identified, of which 25 were considered particularly promising. Biomarker tests, particularly those using sputum or saliva samples and/or available at the point of care, were positively evaluated, with many offering novel approaches to early diagnosis and to determining the cause for acute exacerbations. Several wrist-worn devices and smartphone-based spirometers offering the facility for self-monitoring and early detection of exacerbations were also considered promising. The most promising identified technologies have the potential to improve COPD care and patient outcomes. Further research and evaluation activities should be focused on these technologies.
Introduction
Chronic obstructive pulmonary disease (COPD) is the third leading cause of death globally and presents a significant burden to patients, carers and health services worldwide. 1,2 More than 1.5 million adults are known to be diagnosed with COPD in England and Wales, and a further 3 million adults are estimated to be living with undiagnosed COPD. 3,4 Improving the care and outcomes for people with COPD is a priority for the National Health Service (NHS) in England, which aims to reduce premature mortality from respiratory disease, avoid unnecessary hospital admissions and improve the quality of life and support for patients with long-term conditions and their carers. 5 However, several current issues remain in the diagnosis and monitoring of COPD, some of which could be resolved by technological developments or novel disease biomarkers.
Several authors have noted limitations in the use of spirometry to diagnose COPD due to differing guidelines as to what constitutes COPD, difficulties in technique encountered by frail or cognitively impaired patients, and a lack of awareness and knowledge in primary care leading to delayed diagnosis, especially in non-smoking subgroups. 6 –9 In addition, some commentators have proposed a need for improved diagnostic criteria where asthma and COPD coexist, observing that non-specialist clinicians frequently find it difficult to both diagnose and manage this situation, 8,10,11 whilst others consider that additional emphasis should be placed on early detection. 9,12,13 There is an unmet need for simple, accurate testing approaches that would enable early diagnosis and differentiation from other respiratory diseases. 13 As part of the ongoing care of COPD, patient self-monitoring is increasingly being recognized as beneficial, with evidence indicating this may improve health-related quality of life and the recognition and management of acute exacerbations, 14,15 and self-monitoring is recommended in guidelines from the National Institute for Health and Care Excellence (NICE). 2 However, the most effective approach to self-management in COPD is not known. 14 Finally, several commentators identify the need to determine the cause of acute exacerbations as a research need; this would allow treatment to be tailored to the underlying pathology. 16,17 Correspondingly the NICE Database of Uncertainties about the Effects of Treatments includes questions about the appropriate use of corticosteroids and antibiotics for the management of acute exacerbations of COPD. 18,19 As only around half of exacerbations are associated with a bacterial infection and a only a third of exacerbations demonstrate eosinophilic inflammation, the use of these agents may expose many patients to significant adverse effects and the potential development of antibiotic resistance for no apparent benefit. 20,21 There is an unmet need for novel diagnostic or monitoring approaches, particularly those available at the point of care, to establish the cause of acute exacerbations and to monitor the progression of the disease.
Horizon scanning systems, or early awareness and alert systems, aim to identify significant health technologies prior to launch that may require further assessment or planning prior to adoption. 22,23 A horizon scanning review uses systematic methods to identify, filter, prioritize and present early information on all new and emerging technologies relevant to the area of interest, however, it does not assess the evidence supporting the identified technologies, validate the claims made about them by developers or comprehensively evaluate the potential impact they may have on clinical care. 24,25 We sought to identify new and emerging technologies already in development for the diagnosis or monitoring of COPD that could potentially address the unmet research and clinical practice needs identified above, and then use the views of clinical experts and patients with COPD to establish which of these technologies could be considered the most innovative, acceptable and likely to make an impact on patients and health services in the future. This horizon scanning review forms part of the National Institute for Health Research Horizon Scanning Research and Intelligence Centre work programme. The full report is available on the Centre’s website (http://www.hsric.nihr.ac.uk/news/what-does-the-future-hold-for-copd-diagnostic-and-monitoring-technologies/) and will be used to inform healthcare policymakers, commissioners, researchers, research funders, clinicians and patients about new technologies ‘on the horizon’ for the management of COPD. Ethical approval was obtained from the University of Birmingham’s Internal Ethical Review Committee (reference: IERC2014-5/C1/SF/07).
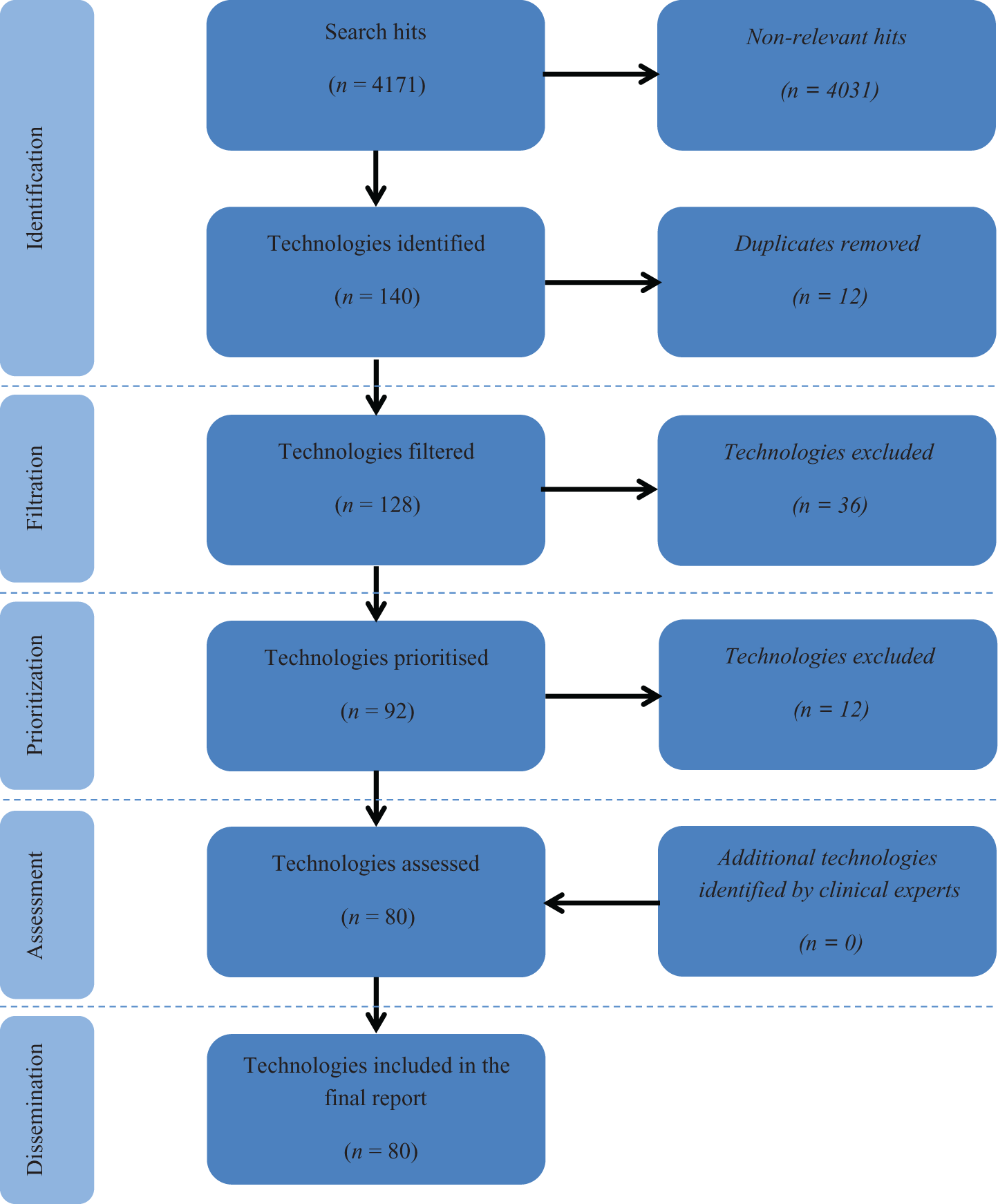
Identification, filtration and prioritization of technologies included in the horizon scanning review. Adapted from the PRISMA flow diagram for systematic reviews. 30
Methods
Identification and filtration of technologies
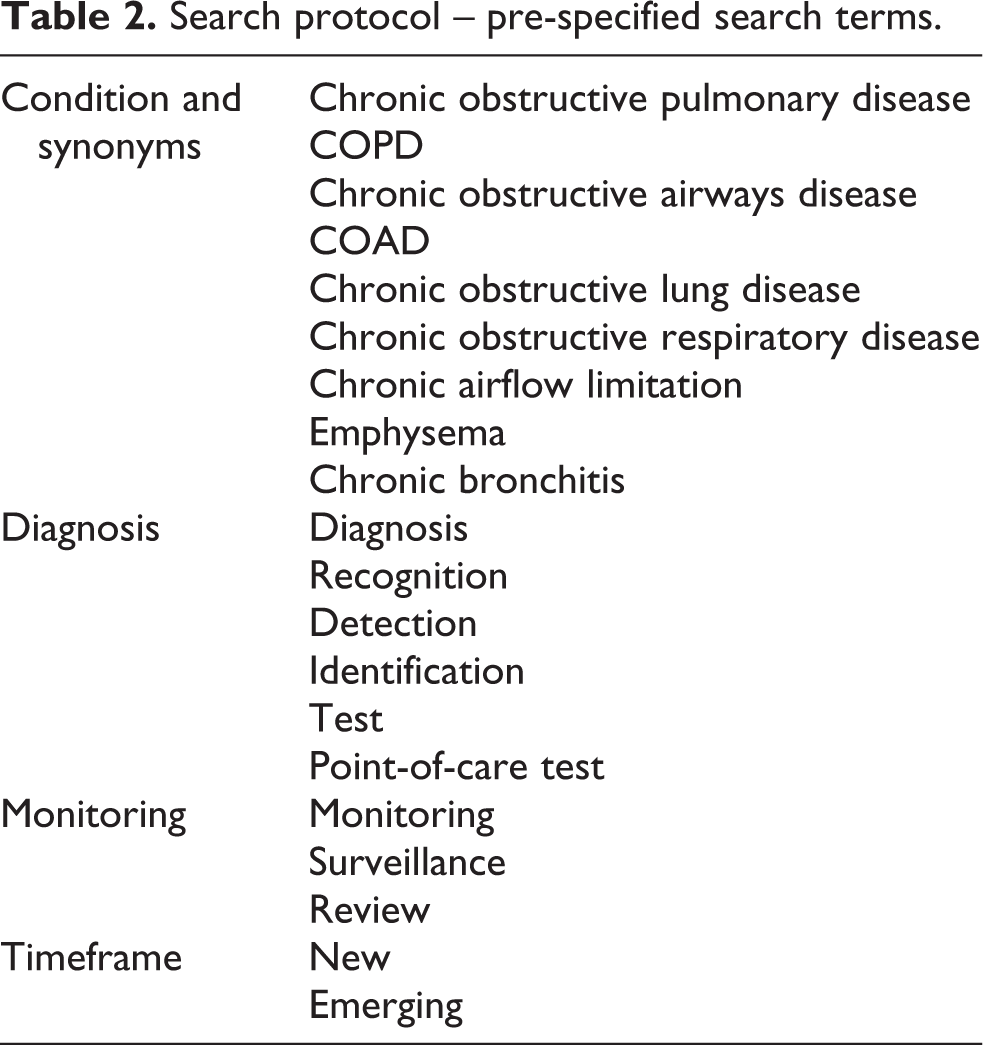
Between January and March 2015, potential technologies for the diagnosis and monitoring of COPD were identified by searching relevant online databases and other websites (Table 1) using pre-specified search terms (Table 2) and eliciting suggestions from clinical experts. Initial search findings were filtered to include only those technologies which were new (already licensed/CE marked or launched in the United Kingdom for ≤24 months – in the launch, early post-marketing or early diffusion phase) or emerging (in development and expected to be licensed/CE marked in the next 18 months – in late phase clinical trials, prelaunch or pre-marketing phase), and results were further prioritized to include only those technologies that demonstrated, or claimed to demonstrate, some degree of innovation (either a completely novel technology, with no direct comparators already marketed, or a significant development from existing marketed products).
Search protocol – pre-specified identification sources.

Search protocol – pre-specified search terms.
External input
Clinical experts and patients with COPD were recruited as expert external advisors to the review. Clinical experts were identified from initial scoping and literature searches and the NICE COPD guideline development and review panel. Fifteen clinical experts were contacted via email and asked to act as review advisors, five of whom subsequently agreed, resulting in an expert panel comprising consultant respiratory physicians, a general practitioner with a special interest in respiratory disease and a clinical nurse specialist. Clinical experts were emailed the table of identified technologies and asked to provide their views on each technology’s level of innovation, potential for impact (on patients and the delivery of health services), potential barriers to adoption and any further comments. They were also asked to identify any additional relevant technologies or relevant research they were aware of that had not already been identified.
Patients with COPD were identified from the patient advisory group to the existing Birmingham Lung Improvement StudieS (BLISS) research programme. 26 Members of the group were approached using a standard email or letter informing them of the review and inviting them to participate. Three patients subsequently agreed and were sent the table of identified technologies and asked to provide their views on each technology’s level of innovation, potential for impact (on patients and the delivery of health services) and acceptability to users, along with any further comments.
Review output
All identified new and emerging technologies for the diagnosis or monitoring of COPD were included in the final report. Clinical expert and patient views were used to identify those technologies that were considered to demonstrate a high degree of innovation and/or a significant potential for impact and adoption by the NHS in England.
Results
Eighty new and emerging technologies for the diagnosis and monitoring of COPD were identified (Table 3, Figure 1). These included 31 biomarkers, 21 telehealth technologies, 6 wearable technologies, 6 imaging technologies, 4 vital sign monitors, 4 questionnaires, 3 spirometers and 5 additional other technologies. Clinical experts and patients provided comments on all identified technologies, providing useful insights into the innovativeness, potential impact, stage of development and probable timeframe for each technology. Of the technologies identified, 25 (31.3%) were considered particularly promising, demonstrating a high degree of innovation and with a significant potential for impact and adoption by the NHS according to expert and patient comments (Table 4).
Technologies identified by the review.

BEAT: COPD -: Biomarkers to Target Antibiotic and Systemic Corticosteroid Therapy in COPD; USFDA: US Food and Drug Administration; COPD: chronic obstructive pulmonary disease; NHS: National Health Service; CRP: C-reactive protein; PCT: procalcitonin; FTIR: Fourier transform infrared; SPOC: saliva point of care; VOC: volatile organic compounds; MRI: magnetic resonance imaging; DOSE: dyspnoea, obstruction, smoking, exacerbation; mPCR: multiplex polymerase chain reaction; SPPB: short physical performance battery; CT: computed tomography; ResPOC: Respiratory Virus Point-Of-Care; DECAF: Dyspnoea, Eosinopenia, Consolidation, Acidaemia and atrial Fibrillation.
Promising technologies – those considered to demonstrate a high degree of innovation, potential for impact, and potential for adoption by the NHS.

FTIR: Fourier transform infrared; SPOC: saliva point of care; COPD: chronic obstructive pulmonary disease; DOSE: dyspnoea, obstruction, smoking, exacerbation; mPCR: multiplex polymerase chain reaction; ADAPT: After DischArge Pulmonary Telehealth; CRP: CRP: C-reactive protein; ResPOC: Respiratory Virus Point-Of-Care; DECAF: Dyspnoea, Eosinopenia, Consolidation, Acidaemia and atrial Fibrillation.
Wearable technologies
All identified wearable technologies were intended to monitor stable COPD through devices worn either across the chest or on the wrist. Whilst devices monitoring respiratory rate, wheeze, blood oxygenation and/or temperature are already available, several new technologies enabled self-monitoring at home with remote access by clinicians, though the feasibility of multiple clinicians interpreting large volumes of recorded data was questioned. Patients commented that users may be unwilling to wear a ‘cumbersome’ looking device and were concerned about ‘having the restriction of a band around [their] chest’. The wrist-based pulse oximeter and BuddyWOTCH devices could offer alternatives to existing fingertip devices, providing continuous monitoring without limiting movement of the user’s hands. In addition, BuddyWOTCH captures physical activity, temperature and heart rate. Both technologies were considered promising, with patients keen to try a more compact, watch style device. Experts considered them ‘potentially useful…for longer term monitoring…in vulnerable groups…[and] for pulmonary rehabilitation’. Concerns were expressed regarding the reliability of wrist-based monitoring; however, a potentially large impact could be expected if these devices demonstrated acceptable performance.
Biomarker technologies
Thirty-one biomarker tests were identified and included in the report, the majority based on blood samples. Alternative samples included sputum, bronchial aspirate and innovatively, saliva and volatile organic compounds from breath samples. Most biomarkers were intended for the diagnosis of COPD, but experts dismissed the majority of blood-based diagnostic biomarkers as being too early in development or ‘possibly as a research tool’ with no demonstrated clinical value, despite some being considered highly innovative. Aα-Val360 was the most promising biomarker in this category, having been proposed as a method to identify patients with early COPD at risk of progression. Experts commented that it was ‘highly innovative…targeting high risk individuals’. Fourier transform infrared spectroscopic monitoring used sputum to rapidly differentiate COPD from other respiratory conditions and determine the risk of an imminent exacerbation; experts commented that this was ‘interesting and if effective then impact and adoption [are] possibly significant’. A further sputum-based biomarker panel to diagnose asthma-COPD overlap was thought to be ‘of interest in secondary care clinics’, whilst experts considered COPD-saliva point of care (SPOC), a novel saliva-based point-of-care test for the diagnosis of COPD incorporating three biomarkers, to have the ‘potential to change practice’.
Several inflammatory blood biomarkers were proposed for the monitoring of COPD for risk of an acute exacerbation. However, fibrinogen and C-reactive protein (CRP) were noted to be ‘non-specific indicators of inflammation’ and therefore ‘unlikely to be much impact or …[high adoption]’. In contrast, procalcitonin was thought to be a ‘more specific marker of infection’. Researchers claimed it was able to differentiate between a bacterial and nonbacterial cause of acute exacerbations, prompting an expert to state ‘…a strong chance of high impact (more appropriate exposure to antibiotics) and adoption’. The Home Use Sputum Test may provide advance warning of an acute exacerbation in the community and experts were very positive, commenting ‘this would be useful and may help identify pathogens early so treatment can be commenced’, and highlighting the potential to ‘reduce antibiotic use’, though also noting ‘identifying a bacterium does not always mean that the cause of symptoms is identified’. Patients commented on its potential to ‘reassure patients’. Other technologies aimed to identify the cause of acute exacerbations in order to better direct specific management. Experts commented that the use of peripheral blood eosinophil count as a biomarker to direct corticosteroid therapy was ‘worth exploring more’, though impact was judged to be ‘unclear, as most exacerbations are in the community and [this biomarker] will need a blood test’. The CRP POC test was considered innovative, providing an accessible fingerprick test in community settings. One expert remarked ‘yes to innovation, yes to potential impact and adoption is a strong possibility if shown to help decision making’.
Telehealth technologies
Telehealth is a popular area of development in technologies for chronic diseases and represented a large proportion of all technologies identified. Many devices were in a late stage of development from established commercial developers and had already obtained a CE mark, making them more likely to come to market within the specified timeframe. However, telehealth was generally viewed negatively by the clinical experts, with comments aimed at the category as a whole rather than in-depth review of each technology. Experts cited a UK multicentre trial of touchscreen telemonitoring for COPD 27 and a systematic review and meta-analysis of telehealth for COPD, 28 commenting on the current high estimates for cost-effectiveness of similar technologies and the lack of evidence that they improve quality of life or reduce mortality. In addition, experts expressed concern about the burden on clinicians, saying ‘the problem with this kind of technology is that you need a healthcare professional to look at the data’.
Imaging technologies
The majority of imaging technologies were considered highly innovative, but clinical experts believed their use would be restricted to research or specialist centres, so that uptake was likely to be very limited, and these technologies had little potential for impact across the whole of the NHS.
Vital sign monitoring technologies
Four devices offered novel methods for monitoring COPD. Both EverOn and the Respiratory Holter technologies monitored respiratory rate, with one expert noting ‘the accurate long-term measurement of respiratory rate in hospital/home may be useful’. However, neither were particularly novel and experts were unsure whether they would be ‘useful in the real world’. Similarly, Capno-Pulse received mixed comments from experts, with some identifying a role for improved non-invasive arterial blood carbon dioxide monitoring and others unclear whether it had potential for impact. However, patients were generally more positive, saying they would be willing to try these devices.
Spirometry technologies
These devices use a smartphone mounted, handheld device for home monitoring of COPD. Most clinicians are aware that monitoring current spirometric parameters does not reliably detect deterioration or exacerbation in COPD, though changes may be evident in asthma and potentially asthma-COPD overlap. 2 However, comments were mostly encouraging, with good potential for adoption when used in conjunction with training and appropriate follow-up. Patients considered these technologies ‘could have [a] considerable impact’ and were ‘definitely’ acceptable to users. Two devices incorporate Bluetooth technology, however, the additional impact of this connectivity was thought to be limited due to the requirement for an interpreting clinician and patients’ ability to respond to the results.
Questionnaire-based technologies
Questionnaires were considered acceptable by patients providing they were not too long as ‘patients do get tired of filling in forms’. Two questionnaires aimed to monitor the severity of exacerbations in order to inform clinical decisions regarding care. The Dyspnoea, Eosinopenia, Consolidation, Acidaemia and atrial Fibrillation (DECAF) score was considered a ‘useful innovation to predict safe early discharge’, whilst experts disagreed about the Dyspnoea, Obstruction, Smoking, Exacerbation (DOSE) index, with one noting that it is ‘well validated…difficult to see why not used more’ and another commenting ‘not clear to me that it is any advance over existing risk assessment tools’. Two questionnaires were aimed at COPD diagnosis. The COPD screening questionnaire includes smoking history and symptoms and aims to facilitate early diagnosis. Again, experts were divided in their opinions. One commented it was ‘a useful way to identify and encourage people to get spirometry’ whereas another said it was ‘not novel, tools already exist’.
Other technologies
Three technologies aimed to diagnose the cause of acute exacerbations in order to direct treatment. The developer of Respiratory Virus Point-Of-Care (ResPOC), a point-of-care test, claims it is capable of identifying respiratory viruses within 1 hour, with an equivalent accuracy to that of standard laboratory tests. Multiplex-polymerase chain reaction uses similar technology to diagnose viral infections using samples of tracheal or nasal aspirate, whilst HIRA-TAN may provide rapid and sensitive identification of bacterial pathogens. All of these three similar technologies received positive comments from experts, noting they were ‘innovative’ with high ‘potential for impact and adoption’.
Discussion
We identified a large number of new and emerging technologies for the diagnosis and monitoring of COPD, almost a third of which were considered to be innovative with significant potential for impact on patients and health services and be appropriate for adoption by the NHS. The technologies included in this horizon scanning review broadly addressed the issues identified as key research and unmet practice needs. Novel biomarkers offered a potential future alternative approach to early diagnosis and the differentiation from other respiratory diseases, including several based on sputum or saliva samples with results available at the point of care. Experts were less favourable about non-COPD-specific biomarkers of inflammation, though plasma fibrinogen has since become the first COPD biomarker to receive Food and Drug Administration approval for use in interventional clinical trials. 29 Many technologies offered the potential to facilitate patient self-monitoring, including vital sign monitors (including wearable technologies), telehealth systems, smartphone-based spirometers and biomarkers, with a home-use sputum test potentially allowing advance warning of an acute exacerbation. Patients emphasized their preference for compact, simple, easy to use and portable monitoring devices that did not interfere with daily life, so that wrist-worn devices were preferred over vest-like wearable devices. Importantly, six innovative technologies were identified with the potential to establish the cause of an acute exacerbation, two of which offered this as at the point of care. These could enable treatment of acute exacerbations to be tailored to the underlying pathology, potentially avoiding the unnecessary use of antibiotics and corticosteroids.
The review used an established horizon scanning review methodology to systematically identify technologies. 22,24 The approach aims to ensure that all technologies appropriate to the review are identified, and this is facilitated by the use of multiple online sources, including specialist media, health technology assessment organizations, regulatory authorities, research funders, subscription-only commercial research and development databases, clinical trial registries and professional and patient groups, in addition to bibliographic databases of published medical literature and specialist journals. However, it is not possible to claim that all possible technologies have been identified as this is dependent on commercial, media and academic reporting, though reassuringly clinical experts did not know of any relevant technologies in development that had not already been identified. A particular limitation of horizon scanning reviews is the restricted information on each technology upon which to base inclusion/exclusion, filtration/prioritization and assessment decisions. It is common for there to be limited publicly accessible information and scientific data available for technologies in development or the early launch stage, and we made no attempt to validate developers claims for their products, all of which require further clinical validation using patient-relevant outcomes and information on costs prior to adoption.
The views of clinical experts and patients with COPD provided essential insights into the place of identified technologies in COPD management and the acceptability of individual technologies to users. For some technologies, there was disagreement as to their potential impact, reflecting the individual’s particular perspective, experience of similar developments and local pathways of care. This was important, as we sought consensus before labelling a technology as particularly promising. Telehealth was a particular source of disagreement in this review, with technologies receiving generally negative comments from clinical experts, citing impracticality, the existing evidence base and cost as a barrier to use, whereas patients were broadly in favour of devices that enabled them to self-monitor and communicate with clinicians. Experts also noted the lack of evidence that variables such as cough frequency or inhaler use were associated with the risk of an acute exacerbation, and that clinical trials of such systems typically measured success in terms such as patient satisfaction, as opposed to conventional clinical parameters, admissions or healthcare utilization. In a field becoming increasingly saturated with similar products, yet experiencing little adoption by the NHS, it seems unlikely that telehealth will become part of routine COPD care until it is shown to improve patient outcomes and a feasible cost-benefit balance is achieved.
Conclusion
This review identified several innovative technologies with the potential for impact in a disease with a significant burden in the United Kingdom and worldwide. The most promising technologies have the potential to improve the early diagnosis of COPD, facilitate patient self-monitoring and establish the cause of acute exacerbations, potentially at the point of care. These technologies have the potential to contribute to addressing key priorities in the NHS Outcomes Framework, namely reducing premature deaths from respiratory disease, enhancing the health-related quality of life of patients with COPD and the quality of life of their carers, ensuring people feel supported to manage their condition and reducing unplanned admissions and time in hospital. 5 In addition, this review identified many technologies in the fields of telehealth and wearable technologies that were considered to be less promising, impractical or poorly directed. This review enables the focus of future research funding and NHS health technology assessment activities to be appropriately targeted, thereby facilitating the adoption of technologies with the potential to make a significant impact on NHS COPD care and the quality of patients’ lives.
Footnotes
Acknowledgements
The authors would like to thank the clinical experts and members of the BLISS Patient Advisory Group (including their co-ordinator, Dr Alexandra Enocson) who contributed to this review.
Author Contribution
DJW conceived the original study idea, LCD and JS contributed to the development of the study design and methods. LCD collected the data and carried out the analysis with assistance from DJW and JS. All authors (LCD, DJW, JS, SH and RM) were involved in the interpretation of the results. LCD and DJW produced the initial draft of the article, which was then circulated repeatedly to all authors for critical revision, before all authors read and approved the final version. All authors had full access to all of the study data and can take responsibility for the integrity of the data and the accuracy of the analysis. DJW is the guarantor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute for Health Research (NIHR), who fund the NIHR Horizon Scanning Research & Intelligence Centre. In addition, RM is supported by the NIHR Cambridge Biomedical Research Centre. The views expressed in this publication are those of the authors and not necessarily those of the NIHR, NHS, or the Department of Health.
