Abstract
Objective
Diabetes-related foot infections (DFIs) are prevalent in patients with diabetes mellitus, often leading to severe complications, including amputations. This study aims to assess the efficacy and safety of systemic antibiotics in DFI treatment.
Research Design and Methods
A systematic review was conducted by searching PubMed, Cochrane databases, and Embase for randomized controlled trials up to August 4, 2024, evaluating the clinical efficacy of systemic antibiotics for DFIs. Primary outcomes were clinical efficacy and safety, comparing different antibiotic classes to penicillins. Subgroup analysis was based on DFI severity.
Results
Of 24 studies, 16 were included in the meta-analysis. Linezolid showed a potential efficacy advantage over penicillins for DFIs but had more adverse effects. Clinical efficacy and safety were comparable across carbapenems and quinolones versus penicillins. Ertapenem showed no significant difference from piperacillin/tazobactam in treating moderate or severe DFIs.
Conclusion
In conclusion, while linezolid may offer a potential efficacy advantage over penicillins in treating DFIs, it is associated with a higher risk of drug-related adverse effects. Penicillins demonstrate comparable clinical efficacy and safety to carbapenems and fluoroquinolones for DFI management. For moderate to severe DFIs, piperacillin/tazobactam and ertapenem are viable options, though treatment should be guided by local antimicrobial resistance patterns.
Keywords
Introduction
Diabetes-related foot infection (DFI) is a common complication of diabetes and is associated with morbidity, prolonged hospitalization and death in the population. 1 According to previous studies, the mortality rate of patients with DFIs can exhibit considerable variation, ranging from 5%–25%, and is contingent upon diverse determinants, including the severity of the infection, comorbidities, and the effectiveness of the treatment modality.2,3 Notably, it remains the most prevalent precipitating event resulting in lower extremity amputation,1,4,5 constituting an independent risk factor for increasing cost of treatment. 6 The contiguous dissemination of pathogens into the underlying bone is an established precursor to the onset of diabetes-related foot osteomyelitis (DFO), a debilitating condition that afflicts a significant proportion (44%–68%) of patients requiring hospitalization for DFIs.7,8 On the other hand, foot ischemia resulting from peripheral artery disease (PAD) not only engenders a noteworthy risk for wound infection but also poses a potential impediment to the therapeutic effectiveness targeting DFI management.9–12 PAD is also acknowledged as a significant risk factor for cardiovascular events, thereby entailing an increased mortality risk. 13 Consequently, the assessment of PAD status, as well as the determination of the presence and severity of concurrent infections, assumes a crucial role in controlling infections among patients living with diabetes.
DFIs are commonly of a polymicrobial nature, involving the presence of aerobic gram-positive cocci, such as Staphylococcus aureus, gram-negative bacilli including Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, as well as anaerobic microorganisms.14,15 The administration of appropriate antibiotics plays a crucial role in managing almost all cases of soft tissue DFIs. As per the current guidelines, the consensus on the optimal empiric antibiotic therapy for DFIs includes penicillins, cephems, carbapenems, fluoroquinolones, and anti-gram positive spectrum antibiotics such as daptomycin, vancomycin, and linezolid. 6 Metronidazole in combination with other antibiotics and clindamycin are also considered potential antibiotic regimens due to their effectiveness against anaerobic pathogens.6,16 However, tigecycline has been shown to be less clinically effective than ertapenem and therefore is not recommended. 17 In cases of moderate to severe DFIs, the administration of broad-spectrum antibiotics in parenteral form is considered reasonable. Furthermore, there is still a lack of clarity regarding the comparative superiority of different antibiotic groups over each other. In light of the above considerations, we conducted a systematic review and meta-analysis with the objective of comparing the clinical efficacy and safety of diverse categories of systemic antibiotics utilized in the treatment of DFIs.
Methods
Inclusion and exclusion criteria
We conducted a systematic search for randomized controlled trials (RCTs) assessing the efficacy of systemic antibiotics (administered intravenously or orally) with varying regimens (including dose, frequency, and duration) in patients with a diagnosis of DFIs with or without osteomyelitis. Our search was limited to studies that provided clear patient inclusion and exclusion criteria. The exclusion criteria for studies were as follows: (1) observational studies, (2) duplicate studies, (3) studies lacking relevant outcomes, such as cure rate and adverse effects, (4) comparison with placebo, topical antibiotics, or antiseptics, (5) evaluation of different treatment durations rather than antibiotic efficacy, (6) non-English language articles, (7) study protocols, and (8) animal studies.
Search strategy and study selection
Relevant studies published until August 4, 2024, were identified from the PubMed, Cochrane, and Embase databases. Unpublished studies were retrieved from the ClinicalTrials.gov registry (https://clinicaltrials.gov/). Keywords were based on antibiotic classification, pharmacological category, and chemical name. The following medical subject heading terms were used: foot ulcer, diabetic foot, bacitracin, framycetin, polymyxins, mupirocin, neomycin, penicillins, cephems, lactams, aminoglycosides, macrolides, quinolones, sulfonamides, trimethoprim, tetracyclines, chloramphenicol, clindamycin, metronidazole, fusidic acid, rifampin, daptomycin, vancomycin, teicoplanin, and glycopeptides (Supplemental Table S1). Article selection was independently performed by two reviewers (MCL and YMH), after which the screening results were compared. Any conflicts were resolved by a third reviewer (PJC) before the final decision. All retrieved abstracts, studies, and citations were reviewed. Additionally, we identified other studies using reference sections from the relevant papers and by correspondence with subject matter experts.
Data extraction
The baseline and outcome data were independently evaluated by two reviewers (MCL and YMH). The study design, population characteristics, infection type, percentage of osteomyelitis and PAD, antibiotic regimens, and cure of tests were extracted. The decisions recorded by the reviewers were compared, and discrepancies were resolved by a third reviewer (PJC) before the final decision.
Methodological quality appraisal
Two reviewers (YMH and MCL) independently assessed the methodological quality of each study using the risk of bias (ROB) 2.0 tool recommended by the Cochrane collaboration. 18 Several domains were assessed, including the bias arising from the randomization, deviations from intended interventions, missing outcome data, outcome measurement, and the selection of the reported result. Bias was rated as a judgment (high, low, or some concern) of individual elements and drew conclusions about the overall risk of bias.
Intervention
In this study, we investigated five distinct groups of antibiotics. Penicillins were defined to encompass piperacillin/clindamycin, piperacillin/tazobactam, ampicillin/sulbactam, penicillin, ticarcillin-clavulanate, and piperacillin. The carbapenems comprised ertapenem and imipenem/cilastatin. Moxifloxacin, levofloxacin, ofloxacin, and clinafloxacin were included in the quinolones. Cefoxitin, ceftriaxone, ceftobiprole, ceftazidime, and cephalexin were all part of the cephems. Any antibiotics that did not belong to the aforementioned categories were classified under the other group.
Outcomes
The primary endpoints were the clinical efficacy and adverse effects. Clinical efficacy was defined as cure or improvement, such as complete remission or regression of symptoms (i.e., purulent discharge, erythema, induration). Adverse effects included total adverse effects and drug-related adverse effects (e.g., diarrhea, headache, nausea, vomiting, and elevated AST/ALT). The secondary endpoints were proportion of amputation, proportion of wound healed or the median day of hospital stay.
Statistical analysis
Data were entered and analyzed using Review Manager version 5.4 (The Cochrane Collaboration, Oxford, England). A meta-analysis was performed according to the PRISMA guidelines. 19 Standard deviations were estimated from confidence interval (CI) limits or standard error. Furthermore, conflicting outcomes were analyzed using the risk ratio (RR) as a summary statistic. The precision levels of the effect sizes were reported as 95% CIs. A pooled estimate of the RR and weighted mean difference was computed using the DerSimonian and Laird random effects model. 20 This choice was predicated on the anticipated heterogeneity among the studies due to variations in study designs, participant characteristics, and intervention protocols. The random effects model assumes that the true effect size varies across studies and accounts for both within-study and between-study variances.
To evaluate the statistical heterogeneity and inconsistency of treatment effects across studies, Cochrane Q tests and I 2 statistics were used. Statistical significance was set at p < .10 for Cochrane Q tests. Statistical heterogeneity across the studies was assessed using I 2 statistics, which quantified the proportion of total outcome variability across the studies. According to the different I 2 values, the heterogeneity across the trials was classified into low (I 2 ≤ 25%), moderate (25% < I 2 < 75%), and high (I 2 ≥ 75%). Publication bias was detected using funnel plots if more than 10 studies were included in the meta-analysis.
Furthermore, subgroup analyses were conducted to assess the individual effects across different severities (Supplemental Table S2), as well as the occurrence of osteomyelitis and PAD.
Results
Characteristics of the included studies and patients
Figure 1 shows the PRISMA flow diagram of our search. A total of 6674 studies were searched from the database. After removing 729 duplicate studies using Endnote, we screened the remaining articles by title and abstract. This process eliminated 2005 non-RCTs, 3571 studies that did not meet our PICO criteria, and 116 studies with unavailable full texts. As a result, 253 studies were eligible for full-text assessment. After further exclusions, including non-English articles, compared with placebo, and compared with antiseptic or topical antibiotics, 24 trials were included in the qualitative synthesis.17,21–43 Finally, 16 trials were included in the meta-analysis.21–36 Flow chart.
Penicillins were principal comparisons in the enrolled studies. There were six trials comparing carbapenems21–26 and penicillins , seven trials comparing quinolones and penicillins,27–33 two trials comparing linezolid and penicillins,34,35 two trials comparing cephems and penicillins,37,38 two trials comparing different penicillins,39,40 one trial comparing a quinolone and a cephalosporin, 41 and three other trials comparing different antibiotics.17,42,43 The duration of treatment is diverse in the enrolled trials, ranging from 7–14 days to 14–28 days.
Baseline characteristics of enrolled RCTs.

N/D: no data, OM: Osteomyelitis, TOC: test of cure; DFI: Diabetic foot infection, CSSSI: Complicated skin and skin-structure infections, SSSTI: Severe Skin and Soft Tissue Infections, UTSA: University of Texas Health Science Center at San Antonio diabetic wound classification system, iv: intravenously; PAD: Peripheral Artery Disease, ABI: ankle/brachial index, PVD: Peripheral vascular disease, VAC: vacuum assisted closure, HTN: Hypertension, CVD: Cardiovascular diseases.
ERT: Ertapenem, TZP: Piperacillin/tazobactam, IMP: Imipenem/cilastatin, AMP/SUL: Ampicillin/sulbactam, MOXI: Moxifloxacin, AMC: Amoxicillin/clavulanate, susp.: suspension, LVX: Levofloxacin, TICC/CA: Ticarcillin-clavulanate, OFX: Ofloxacin,PCN/SUL: Penicillin/sulbactam, CNFX: Clinafloxacin, LZD: Linezolid, DAP: Daptomycin, VAN: Vancomycin, TGC: Tigecycline, PCN: Penicillin, CXT: Cefoxitin, CRO: Ceftriaxone, MET: metronidazole, BPR: Ceftobiprole, CTZ: ceftazidime, CLDM: Clindamycin, CEX: Cephalexin, PIP: Piperacillin.
aPeripheral vascular disease: diminished or absent pulses.
bModerate: Grade 0/1 with stage B/D; Severe: Grade 2/3 with stage B/D.
cModerate-to-severe: Grade 1/2 with stage B/D.
dABI <0.9 or foot pulses barely or not palpable.
eWith OM: treatment duration up to 42 days, TOC 25–27 weeks.
Quality of study
The ROB 2.0 assessment of 24 studies focused on the five domains. The randomization process showed some concerns in seven studies due to insufficient reporting of randomization methods or allocation concealment. However, most trials demonstrated low bias in terms of deviations from intended interventions, with proper adherence to treatment protocols. Missing outcome data was generally well managed across the studies, resulting in a low risk of bias for this domain. The measurement of outcomes, particularly the clinical efficacy of various antibiotic regimens, was generally robust. Nevertheless, 12 studies exhibited some concerns due to potential unblinding or subjective outcome assessment. Reporting bias was minimal, as most studies adhered to their pre-specified protocols and presented clear, detailed results.
Overall, the majority of the studies demonstrate moderate to low bias, with the most significant issues arising from randomization and outcome measurement. The methodological rigor of the studies was mostly satisfactory, making the conclusions credible, though improvements in randomization reporting and blinding could enhance their robustness. The summary of risk of bias was performed on supplemental Figure S1-2.
More than half of the enrolled trials were unclear about the random sequence generation and allocation concealment domains as the methods of sequence generation were not mentioned. Nine trials were open-label, and two trials did not have blind assessors. Most trials used intention-to-treat analysis, and the lost follow-up rate in the two trials was higher than 20%. Most trials reported all outcomes; therefore, selective reporting has a low risk of bias. The summary of risk of bias was performed on supplemental Figure S1-2.
Primary outcomes
Efficacy outcomes
Figure 2 shows the incidence and RR of clinical efficacy in different groups of individual antibiotic comparators. Penicillins were regarded as the control group for meta-analyses as it is the most common comparison in the enrolled trials. The clinical efficacy response rate was 504 out of 692 patients in the carbapenem group and 484 out of 684 patients in the penicillin group. Carbapenems showed similar clinical efficacy compared with penicillins (6 trials, 504/692 vs 484/684, RR 1.05, [95% CI: 0.96-1.13]; p = .28, I
2
= 23%) (Figure 2(A)). In the quinolones versus penicillins, the clinical efficacy response rate was 286 out of 474 in the quinolones group and 271 out of 449 in the penicillins group. The quinolones were equivalent to penicillins in clinical efficacy (7 trials, RR 1.00, [95% CI: 0.90-1.10]; p = .96, I
2
= 0%) (Figure 2(B)). When comparing Linezolid and Penicillins, the clinical efficacy response rate was 182 out of 228 in the linezolid group and 91 out of 133 in the penicillins group. Linezolid had better clinical efficacy compared to penicillins. (2 trials, RR 1.15, [95% CI: 1.01-1.31]; p = .04, I
2
= 0%) (Figure 2(C)). The supplemental Figure S3 displays the results of additional trials that were not included in the meta-analysis. None of the results yielded significant differences between antibiotics, except for the comparison between tigecycline and ertapenem (Figure S3, RR 0.77, [95%CI: 0.71-0.83]). Clinical efficacy.
Safety outcomes
The overall RR of total adverse effects was not significantly different between carbapenems and penicillins (RR 0.90, [95% CI: 0.67-1.12]; p = .46, I
2
= 67%) (Figure 3(A)) or quinolones versus penicillins (RR 1.20, [95% CI: 0.98-1.48]; p = .07, I
2
= 11%) (Figure 3(B)). In the carbapenem group (RR 0.55, [95% CI: 0.27-1.13]; p = .11, I
2
= 0%) (Figure 3(C)), and the quinolone group (RR 1.53, [95% CI: 0.64-3.62]; p = .34, I
2
= 61%) (Figure 3(D)), the risk of drug-related adverse effects did not show a statistically significant difference compared to penicillins. However, linezolid had a higher incidence of drug-related adverse effects compared to penicillins (RR 2.66, [95% CI: 1.47-4.73]; p = .0009) (Figure 3(E)). Adverse effect.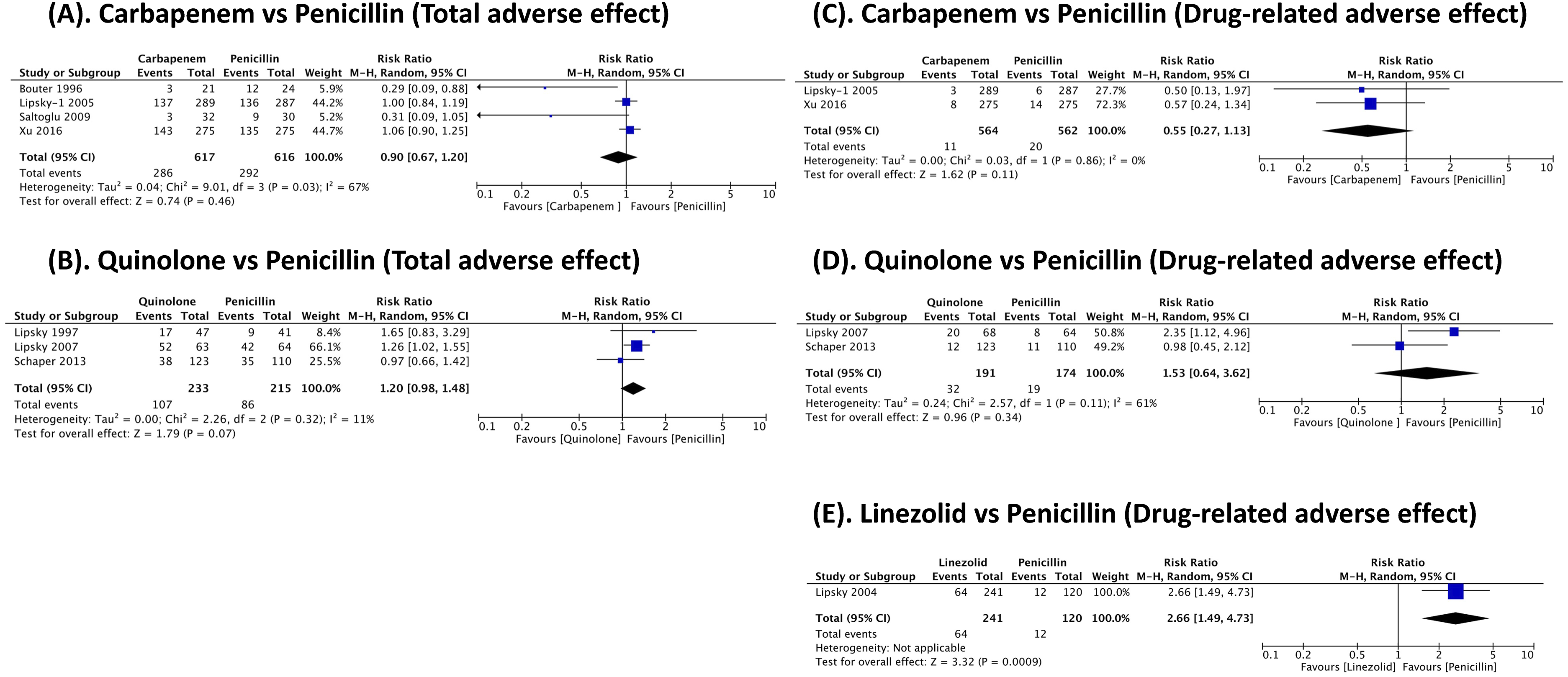
Secondary outcomes
There was insufficient data to perform a meta-analysis on amputation, proportion of wounds healed, and the median hospital stay. Only three trials reported the proportion of amputation, which ranged from 44.4%–68.8%.23,25,37 The treatment regimens in these trials included cefoxitin, ampicillin/sulbactam, imipenem/cilastatin, ampicillin/sulbactam, and piperacillin/tazobactam. Two trials reported the proportion of wound healed.34,42 In one trial, which compared linezolid and ampicillin/sulbactam, the proportion of wound healing was 68% and 56%, respectively. 34 The other trial, which compared clindamycin and cephalexin, reported a proportion of wound healing of 37% and 31%. 42 Only one trial reported the median days of hospital stay. 37 In this trial, the cefoxitin group had a median of 12.1 days, while the ampicillin/sulbactam group had a median of 21.1 days (Supplemental Table S3).
Subgroup analysis
Only two trials comparing ertapenem to piperacillin/tazobactam measuring DFI severities and comparing their efficacies24,26 (Figure 4). In the subgroup analysis, ertapenem did show a comparable efficacies to piperacillin/tazobactam either in severe DFIs (RR 1.04, [95% CI: 0.80-1.35]; p = .78, I
2
= 85%) or moderate DFIs (RR 1.00, [95% CI: 0.95-1.04]; p = .91, I
2
= 0). While grouping both severities, there were no significant differences in efficacies comparing ertapenem to piperacillin/tazobactam (p = .77). In trials encompassing patients with either osteomyelitis or PAD, there existed notable diversity in the antibiotic comparisons conducted, rendering a meta-analysis impractical. Additionally, there was variation in the proportion of patients with osteomyelitis across the trials in this subgroup, and in trials involving patients with PAD, the definitions and proportions of PAD varied among the articles. Table 2 provides information on the percentage of patients with osteomyelitis, the respective definitions or proportions of PAD, and the cure rates for each trial in these subgroups. In studies that included cases of osteomyelitis, there was an observed negative correlation between the prevalence of osteomyelitis and the cure rate (r = −0.667). In contrast, peripheral artery disease (PAD) did not demonstrate a direct influence on the cure rate, irrespective of the presence or absence of osteomyelitis. Subgroup of severity. Subgroup analysis of osteomyelitis and peripheral artery disease (PAD). IMP: Imipenem/cilastatin, AMP/SUL: Ampicillin/sulbactam, TZP: Piperacillin/tazobactam, OFX: Ofloxacin, LZD: Linezolid, CXT: Cefoxitin, TGC: Tigecycline, VAN: Vancomycin, ERT: Ertapenem, PIP: Piperacillin, CLDM: Clindamycin, MOXI: Moxifloxacin, AMC: Amoxicillin/clavulanate, TICC/CA: Ticarcillin-clavulanate, MET: metronidazole, CRO: Ceftriaxone.

Discussion
Penicillins are frequently employed as empirical antibiotics in the treatment of DFIs. In this study, penicillins demonstrated comparable clinical efficacy to carbapenems and quinolones, with the exception of linezolid, which showed superior activity. Notably, ertapenem and piperacillin/tazobactam exhibited similar efficacies in both severe and moderate cases of DFIs, despite the additional anti-pseudomonal coverage provided by piperacillin/tazobactam. In terms of safety outcomes, the incidence of total and drug-related adverse effects for carbapenems and quinolones did not significantly differ from those observed with penicillins, aside from linezolid, which was associated with a significant increase in safety concerns.
According to the Infectious Diseases Society of America (IDSA) and the International Working Group on the Diabetic Foot guideline (IWGDF),6,44,45 gram-positive cocci (GPC), especially Staphylococci, are the most common pathogens causing DFIs. However, causative pathogens may vary due to differences in local epidemiology, resistance patterns, and clinical situations such as individualized risk factors and DFI severity. 46 Therefore, selecting and assessing an appropriate empirical antibiotic is a necessary but challenging issue. In severe infections, which might involve deep tissue, osteomyelitis, or systemic signs of infection that require intensive treatment, empiric parenteral antibiotic therapy with broad-spectrum coverage for gram-positive, gram-negative, and anaerobic bacteria is recommended. Treatment should also consider complicating factors, recent antibiotic use, infection characteristics, and the presence of ischemia or other risk factors.6,44,45
In light of these circumstances, MRSA infections may necessitate thorough examination. According to the CDC’s 2020 report, there has been a significant rise in hospital-onset MRSA (HO-MRSA), healthcare-associated community-onset (HACO), and community-acquired MRSA (CA-MRSA) since 2017. 47 Though the pandemic may have momentarily slowed this trend, MRSA’s growing resistance continues to complicate treatment efforts.47–49 A global study also found a 14.69% prevalence of MRSA in elderly care facilities, underscoring the burden of MRSA in vulnerable populations. 50 Our study provides meaningful insights in this regard, particularly comparing linezolid to penicillins. The findings suggest that linezolid may have a potential efficacy advantage over penicillins, with a statistically significant difference (RR 1.15, [95% CI: 1.01-1.31]; p = .04, I 2 = 0%) (Figure 2(C)). This finding aligns with clinical observations that reported higher cure rates for linezolid compared to penicillins in MRSA cases (72% vs 57%). 35 Whether or not to cover MRSA infection depends on risk factors, including the high local prevalence of MRSA colonization, long-term antibiotic use, hospitalization, presence of osteomyelitis, MRSA nasal carrier, history of previous MRSA infection, diabetes, chronic ulcers, and chronic kidney disease history.6,50–52 Interestingly, one of the studies we included, Lipsky et al., excluded certain MRSA risk factors, yet linezolid still demonstrated therapeutic benefits. 35 Taken together, this suggests that linezolid may play a more significant role in the treatment of DFIs, even in cases where MRSA risk factors are not predominant. On the other hand, the pooled results of our study indicated that linezolid also carried a higher risk of adverse effects (RR 2.26, [95% CI: 1.49-4.73]; p = .0009) (Figure 3(E)), primarily gastrointestinal and hematologic issues. Our study highlights that treatment decisions should consider not only clinical efficacy but also side effects, cost, and resistance potential. While vancomycin remains a commonly used first-line option, linezolid may be an alternative when vancomycin is ineffective. However, careful consideration is needed due to linezolid’s adverse effect profile. The findings contribute to ongoing discussions about MRSA management, underscoring the need for comprehensive treatment strategies that optimize patient outcomes while minimizing risks. Future research is needed to refine these strategies further.
A meta-analysis that compared the efficacy of systemic antibiotics with other systemic antibiotics or placebo in patients with DFIs did not find significant differences among the studied antibiotics. 53 Furthermore, the meta-analysis does not provide clarity regarding the preferred choice of antibiotics for varying degrees of severity in DFIs. While the comparative outcomes of various systemic antibiotics for treating DFIs are quite diverse and often unclear, one RCT 26 revealed that the piperacillin/tazobactam group achieved a higher rate of clinical resolution than the ertapenem group, albeit only in patients with severe DFIs (97.2% vs 91.5%, p < .04). Nevertheless, the findings were inconsistent with another RCT, which showed equivalent clinical outcomes in patients with moderate-to-severe DFIs treated with either ertapenem or piperacillin/tazobactam. 24
To assess the impact of severity, we conducted a subgroup analysis to detect the relationship between clinical efficacy and severity of infection. The clinical efficacy of the piperacillin/tazobactam group was found to be comparable to that of the ertapenem group in moderate or severe DFIs, indicating no significant advantage of one treatment over the other. Fundamentally, piperacillin/tazobactam and ertapenem exhibit differential spectra of activity against various bacterial strains, with piperacillin/tazobactam displaying efficacy against Pseudomonas aeruginosa and ertapenem showing potency against ESBL-producing bacteria. According to Lispsky et al., 24 there were no statistically significant differences observed between the groups treated with piperacillin/tazobactam and ertapenem with respect to eradication of Pseudomonas aeruginosa. 24 As such, the study concluded that the clinical and microbiological outcomes achieved in patients treated with ertapenem were comparable to those obtained with piperacillin/tazobactam. Conversely, Xu et al. 26 reported that piperacillin/tazobactam, despite its known spectrum of activity, demonstrated effectiveness against ESBL-producing bacteria and thus exhibited superior clinical efficacy compared to the ertapenem group. The aforementioned discussion suggests that in cases of more severe DFIs, the likelihood of polymicrobial infections may be elevated, with Pseudomonas aeruginosa typically not being the primary causative agent. However, with the global increasing prevalence of multi-drug-resistant Gram negative bacilli, prudent selection of treatment regimen necessitates an awareness of local patterns of antibiotic resistance.
In addition to the severity of DFIs, other clinical factors should also be considered, as they may affect treatment efficacy. For instance, the presence of DFO may predict unfavorable outcomes. Studies involving patients with DFO showed a significant negative correlation with the cure rate17,23,25,30,35,37 (r = −0.667) (Table 2). Besides, prompt surgical intervention or extended courses of antibiotics may be warranted.6,54 The optimal duration of antibiotic therapy for DFO is a subject of ongoing debate, despite the concurrence in guidelines. According to the IDSA/IWGDF guidelines, 6 antibiotic therapy for DFIs should be adjusted based on infection severity and surgical intervention. The duration of systemic antibiotics is crucial in treating DFIs to ensure complete eradication of infection, reduce the risk of recurrence, and minimize complications. Prolonged or inadequate antibiotic treatment may lead to resistance, making subsequent treatment more challenging and less effective. For patients with mild DFIs, involving only skin or soft tissue, a treatment duration of 1-2 weeks is recommended. For those with moderate to severe infections, especially following surgical debridement, a 10-day course is typically advised. In cases of DFO without bone resection, antibiotics may be needed for up to 6 weeks to ensure adequate resolution of the infection. Clinicians should consider consulting infectious disease or microbiology experts for challenging cases, particularly those involving unusual or highly resistant pathogens. Additionally, transitioning to oral antibiotics may be appropriate, considering factors such as bioavailability, long-term tolerability, accessibility, cost, and clinical diagnostics. Further research is needed to determine the comparative efficacy of these treatment options.
PAD has a well-recognized negative impact on the outcomes of DFIs. Although recent guidelines 55 recommend annual screening for PAD in patients living with diabetes, our analysis found no direct correlation between antibiotic therapy and cure rates in patients with PAD across multiple studies17,21,31,32,37,39 (Table 2). PAD plays a significant role in the pathogenesis of DFIs, and patients with more severe infections or coexisting PAD may require longer antibiotic courses, potentially lasting up to 3-4 weeks based on guideline recommendations. 6 However, meta-analysis on PAD and DFI severity is challenging due to patient heterogeneity and varying classification systems. Further randomized trials and more precise classification methods are needed to better understand these interactions and improve treatment strategies.
In a broad sense, comprehensive and multifactorial assessments of DFIs are necessary, given the complex nature of patients with diabetes mellitus. These assessments should incorporate various clinical presentations, laboratory studies, and other diagnostic evaluations. Definitive treatment of DFIs should be guided by principles of antibiotic stewardship. While our analysis focused on patients who achieved clinical cure or improvement, it is important to acknowledge that the therapeutic efficacy of treatment is not limited to these outcomes alone. Partial improvement or relapse are also relevant outcomes that should be considered in the assessment of treatment efficacy. As such, a more detailed and comprehensive study of these aspects of treatment is needed.
The strength of our study is that only RCTs that compared different systemic antibiotic regimens were included in the meta-analysis to minimize selection bias and confounding factors. Furthermore, more trials were included, and additional results from the severities, and adverse effects were provided. Nevertheless, this study has several limitations. First, heterogeneity could not be avoided because the study design, severity of DFIs, proportion of osteomyelitis and PAD, and regimen of systemic antibiotics varied among trials. Second, not all antibiotic classes were included in the meta-analysis because some comparison groups included only one trial. Finally, the number of trials in each subgroup was insufficient, and most studies did not discuss patients subgroups with severe DFIs, osteomyelitis, and PAD separately. Risk factors and DFI severity are inextricably linked with the choice of systemic antibiotics and therapeutic duration. More reliable assessments and analyses of subgroups of patients are conducive to this field. Further research should consider these perspectives.
Conclusion
In conclusion, linezolid may offer a potential efficacy advantage over penicillins in treating DFIs. Otherwise, penicillins exhibit comparable clinical efficacy and safety to carbapenems and fluoroquinolones for DFI treatment. However, linezolid also presents a higher risk of drug-related adverse effects compared to penicillins. In cases of moderate or severe DFIs, piperacillin/tazobactam and ertapenem can be considered as viable treatment options, although local antimicrobial susceptibilities should be taken into consideration.
Supplemental Material
Supplemental Material - A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections
Supplemental Material for A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections by Mei-Chuan Lee, Yi-Ming Hua, Han Siong Toh, Hui-Chen Su and Po-Jung Chen in Diabetes & Vascular Disease Research.
Supplemental Material
Supplemental Material - A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections
Supplemental Material for A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections by Mei-Chuan Lee, Yi-Ming Hua, Han Siong Toh, Hui-Chen Su and Po-Jung Chen in Diabetes & Vascular Disease Research.
Supplemental Material
Supplemental Material - A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections
Supplemental Material for A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections by Mei-Chuan Lee, Yi-Ming Hua, Han Siong Toh, Hui-Chen Su and Po-Jung Chen in Diabetes & Vascular Disease Research.
Supplemental Material
Supplemental Material - A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections
Supplemental Material for A systematic review and meta-analysis of randomized controlled trials of systemic antibiotics for diabetes-related foot infections by Mei-Chuan Lee, Yi-Ming Hua, Han Siong Toh, Hui-Chen Su and Po-Jung Chen in Diabetes & Vascular Disease Research.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
