Abstract
Recently, clear benefits have been shown from long-term noninvasive ventilation (NIV) in stable chronic obstructive pulmonary disease (COPD) patients with chronic hypercapnic respiratory failure. In our opinion, these benefits are confirmed and nocturnal NIV using sufficiently high inspiratory pressures should be considered in COPD patients with chronic hypercapnic respiratory failure in stable disease, preferably combined with pulmonary rehabilitation. In contrast, clear benefits from (continuing) NIV at home after an exacerbation in patients who remain hypercapnic have not been shown. In this review we will discuss the results of five trials investigating the use of home nocturnal NIV in patients with prolonged hypercapnia after a COPD exacerbation with acute hypercapnic respiratory failure. Although some uncontrolled trials might have shown some benefits of this therapy, the largest randomized controlled trial did not show benefits in terms of hospital readmission or death. However, further studies are necessary to select the patients that optimally benefit, select the right moment to initiate home NIV, select the optimal ventilatory settings, and to choose optimal follow up programmes. Furthermore, there is insufficient knowledge about the optimal ventilatory settings in the post-exacerbation period. Finally, we are not well informed about exact reasons for readmission in patients on NIV, the course of the exacerbation and the treatment instituted. A careful follow up might probably be necessary to prevent deterioration on NIV early.
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic disease with a high mortality and morbidity worldwide. According to the World Health Organization, it is the only chronic disease with increasing mortality (see http://www.who.int/respiratory/copd/burden). Patients with end-stage COPD frequently develop chronic respiratory failure (CRF) which is associated with end of life. In that stage of disease, treatment options are limited as until now, only long-term oxygen therapy has been proven to prolong life in these patients [Nocturnal Oxygen Therapy Trial Group, 1980]. However, once patients develop advanced hypercapnic respiratory failure, giving oxygen may not be sufficient, and support of ventilation might be necessary.
While there is no doubt that applying long-term nocturnal noninvasive ventilation (NIV) improves outcomes in patients with restrictive chest wall diseases and neuromuscular diseases [Simonds and Elliott, 1995; Leger et al. 1994], evidence for long-term application in COPD patients has been lacking for a long time. However, in the past 7 years, with a change in ventilatory strategy, clear benefits have been achieved also in COPD patients with CRF, though in stable disease [Windisch et al. 2002, 2005; Dreher et al. 2010, 2011]. Interestingly, until now, it has not been proven that nocturnal NIV would also improve the, for many patients, rather miserable situation after an exacerbation. In this review we will first briefly summarize the results of studies in stable hypercapnic COPD patients, while thereafter we will discuss the results of post-exacerbation studies more extensively.
Nocturnal NIV in COPD patients with stable chronic hypercapnic respiratory failure
For 20 years now, studies have been investigating NIV in stable COPD patients with CRF [Casanova et al. 2000; Clini et al. 2002; Strumpf et al. 1991; Gay et al. 1996; Lin, 1996; McEvoy et al. 2009; Meecham Jones et al. 1995]. Most initial randomized controlled trials (RCTs) did not show clear benefits [Casanova et al. 2000; Clini et al. 2002; Strumpf et al. 1991; Gay et al. 1996; Lin, 1996; McEvoy et al. 2009]. Last year, an updated meta-analysis of individual patient data was published comparing NIV with conventional management in patients with COPD and stable CRF, showing that NIV in patients with stable COPD does not improve lung function, gas exchange, sleep efficiency or 6-minute walking distance [Struik et al. 2014a]. However, inspiratory pressures above 18 cm H2O, a better compliance to the therapy, and a higher baseline arterial carbon dioxide levels (PaCO2), were associated with significantly more reduction in PaCO2 with therapy [Struik et al. 2014a].
The hypothesis that probably higher pressures are needed to improve gas exchange and thus to expect also clinically meaningful outcomes has been investigated by Windisch and coworkers [Windisch et al. 2002, 2005; Dreher et al. 2010, 2011]. Although the studies did not contain a control group getting standard care, they indeed showed that with higher pressures and a more controlled form of NIV aimed at a maximal reduction in PaCO2 (so called high-intensity NIV), gas exchange but also lung function and health-related quality of life (HRQoL) improved more, and that this high-intensity NIV was well tolerated [Windisch et al. 2002, 2005; Dreher et al. 2010, 2011]. These promising results have now been confirmed in long-term RCTs [Duiverman et al. 2008, 2011; Köhnlein et al. 2014]. In addition, it has been shown that high-intensity NIV improves the outcomes of pulmonary rehabilitation in severe stable hypercapnic COPD, in terms of improvement in gas exchange, pulmonary function, exercise tolerance and HRQoL [Duiverman et al. 2008, 2011]. In 2014, the study of Köhnlein and colleagues showed, next to benefits in gas exchange, HRQoL and lung function, also a substantial survival benefit [Köhnlein et al. 2014].
With regards to the prevention of exacerbations, the only RCT showing any benefit of NIV in stable COPD patients was the Italian multicentre study of Clini and colleagues [Clini et al. 2002]. The total hospital admission rate decreased by 45% in the NIV group, and increased by 27% in the control group compared with the period before the start of the study. Furthermore, intensive care unit admissions decreased more in the NIV group (by 75%) than in the control group (by 20%). However, as this was not their primary outcome, these results were only hypothesis generating. Other studies in stable COPD patients could not reproduce these results, also because hospital admission did occur very infrequently [Duiverman et al. 2011; Köhnlein et al. 2014].
To conclude, although former studies and the most recent meta-analysis have shown that nocturnal NIV in stable hypercapnic COPD patients does not improve outcomes, the more recent trials using high-intensity NIV have shown important and relevant outcomes. In our opinion, these benefits are confirmed and nocturnal NIV should be considered in COPD patients with chronic hypercapnic respiratory failure in stable disease, preferably combined with pulmonary rehabilitation.
Nocturnal NIV in COPD patients with prolonged hypercapnia after acute respiratory failure due to a COPD exacerbation
During the natural history of COPD, exacerbations are an important contributor to morbidity and mortality [Global Initiative for Chronic Obstructive Lung Disease, 2015; Vestbo, 2004]. Unfortunately, patients often do not recover to baseline levels of lung function and symptoms and therefore COPD exacerbations are an important contributor to lung function decline, resulting in reduced physical activity, poorer quality of life and increased risk of death [Donaldson et al. 2002, 2005; Seemingal et al. 1998; Soler-Catalu et al. 2005].
The best predictor of getting an exacerbation is the occurrence of prior exacerbations, indicating that there is a group of patients susceptible to exacerbations, irrespective of lung function [Hurst et al. 2010]. Despite this, in patients with severe airflow obstruction, there are more ‘frequent exacerbators’ and exacerbations are often more severe, as indicated by more hospital admissions [Hurst et al. 2010].
NIV has become an established treatment in acute hypercapnic respiratory failure (AHRF) in patients with a COPD exacerbation [Ram et al. 2004]. However, next to a high in-hospital mortality [Ram et al. 2004; Stefan et al. 2015], after discharge 60–80% of the patients are re-admitted within 1 year and 30–49% die within this first year after their hospital admission for AHRF [Chu et al. 2004; Echave-Sustaeta et al. 2010]. These disappointing outcomes raised the question of whether providing long-term NIV to patients who recover from their exacerbation but remain hypercapnic might improve outcomes.
Despite the fact that evidence has been lacking, in several countries patients suffering from frequent exacerbations with AHRF receive domiciliary NIV. This different attitude towards the initiation of domiciliary ventilation after an exacerbation might also explain the huge differences in prevalence of domiciliary ventilation in COPD which has been found between the different countries [Lloyd-Owen et al. 2005]. The initiation of domiciliary long-term NIV has consequently also been investigated in studies.
In 2003, Tuggey and colleagues performed a retrospective economic analysis of domiciliary NIV in a highly selected group of 13 patients having frequent exacerbations, treated in their clinic [Tuggey et al. 2003]. They showed that there was a significant reduction in the rate of admission (from a mean of five admissions in the year before NIV to a mean of two admissions in the year after NIV was initiated), and the total length of hospital stay per patient (17 days
A similar situation was investigated in the USA, where chart reviews were conducted and compared in patients who were discharged with NIV
The third study to be discussed is from Funk and colleagues. They had to deal with the same situation, as patients were in their centre routinely evaluated for long-term NIV if they remained hypercapnic after an episode of AHRF requiring mechanical ventilation. For study purposes, these patients were randomised to continue or withdraw NIV after 6 months standard home NIV implemented after their exacerbation [Funk et al. 2011]. The main problem interpreting the results of this study is that the main part of primary end point, that is, the probability of clinical worsening defined as resumption of NIV due to severe dyspnoea or progressive hypercapnia, could by definition only be fulfilled in the withdrawal group. Looking more precisely into their results, it becomes clear that the part of the primary end point that could be fulfilled in both groups, ICU admission, was not different, occurring in two patients in the ventilation group
Two RCTs have been conducted including a control group not receiving home NIV after an acute exacerbation with AHRF [Cheung et al. 2010; Struik et al. 2014b]. A RCT with ‘sham ventilation’ as control [continuous positive airway pressure (CPAP) 5 cm H2O] was published by Cheung and colleagues [Cheung et al. 2010]. They showed that when home NIV was provided after an exacerbation with AHRF, the proportion of patients developing recurrent AHRF was 38.5%
A recently published large multicentre RCT by Struik and colleagues could not demonstrate an improvement in time to readmission or death by adding NIV for 1 year in patients with prolonged hypercapnia after an episode of NIV for AHRF despite the use of higher inspiratory pressures indeed leading to more improvement in gas exchange, and the frequent addition of pulmonary rehabilitation post-exacerbation [Struik et al. 2014b]. One year after discharge, 56%
Summarizing these publications there are several important items to be discussed. An important issue when comparing results of these different trials is that different end points were used. In the study of Struik and colleagues the primary end point (readmission or death) was reached quite often also in the NIV group (65% in the NIV group
Second, it seems that the studies, although they have all included COPD patients after an exacerbation, included patients in different disease stages, with or without comorbidity or pre-existent CRF. This might have influenced outcomes. In the retrospective analysis of Galli and colleagues, the standard treatment group performed worse (75% re-admitted or death in 6 months) compared with the other studies [Galli et al. 2014]. In this study comorbidities, also cardiac comorbidities, were very frequent while in the other studies these patients were excluded [Struik et al. 2014b] or rarely there [Funk et al. 2011; Cheung et al. 2010]. Although this might have influenced outcomes negatively in the control group in the study of Galli and colleagues, it probably represents real life best. On the other hand, patients who exacerbate with AHRF but do not suffer from CRF, probably do not need chronic nocturnal NIV as we know that some of these patients return to normocapnia after recovery [Costello et al. 1997]. The more stable patients from Funk and colleagues, with proven CRF, had a better prognosis when NIV was initiated [Funk et al. 2011]. On the other hand, in the study of Struik and colleagues, 26% of the patients in the standard treatment group became spontaneously eucapnic after 3 months, thus probably representing a part of the patients that do not need long-term NIV at all [Struik et al. 2014b]. However, one should consider that if the authors would have been able to preselect the chronic hypercapnic patients, as was done in the study of Funk and colleagues, this might have resulted in a higher event rate in especially the standard treatment arm. Moreover, the NIV group might have performed better as the CRF would be counterbalanced by the NIV. What was shown in the study of Struik and colleagues [Struik et al. 2014b], however, was a comparable deterioration rate in the control group compared with the studies of Funk and colleagues and Cheung and coworkers [Funk et al. 2011; Cheung et al. 2010], but a higher event rate in the NIV group. The latter cannot be explained by this patient selection issue. Overall, it has been hypothesized that the indication for NIV post-exacerbation should be set later as this brings the opportunity to select the patients who suffer from acute CRF. However, this issue of timing remains challenging, as we should select the patients that are suffering from CRF, have deteriorated this time and will deteriorate in the future because of acute CRF, while we do not want to wait too long as event rates are high and events usually also occur during the first 100 days after the exacerbation [Galli et al. 2014; Funk et al. 2011; Cheung et al. 2010]. Future studies should try to incorporate these points by (a) selecting patient with more severe remaining hypercapnia and (b) lengthening the time frame (an overview of the previous RCTs regarding these aspects is given in Table 2).
Studies investigating NIV in COPD patients after an acute exacerbation with AHRF.
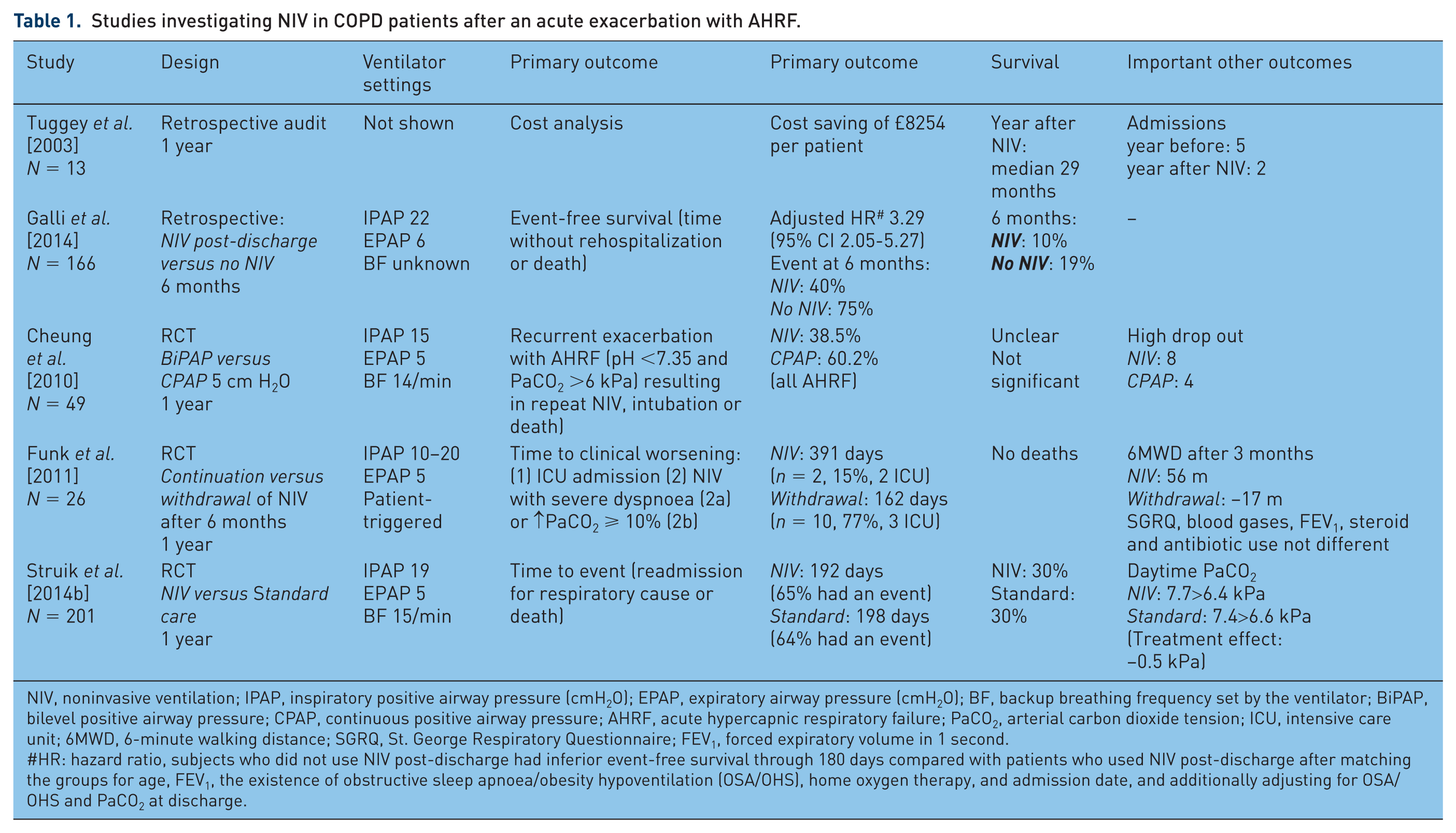
NIV, noninvasive ventilation; IPAP, inspiratory positive airway pressure (cmH2O); EPAP, expiratory airway pressure (cmH2O); BF, backup breathing frequency set by the ventilator; BiPAP, bilevel positive airway pressure; CPAP, continuous positive airway pressure; AHRF, acute hypercapnic respiratory failure; PaCO2, arterial carbon dioxide tension; ICU, intensive care unit; 6MWD, 6-minute walking distance; SGRQ, St. George Respiratory Questionnaire; FEV1, forced expiratory volume in 1 second.
#HR: hazard ratio, subjects who did not use NIV post-discharge had inferior event-free survival through 180 days compared with patients who used NIV post-discharge after matching the groups for age, FEV1, the existence of obstructive sleep apnoea/obesity hypoventilation (OSA/OHS), home oxygen therapy, and admission date, and additionally adjusting for OSA/OHS and PaCO2 at discharge.
Timing of initiation of NIV after an exacerbation in completed and ongoing randomized controlled trials.

NIV, noninvasive ventilation; AHRF, acute hypercapnic respiratory failure; PaCO2, arterial carbon dioxide tension.
Status of the trials not published yet retrieved from ClinicalTrials.gov, and through contact with study coordinators.
Finally, differences in outcomes and reported benefits of NIV could theoretically be a matter of differences in ventilator settings providing insufficient ventilatory support. From the studies performed in CRF we have learnt that higher inspiratory pressures are necessary to improve gas exchange. However, we are insufficiently informed about probable disadvantages of this high-intensity NIV [Lukácsovits et al. 2012]. In the study of Struik and colleagues the highest pressures were used indeed leading to improved gas exchange while clear benefits in terms of survival or readmission rates did not result [Struik et al. 2014b]. However, it should also be noted that in the NIV group, there were patients that remained severely hypercapnic, which might indicate that the improvement in ventilation with these settings was still suboptimal [Struik et al. 2014b].
In conclusion, there is at the moment insufficient evidence to support the use of home nocturnal NIV in patients with prolonged hypercapnia after a COPD exacerbation with AHRF. However, some uncontrolled trials might have shown some benefits in patients that remain hypercapnic (long enough) after an acute exacerbation with AHRF. The challenge will be to select, preferably beforehand, the patients that will benefit most. As this is not always possible in clinical practice, studies mainly focus on adequate timing post-exacerbation. The data of a large British RCT, a French RCT and a Danish RCT are expected which include patients who remained hypercapnic after different time periods (Table 2). Furthermore, there is insufficient knowledge about the optimal ventilatory settings in the post-exacerbation period. Finally, we are not well informed about exact reasons for readmission in patients on NIV, the course of the exacerbation and the treatment instituted. A careful follow up might be necessary to prevent early deterioration on NIV [Funk et al. 2011].
Overall summary
Nocturnal NIV is an evidence-based therapy for the treatment of CRF in stable COPD patients improving survival and HRQoL (evidence level B). In patients who remain hypercapnic after an acute COPD exacerbation with ARF, there is at the moment insufficient evidence to support the continued use of home nocturnal NIV. However, further studies are necessary to select the patients that optimally benefit, select the right moment to initiate home NIV, select the optimal ventilatory settings and to choose optimal follow-up programmes.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.D. is the recipient of an European Respiratory Society Long-term Fellowship (LTRF 2014-3126) W.W. received personal speaking fees from companies dealing with mechanical ventilation. In addition, the Merheim Hospital in Cologne received an open research grant for two years from Weinmann/Germany and Vivisol/Germany. J.S. received speaking fees and travel funding from companies which are involved in mechanical ventilation and monitoring of gas exchange. The study group of J.S. received open research grants from Breas Medical, Heinen und Löwenstein, Respironics, ResMed Germany, Weinmann, Vitalaire and Vivisol. P.W. has recieved a research grant and personal speaking fees from Philips/Respironics, The Netherlands.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
