Abstract
Although urinary albumin is the well-known non-invasive marker for diabetic nephropathy, its sensitivity is relatively low. To select more adequate marker, we examined whether urinary tubular markers were more sensitive than albumin using spontaneous type 2 diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) rats. The OLETF rats exhibited histopathological alterations in glomeruli and tubules at 14 weeks of age, but there were no significant differences in the urinary albumin between OLETF and control, Long-Evans Tokushima Otsuka (LETO), rats at 10–16 weeks of age. In the OLETF rats, urinary excretions of N-acetyl-β-
Introduction
Diabetic nephropathy, one of the common microvascular complications of diabetes mellitus, is the main cause of mortality and morbidity. 1 About 40% of patients with type 2 diabetes are at risk of developing chronic kidney disease (CKD). 2 Although microalbuminuria is used as a marker of early-stage diabetic nephropathy, several studies have reported that type 2 diabetic patients with normoalbuminuria have already lower glomerular filtration rate (GFR).3–5 Moreover, the UK Prospective Diabetes Study demonstrated that 51% of type 2 diabetic patients who progressed to CKD had no preceding albuminuria. 6 Therefore, more sensitive markers than urinary albumin are needed to accurately detect early stage of nephropathy in type 2 diabetic patients.
Traditionally, glomerular dysfunction is thought to be a major factor for the development and progression of diabetic nephropathy.7,8 However, it is increasingly appreciated that the renal tubules play a role in the pathogenesis of diabetic nephropathy, due to long exposure to a variety of metabolic and haemodynamic injuring factors. 9 In general, tubular injury is associated with albuminuria via the reduction of albumin reabsorption in the damaged proximal tubules. 10 In addition, it is reported in a cross-sectional study that urinary tubular markers, such as kidney injury molecule-1 (Kim-1) and neutrophil gelatinase–associated lipocalin (NGAL), are independently associated with albuminuria at the early stage of nephropathy in patients with type 2 diabetes. 11 Kim-1 is a type 1 membrane protein expressed at negligible level in normal rats and is massively induced in the tubules after ischaemic or toxic injury in rats.12,13 NGAL, a small protein that belongs to the lipocalin protein family, is produced in epithelial cells and neutrophils in most tissues and is a marker of renal tubular injury. 14 However, it remains unclear whether these biomarkers would be more sensitive than albuminuria during the development of type 2 diabetic nephropathy.
Previously, we showed that urinary concentration of vanin-1, which is an epithelial glycosylphosphatidylinositol-anchored pantetheinase,15,16 was elevated before conventional markers in rats with the nephrotoxicant- and drug-induced renal tubular injury.17,18 A recent study showed that the protein level of renal vanin-1 increased in streptozotocin-induced type 1 diabetic rats, and urinary vanin-1 was detected in patients with type 1 diabetic nephropathy.
19
However, it remained unknown whether urinary vanin-1 is a more sensitive marker than urinary albumin during the course of type 2 diabetes. The purpose of this study was to evaluate a potential utility of urinary vanin-1, Kim-1, N-acetyl-β-
Materials and methods
Animals
All animal procedures were approved by the Animal Care and Use Committee of Jichi Medical University (Shimotsuke, Japan) and performed in accordance with the guidelines for animal research. Male spontaneous type 2 diabetic OLETF rats (
Patients
A total of 32 patients were recruited from the outpatient clinic at Kanazawa University Hospital (Kanazawa, Japan) (Table 1). Nine patients were non-diabetic and treated for endocrine diseases, including thyroid tumour and hypothyroidism, whereas 23 patients were treated for type 2 diabetes. A spot urine sample was obtained from all participants. The levels of urinary albumin excretion (UAE) were lower than 20 mg/g creatinine in all non-diabetic and 12 diabetic patients. On the other hand, albuminuria, defined as microalbuminuria (UAE 30–300 mg/g creatinine) or macroalbuminuria (UAE ≥ 300 mg/g creatinine), was detected in the remaining 11 diabetic patients (microalbuminuria,
Characteristics of patients with and without type 2 diabetes.
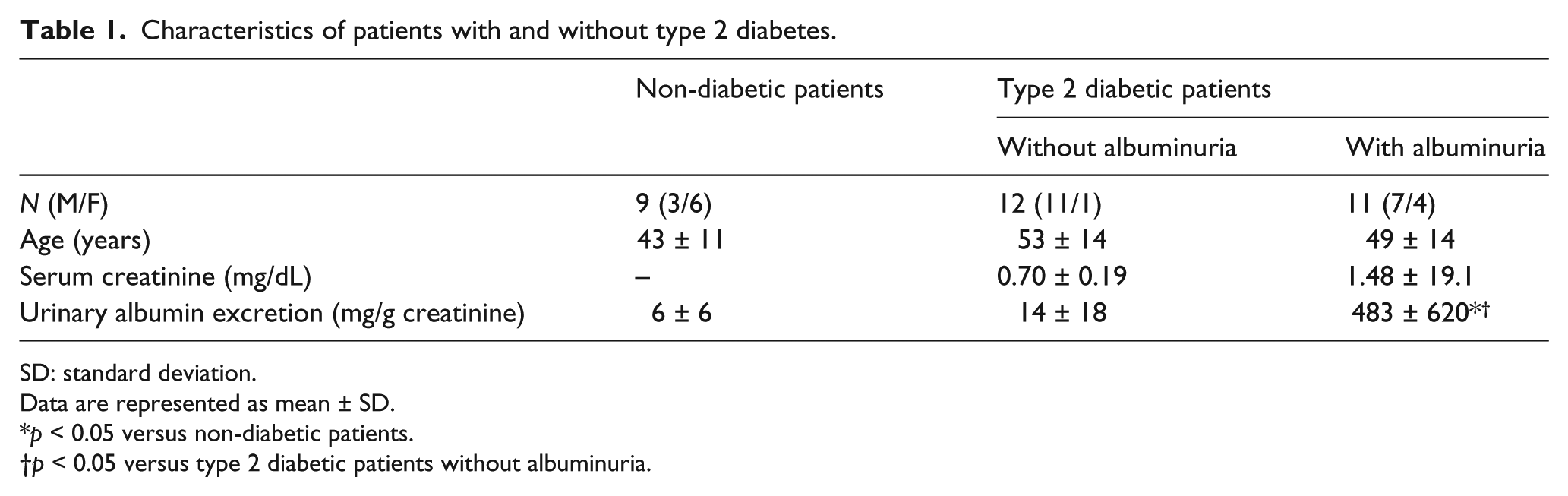
SD: standard deviation.
Data are represented as mean ± SD.
The clinical study was approved by the Ethics Committee of Kanazawa University (Kanazawa, Japan) and was conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent.
Laboratory measurements
After urine and blood samples were centrifuged at 1000
Measurement of vanin-1, Kim-1 and NGAL protein in the kidney
Sample lysates were prepared from the kidneys of rats at 14 and 50 weeks of age. After measurement of total protein concentration using bicinchoninic acid assay (BCA) protein assay kit (Thermo scientific, Rockford, IL, USA), 25 µg of total protein was loaded onto the microplate for ELISA of vanin-1, Kim-1 and NGAL.
Histopathology
Kidney was fixed in 10% formalin/phosphate-buffered saline overnight, dehydrated by passing through an ascending ethanol series, and embedded in paraffin wax. The paraffin-embedded tissue sections (4 µm) were deparaffinized in xylene, rehydrated with a series of alcohol washes and then stained with periodic acid-Schiff (PAS) reagent and haematoxylin. Renal tissues of LETO and OLETF rats were examined with a light microscope. Tubular damage (atrophy and dilatation) was graded from 0 to 4 (0 = normal; 1 = mild; 2 = moderate; 3 = severe and 4 = very severe) 21 with a minor modification. Each score was evaluated in a blind manner.
Statistical analysis
Data are expressed as the means ± standard error of the mean (SEM). Variables were compared using the unpaired
Results
Time courses of body weight and blood glucose in rats
Because we focused on the early phase in the course of type 2 diabetes, we investigated the changes of the biomarkers in animals up to 22 weeks of age. As shown in Figure 1, animals gained body weight in an age-dependent manner, and the OLETF rats exhibited the larger body weight than the LETO rats. In the OLETF rats, blood glucose concentration was significantly higher than that in the LETO rats at 14 weeks of age, and the difference between these animals became more prominent at 22 weeks of age.
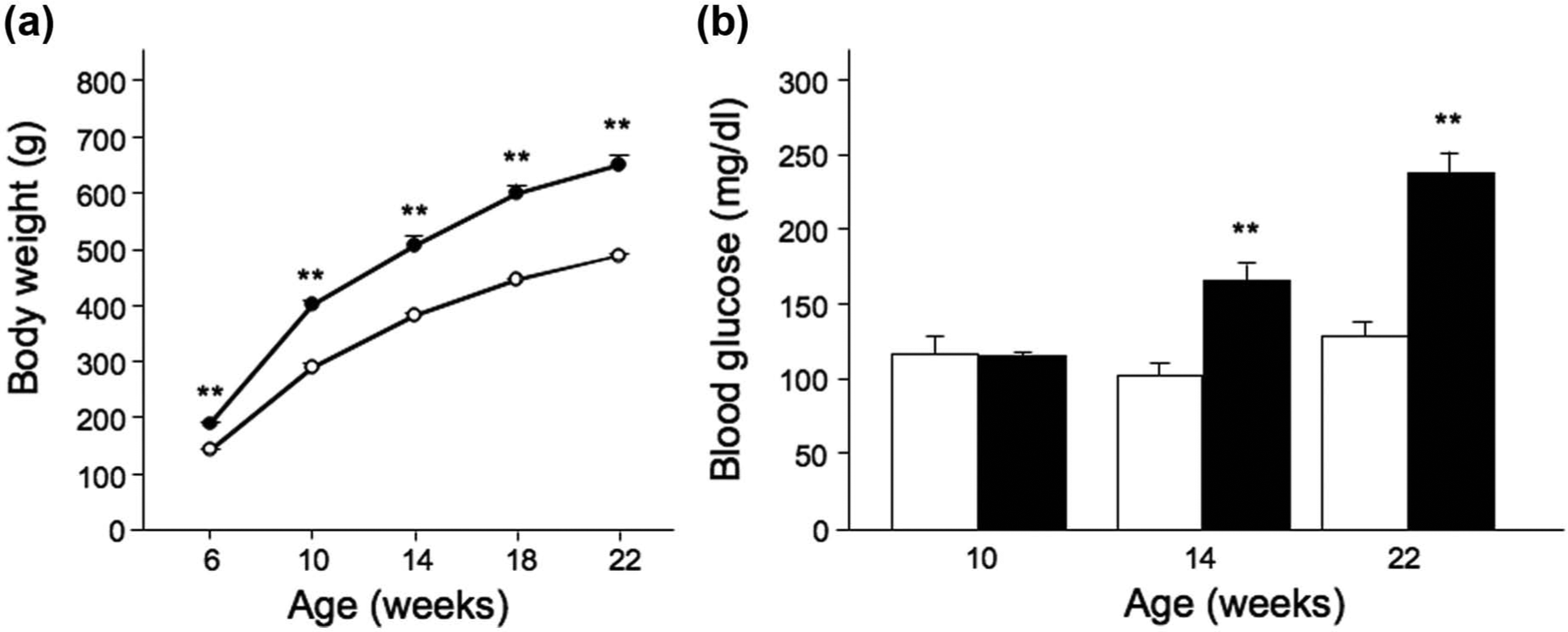
Changes in body weight and blood glucose concentration: (a) body weight in LETO (
Time courses of renal function and histology in rats
There were no significant differences in serum creatinine between OLETF and LETO rats at 10 and 22 weeks of age (Figure 2(a)). However, serum creatinine at 50 weeks of age was significantly higher in the OLETF than in the LETO rats (online Supplementary Table 1). Creatinine clearance (CCr) in the OLETF rats was similar to that in the LETO rats at 10 weeks of age, and subsequently, hyperfiltration was detected in the OLETF rats at 22 weeks of age (
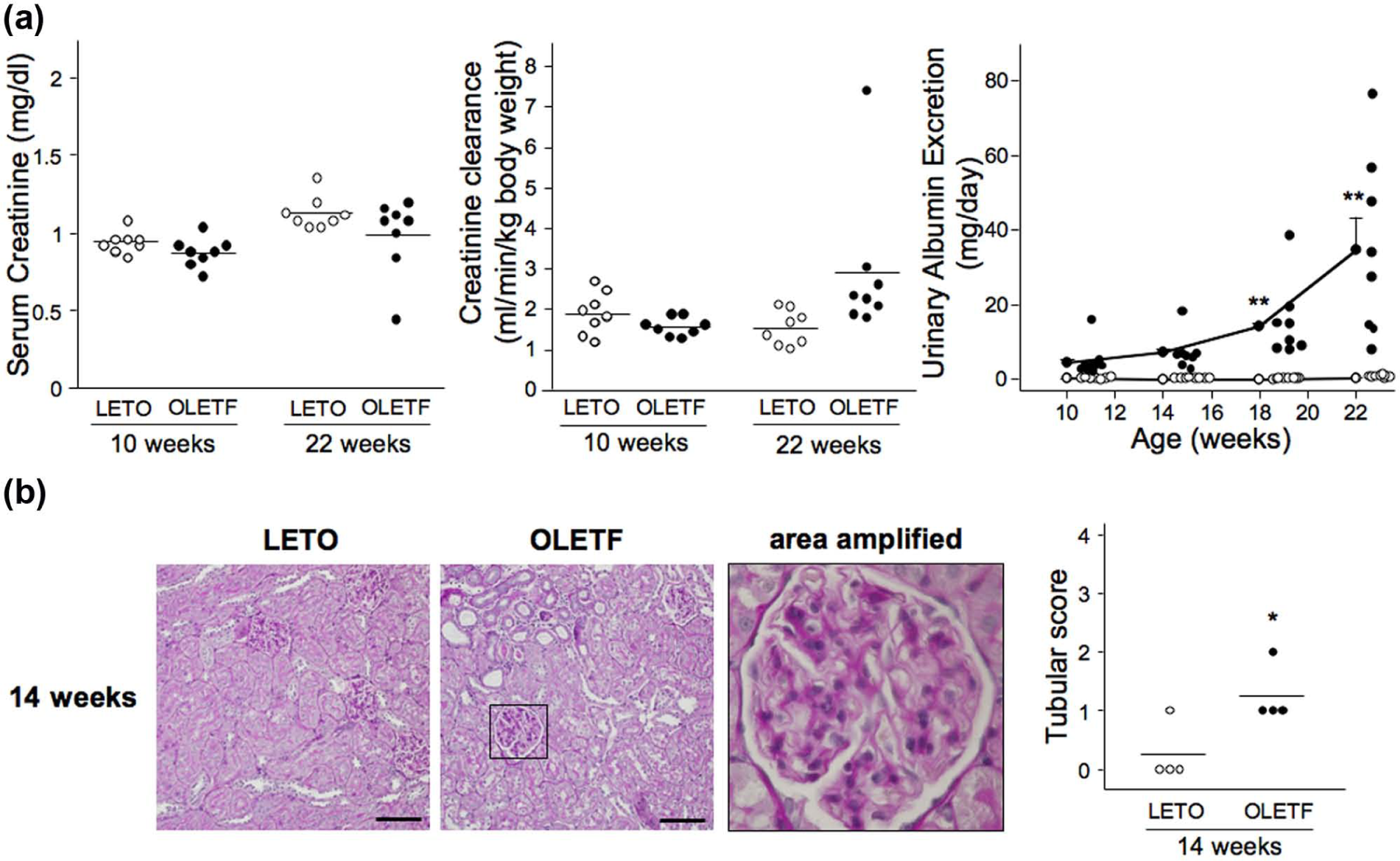
Changes in renal parameters and histopathology: (a) blood samples were taken from tail veins of animals at 10 and 22 weeks of age. Animal was placed individually in a metabolic cage, and 24-h urine samples in LETO (
There was no significant difference in the UAE between OLETF and LETO rats at 10–14 weeks of age. Importantly, the OLETF rats had already exhibited histopathological alterations in glomeruli with mesangial sclerosis and in tubular damage assessed as dilatation and atrophy at 14 weeks of age (Figure 2(b)).
Time courses of urinary tubular biomarkers in rats
Next, we investigated whether urinary tubular biomarkers, vanin-1, Kim-1, NGAL and NAG, were more sensitive than urinary albumin in the OLETF rats. Figure 3(a) showed that urinary vanin-1 in the OLETF rats was significantly higher at 18 weeks of age than that in the LETO rats, but the increase was transient. On the other hand, urinary excretion of Kim-1 significantly increased in the OLETF rats at 14 weeks of age, which continued up to 22 weeks of age. In addition, urinary Kim-1 significantly elevated at 50 weeks of age in the OLETF rats (online Supplementary Table 1). There was no significant increase in urinary NGAL throughout the study period (Figure 3(a)). Urinary NAG elevated only at 22 weeks of age in the OLETF rats.
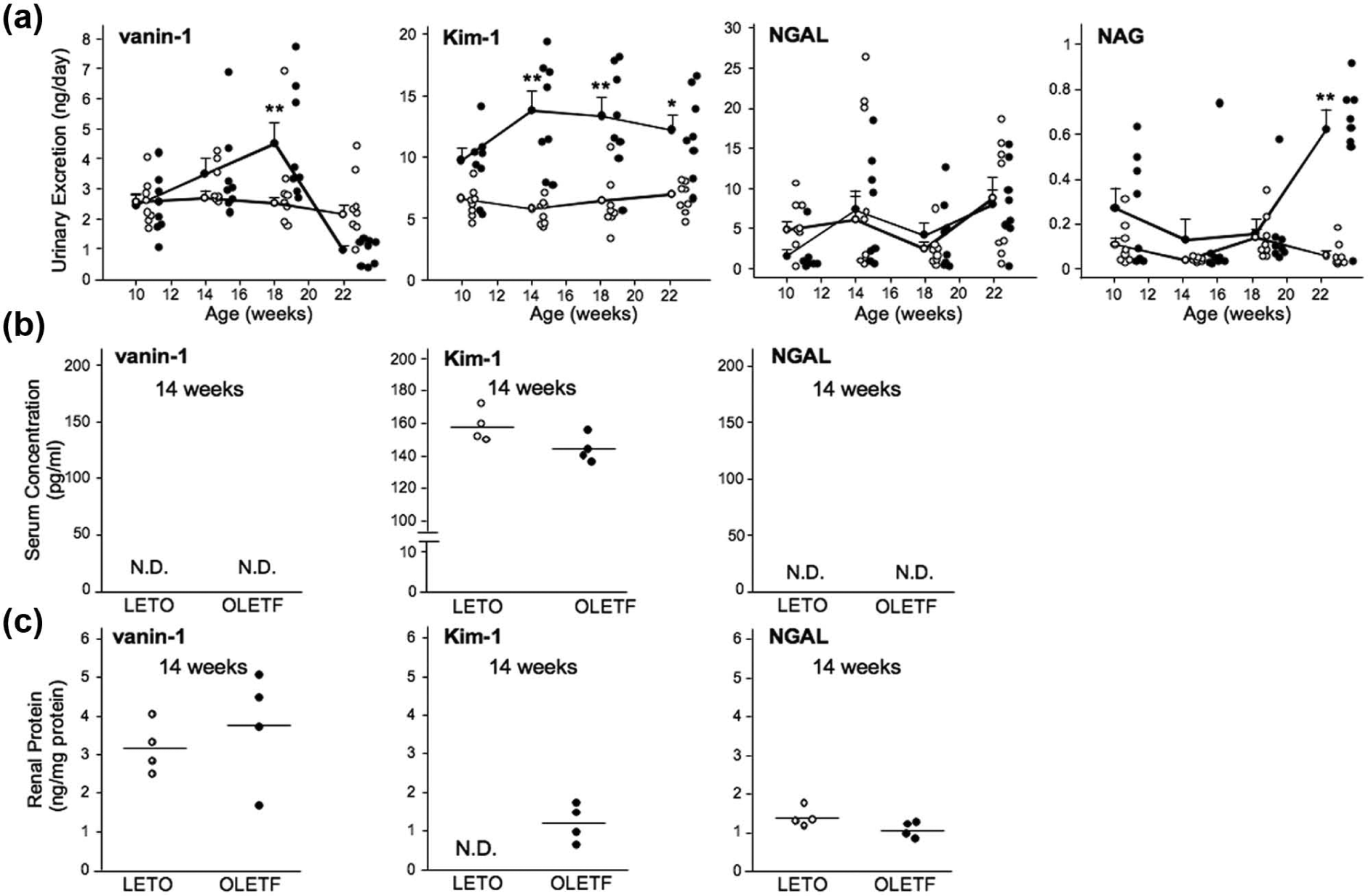
Urinary excretion and serum and renal concentrations of the biomarkers: (a) animal was placed individually in a metabolic cage, and 24-h urine samples in LETO (
Because urinary Kim-1 started to increase at 14 weeks of age, we examined its serum concentration and renal protein expression in the OLETF rats at this point. Serum concentration of Kim-1 was detected in the OLETF rats without significant increase compared to that in the LETO rats (Figure 3(b)). Renal protein expression of Kim-1 could be detected in the OLETF but not in the LETO rats (Figure 3(c)). Serum concentrations of vanin-1 and NGAL were undetectable not only in the LETO but also in the OLETF rats at 14 weeks of age. The protein expressions of vanin-1 and NGAL in the kidney of the OLETF rats were similar to those in the LETO rats at 14 weeks of age (Figure 3(c)).
Urinary tubular biomarkers in humans
Finally, we investigated whether the urinary tubular biomarkers were also detectable in type 2 diabetic patients with and without albuminuria (Table 1). As shown in Figure 4, urinary vanin-1 was not detected in the non-diabetic and diabetic patients without albuminuria. On the other hand, vanin-1 was detected in 1 of 7 diabetic patients with microalbuminuria and 2 of 4 patients with macroalbuminuria. Urinary human KIM-1 tended to be higher in the 23 diabetic patients than in non-diabetic patients (
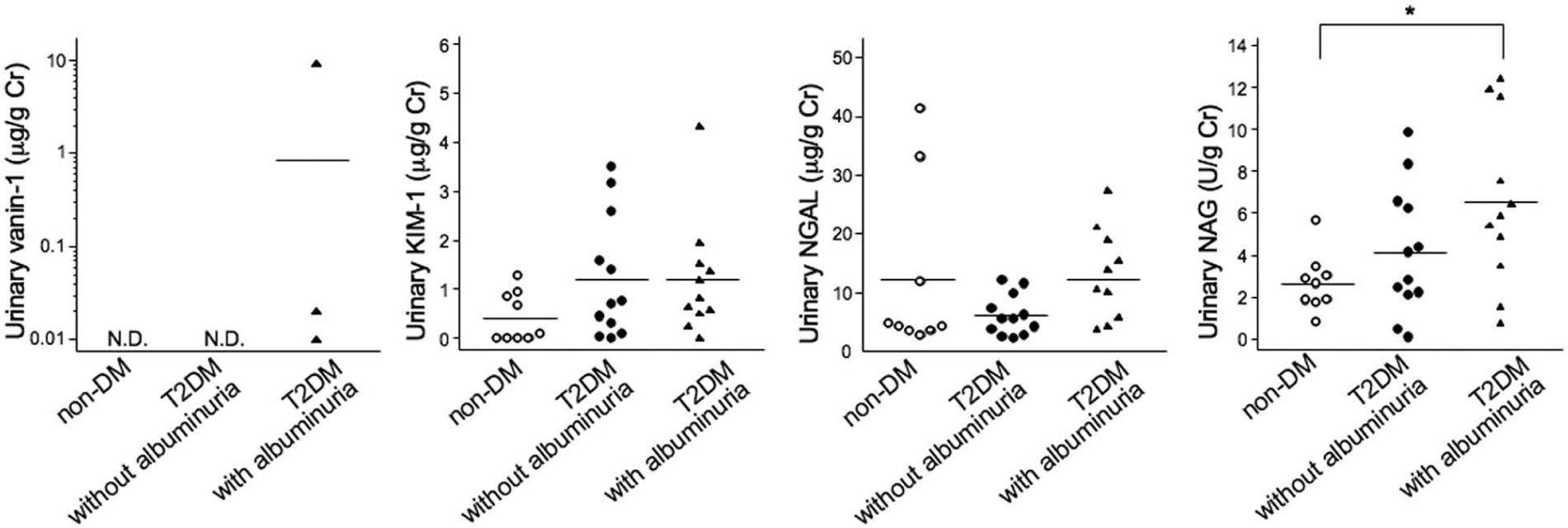
Newly developed urinary biomarkers in patients with and without type 2 diabetes. A spot urine sample was obtained from non-diabetic patients (non-DM) (
Discussion
In this study, urinary vanin-1 and Kim-1, both tubular damage markers, showed significant elevation before overt albuminuria in the OLETF rats with histopathologically proven renal tubular injury. Urinary Kim-1 concentration continued to be higher after appearance of albuminuria, whereas the elevation of urinary vanin-1 was transient. Microalbuminuria appears when significant glomerular damage has occurred, 22 but is not necessarily reflected in renal impairment because nephropathy sometimes occurs in the normoalbuminuric patients.6,23 Therefore, urinary Kim-1 without albuminuria could facilitate detection of renal tubular injury in type 2 diabetes.
Several studies have shown that urinary tubular markers were elevated in animal models of type 1 diabetic nephropathy19,24 and in type 1 diabetic patients with albuminuria.25–27 In streptozotocin-induced type 1 diabetic mice, urinary NGAL concentration was found to be markedly increased compared to that of control mice. 24 On the other hand, this study revealed that urinary NGAL was not elevated in the early stage of nephropathy of type 2 diabetic OLETF rats. In type 1 diabetes, major structural changes involve the glomerulus predominantly, whereas light microscopy studies have shown that a substantial proportion of type 2 diabetic patients have more advanced tubular and vascular than glomerular lesions. 28 Urinary NGAL reflects defects in glomerular filtration barrier, proximal tubule reabsorption and distal nephrons in type 1 diabetic animals. 24 Because type 2 diabetic animals were used in this study, influence of diabetes on urinary NGAL might be small. Unlike NGAL, Kim-1 is expressed on the apical membrane of proximal tubules, and its ectodomain is cleaved and released into the lumen of the tubules. Possibly due to renal localization of Kim-1, urinary Kim-1 reflected apparent histopathologically proven renal tubular injury in type 2 diabetic rats.
Previously, we showed that urinary vanin-1 was detected earlier than other biomarkers including Kim-1 in rats with the nephrotoxicant- and drug-induced acute kidney injury.17,18 In this study, urinary Kim-1 was detected earlier than vanin-1. This difference might be at least partly due to the difference of pathophysiological conditions between the type 2 diabetic nephropathy and drug-induced acute kidney injury. Specifically, chronic exposure of renal cells to high glucose concentrations is known to activate the p38 Mitogen-Activated Protein Kinase (MAPK) pathway. 29 Although vanin-1 is not associated with p38 MAPK pathway, the cleavage of Kim-1 is accelerated by p38 MAPK activation. 30 The present finding that glucose concentration in the OLETF rats was significantly higher than that in the LETO rats at 14 weeks of age might lead to early detection of urinary Kim-1 in this animal model.
In this study, the findings obtained in animal study were confirmed by the clinical data. Urinary vanin-1 was detected in a subset of type 2 diabetic patients with albuminuria. Urinary NGAL was not increased in type 2 diabetic patients, even in those with albuminuria. In addition, urinary human KIM-1 tended to be elevated in diabetic patients compared to non-diabetic patients, although there was no significant difference between diabetic patients with and without albuminuria. Nauta et al. 27 also showed that urinary KIM-1 increased in diabetic patients, regardless of development of albuminuria. Recently, Kim et al. 11 reported that urinary KIM-1 levels were significantly higher in type 2 diabetic patients with albuminuria than in non-diabetic control subjects, but such an elevation was not detected in diabetic patients without albuminuria. In addition, urinary NGAL levels were significantly higher in diabetic patients with macroalbuminuria, but not in those with microalbuminuria. 11 The discrepancies between our and their results might be, at least in part, due to the differences in degree of tubular injury in diabetic patients with and without albuminuria. The data from these clinical and animal studies suggest that urinary Kim-1 might be detectable before the development of albuminuria.
In summary, we found that urinary Kim-1 and vanin-1 were elevated before appearance of albuminuria in type 2 diabetic OLETF rats. The increase of urinary vanin-1 was transient, whereas that of Kim-1 continued even after the progression of nephropathy. Therefore, urinary Kim-1 would be a sensitive biomarker of nephropathy before overt albuminuria in these type 2 diabetic animals. Merit of urinary Kim-1 in diabetic patients remains to be determined in further clinical studies.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was supported by a Grant-in-Aid for Scientific Research (24790535) from the Ministry of Education, Culture, Sports, Science and Technology, Japan (K.H.); by a Grant-in-Aid for Japan Research Foundation for Clinical Pharmacology (K.H.) and by a Grant-in-Aid for Research on Advanced Medical Technology from the Ministry of Health, Labor and Welfare of Japan (A.F. and H.A.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
