Abstract
The aim of this study is to screen the cognitive function during exacerbation of chronic obstructive pulmonary disease (COPD) and investigate whether there is any association between cognitive function and functional impairment, disease severity, or other clinical parameters. Age and sex-matched 133 subjects with COPD exacerbation, 34 stable COPD subjects, and 34 non-COPD subjects were enrolled in this study. For the purpose of this study, mini–mental state examination (MMSE) and Hospital Anxiety and Depression scale were performed. Six-minute walk distance (6MWD) was recorded, and BODE index was calculated. COPD subjects with exacerbation had the lowest MMSE scores (p = 0.022). Frequency of subjects with MMSE score lower than 24 is 22.6, 8.8, and 8.8% in the COPD subjects with exacerbation, stable COPD, and non-COPD control subjects, respectively. The COPD subjects with exacerbation who had MMSE scores lower than 24 were older and less educated. Subjects with COPD exacerbation had shorter 6MWD than that of stable COPD and non-COPD subjects. After controlling for the impact of age and educational level on MMSE, there was no association between 6MWD and MMSE scores in subjects with COPD exacerbation. Cognitive impairment is an important comorbidity during COPD exacerbation. Functional capacity is also lower in exacerbation. However, no association was found between cognitive impairment and functional capacity during exacerbation.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a multisystem disease that affects musculoskeletal, cardiovascular, psychological, and cognitive function besides pulmonary function leading to functional limitations and disability worldwide. 1,2 Non-respiratory impairments have even greater impact on disability. 2 Cognitive dysfunction is associated with increased length of stay, mortality, and dependency in activities of daily living. 1
Most studies investigating cognitive function in COPD are conducted in the stable period. 3 –9 Cognitive dysfunction was present in 43% of subjects with COPD following exacerbations requiring mechanical ventilation. 10 Hypoxemia, hypercapnia, smoking, immobilization, depression, and comorbidities are possible suggested causes for cognitive impairment, 1,4,9 although some could not find any cause. 6,8,10 Cognitive assessment of subjects with COPD in the exacerbation period is scarce. 10 –13
In a recent prospective longitudinal study by Dodd et al., cognitive impairment observed in patients with COPD exacerbation was independent of disease severity and hypoxemia. 13 Cognitive dysfunction was associated with worse health status and did not improve with recovery after 3 months. They did not investigate the association between the cognitive impairment and functional capacity.
Many neuropsychological tests are used to evaluate the cognitive function. For screening, Mini–mental state examination (MMSE) is one of the widely used tools, which can also be used in COPD. 2,3,10,11 It is relatively simple and less time consuming, which is an advantage while assessing a subject recovering from acute exacerbation.
The aim of this study is to screen the cognitive function during exacerbation of COPD and investigate whether there is any association between cognitive function and functional impairment, disease severity, or other clinical parameters.
Methods
Setting
Data were collected retrospectively from the pulmonary rehabilitation registry and electronic medical records between January 2010 and December 2011. The study was approved by Gazi University ethical committee (2013-25901600-2183). Medical and rehabilitation records of 172 consecutive subjects with COPD hospitalized for exacerbation at Department of Pulmonary Medicine of Gazi University were reviewed.
Participants
COPD was diagnosed according to Global Initiative for Chronic Obstructive Lung Disease criteria. 14 A total of 172 records of subjects with COPD with exacerbation were initially evaluated. Twenty-three subjects were excluded because of illiteracy (n = 14), psychiatric illness (n = 3), dementia (n = 3), and other neurological diseases (n = 3; multiple sclerosis, stroke, and brain metastasis). Of the remaining 149 subjects, 11 subjects were hospitalized more than once and 5 had missing data leaving 133 subjects in the study population.
A control group of 34 stable subjects with COPD and another control group of 34 subjects without COPD were recruited among outpatients who were admitted to the physical medicine and rehabilitation department for other causes. They were matched to the subjects with COPD exacerbation by age, gender, and educational status. Patients with Alzheimer’s disease, cerebrovascular disease, hypothyroidism, obstructive sleep apnea, and who were on medications affecting the cognitive status were not included.
Measures
Demographic features, systemic comorbidities (cardiovascular disease, hypertension, diabetes mellitus, cancer, neuropsychological, gastrointestinal, and musculoskeletal disorders), medications used, and history of fall in the previous year were recorded. Smoking status was defined as past, current, or never smokers. Spirometric stage of COPD, number of respiratory infections that required antibiotics and steroids in the previous year, and history of mechanical ventilation and long-term oxygen therapy (LTOT) were recorded. Body mass index (BMI) was calculated (kg/m2).
Spirometric lung function assessments (Sensormedics Vmax Series 20C Respiratory Analyzer, Sensormedics Corporation, Yorba Linda, California, USA) of COPD subjects were performed according to American Thoracic Society’s guidelines. Forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC), and FEV1/FVC ratios were recorded. Arterial blood gases were measured at rest (Radiometer ABL 735 blood gas analyzer, Denmark). Arterial oxygen pressure, arterial carbondioxide pressure, arterial oxygen saturation, and pH values were included in the analysis. Complete blood count and biochemistry tests, including hemoglobin and albumin levels were analyzed.
Turkish version of MMSE was used to assess cognitive function in several domains 15 ; orientation to time and place; immediate and delayed recall; attention calculation; language consisting of naming, verbal fluency, comprehension and ability to follow instructions, reading, and writing; and constructional ability. Maximum total score is 30, and scores less than 24 indicate cognitive dysfunction. Depression and anxiety levels were measured using Hospital Anxiety and Depression scale. 16 Reliability and validity study of Turkish version of Hospital Anxiety and Depression scale defined cutoff scores for anxiety and depression subscales as 10 and 7, respectively. 17
Six minute walk test was performed in COPD subjects in a 30-meter corridor. Distance walked in 6 minutes (6MWD) was recorded. The BODE index, a composite measure, consisted of the following components 18 : B: BMI, O: the severity of airflow obstruction (FEV1), D: severity of dyspnea (modified Medical Research Council Dyspnea Scale), and E: exercise capacity (6MWD). Each component is scored separately before adding up to total BODE score, which ranges from 0 to 10. Higher scores indicate greater disease severity.
Statistical analysis
Statistical analysis was performed using SPSS for Windows 15.0 (Worldwide Headquarters SPSS Inc.). Shapiro–Wilk test results and graphical inspection of the data were used to assess the normality of the variables. One-way analysis of variance (ANOVA) with Scheffé post hoc test was used to compare the normally distributed continuous variables among more than two groups. Levene statistic was used to test whether the homogeneity of variances of the variables was violated. Welch ANOVA with Games-Howell post hoc test was used, for which the homogeneity of variances of the variables was violated. Kruskal–Wallis test with Mann–Whitney U test as a post hoc test was used to compare the non-normally distributed continuous variables among more than two groups. Student’s t-test was used to compare the normally distributed continuous variable, where Mann–Whitney U test was used to compare the non-normally distributed continuous variables among two COPD groups. For categorical variables, Pearson χ 2 test, Fisher–Freeman–Halton exact test, and Fisher’s exact test were carried out where appropriate. A statistically significant difference was defined as p < 0.05.
Data are presented as mean ± (standard deviation (SD) or median (Q1–Q3)) and percentage in the tables.
Results
Demographic, clinical, and laboratory features are reported in Table 1. Age, gender, educational status, BMI, and comorbidities were similar among groups. Duration of COPD, number of LTOT users, and smokers were not different between COPD subjects with and without exacerbation. Disease severity is higher in the COPD exacerbators as defined by FEV1 and history of infections and hospitalizations. Exacerbators had shorter 6MWD and higher BODE scores. Albumin levels were the lowest in COPD subjects with exacerbation.
Demographic, clinical, and laboratory features of the subjects.
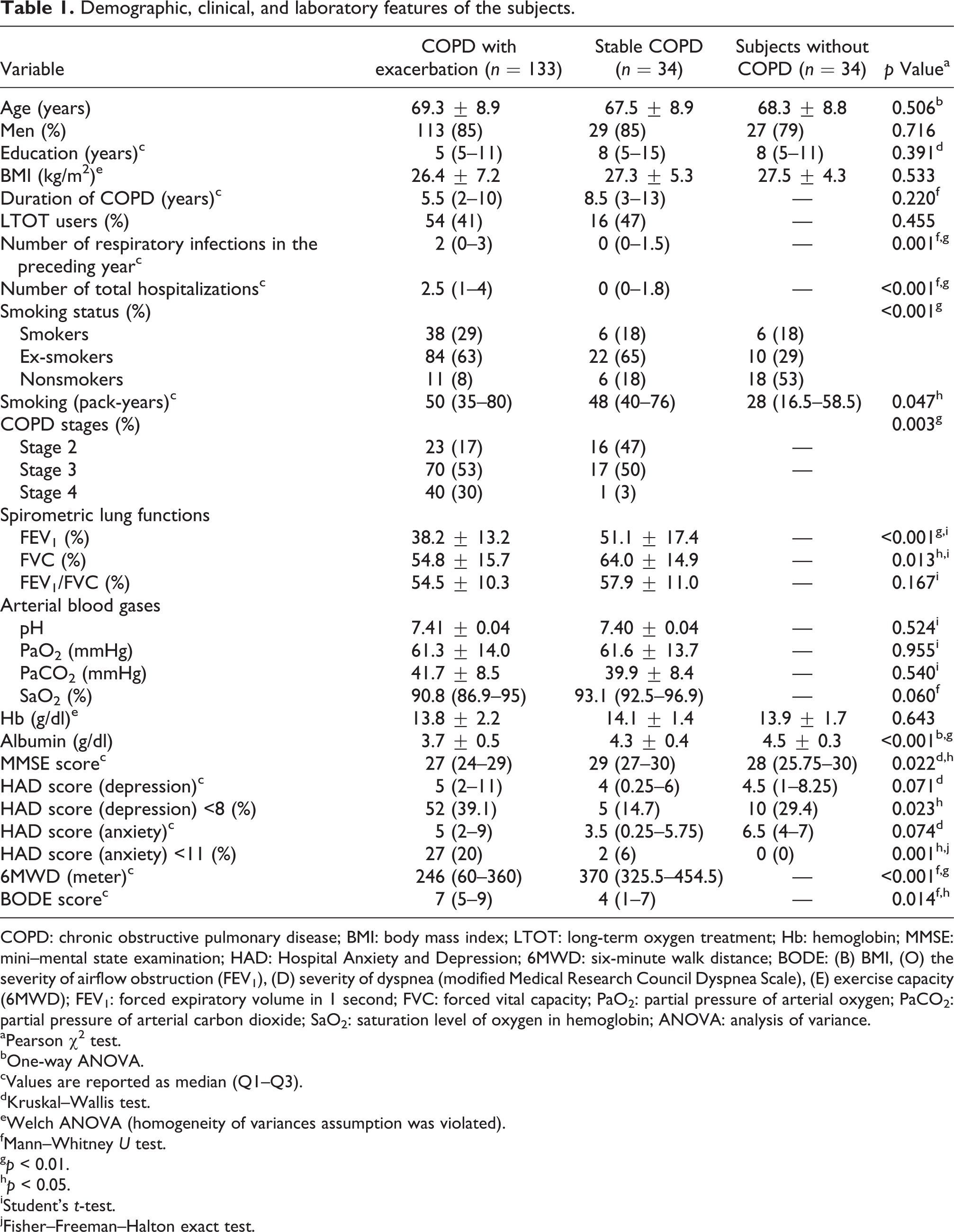
COPD: chronic obstructive pulmonary disease; BMI: body mass index; LTOT: long-term oxygen treatment; Hb: hemoglobin; MMSE: mini–mental state examination; HAD: Hospital Anxiety and Depression; 6MWD: six-minute walk distance; BODE: (B) BMI, (O) the severity of airflow obstruction (FEV1), (D) severity of dyspnea (modified Medical Research Council Dyspnea Scale), (E) exercise capacity (6MWD); FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; PaO2: partial pressure of arterial oxygen; PaCO2: partial pressure of arterial carbon dioxide; SaO2: saturation level of oxygen in hemoglobin; ANOVA: analysis of variance.
aPearson χ 2 test.
bOne-way ANOVA.
cValues are reported as median (Q1–Q3).
dKruskal–Wallis test.
eWelch ANOVA (homogeneity of variances assumption was violated).
fMann–Whitney U test.
g p < 0.01.
h p < 0.05.
iStudent’s t-test.
jFisher–Freeman–Halton exact test.
Mean length of hospital stay for exacerbation was 13.6 ± 7.8 days. MMSE was administered to subjects with exacerbation 6.7 ± 2.7 days after admission to hospital. Subjects with COPD exacerbation had the lowest MMSE score among all groups. There was no significant difference in MMSE scores between stable COPD subjects and non-COPD subjects. When MMSE domains were analyzed separately, impairments in the following domains were higher in COPD subjects with exacerbation: orientation to time, attention calculation, reading, writing, and constructional ability (Table 2).
Differences in the percentage of impairment in each of the MMSE domains between groups.
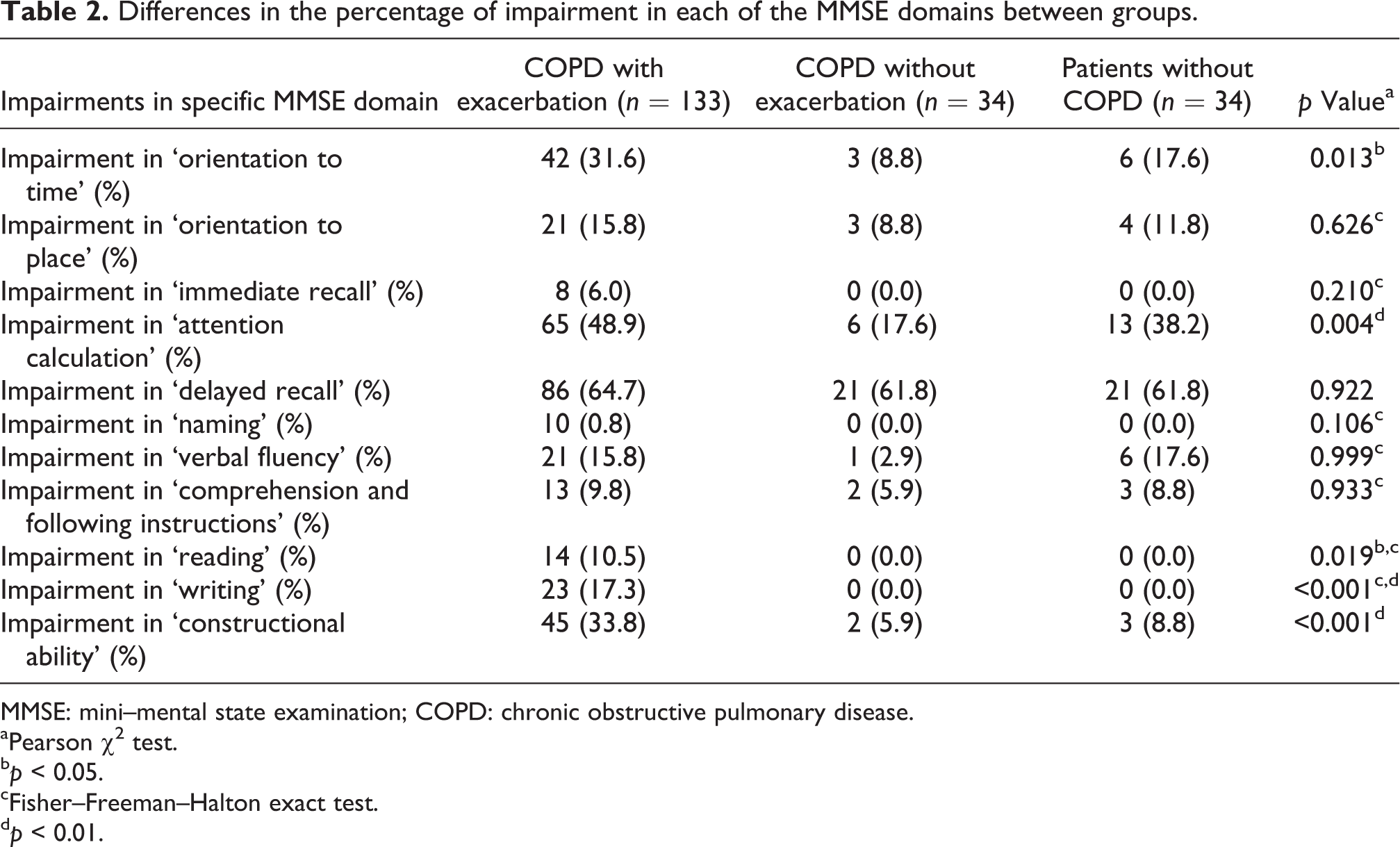
MMSE: mini–mental state examination; COPD: chronic obstructive pulmonary disease.
aPearson χ 2 test.
b p < 0.05.
cFisher–Freeman–Halton exact test.
d p < 0.01.
Thirty (22.6%) COPD subjects with exacerbation had MMSE lower than 24. Subjects with lower cognitive scores were older and less educated (Table 3). Although arterial blood gases and clinical parameters were similar between groups, cognitively impaired subjects had shorter 6MWD, higher BODE scores, and higher prevalence of fall history. The prevalence of depression was high in the subjects with lower MMSE scores.
Comparison of demographic and clinical features in the COPD patients with exacerbation divided according to a cutoff of 24 points on MMSE.
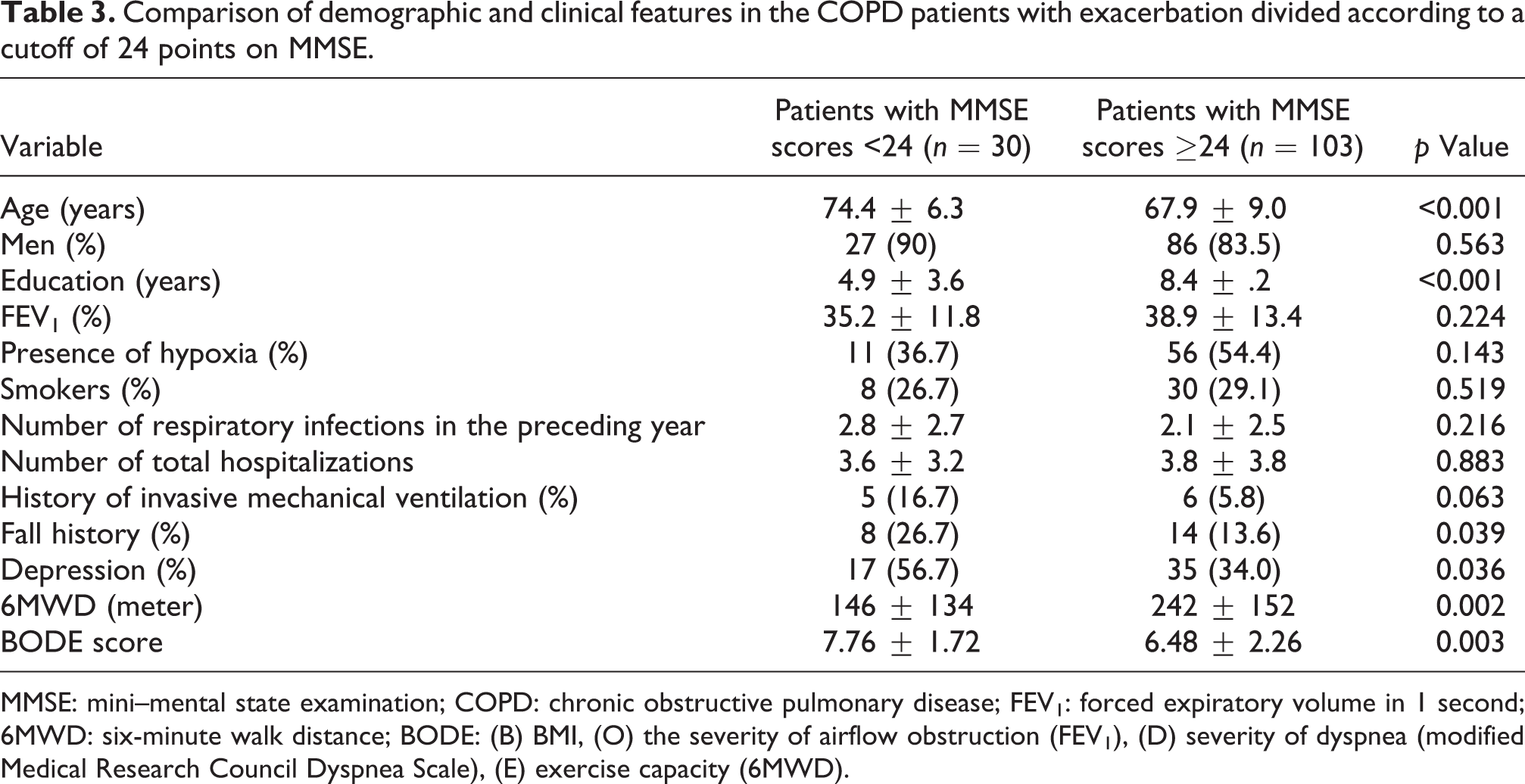
MMSE: mini–mental state examination; COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in 1 second; 6MWD: six-minute walk distance; BODE: (B) BMI, (O) the severity of airflow obstruction (FEV1), (D) severity of dyspnea (modified Medical Research Council Dyspnea Scale), (E) exercise capacity (6MWD).
Table 4 demonstrates comparison of the same parameters after the implementation of age and educational level-adjusted MMSE scores using population-based normative values. 19 Thereafter, 6MWD, BODE scores, prevalence of fall history, and depression were similar between exacerbated COPD subjects with and without cognitive impairment.
Comparison of demographic and clinical features in the COPD patients with exacerbation divided according to population-based norms on MMSE score18.

MMSE: mini–mental state examination, COPD: chronic obstructive pulmonary disease, FEV1: forced expiratory volume in 1 second, 6 MWD: six-minute walk distance, BODE: (B) BMI, (O) the severity of airflow obstruction (FEV1), (D) severity of dyspnea (modified Medical Research Council Dyspnea Scale), (E) exercise capacity (6MWD).
aCutoff value of each subject is determined according to their age and education.
bPearson χ 2 test.
cValues are reported as median (Q1–Q3).
dFisher’s exact test.
Discussion
Summary of main findings
The COPD subjects hospitalized for exacerbation demonstrated lower MMSE scores than stable COPD and non-COPD subjects in this study. Spirometric results, hypoxemia, and the number of infectious exacerbations in the previous year did not seem to predict cognitive dysfunction during exacerbation in the present study. COPD subjects with exacerbation also had lower functional capacity. No association was found between cognitive impairment and functional capacity during exacerbation.
Prevalence of cognitive impairment in COPD
Antonelli-Incalzi et al. found that 35.5% of stable COPD subjects had cognitive impairment using MMSE. 3 This ratio is higher than what we observed in stable COPD subjects in our study (8.8%). Hung et al. reported that non-severe COPD subjects had comparable cognition scores with those without COPD, but severe COPD subjects had lower cognitive performance. 8 Also, Meek et al. showed that MMSE scores were in normal limits in COPD patients. 20 Our study also showed that MMSE scores of stable COPD subjects were similar to those of non-COPD subjects. However, these results might be explained by the limited ability of MMSE in distinguishing the subtle changes in cognitive functions that may be observed in stable COPD. Using neuropsychological tests, Dodd et al. found that the frequency of mild to moderate cognitive impairment was greater in subjects with stable COPD than the control subjects. 13
MMSE was used to screen the cognitive function during exacerbation period. 10,11 Detailed neuropsychological tests might be time consuming and difficult to perform in the hospitalized COPD subjects although they were performed successfully in a recent study by Dodd et al. MMSE still stands as a good alternative for screening.
The prevalence of cognitive impairment in patients with exacerbation was 22.6% in our study when a cutoff point of 24 was used to compare our results with other studies in the literature. There are few studies addressing the cognitive function during exacerbations. 10 –13 Ambrosino et al. found that 43% of subjects requiring mechanical ventilation during exacerbations had cognitive impairment. 10 The age and gender were comparable with that of ours, but mean MMSE scores were lower (24.1 ± 3.7). In another study, elderly COPD subjects hospitalized for nonacidotic exacerbation had mean MMSE scores (20.7 ± 6.8) lower than those observed in our study. 11 They were older (81.7 years), but airway obstruction (FEV1 = 67.5%) was less severe than that of our population. They aimed to investigate predictors of mortality, and there was no control group to compare cognitive status. Due to the absence of control group, the effect of advanced age on cognitive deterioration cannot be excluded. Dodd et al. observed similar MMSE scores (27 ± 4) to our results in a smaller group of COPD subjects with exacerbation. 13 In both studies, no relationship was found among lung functions and lower MMSE scores in accordance with our findings. 10,13
Ambrosino et al. concluded that exacerbations had an influence on cognitive deterioration. 10 On the other hand, Dodd et al. reported that it is uncertain that the cognitive deficit is a result of acute exacerbation or chronic differences in characteristics of both patient groups since cognitive dysfunction did not improve with recovery over 3 months. 13
Risk factors for cognitive impairment
Many mechanisms are proposed to elucidate the cognitive decline in COPD. In the present study, older age and fewer education years were predicting factors for cognitive decline during exacerbation. However, when population-based norms were used to determine cognitive impairment, age and education years did not predict cognitive decline. Lung function did not explain the cognitive impairment similar to a number of studies showing that cognitive decline was not correlated with FEV1. 4 –6 These studies included less severe COPD subjects who were tested by MMSE for cognitive assessment. When subjects with advanced COPD were involved and detailed neuropsychological tests were performed, cognitive dysfunction was associated with COPD severity. 21
The effects of hypoxemia and LTOT on cognition are not clearly proved. Although some studies included only stable COPD patients with hypoxemia 3 –6 and suggest that hypoxemia might be associated with cognitive impairment, 3 –5 others suggest that hypoxemia alone is not responsible for cognitive dysfunction. 1,6,13 Thakur et al. reported that LTOT decreases the risk of cognitive impairment, 4 while Pretto and McDonald observed that acute reversal of hypoxemia did not improve cognitive function in stable hypoxemic COPD patients. 7 We could not find any association between MMSE scores and hypoxemia during exacerbation either.
Effects of smoking on cognition are controversial. Smoking is found to improve cognitive performance. 22 It is suggested to enhance attention, memory, and executive function through stimulation of neurotransmitter release. In contrast, smoking is shown to speed yearly cognitive decline in the elderly. 23 Higher pack-year smoking was associated with higher rate of decline. Smoking is supposed to interfere with cognition through increasing cerebral atherosclerosis by direct neurotoxic effect or inducing cerebral hypoxia with impairment of lung function. 1 However, we did not find any association between smoking and cognitive impairment in COPD subjects during exacerbation.
In our study, cognitively impaired exacerbators had shorter 6MWD and higher prevalence of fall history, which did not persist after age and educational level adjustments were applied. Older and less educated subjects tended to have both lower cognitive performance and lower functional capacity. The results did not support our hypothesis that cognitive performance might be associated with functional capacity. Although patients in relatively good health are expected to have both higher levels of physical activity and cognitive performance, there might be some relationship between physical activity and cognitive function. Lautenschlager et al. report that higher cognitive performance motivates participation in exercise activity, and on the other side, physical activity improves cognitive function in subjects with memory impairment. 24 Physical activity is thought to stimulate synaptogenesis and cerebral blood perfusion. Activity-associated environment enrichment also may affect neuronal plasticity. This effect of physical activity is also shown to be reflected to COPD subjects through pulmonary rehabilitation. 1,25 As far as we know, this is the first study investigating the association between cognitive impairment and functional capacity in COPD subjects during exacerbation.
Systemic comorbidities like hypertension and diabetes mellitus are prevalent in COPD subjects, and these vascular diseases also have impact on cognitive impairment. 14 However, in a number of studies, 4 the specific role of cardiovascular comorbidities could not be established, and in the others, subjects with cardiovascular disease were excluded from the study. 6,9 Shared risk factors for both COPD and cardiovascular disease, like smoking, are thought to mediate the effects on cognition. Interaction of depressive symptoms with cognitive function should also be considered. Depression did not explain development of cognitive impairment in some studies, 5,6,11 while it was not even evaluated in others. 10,12,13 In our study, depression did not explain cognitive decline in COPD exacerbation.
Specific areas of cognitive deficit in COPD include attention, memory, motor, and executive function. 1,21 Defective constructional abilities and secondary verbal memory were found to be causes for physical dependence in hypoxemic COPD subjects. We also found that attention, writing, reading, and constructional ability domains were the most severely impaired in the subjects with exacerbation.
Strengths and limitations of the study
Although large sample size and assessment of cognitive status and functional capacity in subjects with COPD exacerbation are the strengths of our study, some limitations exist. The present findings may not be completely valid for stable COPD subjects since the number of stable COPD subjects is few. Subjects with stable COPD are not perfectly matched with exacerbators in terms of spirometric severity. Another limitation is the cross-sectional design. MMSE of the same subjects in a longitudinal time interval would demonstrate the impact of exacerbation more precisely. MMSE is not an appropriate tool to diagnose subtle cognitive impairment; however, we believe that during the exacerbation period, shorter time required to perform the test increases the accuracy of test results.
Theoretically it is possible that cognitively impaired subjects are at higher risk for exacerbations. We could not exclude the proposition that lower baseline cognitive status might influence compliance with medical and oxygen therapy increasing the risk of exacerbation, as exacerbators have higher rate of respiratory infections and hospitalizations.
Implications for future research and clinical practice
Cognitive impairment is observed in COPD subjects with exacerbation. Older age and educational level did not predict cognitive impairment when population-based norms were used. There was no association between functional capacity and cognitive impairment. Impairments in orientation to time, reading, writing, attention, and constructional ability were higher in the COPD subjects with exacerbation. Cognitive deficit areas may be taken into account while selecting the educational materials and methods. Appropriate compensatory and supportive approaches, like more active caregiver participation, and more visual and auditory education materials may also be considered.
Footnotes
Authors’ notes
OOT takes the responsibility for the integrity of the work as a whole; contributed to study design, induction and treatment of subjects into the study, acquisition, analysis, and interpretation of the data; and responsible for the drafting, review, and final approval of the manuscript. SOB contributed to the induction of subjects into the study, acquisition, analysis and interpretation of the data, review, and approval of the manuscript. NK contributed to the induction and treatment of subjects into the study, review, and approval of the manuscript. GKK contributed to the analysis and interpretation of the data, review, and approval of the manuscript. The abstract of the research article was previously submitted for presentation in the 23rd National Physical Medicine and Rehabilitation Congress held at Antalya, Turkey from May 11 to 15, 2011.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
