Abstract
Objective
This study investigated the expression and diagnostic significance of LINC00861 in DN and explored its functional mechanisms in high glucose-induced cell damage.
Methods
We enrolled 285 participants: 92 healthy controls, 98 uncomplicated T2DM patients, and 95 DN patients. Serum LINC00861 and miR-378a-3p levels were measured via qRT-PCR. Correlations with clinical parameters were assessed using Pearson analysis. Logistic regression identified risk factors for DN progression, and ROC curves evaluated diagnostic accuracy. In vitro, LINC00861 was knocked down to assess its effects on proliferation, inflammation, and oxidative stress under high glucose. Targeting of miR-378a-3p was confirmed by dual-luciferase and RIP assays.
Results
The LINC00861 expression was significantly elevated in DN patients compared to T2DM and healthy controls, and correlated positively with FBG, HbA1c, and albuminuria, but negatively with eGFR. Multivariate analysis identified LINC00861 as an independent risk factor for DN. The AUC for distinguishing T2DM from DN was 0.918. miR-378a-3p expression was significantly lower in DN patients than in those with T2DM or healthy controls. LINC00861 silencing may alleviate HG induced aberrant proliferation, inflammatory responses, and oxidative stress, possibly via the regulation of miR-378a-3p.
Conclusion
The LINC00861 is upregulated in DN and may facilitate renal injury by sponging miR-378a-3p, indicating its potential as a diagnostic biomarker and therapeutic target.
Introduction
Diabetic nephropathy (DN), a prevalent microvascular complication of diabetes, represents a major cause of end-stage renal disease (ESRD) worldwide. Its increasing incidence adversely affects both the quality of life and overall survival of patients.1–4 At present, the early detection of DN remains difficult due to insufficiently sensitive and reliable biomarkers. Moreover, the pathogenic mechanisms underlying DN have not been completely clarified, which contributes to a scarcity of effective therapeutic interventions and suboptimal clinical outcomes.5–7 The pathology of DN involves a multifactorial process characterized by oxidative stress, inflammatory activation, and renal fibrosis, all promoted by chronic hyperglycemia. 6 Furthermore, the accumulation of reactive oxygen species (ROS) and metabolic imbalances within the renal microenvironment aggravate injury to the glomerular filtration barrier and tubular structures, ultimately leading to proteinuria and reduced glomerular filtration rate.6,8,9 Thus, elucidating the molecular mechanisms of DN and discovering new diagnostic markers and therapeutic targets hold significant clinical value.
Recent evidence highlights the critical regulatory functions of long non-coding RNAs (lncRNAs) in the pathological processes of DN.10,11 Studies based on renal biopsy specimens indicate that lncRNAs participate in the modulation of inflammatory and apoptotic pathways triggered by hyperglycemia, affecting macrophage polarization and injury to renal tubular epithelial cells.12,13 Furthermore, in vitro investigations support that lncRNAs regulate the production of inflammatory mediators through miRNA-mRNA interactions and contribute to processes such as renal fibrosis and tissue repair.13–15 Notably, LINC00861 is dysregulated in individuals with inadequate glycemic management, indicating its potential utility as a diagnostic marker in DN. 16 Additionally, LINC00861 also modulates endoplasmic reticulum stress, which is recognized as a pivotal mechanism in renal cell damage induced by high glucose conditions.17–19 Moreover, microRNAs including miR-378a-3p have been implicated in renal impairment. For instance, under ischemia-reperfusion conditions, miR-378a-3p promotes ferroptosis through suppression of GPX4 and SLC7A11, intensifying kidney injury. 20 Although the crosstalk between lncRNAs and miRNAs is emerging as a fundamental aspect of DN pathogenesis, the precise role of LINC00861 and particularly its potential influence on mesangial cells via miR-378a-3p remains elucidated.
This study was designed to comprehensively assess the expression profile, diagnostic potential, and molecular mechanisms of linc00861 in serum samples from individuals with diabetic nephropathy (DN). Utilizing a combination of clinical specimens and cellular models, we aimed to elucidate whether linc00861 influences high glucose-induced mesangial cell proliferation, inflammatory cytokine secretion, and oxidative stress by targeting miR-378a-3p. The outcomes of this investigation may offer novel perspectives for the early detection and targeted treatment of DN. Furthermore, this work could enhance the current comprehension of DN pathogenesis and support the theoretical basis for novel non-invasive diagnostic approaches.
Method
Study participants
This study enrolled a total of 285 participants from the Departments of Endocrinology and Nephrology. The cohort was categorized into three groups: 92 healthy controls, 98 patients diagnosed with type 2 diabetes mellitus (T2DM) based on the 2020 American Diabetes Association (ADA) criteria who had preserved renal function and no albuminuria, and 95 patients with T2DM and comorbid DN. Exclusion criteria comprised: (1) type 1 diabetes; (2) acute infections, autoimmune disorders, or malignant tumors; (3) impaired liver function, defined as alanine aminotransferase levels exceeding twice the upper limit of normal; (4) recent administration of anti-inflammatory or immunosuppressive medications; and (5) current pregnancy or breastfeeding. We retrospectively collected data on age, gender, body mass index (BMI), duration of disease, fasting blood glucose (FBG), glycated hemoglobin (HbA1c), systolic blood pressure (SBP), diastolic blood pressure (DBP), total cholesterol (TC), triglycerides (TG), estimated glomerular filtration rate (eGFR), and albuminuria. The protocol was approved by the Ethics Committee of The Third Xiangya Hospital of Central South University and conformed to the principles outlined in the Declaration of Helsinki. Written informed consent was provided by all subjects. In addition, all serum samples used for RNA extraction in this study were collected from fasting venous blood after a minimum of 8 hours of fasting.
Cell culture and transfection
Human mesangial cells (HMCs) were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai) and grown in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, USA) containing 10% fetal bovine serum (FBS; Gibco, USA) and 1% penicillin-streptomycin (Gibco, USA). The cells were incubated at 37 °C in a humidified environment with 5% CO2. To simulate diabetic nephropathy (DN) condition, HMCs were exposed to 30 mmol/L D-glucose for 24 hours; the control group received 5 mmol/L D-glucose. 30 mmol/L mannitol was used as the osmotic control.
For transfection, cells plated in 6-well plates were transfected with si-LINC00861 using Lipofectamine 2000 (Thermo Fisher Scientific, USA), following the supplier’s protocol. Cells transfected with si-NC were used as the negative control. To investigate the role of miR-378a-3p, a miR-378a-3p inhibitor was synthesized. Cells with stable knockdown of LINC00861 were then co-transfected with the miR-378a-3p inhibitor. After 48 hours of transfection, cells were collected for further analysis.
qRT-PCR for LINC00861 detection in serum and HMCs
Total RNA was isolated from serum samples and HMCs using the miRNeasy Serum/Plasma RNA Extraction Kit (Qiagen, Germany). The concentration and purity of RNA were assessed on a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA). Subsequent reverse transcription was carried out using the PrimeScript RT Kit (Takara, Japan) to synthesize cDNA. Quantitative real-time PCR (qRT-PCR) was then performed using the SYBR Premix Ex Taq II Kit (Takara, Japan) on a StepOnePlus Real-Time PCR System (Thermo Fisher Scientific, USA). The expression level of LINC 00861 was normalized and calculated via the 2−ΔΔCt method.
Enzyme-linked immunosorbent assay (ELISA) for inflammatory factor detection
The concentrations of IL-1β, IL-6, and TNF-α in supernatants from HMCs cultures were quantified using commercial ELISA kits purchased from R&D Systems (USA), following the protocols provided by the manufacturer.
Measurement of ROS and malondialdehyde (MDA) levels and superoxide dismutase (SOD) activity
Intracellular ROS were quantified with a fluorescence-based assay kit (Abcam, USA). MDA concentration and SOD activity were measured using commercial kits (Sigma, USA). For ROS detection, HMCs were plated onto 96-well plates at 2×104 cells per well and exposed to either high glucose (HG) or normal glucose (NG) medium for 24 hours. According to the manufacturer’s protocol, the cells were incubated with ROS sensitive fluorescent dye, and the fluorescence intensity was recorded with a microplate reader (Molecular Devices). Results are presented relative to the control group. For MDA and SOD analysis, HMCs were seeded in 24-well plates at a density of 5×104 cells per well and treated under HG or control conditions for 24 hours. Subsequently, the cells were harvested and lysed. MDA content and SOD activity were determined following the instructions provided with the respective kits.
CCK8 assay
The proliferation potential of transfected HMCs was evaluated with a Dojindo Cell Counting Kit-8 (CCK-8, Japan). Cells were plated into 96-well plates at 5×103 cells per well and maintained for 24, 48, and 72 hours. At the indicated time points, 10 μL of CCK-8 reagent was introduced into each well, and the plates were incubated at 37°C for 2 hours. Absorbance readings were taken at 450 nm using a Bio-Rad microplate reader (USA).
Dual-luciferase reporter assay
Based on lncRNA SNP2 predictions, a putative binding site for miR-378a-3p was detected in the 3′UTR of LINC00861. To experimentally confirm this binding, a dual-luciferase reporter assay was conducted. The wild-type 3′UTR segment of LINC00861 (LINC00861-WT) and a corresponding mutant (LINC00861-Mut) were each inserted into the psiCHECK™-2 vector (Promega, Milwaukee, USA). These constructions were then co-transfected into HMCs, and relative luciferase activity was quantified using the detection kit provided by Promega.
RNA immunoprecipitation (RIP) analysis
RIP experiments were conducted in HCMs with the Magna RIP RNA-Binding Protein Immunoprecipitation Kit (Millipore). Cell lysates prepared using RIP lysis buffer were immunoprecipitated with antibodies against Ago2 or IgG in RIP buffer. The resulting RNA protein bead complexes were washed with RIP wash buffer and treated with Proteinase K. RNA was subsequently isolated from the immunoprecipitated and quantified via qRT-PCR.
Data analysis
Statistical analyses were carried out with SPSS 26.0 and GraphPad Prism 9.0. Measurement data are reported as mean ± standard deviation (x ± s). For comparisons across multiple groups, one-way ANOVA was applied, with subsequent pairwise comparisons performed using t-tests. Categorical variables are summarized as number (percentage) and analyzed with the chi-square test. The association between LINC00861 and clinical biochemical indicators was assessed via Pearson correlation analysis. Independent risk factors associated with the progression fromT2DM to DN were identified through logistic regression. The diagnostic value of LINC00861 was evaluated using receiver operating characteristic (ROC) curve analysis. A two-side
Results
Baseline characteristics of study participants
Comparison of baseline clinical characteristics among healthy controls, T2DM, and DN patients.
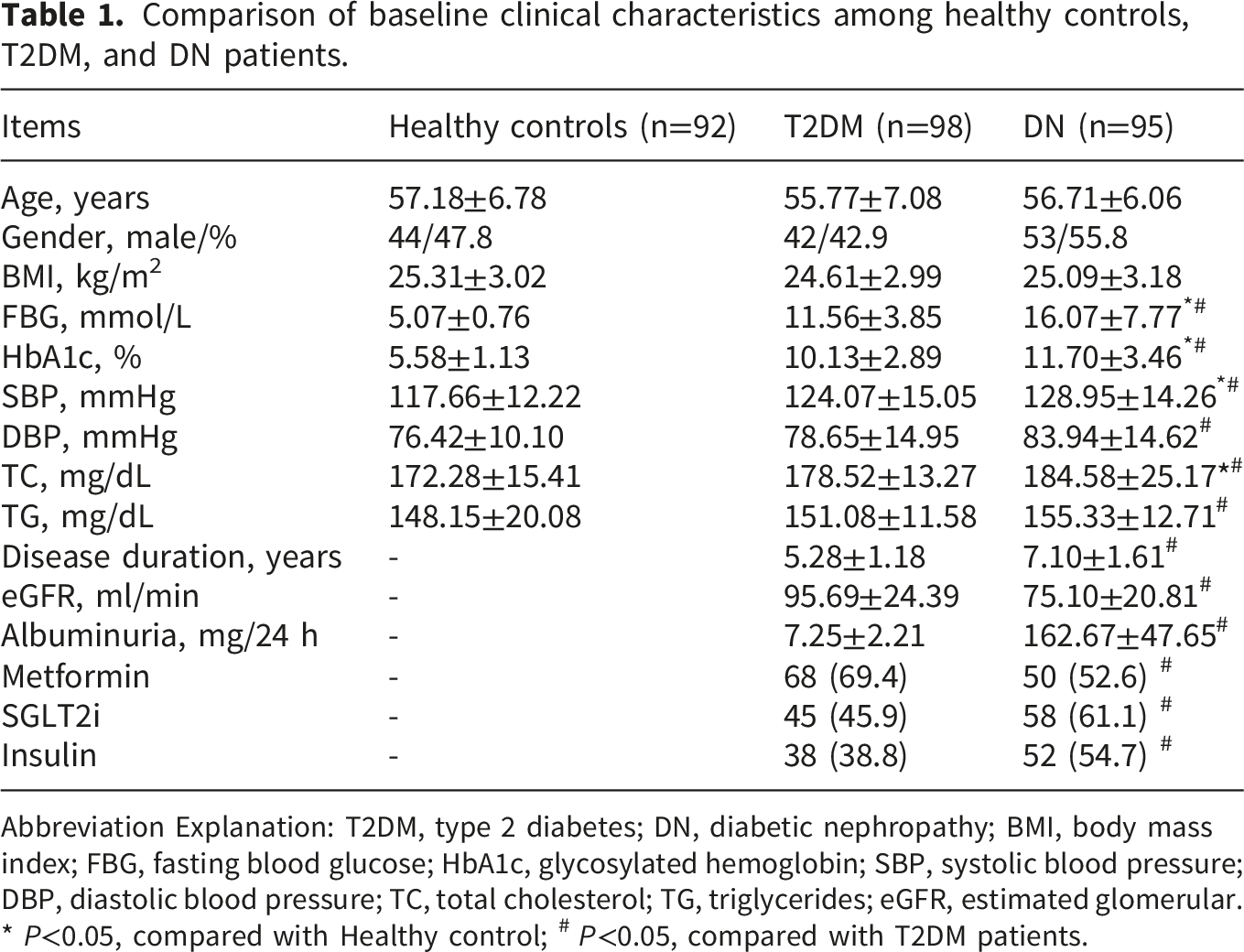
Abbreviation Explanation: T2DM, type 2 diabetes; DN, diabetic nephropathy; BMI, body mass index; FBG, fasting blood glucose; HbA1c, glycosylated hemoglobin; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; eGFR, estimated glomerular. *
Expression level and diagnostic efficacy of LINC00861 in DN patients: A ROC curve analysis
As illustrated in Figure 1(a), the expression levels of LINC00861 exhibited significant differences among the HC, T2DM, and DN groups. Relative to the HC group, LINC00861 was markedly up regulated in the T2DM group. Moreover, its expression was further elevated in the DN group compared to the T2DM group, with all intergroup comparisons reaching high statistical significance. Figure 1(b) displays the ROC curve evaluating the efficacy of LINC00861 in distinguishing T2DM from DN. The AUC was 0.918. At a sensitivity of 87.4%, the specificity was 81.6%, demonstrating that LINC00861 possesses strong diagnostic value for differentiating DN from T2DM. Expression and diagnostic value of LINC00861 in DN. (a) Relative expression levels of LINC00861 in HC, T2DM, and DN groups. (b) ROC curve analysis of LINC00861 for discriminating DN from T2DM.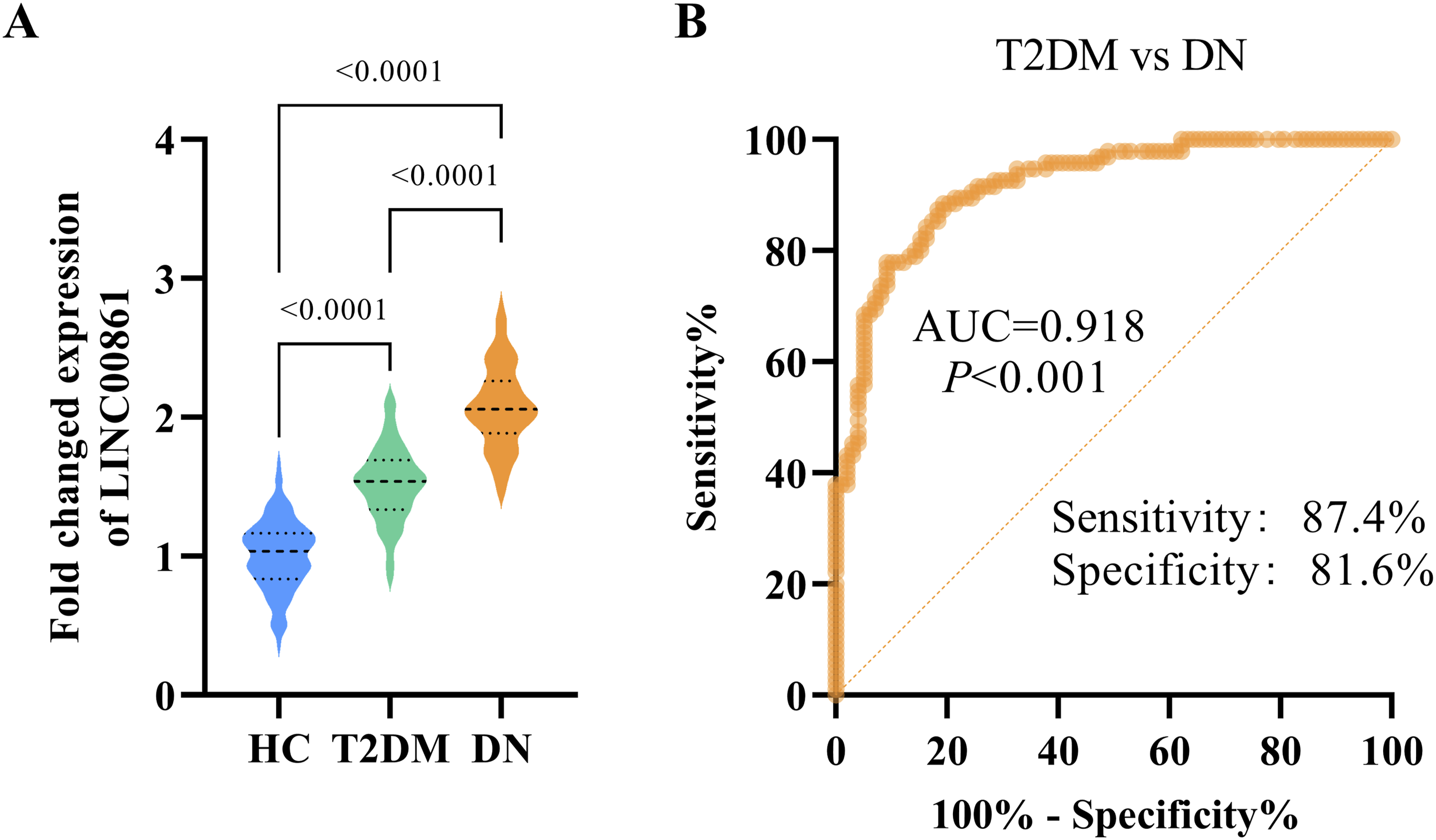
Independent risk factors for DN
Univariate and multivariate logistic regression analysis of factors associated with DN in patients with T2DM.

Abbreviation Explanation: T2DM, type 2 diabetes; DN, diabetic nephropathy; BMI, body mass index; FBG, fasting blood glucose; HbA1c, glycosylated hemoglobin; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides.
Correlation between LINC00861 and clinical indicators
Correlation between LINC00861 expression and clinical parameters in patients with DN.

Abbreviation explanation: DN, diabetic nephropathy; FBG, fasting blood glucose; HbA1c, glycosylated hemoglobin; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglycerides; eGFR, estimated glomerular.
Knockdown of LINC00861 ameliorates high glucose-induced cellular injury by suppressing its expression
As depicted in Figure 2, silencing LINC00861 markedly attenuated the phenotypic alterations triggered by HG treatment in cultured cells. Compared with the control group, the mannitol group showed no significant differences, indicating that all subsequent changes resulted from the specific effects of D-glucose rather than alterations in osmotic pressure. According to Figure 2(a), LINC00861 expression was substantially upregulated under HG conditions relative to the control. Transfection with si-LINC00861 under HG conditions resulted in a pronounced reduction in its expression compared to both the HG group, confirming the efficacy of LINC00861 knockdown in counteracting HG mediated upregulation. CCK-8 assays (Figure 2(b)) revealed a consistent increase in optical density (OD) values across multiple time points in HG treated cells relative to controls. While no notable difference was detected between the HG+si-NC and HG groups, OD values were significantly suppressed in the HG+si-LINC00861 group compared to the other HG-exposed groups, implying that LINC00861 depletion impedes HG enhanced cell proliferation. With respect to oxidative stress parameters (Figure 2(c)–(e)), HG stimulation resulted in significantly diminished SOD activity, along with elevated MDA content and ROS levels compared to the control. Conversely, the HG+si-LINC00861 group showed a significant recovery of SOD activity and a reduction in both MDA and ROS levels relative to the HG and HG+si-NC groups, indicating that LINC00861 knockdown effectively alleviated oxidative stress under HG conditions. Figure 2(f) illustrates that exposure to HG led to a significant rise in the secretion of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β, relative to the control. No discernible differences were observed between the HG+si-NC and HG groups. In contrast, the HG+si-LINC00861 group exhibited a marked reduction in the levels of these cytokines, supporting the conclusion that LINC00861 silencing mitigates HG-induced inflammatory responses. In conclusion, linc00861 knockdown effectively reverses the HG-induced overexpression of LINC00861 and ameliorates its downstream effects, including enhanced proliferation, inflammatory activation, and oxidative stress. Silencing LINC00861 attenuates HG induced phenotypic alterations in cultured cells. (a) Relative expression level of linc00861 detected by qRT-PCR. (b) Cell proliferation analyzed by CCK-8 assay. The OD values were measured at multiple time points. (c–e) Oxidative stress-related parameters. (f) Secretion levels of pro-inflammatory cytokines TNF-α, IL-6, and IL-1βdetermined by ELISA.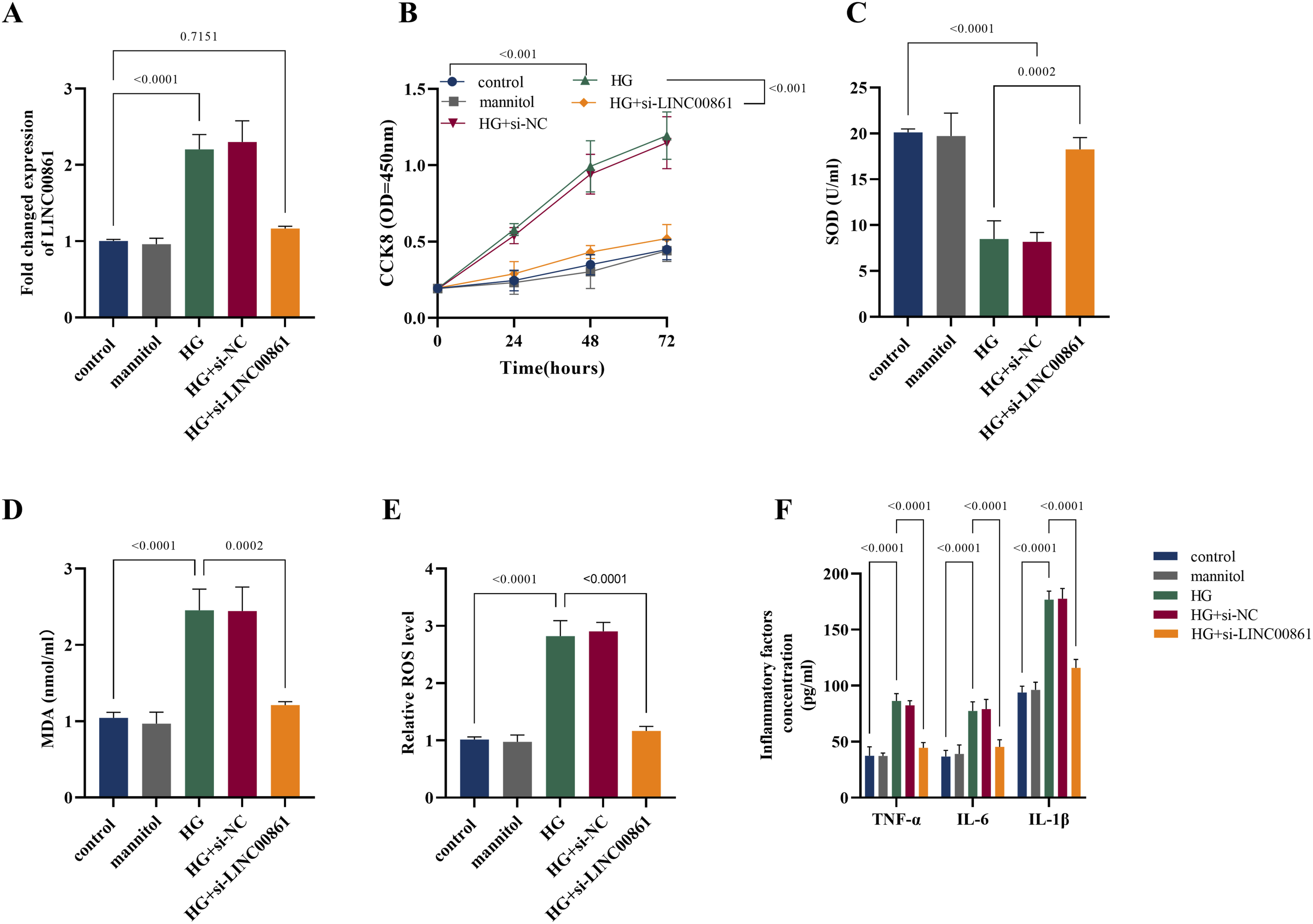
Validation of the targeting relationship between LINC00861 and miR-378a-3p
Bioinformatic prediction (Figure 3(a)) suggested potential complementary binding sites between LINC00861 and miR-378a-3p. Using a dual-luciferase reporter assay (Figure 3(b)), we observed that the miR-378a-3p mimic markedly reduced luciferase activity in the wild-type (WT) LINC00861 reporter construct, whereas the miR-378a-3p inhibitor caused a non-significant increase. By contrast, the mutant (MUT) reporter construct showed no notable changes in luciferase activity upon either treatment, supporting a specific binding interaction between LINC00861 and miR-378a-3p. RIP assays (Figure 3(c)) further demonstrated significant enrichment of both LINC00861 and miR-378a-3p by an Ago2-specific antibody compared to the IgG control, confirming their association within the Ago2-containing RNA-induced silencing complex (RISC). Clinical sample analysis revealed a progressive decline in miR-378a-3p expression from HC through T2DM to DN (Figure 3(d)). Correlation analyses revealed a significant inverse relationship between LINC00861 and miR-378a-3p expression in both T2DM and DN patients (Figure 3(e) and (f)). Specifically, in patients with diabetes, decreased expression levels of miR-378a-3p were associated with gradually elevated expression levels of LINC00861, showing an inverse correlation with a correlation coefficient of 0.8024. Similarly, in patients with diabetic nephropathy compared to those with diabetes alone, miR-378a-3p expression further declined, while LINC00861 expression continued to rise, resulting in a strengthened inverse correlation with a correlation coefficient of 0.8191. In the HG induced model, silencing LINC00861 reversed the HG mediated downregulation of miR-378a-3p. This effect was abolished upon miR-378a-3p inhibition, indicating that LINC00861 contributes to HG associated molecular regulation by suppressing miR-378a-3p (Figure 3(g)). Our results indicate that LINC00861 functions as a competing endogenous RNA (ceRNA) by sponging miR-378a-3p. LINC00861 directly binds to miR-378a-3p and acts as a molecular sponge. (a) Bioinformatic prediction of putative binding sites between LINC00861 and miR-378a-3p. (b) Dual-luciferase reporter assay showing that miR-378a-3p mimic significantly reduced luciferase activity of the WTLINC00861 reporter, but not the MUT construct. (c) RIP assay confirmed enrichment of both LINC00861 and miR-378a-3p in Ago2-immunoprecipitated complexes compared to IgG control. (d) Comparison of miR-378a-3p Expression Levels among HC, T2DM, and DN. (e and f) Correlation analysis between LINC00861 and miR-378a-3p expression in T2DM (e) and DN (f), respectively. (g) Knockdown of LINC00861 under HG conditions increased miR-378a-3p expression, an effect reversed by miR-378a-3p inhibition.
miR-378a-3p mediates the protective effect of LINC00861 knockdown against HG induced damage
Under HG stimulation, a significant promotion of cell proliferation was observed, as reflected by elevated OD values in CCK-8 assays (Figure 4(a)). Concurrently, the secretion of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β, was substantially upregulated (Figure 4(b)). Moreover, oxidative stress markers were markedly disturbed, characterized by decreased SOD activity, increased MDA content, and elevated ROS levels (Figure 4(c)–(e)). Knockdown of linc00861 effectively attenuated HG induced hyperproliferation, suppressed the production of inflammatory cytokines, and mitigated oxidative stress, as demonstrated by enhanced SOD activity, reduced MDA content, and decreased ROS generation. However, these protective effects were largely abolished upon co-transfection with a miR-378a-3p inhibitor. Collectively, these results indicate that LINC00861 silencing may alleviate HG-induced aberrant proliferation, inflammatory responses, and oxidative stress, possibly via the regulation of miR-378a-3p. Knockdown of linc00861 attenuates HG-induced hyperproliferation, inflammation, and oxidative stress via miR-378a-3p. (a) Cell proliferation was assessed by CCK-8 assay under HG conditions. (b) Secretion levels of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) were measured by ELISA. (c–e) Oxidative stress markers: SOD activity (c), MDA content (d), and ROS levels (e).
Discussion
DN represents a serious and frequent microvascular complication of diabetes, with a multifactorial pathogenesis encompassing various pathological mechanisms, including inflammation, oxidative stress, and fibrosis.5,21 Current standard diagnostic measures, such as albuminuria and eGFR, although clinically useful, exhibit limitations related to low sensitivity and vulnerability to confounding factors. Often, detectable changes in these markers manifest only after considerable structural kidney damage has taken place. Consequently, the discovery of novel biomarkers exhibiting improved sensitivity and specificity, along with a deeper investigation into their functional mechanisms in DN pathogenesis, is essential to facilitate early detection and advance targeted treatment approaches.
This study investigated the expression patterns of LINC00861 in serum from DN patients, evaluated its diagnostic potential, and explored its regulatory mechanisms in renal cellular injury, offering novel experimental insights and theoretical foundations for early detection and management of DN. qRT-PCR results indicated that serum LINC00861 levels were markedly elevated in DN patients relative to individuals with uncomplicated T2DM and healthy subjects, implying a potential involvement of LINC00861 in DN initiation and advancement. According to ROC curve analysis, LINC00861 displayed robust discriminative ability in differentiating DN patients from those with T2DM only, supporting its potential utility as a circulating biomarker.22,23 Furthermore, multivariate logistic regression identified LINC00861 as an independent risk factor associated with DN development in T2DM patients, highlighting its critical role in disease progression,a finding consistent with the recognized importance of lncRNAs in diabetic kidney disease. 14 Correlation analyses revealed a positive association between LINC00861 expression and albuminuria, and an inverse correlation with eGFR, suggesting that LINC00861 expression correlates with renal impairment severity and reinforces its functional contribution in DN pathogenesis. Together, these results position LINC00861 as a key molecular integrator of metabolic dysregulation, with dynamic expression monitoring offering promise for early risk stratification in DN.
This study reveals an upregulation of LINC00861 expression in patients with T2DM, with a further significant increase observed in those developing DN. These findings suggest that its expression is influenced by hyperglycemic metabolic conditions. Moreover, LINC00861 levels demonstrate a stepwise elevation from healthy controls to T2DM patients and then to DN patients, showing significant correlations with clinical indicators of renal injury. This supports a close association between its expression abundance and the progression from diabetes to nephropathy. More importantly, functional experiments indicate that modulating LINC00861 expression in renal cells specifically regulates key molecules involved in core pathological processes of DN, such as oxidative stress and inflammation. These results establish an active role in driving kidney injury under diabetic conditions. Therefore, we propose that LINC00861 holds dual significance. On one hand, its gradual increase in expression may serve as a dynamic risk marker for monitoring the transition from diabetes to nephropathy. On the other hand, it plays a crucial pathogenic role locally in the kidney, acting as a key link connecting metabolic disturbances to end-organ damage. This provides a theoretical basis for its potential role in the treatment of diabetic kidney disease.
In vitro, studies revealed that exposure to a HG milieu markedly upregulates LINC00861 expression in HMCs. This upregulation was associated with enhanced secretion of pro-inflammatory mediators, aggravated oxidative stress parameters, and increased cellular proliferative capacity, suggesting a role for LINC00861 in renal cell damage under hyperglycemic conditions. Therefore, our observations support a key theory in diabetic nephropathy “metabolic memory” which suggests that a history of poor glycemic control can exert a long-term impact on renal health.24,25 Functional assays, including gain- and loss of function studies, established that LINC00861 directly modulates inflammatory processes: knockdown of LINC00861 attenuated the high glucose-triggered inflammatory cytokine release, whereas its overexpression amplified such effects. This regulatory pattern parallels the functions of established inflammatory mediators in DN.26,27 Of particular significance, LINC00861 also promoted mesangial cell proliferation, implying its potential involvement in accelerating renal injury through dual pro-inflammatory and pro-proliferative mechanisms. This functional profile aligns closely with key pathological hallmarks of DN, namely mesangial matrix expansion and fibrosis. 28
At the mechanistic level, this work employed a combination of silico analysis, luciferase-based reporter gene assays, and RIP to confirm the direct binding between LINC00861 and miR-378a-3p, indicating that LINC00861 may function as a competitive endogenous RNA (ceRNA) to repress miR-378a-3p expression. Rescue assays demonstrated that suppressing miR-378a-3p abolished the protective outcomes resulting from LINC00861 knockdown, thereby reinforcing the conclusion that LINC00861 promotes inflammatory responses, oxidative stress, and cellular proliferation through sponging miR-378a-3p. Together, these results offer valuable insights into the regulatory mechanism of LINC00861 in DN.
The findings of this study underscore the pivotal involvement of inflammatory processes in the development and progression of DN. Patients with DN exhibited markedly increased serum concentrations of IL-1β, IL-6, and TNF-α, which aligns with earlier reports implicating the TLRs/MyD88 axis 29 and NLRP3 inflammasome activation. 30 A positive association was observed between LINC00861 expression and the levels of these pro-inflammatory cytokines, indicating a possible regulatory role for LINC00861 in mechanisms such as macrophage polarization or NF-κB signaling. 5 Moreover, upregulation of linc00861 enhanced cellular proliferation under high glucose conditions, consistent with maladaptive repair processes that drive renal fibrosis in DN. 31 Collectively, these results suggest that LINC00861 facilitates disease advancement by amplifying inflammatory pathways.
Notwithstanding its findings, the present investigation is subject to several limitations. The clinical samples analyzed were obtained from a single center, and the cohort size was relatively limited, which may restrict the generalizability of the results. Thus, further direct validation using a larger collection of human tissue samples is warranted.
In conclusion, our work provides evidence that LINC00861 is elevated in the serum of DN patients, demonstrates considerable diagnostic accuracy, and shows a strong correlation with markers of renal impairment. At the mechanistic level, LINC00861 acts as a molecular sponge for miR-378a-3p, thereby modulating downstream signaling events that exacerbate inflammatory activation, oxidative stress, and proliferation in mesangial cells, which collectively contribute to DN progression. These insights not only highlight the potential of LINC00861 as an early diagnostic biomarker but also offer a conceptual framework for targeting the LINC00861/miR-378a-3p axis in future therapeutic strategies.
Footnotes
Ethical considerations
The study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of The Third Xiangya Hospital of Central South University before the study began.
Consent to participate
The written informed consent has been obtained from the participants involved.
Author contributions
Conceptualization, H.L., M.P., X.H., D.F., Q.X.; Data curation, H.L., M.P., X.H., D.F., Q.X.; Formal analysis, M.P., X.H., D.F.; Funding acquisition, D.F.; Investigation, M.P., X.H.; Methodology, H.L., M.P., X.H., D.F., Q.X.; Project administration, D.F., Q.X.; Resources, M.P., X.H.; Software, H.L., M.P., X.H., D.F.; Supervision, D.F., Q.X.; Validation, M.P., X.H.; Visualization, M.P., X.H.; Roles/Writing - original draft, M.P., X.H.; Writing - review & editing, H.L., D.F., Q.X.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
