Abstract
Most severe pulmonary exacerbations (PExs) in adult patients with cystic fibrosis (CF) are treated with 2 week of intravenous (IV) antibiotics. At occasions, the treatment is extended. The morbidity and the cost of extending the treatment are considerable. Risk factors and the outcome of extending the course of treatment have not been formally investigated. This was a prospective study. Decision to extend the course of antibiotics was made in patients who were not deemed to have responded to the initial 14 days of treatment. Risk factors examined for extending the course were site of treatment (home or hospital), CF symptom score, body mass index (BMI), forced expiratory volume in the first second (FEV1) and C-reactive protein (CRP) at days 1 and 14 of the treatment. The following outcome measures were assessed for PExs requiring prolongation of treatment: FEV1, BMI, CF symptom score, CRP and number of days until the following PExs. PExs that were treated with 14 day course were used for comparison. Of all the PExs, 22.9% needed extension of treatment beyond day 14. Compared with PExs needing 14 days of antibiotics, CF symptom score, FEV1 and CRP at day 14 were worse in those who had to have the course extended. Extending the course of IV antibiotics to 21 days improved symptom score, but not any of the other outcome measures, including the number of days until the next PEx. Extending the course beyond 21 days did not result in improvement in any outcome measure. PExs in patients with worse lung disease and greater residual symptoms and lung inflammation at day 14 of antibiotic treatment were associated with the extension of the course of IV antibiotics. Prolonging the treatment to 21 days improved symptoms, but did not result in improvement in any other short-term or lung outcome measures.
Introduction
The aim of managing pulmonary exacerbations (PExs) in patients with cystic fibrosis (CF) with intravenous (IV) antibiotics is to return patients’ symptoms and lung function tests to pre-exacerbation status. There are no randomised controlled trials on the optimal duration of IV treatment. Nevertheless, most CF guidelines and book chapters recommend 10–14 days courses of antibiotics. 1 –3 Many authors suggest that the extension of the IV courses of antibiotics can be made for those patients who are older and sicker. In practice, extending the course of IV antibiotics is made when the patient is not deemed to have gained the expected benefit at day 14.
The lungs in most adult patients with CF are chronically infected with Pseudomonas aeruginosa, and therefore, most PExs are treated with a combination of at least two antibiotics. Due to the chronicity of the lung infection, the severity of structural damage and the poorer penetration of drugs through thick mucosa, larger doses of antibiotics are required to treat CF PExs than in other groups of patients with lung infections. 1,3
Frequent and longer courses of IV treatment, particularly with aminoglycosides, has now been shown to be associated with renal impairment, 4,5 autotoxicity 6,7 as well as increased rate of antibiotic allergy. 8 In addition, extension of duration of IV antibiotic treatment is associated with added volume of work to patients, CF care workers and in incremental cost pressure of CF care. Given these considerations, the decision to extend the treatment for more than 14 days needs to be made with care.
Two retrospective studies 9,10 found that the administration of antibiotics for 14 days in adolescent and young adult patients with CF resulted in clinical recovery in 72% and 75% of patients. A recent guideline committee for the treatment of CF PExs that studied the literature of management of PExs found no evidence for the optimal duration of treatment of CF PExs. 11 The committee recommended that the future studies should examine short- and long-term subjective and objective outcome of management of PExs according to the duration of treatment.
We conducted a prospective research programme on CF PExs in adult patients with CF between 2007 and 2010. The diagnosis of PExs and the decision for treatment was formally predefined. The programme investigated both objective and subjective parameters of PExs. One of the aims of this research was to examine the characteristics of PExs that required more than 2 weeks of IV antibiotics and to investigate whether the extension of the IV course beyond 2 weeks resulted in subjective and clinically objective benefits.
Our hypothesis was that the patients who needed extension of antibiotics were those with more severe structural lung abnormalities and those with residual lung infection at the end of the course. We also postulated that the extension of antibiotics would improve several outcome measures including delaying the following PEx.
Patients and methods
The research programme included a 2-year recruitment period and a 1-year follow-up. All patients with CF were treated and managed in the same CF unit. All medical records were kept within the Department of Respiratory Medicine and were readily available.
The programme was approved by the Southmead Research Ethics Committee (Bristol, UK) and all the patients with CF signed an informed consent prior to being enrolled in the study.
Inclusion criteria were consenting adult patients with CF receiving full care in our CF unit who were deemed to need IV antibiotics for the treatment of acute PExs. Patients whose care needed to be shared with adjacent smaller units, those with PExs treated with oral courses of antibiotics and patients who had received lung transplant were excluded from the study. Because of the small number of patients in our unit, patients infected with Burkholderia cepacia complex were also excluded. Episodes in which IV antibiotics were initiated for reasons other than PExs were also excluded from the study.
Throughout the programme, definition of PExs was dependent on the decision to treat. We elected a definition of PExs, which was similar to that used for patients with chronic obstructive airways disease. 12 PExs were defined as ‘an event in the course of the disease when there is an increase in symptoms beyond day-to-day fluctuations for the patient, which instigate treatment with IV antibiotics’.
Upon the decision to start treatment with IV antibiotics, site of treatment (hospital or home) was decided as a consensus between the patients and the CF team. Patients were considered to have been treated ‘at home’ if they received more than 80% of the treatment at home. If patients were treated in hospital or if the treatment was started in hospital but completed at home the site of treatment was classified under ‘other’. As part of the research programme, all patients treated at home received two structured visits every week from CF nurses or CF physiotherapists throughout the course. At each visit, emphasis was made on regular physiotherapy, adherence to treatment and advice on physical rest and proper nutrition.
At the start of the treatment and on day 14, patients underwent the following assessments: CF symptom scoring, spirometry and measurement of body mass index (BMI) and serum C-reactive protein (CRP). For those needing extension of treatment, assessment were also repeated at days 21 and 28 of the treatment.
Extension of treatment was based on the views of the treating physician together with the members of the CF multidisciplinary team in consultation with the patient. Subsequently, extensions were made for 1 week at a time until the patient was deemed to have achieved sufficient clinical improvement to stop the treatment.
All patients were given at least dual IV antibiotics. The choice of antibiotics was made according to a previous response to and/or results of bacterial sensitivity on previous sputum culture. Only in a small number of patients, the choice of antibiotics was changed during the treatment. In the vast majority of patients, extension of the treatment was made with the same set of antibiotics. All the patients received a combination of two antibiotics: aminoglycoside usually tobramycin or amikacin and another antibiotic of the following: ceftazidime, aztreonam, piperacillin-tazobactam or -meropenem. The doses were similar for all the patients. For those in whom the treatment was extended, the same antibiotics were given as most experienced partial improvement. No changes to other drugs such as inhaled or other medications were made throughout the period of exacerbation.
For the purpose of analyses, patients were divided into two groups; group 1: those who had treatment of 14 days and group 2: those who needed more than 14 days of treatment.
The respiratory symptom score was assessed at the start and at the end of the IV course of antibiotics. The symptom score has been previously described and validated. 13 In short, this was a 20-point score that evaluated four symptoms: cough, sputum, breathlessness and fatigue. Each symptom was scored from 0 (no symptoms) to 4 (severe symptoms). The total symptom score was the summation of the individual scores of the four symptoms. The minimal clinically important difference was 1 point change. The score was found to closely correlate with spirometry values, which was sensitive to change over the course of IV treatment and the change of score with antibiotics correlated with changes in spirometry values.
Spirometry was performed by qualified and experienced lung physiologists using daily calibrated lung function equipments according to the recommendations of the British Thoracic Society/Association of Respiratory Technicians and Physiologists guidelines. 14 For the purpose of this study, the actual values rather than the percentage of predicted values were selected for the analyses. This is mainly because the reference values for this group of patients remain continuous.
Risk factors assessed were age, gender, site of treatment (home or others), forced expiratory volume in the first second (FEV1), BMI and CRP. Other factors compared in the two groups were day 14 FEV1, CF symptom score, BMI and CRP. Outcomes of extending the course were days until the following exacerbation, change in FEV1, CRP, CF symptom score and change in BMI.
Days until the next exacerbation were calculated from the end of the treatment until the start of the next IV antibiotic treatment for an acute PEx or until 365 days had lapsed without the need for IV treatment, whichever was shorter. For the purpose of analysis, if patients did not have PEx, time until the next exacerbation was considered to be 365 days.
Statistical analysis
The statistical analysis for the research programme was made with the support of the Department of Biostatistics at the University Hospitals NHS Trust, Bristol. Prism 4 and Graph Pad computer package was used.
Standard statistical analyses were applied. Basic data were observed for normality of distribution. As a result, comparisons of FEV1, CRP and BMI at the start and at the end of treatment were made using paired t test. As data on symptom score were both categorical and not normally distributed, Wilcoxon matched paired test was used for comparing symptom score.
For similar reasons, comparison of groups 1 and 2 was made using unpaired Student’s t test for FEV1, BMI and CRP and using Mann-Whitney U test for symptom score and for time until the next PExs.
Results
A total of 168 PExs in 58 adult patients with CF (average 2.9 PExs per patient) were analysed. The majority of PExs (n = 129; 77.1%) were treated for 14 days (group 1), and 39 (22.9%) PExs were not deemed to have attained sufficient recovery and therefore needed extension of treatment (group 2). Of the 39 PExs, 28 PExs needed extension for 21 days and 11 PExs required extension of treatment for over 21 days mostly for 28 days. Table 1 shows a breakdown of the number of exacerbation per duration of treatment. Figure 1 shows a histogram for the distribution of duration of treatment in both the groups.

A histogram of the distribution of the duration of the treatment in the both the study groups 1 and 2.
Number of PExs in the two study groups and the subgroups of group 2

PExs: pulmonary exacerbations.
Age and gender did not differ for both the groups. The mean age (SD) for groups 1 and 2 was 25.2 (6.6) and 27.5 (6.9) years, respectively, p = 0.062. The distribution of female-to-male did not differ between the two groups; 67:60 for group 1 and 17:24 for group 2; p = 0.21.
The site of treatment affected the duration of the treatment. Patients in group 2 had their treatment administered at home more frequently than patients in group 1 (odds ratio 2.16; 95% confidence interval 1.02–4.58, p = 0.04).
Table 2 shows factors associated with the extension of the treatment at the start and at day 14 of treatment. Compared with group 1, patients in group 2 had worse symptom score, but not worse FEV1 or CRP at the start of treatment (day 1). In addition, compared with group 1, group 2 had worse FEV1, worse symptom score and greater CRP values at day 14.
Comparison of the characteristics of pulmonary exacerbations at day 1 and day 14 of the treatment of the two study groups
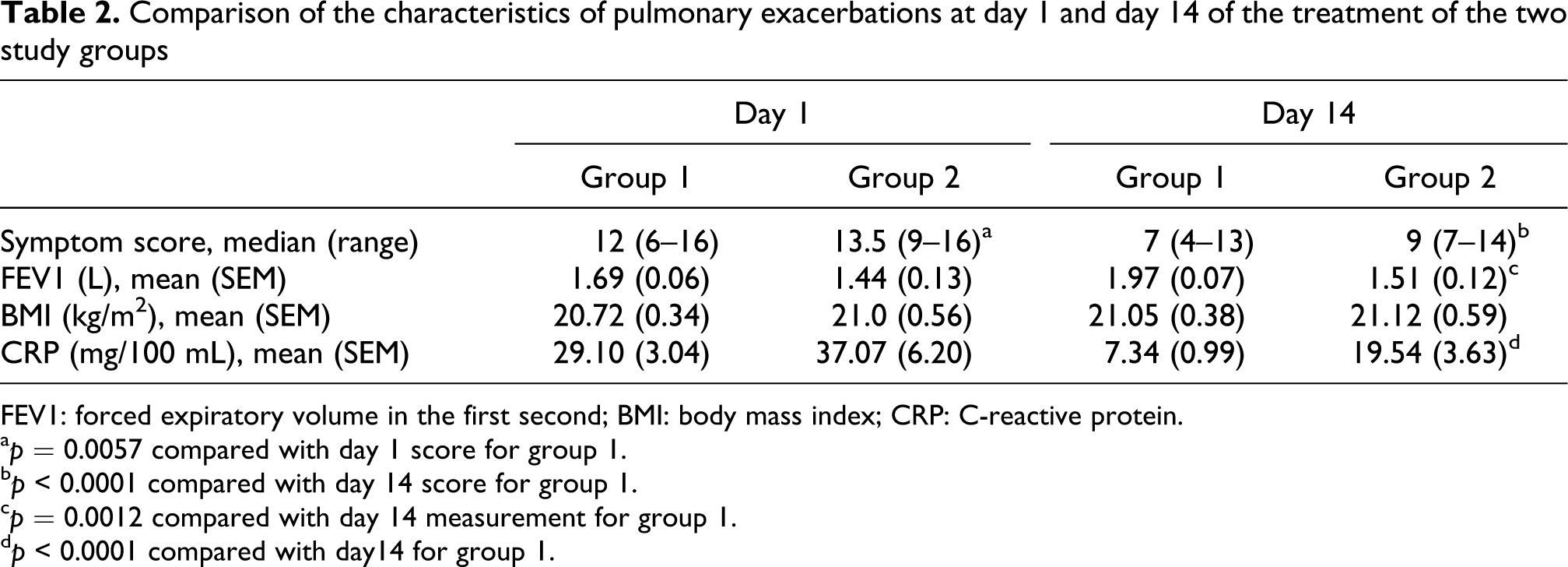
FEV1: forced expiratory volume in the first second; BMI: body mass index; CRP: C-reactive protein.
a p = 0.0057 compared with day 1 score for group 1.
b p < 0.0001 compared with day 14 score for group 1.
c p = 0.0012 compared with day 14 measurement for group 1.
d p < 0.0001 compared with day14 for group 1.
Table 3 shows the outcome of extending IV antibiotics. Small but significant improvement was noted in CF symptom score but not in FEV1, CRP or BMI as a result of extending the treatment. There was no improvement in any of the measurements from extending antibiotic treatment beyond 21 days.
Main outcome measurements at days 21 and 28 for group 2 using data at day 14 as comparatorsa

FEV1: forced expiratory volume in the first second; BMI: body mass index; CRP: C-reactive protein.
aThe three pulmonary exacerbations where the treatment was extended for over 28 days were not included in the analyses.
b p = 0.006 compared with day 14.
The days until the following exacerbation did not differ between groups 1 and 2; median (range) 90 (6–365) days for group 1 and 77 (12-365) days for group 2; p = 0.50.
Discussion
In this study, we prospectively examined risk factors associated with extending the course of IV antibiotics beyond the conventional period of 14 days in adult patients with CF with PExs. Unlike previous studies, our analysis assessed a range of risk factors including objective factors such as FEV1 and CRP as well as subjective measures including CF symptom scores. Furthermore, our research evaluated whether extending the treatment would result in long-term improvement of health outcome measures including days until subsequent exacerbations.
In clinical practice, extension of antibiotics during the study period was made on the lack of clinical response to treatment at day 14 as perceived by the caring team and/or by the patient. When this research programme was designed, it was purposefully aimed that the decision to treat or to extend the length of the treatment was made on clinical grounds without the interference of the researchers.
In our study, 22.9% of patients needed extension of treatment. This is a similar proportion to another research, where the risk factors for the failure of recovery of FEV1 in patients with CF after IV treatment were examined. 10 In their study, FEV1 did not return to the baseline after IV treatment in 25% of PExs. Unlike our study, however, risk factors for nonrecovery included female sex. Other risk factors were concomitant allergic bronchopulmonary aspergillosis (ABPA) and chronic infection with Methicillin-resistant Staphylococcus aureus (MRSA), P. aeruginosa and B. cepacia. The authors suggested a more aggressive treatment for the patients with one or more risk factors.
In our research, factors that separated out the two groups were seen at the end of the planned treatment (day 14). Greater residual symptom score (worse symptoms), greater CRP values as well as smaller FEV1 value at day 14 were associated with the decision to extend the treatment. Conceivably, these factors might represent patients who remained ill and would either demand or mitigate the clinical decision to treat for further period.
Extending the treatment beyond day 14 in our study resulted in a small improvement in symptom score at day 21, but not in other objective parameters such as FEV1, CRP and BMI. Extending the course of treatment beyond 21 days has not resulted in change in any of the outcome measures albeit in a small number of PExs.
During the analysis of this study, another retrospective research was published reporting that a longer duration of treatment was needed in patients with more reduced FEV1. The study found that, despite longer duration of treatment, complete recovery of lost FEV1 did not happen with the extension of treatment. 15 The same study also did not show difference in FEV1 recovery according to the site of treatment (see below). Unlike our study, subjective parameters such as quality of life measures were not assessed. Furthermore, a longer term outcome such as the time until the subsequent exacerbation was not examined.
As our study was prospective, we were able to add another dimension in examining factors at the end of exacerbation that were associated with the extension of treatment. Our analysis found that the treatment extension for patients in group 2 occurred in patients who were more symptomatic at the start of treatment (day 1) compared with group 1. Otherwise, the two groups of PExs were comparable with respect to FEV1, BMI and CRP at day 1 of the treatment.
By starting antibiotics for CF PExs, it is implicitly postulated that the accumulation of bacteria in the lungs plays an important part in the aetiology of PExs. Therefore, antibiotics are provided to reduce the bacterial load in a process that often seemed to be similar to ‘cutting the grass’. However, it was notable that the extension of the IV antibiotics treatment did not result in extending time until the next exacerbation. This would suggest that the factors other than the bacterial growth might be associated with proximity and frequency of PExs and that the longer courses of IV antibiotics may not necessarily influence the duration needed for bacterial factors (probably the speed of intrapulmonary bacterial growth) or inflammatory factors to cause further PExs.
Our study showed that the treatment of CF PExs at home was one risk factor for extending treatment beyond day 14. With improvement of knowledge of CF and with the advent of devices allowing long-term venous access together with more familiarity of self administration of IV antibiotics, home treatment of PExs is now provided more frequently than before. This approach is more attractive as it causes less disruption to patients’ lives, can save hospital bed days and is thought to be less costly than hospital treatment. 16,17
However, the efficacy of home IV treatment compared with hospital treatment for CF PExs was examined in several studies that reported different outcome. Some retrospective studies found equal outcome of care for home treatment compared with hospital treatment. 18,19 This was supported by a recent retrospective analysis of 1535 adolescent and adult patients with CF, where the site of the treatment did not affect the return to baseline of FEV1. 15
In contrast, carefully planned studies in adult 20 and in paediatric patients with CF 21 reported less favourable results for home treatment of PExs with respect to lung function tests and BMI compared with either hospital treatment or treatment that was started in hospital and completed at home.
We previously outlined advantages and disadvantages of home treatment and noted the variation in the literature with regards to the outcome. 22 We also highlighted that, to date, there had been no studies to show a superior objective outcome for home treatment. Two Cochrane reviews had already supported this conclusion although it admitted that only a few studies were included in their analyses. The reviewers demanded more randomised trials. 23,24 Factors such as closer observations and more intense treatment such as physiotherapy and dietetic input in hospital were thought to be the cause of better outcome.
As a result, we established a home management programme with intensive support from the CF team during PExs. Despite the augmentation of home support, the current findings are the first in a series of results to enforce the inferior outcome of home treatment. To our knowledge, this study is the first to show that the IV antibiotic courses provided at home (even with the team’s support) as opposed to those provided wholly or partially in hospital are more likely to be associated with extension of the course of the treatment beyond day 14.
Extending IV treatment have a significant impact on manpower input from the CF team, the inconvenience for patients taking IV antibiotics for longer period as well as on drug and equipment expenditure. These would offset the perception of home treatment being cheaper and more convenient than hospital treatment and would add to the controversy of the advantage of home treatment.
This study had its limitations. The number of patients was relatively modest. Given the limited number of patients with CF available in most adult CF units and the unique approach for home support adapted in this research programme, our study, like other studies, 20,21 treated and analysed each PEx as a separate event. Similarly, due to the modest number of patients, factors that might contribute to the extension of antibiotics such as CF related diabetes and ABPA were not examined. Another limitation is that the study by its nature was observational albeit addressed a common practice in CF units worldwide. Given the variability in disease severity amongst patients and the impact of IV treatment, it is not possible to design a prospective work in which patients are divided into those who take antibiotics for 2 weeks and those who take them for longer period.
Alternatives to extending the courses in unresponsive CF treatment need to be individualised. This may include watchful waiting and clinical follow-up without extending the course of the treatment as some PExs may recover after day 14 without the extension of the treatment. Treating high-risk groups with more than two antibiotics may need to be considered. A third option is to add anti-inflammatory agents such as oral corticosteroids to high-risk group. The latter is a practice adapted by many UK physicians, 25 but has only examined in a small randomised trial. 26 Obviously, the outcome of each of these options needs to be investigated. Another choice is to change the set of IV antibiotics at 2 weeks or to tailor antibiotic choice to in vitro sensitivity, which would become available at the 2-week point. This is not withstanding the previous studies, which showed that there was no difference in the outcome of treating exacerbations when there was or was not a synchronisation between the prescribed antibiotics and in vitro sensitivity profile. 27,28
In the CF community, there is an emphasis on introducing much needed new therapeutic agents acting on several aspects of the CF syndrome. This includes testing new antimicrobial agents, anti-inflammatory treatments and mucolytic agents. This effort is laudable, but recent studies including our study highlight the need to look back at the basic principles of management of CF exacerbations in methodical ways. This examination should include the effect of the site of the treatment (hospital or home), the duration of the IV course for PExs and factors associated with shorter times until the next PEx. These questions are better answered in the form of multicentre prospective studies that include sufficient number of patients and that aimed to validate methods of assessment of outcome measures comprising patient-reported outcomes.
Footnotes
Funding
This research programme was supported by an unrestricted grant from the David Telling Research Trustees, Bristol.
Conflict of interest
The authors declared no conflicts of interest
