Abstract
Visceral adiposity is a pivotal pathogenic driver of type 2 diabetes mellitus (T2DM) and its vascular complications. We performed two-sample Mendelian randomization (MR) analyses to examine the causal relationships between visceral adipose tissue (VAT) accumulation and diabetes complications. Single nucleotide polymorphisms (SNPs) specific to VAT accumulation were identified through large-scale genome-wide association studies (GWAS) at genome-wide significance (P < 5 × 10-8), using the inverse variance weighted (IVW) approach for primary analyses. The MR-IVW random-effects model revealed significant associations between genetically predicted VAT accumulation and increased risks for T2DM complications: multi-organ (OR = 2.58, 95% CI, 2.21–3.03), neurological (OR = 3.58, 95% CI, 2.34–5.47), retinopathy (OR = 2.90, 95% CI, 2.21–3.81), nephropathy (OR = 3.14, 95% CI, 2.23–4.45), and peripheral vascular (OR = 3.32, 95% CI, 2.31–4.76). Consistency across complementary MR methods supported the causal inference. This study provides genetic evidence that VAT accumulation is causally associated with increased risk of multiple T2DM complications, including neurological, ocular, kidney, and peripheral vascular outcomes. Future research should prioritize mechanistic studies to elucidate VAT-driven inflammatory and lipotoxic pathways in diabetic complications.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a global health crisis, projected to affect over 780 million people by 2045. 1 T2DM leads to significant damage to various organs, including the cardiovascular system, brain, kidneys, eyes, and peripheral nerves, significantly impacting quality of life and healthcare costs. 2 Adipose tissue comprises two main compartments: visceral adipose tissue (VAT) and subcutaneous adipose tissue, each with distinct metabolic effects. 3 Growing evidence suggests that VAT accumulation has more adverse effects on cardiovascular and cerebrovascular diseases and diabetic complications compared to subcutaneous fat.4–6 This pathogenic fat depot releases inflammatory cytokines and hormones that worsen insulin resistance, but direct evidence linking it to diabetes complications remains limited. Conventional observational studies are limited in establishing causal relationships due to potential confounding by dietary patterns, lifestyle factors, and medication use. Mendelian randomization (MR) overcomes confounding biases inherent in observational studies by using genetic variants as instrumental variables, emulating randomized controlled trial principles.7,8 The present study conducted a two-sample MR analysis to evaluate the causal links between genetic susceptibility to VAT accumulation and diabetes-related organ damage.
Research design and methods
Data acquisition
We conducted a two-sample MR analysis to investigate the potential causal relationship between VAT and complications associated with T2DM. Figure 1 illustrates our analytical framework. The MR design is based on three fundamental assumptions.
8
Genetic instrumental variables (IVs) for VAT accumulation were derived from a large-scale Genome-Wide Association Study (GWAS) conducted by Karlsson et al..
9
This study utilized data from the UK Biobank creating two sub-cohorts that enabled the estimation of predicted VAT accumulation in 325,153 individuals of White British ancestry. The summary data for predicted VAT from this GWAS can be accessed at UK Biobank GWAS Summary Statistics. Outcomes related to T2DM complications were obtained from the publicly available GWAS of European descent in FinnGen R9 release, which can be found at FinnGen R9 Summary Statistics.
10
Flow chart of Mendelian randomization analysis.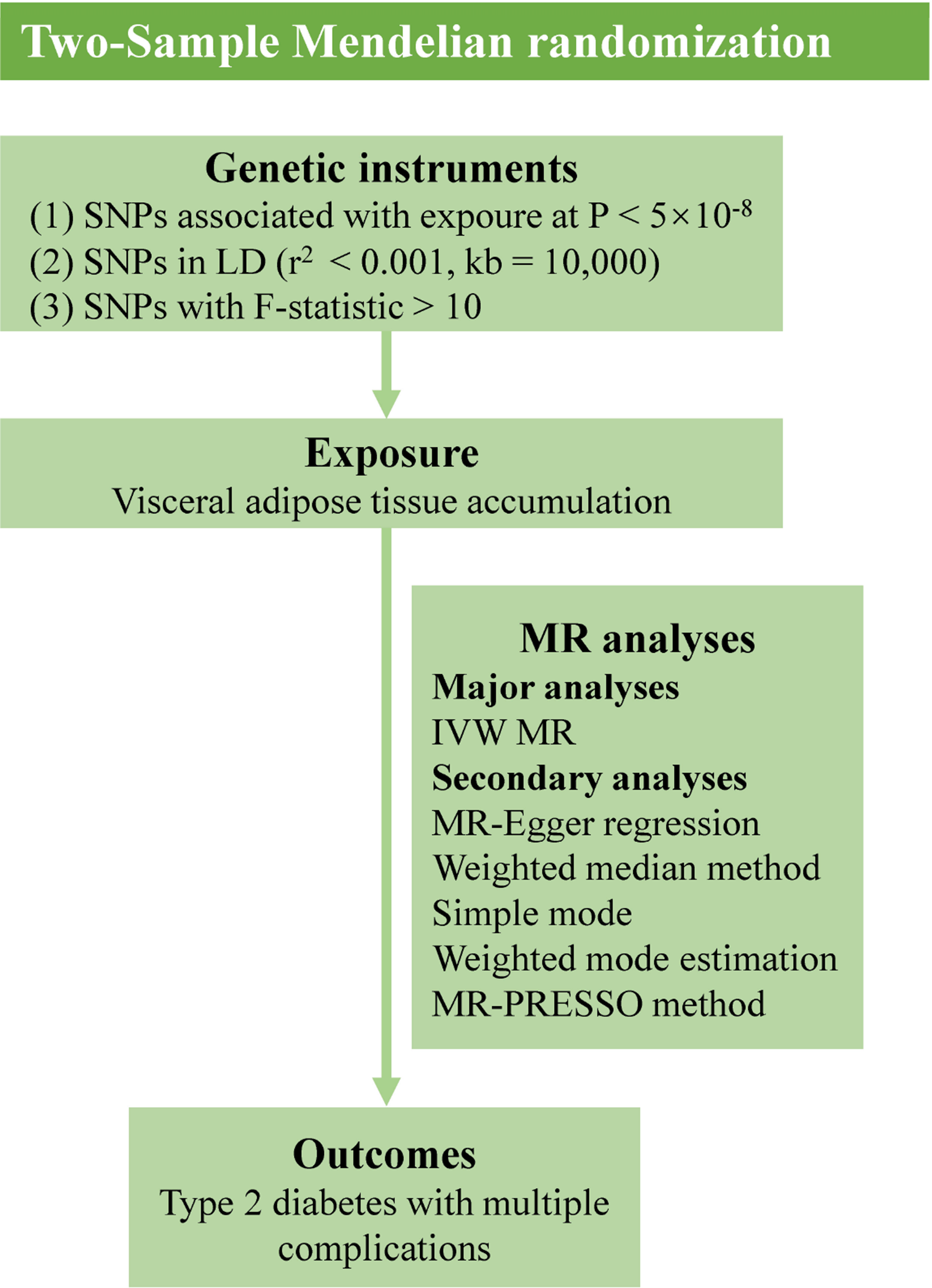
Mendelian randomization
To minimize bias from sample overlap, we selected VAT data from the UK Biobank and T2DM data from the FinnGen study. We identified single-nucleotide polymorphisms (SNPs) associated with VAT at genome-wide significance (P < 5 × 10-8). These SNPs were further clumped using the PLINK clumping algorithm, applying a linkage disequilibrium (LD) threshold of r2 < 0.001 within a 10,000-kb window to ensure independence among the selected SNPs. The instrument strength of selected SNPs was assessed by calculating the F-statistics. An F-statistic greater than 10 indicates that the correlation between the IVs and VAT is sufficiently strong to mitigate bias from weak IVs.
Statistical analysis
The inverse-variance weighted (IVW) method served as the primary approach in our MR analysis, combining SNP-specific causal estimates for T2DM complications after excluding outlier SNPs. Additionally, we complemented the robustness of the IVW results with other MR methods, including MR-Egger, weighted median, simple mode, and weighted mode estimation. Several sensitivity analyses were performed to ensure the stability of the results. Heterogeneity among SNPs was evaluated using Cochran’s Q, where P < 0.05 was considered statistically significant. MR-Egger regression was used to assess potential directional pleiotropy, where an intercept term with P < 0.05 suggests bias. We also used an MR-Pleiotropy RESidual Sum and Outlier (MR-PRESSO) method to identify and correct for observed outlier SNPs and potential horizontal pleiotropy. 11 All MR analyses were performed in the R environment (V.4.0.2; R Foundation for Statistical Computing, Vienna, Austria) using “TwoSampleMR (v0.5.6)” and “MR-PRESSO (v1.0)” packages.
Results
Following the exclusion of SNPs exhibiting linkage disequilibrium, palindromic SNPs, and duplicated SNPs (Supplemental Table S1), we identified 203 genetic variants to serve as instrumental variables for investigating the causal relationship between VAT accumulation and diabetic complications. The harmonized data sets derived from the MR analysis are available in the Supplementary Dataset.
Associations of genetically predicted visceral adipose tissue and T2DM with multiple complications.

aThe Mendelian randomization analysis was performed based on random-effects inverse-variance weighted model.
Abbreviations: T2DM, type 2 diabetes; SNPs, single nucleotide polymorphisms; OR, odds ratio; CI, confidence interval; IVW, inverse-variance weighted.
MR-Egger intercepts (P > 0.05) indicated the absence of unbalanced horizontal pleiotropy (Supplemental Table S3
Discussion
The present two-sample MR study provides genetic evidence supporting a causal role of VAT accumulation in the increased risk of developing organ complications in individuals with T2DM.
The relationship between visceral fat accumulation and diabetes has received widespread attention in recent studies. VAT accumulation is not only associated with the onset of diabetes but also closely linked to an increased risk of various complications. Research indicates that VAT accumulation can lead to increased insulin resistance, thereby exacerbating diabetes.12,13 First, an earlier study underscored VAT as a critical determinant of atherosclerotic risk in T2DM by finding a positive correlation between VAT area and carotid atherosclerosis severity, 14 suggesting its direct involvement in vascular pathology. Moreover, the visceral-to-subcutaneous fat ratio (V/S ratio) has been identified as an independent predictor of major adverse cardiovascular events, 15 further highlighting the superior prognostic value of fat distribution metrics over absolute adiposity measures. Second, VAT accumulation is also closely related to metabolic dysregulation and risk stratification in individuals with diabetes. Studies have shown that the Visceral Adipose Index (VAI) is an effective indicator for assessing cardiovascular metabolic risk.16,17 Multiple longitudinal studies have indicated the superior predictive validity of the VAI in identifying high-risk individuals for prediabetes and incident type 2 diabetes compared to conventional anthropometric measures.18,19 Third, VAT accumulation is also related to microvascular and macrovascular complications in patients with diabetes. Studies have shown that an increase in VAT accumulation is associated with the increased incidence of retinopathy in Japanese type 2 diabetes patients, specifically white matter lesions and lacunar infarcts—independent of traditional risk factors.19–21 Emerging evidence from cross-sectional studies further underscores the clinical relevance of VAT in increased urinary albumin excretion, diabetic nephropathy,22,23 and diabetic retinopathy. 24 Finally, studies implicate visceral adiposity in cerebrovascular pathology, extending beyond stroke risk to cerebral microvascular dysfunction. Multimodal imaging studies demonstrate that visceral fat accumulation correlates with impaired cerebral blood flow autoregulation and vascular reactivity, 25 while independently associating with small vessel disease markers. 26 Therefore, in-depth analysis of the relationship between visceral fat and diabetes complications will help to improve management and reduce the risk of complications in individuals with diabetes.
A number of potential mechanisms link VAT to diabetes complications. One key mechanism linking VAT to diabetes complications is the release of bioactive substances from this highly metabolically active tissue. These substances, including adipokines and pro-inflammatory cytokines,27,28 play a crucial role in the development of vascular complications.27,29,30 Moreover, hypoxia-inducible factor-1 alpha (HIF-1α) plays a critical role in adipose tissue remodeling, 31 levels of which are increased in obesity and trigger a profibrotic reaction and the infiltration of M1 macrophages, which contribute to systemic inflammation and endothelial dysfunction. 32
When interpreting our results, several limitations should be noted. First, undetected sample overlap between VAT-associated SNPs and diabetic complications may introduce bias, despite filtering weak instruments (F-statistics >10). Second, participant overlap between VAT accumulation and diabetic complications GWAS sources cannot be fully excluded, potentially inflating causal estimates, despite harmonizing ancestry-matched cohorts. Third, European-derived genetic data limit generalizability to populations with distinct adiposity patterns. Finally, clinical translation requires accessible imaging for precise VAT accumulation quantification, while heterogeneity in complication phenotyping may attenuate causal effect estimates.
Conclusion
In conclusion, the current study provides evidence of a positive causal effect of VAT accumulation in T2DM complications. These findings underscore VAT as a therapeutic target for mitigating diabetic end-organ damage, while also calling for future studies to fully understand the mechanisms involved.
Supplemental Material
Supplemental material - Visceral Adipose Tissue Accumulation and Risk of Organ Complications among Individuals with Type 2 Diabetes Mellitus: A Two-Sample Mendelian Randomization Study
Supplemental material for Visceral Adipose Tissue Accumulation and Risk of Organ Complications among Individuals with Type 2 Diabetes Mellitus: A Two-Sample Mendelian Randomization Study by Xingji Lian, Xiaomin Zeng, Bing Zhang, Yan Huang, Xiaohui Peng, Yanhui Wang, Chongzhe Yang, Yuyu Huang in Diabetes & Vascular Disease Research.
Supplemental Material
Supplemental material - Visceral Adipose Tissue Accumulation and Risk of Organ Complications among Individuals with Type 2 Diabetes Mellitus: A Two-Sample Mendelian Randomization Study
Supplemental material for Visceral Adipose Tissue Accumulation and Risk of Organ Complications among Individuals with Type 2 Diabetes Mellitus: A Two-Sample Mendelian Randomization Study by Xingji Lian, Xiaomin Zeng, Bing Zhang, Yan Huang, Xiaohui Peng, Yanhui Wang, Chongzhe Yang, Yuyu Huang in Diabetes & Vascular Disease Research.
Footnotes
Ethical approval
Ethical review and approval were waived for this study because this study was conducted on summary-level genetic data from publicly available databases of large-scale genome-wide association studies.
Author Contributions
Study concept and design: Xingji Lian, Xiaomin Zeng, Chongzhe Yang and Yuyu Huang. Drafting of the manuscript: Xingji Lian, Xiaomin Zeng. Critical revision of the manuscript for important intellectual content: Bing Zhang, Chongzhe Yang and Yuyu Huang. Statistical analysis: Yan Huang. Obtained funding: Xingji Lian, Bing Zhang, Chongzhe Yang and Yuyu Huang. Administrative, technical, or material support: Xingji Lian, Xiaohui Peng, Yanhui Wang, Bing Zhang. Study supervision: Chongzhe Yang and Yuyu Huang.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was supported by the National Natural Science Foundation of China (82170234), the China International Medical Foundation (Z-2019-42-1908-2), Health Science and Technology General Guidance Program of Guangzhou (20241A011019), KRT Plan of Guangdong Medical Development Foundation (K-20240111), and Guangzhou Planned Project of Science and Technology (2024A04J3999, 202201020512 and 2024A03J1178).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Only publicly available data were used in this study, and data sources and handling of these data were described in the Materials and Methods and supplementary files. The datasets analyzed for this study are publicly available through the IEU OpenGWAS database (![]() ) under token-authenticated access. Further inquiries can be directed to the corresponding author.
) under token-authenticated access. Further inquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
