Abstract
Bronchiectasis is a chronic inflammatory lung disease, which has similarities to chronic obstructive pulmonary disease (COPD). Comorbidities of COPD include increased risk of cardiovascular (CV) disease, loss of bone mineral density (BMD) and loss of skeletal muscle mass and function, all linked to systemic inflammation. The potential for such comorbidities has not been explored in bronchiectasis. We hypothesised that patients with bronchiectasis would have similar increased comorbidities. A total of 20 patients with noncystic fibrosis bronchiectasis were compared to 20 controls similar in age, gender and smoking exposure. Assessments included aortic pulse wave velocity (PWV; (a measure of arterial stiffness and an independent predictor of CV risk), blood pressure (BP) as well as levels of interleukin-6 (IL-6), albumin, fasting glucose and lipids. Body composition (fat free mass index (FFMI)), BMD, the 6-min walk distance (6MWD) and self-reported physical activity were also determined. Patients with bronchiectasis had increased aortic PWV, 10.5 (3.0) m/second, when compared with controls, 8.8 (1.6) m/second (p < 0.05), despite similar central and peripheral BP and lipid profile. Patients also had increased IL-6 and reduced albumin and glucose. Although mean body mass index, FFMI and BMD were similar in patients and controls, only 20% of patients had a healthy BMD compared with 50% of controls. Patients had reduced 6MWD and reported less physical activity (p < 0.05). Patients with bronchiectasis had increased arterial stiffness (an indicator of increased CV risk), increased inflammation, reduced exercise capacity and bone thinning. These additional comorbidities require further evaluation for their management in these patients.
Introduction
Bronchiectasis is a chronic lung disease, defined by permanent dilatation and destruction of bronchi and bronchioles. 1 In common with chronic obstructive pulmonary disease (COPD), alpha-1-antitrypsin and cystic fibrosis (CF), the pathology has been linked to repeated infection and a chronic inflammatory state, 2 and patients with bronchiectasis commonly present with airflow obstruction. 3 Patients with COPD have a number of comorbidities that include an increased risk of cardiovascular (CV) disease, altered body composition and loss of bone mineral density (BMD). 4,5 In patients with COPD, increased aortic pulse wave velocity (PWV), a measure of arterial stiffness and an independent predictor of CV events, is increased and has been related to airways obstruction and systemic inflammation. 6 More recently, in patients with alpha-1-antitrypsin deficiency, increased aortic PWV and loss of BMD have been found. 7 Also in patients with CF, a genetic disorder that leads to diffuse bronchiectasis in younger individuals, there is evidence of arterial stiffening 8 and loss of BMD and skeletal muscle mass. 9
Patients with chronic respiratory disease often present with a number of risk factors for the development of these conditions, such as impaired lung function and oxygen saturations, chronic inflammation and reduced physical activity. However, bronchiectasis is not associated with exposure to cigarette smoke, unlike COPD. There is evidence of hypertension and CV disease in 19% and 20% of patients with bronchiectasis, respectively, 10 and cardiac dysfunction has also been reported. 11 However, there have been no studies of arterial stiffness, body composition or BMD in this population.
Therefore, the aim of this study was to identify comorbidities in patients with bronchiectasis and relationships between arterial stiffness and CV risk factors such as impaired pulmonary function, systemic inflammation and reduced exercise capacity. It was hypothesised that patients would have increased aortic PWV, reduced exercise capacity, skeletal muscle mass and BMD compared with controls, free from respiratory disease, similar in age and sex.
Methods
Subjects
Patients with non-CF bronchiectasis were recruited from respiratory outpatients and compared with control subjects, similar in age, sex and smoking status. Bronchiectasis was confirmed on high-resolution computed tomography and showed that 16 patients had bilateral bronchiectasis, two had unilateral and two had a cylindrical disease pattern. Patients were clinically stable, defined as no change in medication and no exacerbation of respiratory symptoms 1 month prior to recruitment. Participants with hypertension or taking antihypertensive medication were included in order to maximise recruitment and generalisability to the wider population. Smokers and past smokers were also included for this reason but patients and controls were matched for smoking exposure. All subjects gave written, informed consent, and the study was approved by the South West Wales Research Ethics Committee (08/WMW02/48). Subjects were excluded from the study if they had known heart disease, cor pulmonale, malignancy or any other inflammatory or metabolic condition or were receiving oral corticosteroids.
Anthropometry, pulmonary and physical characteristics
All participants performed spirometry (Vitalograph, UK) from which forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC) and their ratio (FEV1/FVC) were determined according to American Thoracic Society and European Respiratory Society guidelines. 12 Resting oxygen saturation was determined by pulse oximetry (Pulsox-3iA, Konica-Minolta, Japan).
Height and weight were measured and used to calculate body mass index (BMI). All subjects except two controls, who declined, underwent a dual energy x-ray absorptiometry scan (Discovery bone densitometry system Hologic, Bedford, Massachusetts, USA) to determine body composition (fat free mass (FFM)) and BMD at the hip and lumbar spine. Whole body FFM was corrected for height 2 and expressed as the FFM index (FFMI). The BMD was expressed as T-scores, and osteoporosis was defined as a T-score less than −2.5 and osteopenia as T-score between −1.0 and −2.5. 13 Exercise capacity was assessed in all subjects using the 6-min walk distance (6MWD), and all subjects completed a physical activity questionnaire of self-reported activity over the last month. 14
CV measurements
All assessments were undertaken with subjects having fasted and abstained from caffeine, tobacco and inhaled short-acting β2 agonists for at least 6 h and 12 h following long-acting β2 agonists or anticholinergics. After 10 min of rest, peripheral systolic and diastolic blood pressures (BPs) were determined and used to calculate mean arterial pressure (MAP) (OMRON Corporation, Kyoto, Japan). Applanation tonometry was used to measure pulse waveforms at the radial, carotid and femoral sites using a high-fidelity micromanometer (Millar instruments, Texas, USA). PWV was derived using the R wave of a simultaneous electrocardiogram recording. Aortic PWV was calculated as the speed of the pulse wave travelling between the carotid and femoral pulse and brachial PWV was determined using the radial and carotid pulse (SphygmoCor; AtCor Medical, Sydney, Australia). The radial pulse wave was used to generate a central arterial waveform, and augmentation index (AIx) (corrected to heart rate of 75 bpm) was calculated as the difference between the first and reflected systolic peak as a percentage of pulse pressure. 15
Biochemistry
Fasting blood was analysed to determine the levels of glucose, lipids and albumin according to standard hospital practice, and in patients arterialised earlobe gas was analysed. The inflammatory mediator, IL-6, was measured by enzyme-linked immunosorbent assay (R&D Systems Europe, Abingdon, UK). 4
Data analysis
Data analysis was performed with the Statistical Package for the Social Sciences (SPSS, Chicago, Illinois, USA) software version 16.0. Positively skewed data were log10 transformed. Analyses included the independent t test, Pearson’s correlations and stepwise multiple regression. The Chi- square test was used for nominal data comparisons. The significance level was set at p < 0.05. The sample size calculation was based on the primary outcome measure of aortic PWV, which has been reported to have a SD of 2 m/second in patients with COPD. In order to detect a 15% difference between patients and controls with a significance of 0.05 and a power of 0.8, 18 individuals were required per group.
Results
Study population
A total of 20 patients and 20 controls similar in age, sex and smoking history were recruited (Table 1). The aetiology of bronchiectasis was variable; 13 patients developed the disease following an infection (six during adulthood and seven had whooping cough or pneumonia during childhood) and seven patients had idiopathic bronchiectasis. Although bronchiectasis was the primary diagnosis, 13 patients had evidence of airflow obstruction on spirometry. Two patients had previously undergone a lobectomy for bronchiectasis, and in total, six patients had a documented history of a number of pulmonary pathogens, which comprised of the following: four Pseudomonas aeruginosa, four Streptococcus pneumoniae, two Haemophilus influenzae and three Candida spp. The patients mean (SD) resting oxygen saturations were 96 (2)%, and arterial oxygen level was 11 (2.4) kPa. No patient met the UK criteria for long-term oxygen therapy (7.3 kPa).
Subject characteristicsa

6MWD: 6-min walk distance; BMD: bone mineral density; BMI: body mass index; FEV1: forced expiratory volume in 1 second; FFMI: fat free mass index; FVC: forced vital capacity; METs: metabolic equivalents; SpO2: peripheral oxygen saturations.
aData are presented as mean (SD) unless stated otherwise.
bMedian (range).
c n = 18.
Medication
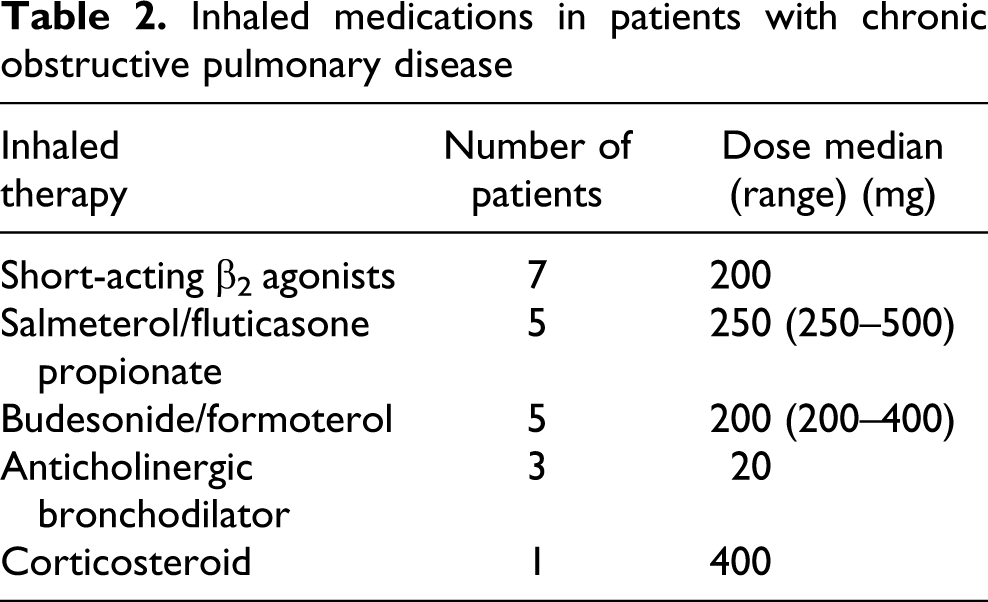
Of the patients, seven were prescribed short-acting inhaled β2 agonists, 10 inhaled combination therapy (salmeterol/fluticasone propionate or budesonide/formoterol), three inhaled anticholinergic bronchodilator and one inhaled corticosteroids; these were the patients who had airflow obstruction (Table 2). Seven patients were taking prophylactic antibiotics (four azithromycin), and 15 of the patients had previously been prescribed short courses of oral corticosteroids for a respiratory exacerbation. As it was not possible to accurately identify previous exposure to oral corticosteroids, patients on permanent oral corticosteroids were excluded. None of the control subjects was being treated with antibiotics or had taken oral corticosteroids in the past. Six patients were receiving antihypertensives, while none of the controls had been diagnosed or medicated for hypertension. Three of the patients and three controls were taking statins.
Inhaled medications in patients with chronic obstructive pulmonary disease
CV and biochemical data
Patients with bronchiectasis had greater aortic PWV (10.5 (3.0) vs. 8.8 (1.6)) and heart rate than controls, although peripheral and central BPs were similar (Table 3), and a similar proportion of patients (n = 12) and control subjects (n = 10) had elevated BP (systolic BP >140 mmHg or diastolic BP >90 mmHg). 16 More patients were on treatment for hypertension than controls, and those patients on antihypertensives had greater PWV 12.3 (3.3) than patients not on antihypertensive medication 9.5 (2.4) m/second (p = 0.045). Patients with bronchiectasis had increased interleukin-6 (IL-6) as well as reduced albumin and glucose (p < 0.05), although total cholesterol was similar.
Haemodynamic and biochemical data in patients and controlsa

AIx: augmentation index; DBP: diastolic blood pressure; HR: heart rate; IL-6: interleukin 6; MAP: mean arterial pressure; PWV: pulse wave velocity; SBP: systolic blood pressure.
aData are presented as mean (SD) unless stated otherwise.
bGeometric mean.
In patients, aortic PWV was related to age (r = 0.728, p < 0.001), heart rate (r = 0.450, p = 0.047) and inversely related to FEV1% predicted (r = −0.482, p = 0.032) and oxygen saturation (r = −0.548, p = 0.01). In all subjects pooled, aortic PWV also related to age (r = 0.69, p < 0.001), FEV1% predicted (r = −0.552, p < 0.001), heart rate (r = 0.405, p = 0.010), 6MWD (r = −0.45, p = 003), log10 IL-6 (r = 0.389, p = 0.013) as well as central and peripheral MAP (r = 0.396, p = 0.011 and r = 0.382, p = 0.015).
Stepwise linear regression in all subjects, with aortic PWV as the dependent variable, was carried out using the related parameters above and found that only age was a predictor of aortic PWV (R 2 = 0.529, p < 0.001).
Physical characteristics
Patients and controls had similar mean BMI, total FFMI and BMD at the hip and lumbar spine (Table 1). However, fewer patients had an accepted healthy BMI (20–25 kg/m2) and fewer had a normal BMD when compared with controls (Figures 1 and 2). Overall 80% of patients had either osteoporosis or osteopenia compared with 50% of controls subjects (p = 0.05). Patients had less 6MWD and reported less physical activity (p < 0.05).
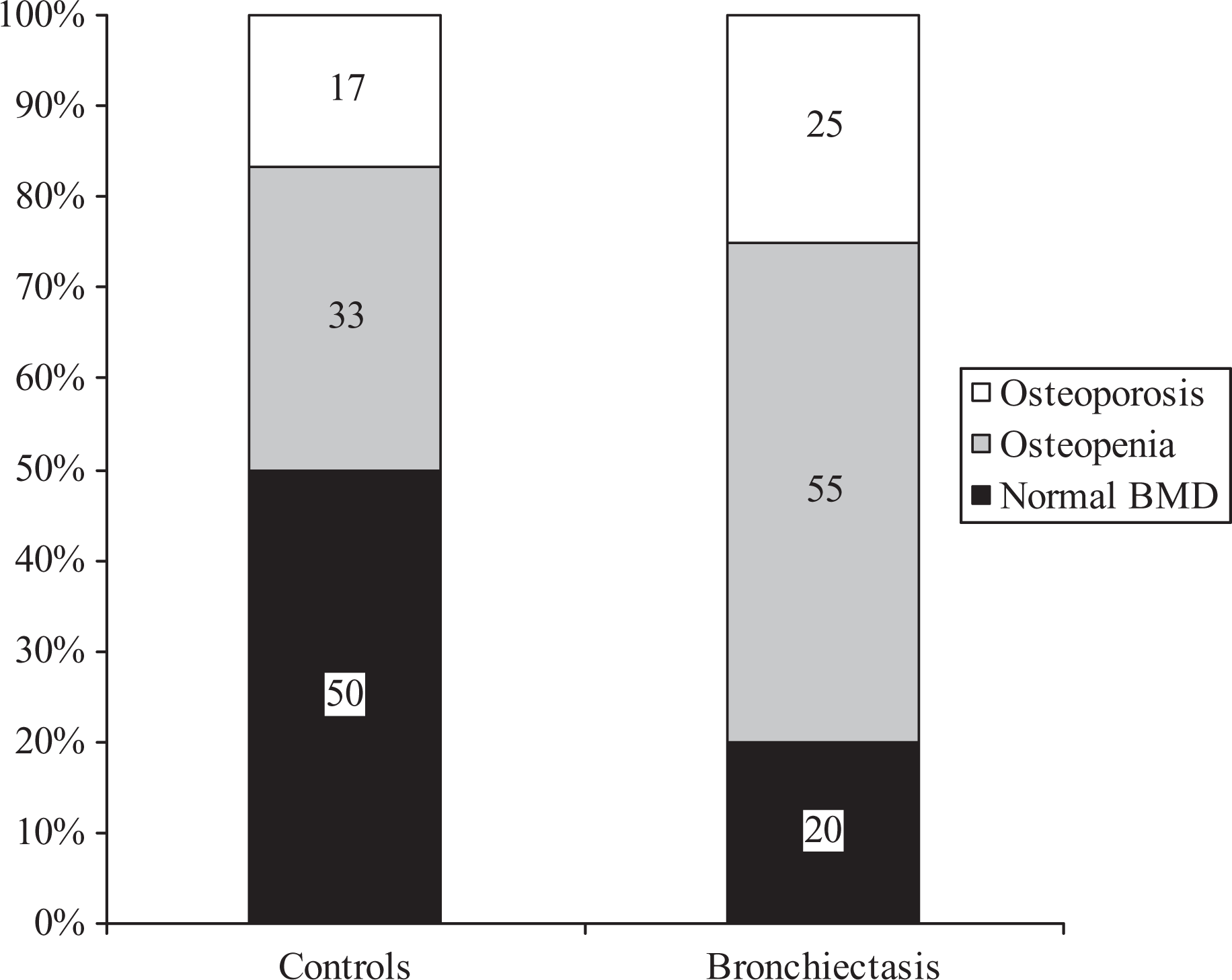
Osteoporosis or osteopenia in patients with bronchiectasis and control subjects. Numbers on the chart represent the number of individuals in each classification. Osteoporosis defined as a T-score less than −2.5. Osteopenia defined as T-score between −1.0 and −2.5.

Aortic PWV in patients with COPD and controls treated and not treated for hypertension. COPD: chronic obstructive pulmonary disease; PWV: pulse wave velocity.
Discussion
In this study, in patients with bronchiectasis, we found evidence of a range of comorbidities similar to those reported in COPD and CF.4,6,7,17,18 Patients with bronchiectasis had a mean aortic PWV, over 1 m/second greater than controls, which indicates increased arterial stiffness. This was in the context of other CV risk factors such as mean BP and cholesterol and both groups had a low exposure to cigarette smoke. An increase of 1 m/second aortic PWV corresponds to an adjusted risk increase in 15% in CV mortality, 2 indicating a potentially clinically relevant increased risk of CV disease in patients with bronchiectasis. More patients were on treatment for hypertension than controls, and such patients had greater PWV, which may reflect a stiffer aorta due to established hypertension. Patients with bronchiectasis also had increased systemic inflammation, increased heart rate and evidence of bone thinning and impaired exercise capacity, as measured by reduced 6MWD and physical activity score, when compared with controls. The mechanism of increased systemic inflammation is likely related to the pathology of repeated infection in bronchiectasis 2 and raised heart rate may be a consequence of impaired pulmonary function, as in COPD. 6
In the present study, aortic PWV was related directly to heart rate and inversely to oxygen saturation. The relationship of aortic PWV with heart rate is in debate. Although it has been suggested that heart rate can influence the assessment of PWV, this is not supported by a consensus document. 15 An alternative suggestion is that increased heart rate may indicate increased sympathetic drive that has been shown to predict PWV. 19 The inverse relationship between oxygen saturation and aortic PWV may by explained by the increased oxidative stress, heighted inflammation and endothelial dysfunction. 20
Increased aortic PWV indicates aortic stiffness that results in an early arrival of the reflected pulse wave that returns to the heart during systole rather than diastole. The result is an increase in central systolic pressure, reduced ventricular perfusion and increased CV risk, which could contribute to the presence of CV disease in patients with bronchiectasis. 10 In addition, stiff arteries increase afterload and could contribute to the development of cardiac dysfunction that has been identified in patients with bronchiectasis. 11 In the present study, there was no difference in brachial PWV or AIx, which may be a consequence of the different properties of central and peripheral arteries. The utility of both these measures have not been established in this age group of subjects. 15,21
Although there is a paucity of evidence of comorbidities in bronchiectasis, the presence of comorbidities including increased aortic PWV and loss of BMD has now been identified in a number of chronic respiratory diseases including COPD 6 and alpha-1 antitrypsin deficiency. 7 Similarly, in patients with CF, a genetic cause of bronchiectasis, increased AIx, an indicator of arterial stiffness in individuals younger than 50 years, has been reported. 8 Similar to these studies, the present investigation showed that both age and FEV1% predicted were related to aortic PWV. When the results for all subjects were pooled, aortic PWV was additionally related to central and peripheral MAP, heart rate, 6MWD and Log10 IL-6; however, only age was a predictor of aortic PWV. These associations may not have been apparent in patients alone due to the small numbers, but they could contribute to the development of increased arterial stiffness. Although the mechanism has yet to be identified, population studies have linked impaired lung function and CV disease independent of age and smoking history. 22 Lung function has also been linked with increased arterial stiffness. 23 It has been suggested that a chronic systemic inflammatory state may underlie this association and cross-sectional studies have shown such link between a chronic inflammatory state and the development of CV disease, through the reduction in vasodilators such as nitric oxide. 24 It has been shown that the induction and reversal of acute systemic inflammation resulted in corresponding changes in PWV. 25
We also identified that the patients with bronchiectasis were in suboptimal physical condition. Although mean BMI and BMD was similar between patients and control subjects, fewer patients were in the optimal range for BMI (20–25 kg/m2); mean albumin and glucose were reduced suggesting suboptimal nutrition, and more patients had osteoporosis or osteopenia when compared with controls’; a quarter of the patients with bronchiectasis had osteoporosis and more than half had osteopenia. It is interesting that 50% of the control group had osteoporosis or osteopenia, which may be attributed to the majority of participants being postmenopausal females.
This highlights a limitation of using mean figures to compare data and is in agreement with the increased prevalence of osteopenia and osteoporosis children with bronchiectasis compared with control subjects similar in age, sex and z-score. 26 Impaired nutritional status has previously been identified in patients with more severe bronchiectasis. 27 In the present study, only two patients were underweight; however, mean levels of albumin were reduced when compared with control subjects, which may indicate suboptimal nutritional status or the nutritional impacts of chronic systemic inflammation and has the implications for physical function.
The mechanism of physical impairments is likely to be multifactorial in patients with bronchiectasis. Osteoporosis is a common feature in patients with COPD and CF where it has been linked with systemic inflammation, use of corticosteroids and reduced BMI, FFM and reduced physical activity, 18,28 which were the factors present in patients in this study on bronchiectasis. Reduced physical activity is a consequence of dyspnoea, 29 which has physical, metabolic and psychological implications, and may contribute to increased CV risk as well as loss of BMD and reduced quality of life. 30
Limitations of this study
As there has been no previous research focused on CV and other comorbidities in bronchiectasis, the sample size was based on previous work in patients with COPD, and as this study suggests less clear cut prevalence of comorbidities, future studies may need more patients to give a better insight in bronchiectasis. In this prospective study, although patients and controls were similar in age, gender and smoking status, the hypertensive status of patients and controls was not known before assessment and differed in patients and controls. In addition, they differed in lung function, indeed 10% had undergone a lobectomy and 75% had been prescribed oral corticosteroids, which may have affected the results. It is recognised that spirometry does not fully quantify bronchiectasis; future studies could use alternative methods when these are validated. Given the small numbers, regression was only performed in pooled data for patients and controls using variables that were correlated; therefore, this analysis should be interpreted with caution. The cross-sectional nature of the study does not allow causal relationships to be identified. Nevertheless, this exploratory study identified some abnormalities that require further investigation.
Conclusion
Patients with bronchiectasis had increased aortic stiffness compared with controls similar in age, BMI, smoking history, BP and cholesterol. Aortic PWV was related to age and heart rate, and inversely related to lung function and oxygen saturation. The patients also had reduced exercise capacity and more patients had osteoporosis. These represent additional comorbidities that require further investigation to optimise outcomes for patients.
Footnotes
Acknowledgements
The authors thank the patients and volunteers who participated in the study and Gareth Dunseath and Maggie Munnery for technical assistance.
Conflict of interest
The authors declared no conflicts of interest.
Funding
The author (NSG) was funded by the Research Capacity Building Collaboration RCBC Wales.
