Abstract
Background
The frequency of type 2 diabetes mellitus (T2DM) is rising annually. Coronary heart disease (CHD) is a prevalent complication affecting individuals with T2DM.
Objective
The aim of this investigation was to assess the level of DBH-AS1 in T2DM with CHD, and to determine its potential role in forecasting the occurrence of significant cardiovascular events.
Methods
The DBH-AS1 levels were detected by qRT-PCR. The diagnostic value of DBH-AS1 was assessed through receiver operating characteristic (ROC) curve analysis. Logistic regression was conducted to identify the risk factors for cardiovascular events among patients with T2DM with CHD. Cell proliferation was detected by Cell Counting Kit-8 (CCK-8) assay, apoptosis was detected by flow cytometry, and the concentration of inflammatory factors was detected by Enzyme Linked Immunosorbent (ELISA) kit.
Results
DBH-AS1 was down-regulated in serum of both T2DM with CHD and cardiovascular events patients. Of the cardiovascular events that occurred, major events included recurrent angina (20%), cardiovascular death (7.5%), acute myocardial infarction (23.75%), severe arrhythmia (22.50%), acute heart failure (18.75%) and stroke (7.5%). And DBH-AS1 had a predictive value for each adverse of cardiovascular events. DBH-AS1 regulated the expression of miR-483-5p and affected the proliferation, apoptosis, and secretion of inflammatory factors of HCAECs.
Conclusion
DBH-AS1 may serve as a predictor for the occurrence of cardiovascular events in T2DM with CHD patients.
Introduction
Type 2 diabetes mellitus (T2DM) accounts for approximately 90% of all diabetes diagnoses, predominantly affecting adults and the elderly population. As dietary patterns have evolved, there has been a marked increase in the consumption of foods that are high in sugar, fat, and calories, which has led to a consistent annual rise in the prevalence of T2DM. 1 Individuals with T2DM typically experience prolonged periods of elevated blood glucose levels, which can predispose them to vascular damage and elevate their risk of developing cardiovascular conditions. 2 Coronary heart disease (CHD) is a prevalent complication affecting individuals with T2DM. Statistical analyses indicate that over 60% of deaths in T2DM patients are attributed to CHD. 3 Patients with T2DM and CHD are exposed to a multitude of cardiovascular risk factors. Identifying biological markers that can reliably forecast the onset of significant adverse cardiovascular events could enable healthcare providers to accurately monitor the progression of the disease in patients and enhance their outcome prospects. 4
Long non-coding RNAs (lncRNAs) primarily modulate gene expression. An increasing number of lncRNAs have been pinpointed as pivotal in the context of diabetes mellitus, encompassing associations with type 2 diabetes. 5 One investigation identified 441 lncRNAs that exhibited altered expression levels in patients with T2DM, with 366 genes showing increased expression and 75 genes demonstrating decreased expression. 6 In addition, lncRNAs have been implicated in the pathogenesis of various cardiovascular diseases.7,8
It has been documented that lncRNA DBH-AS1 serves as a biomarker in a range of tumorigenic conditions, including melanoma, 9 hepatocellular carcinoma, 10 and non-small cell lung cancer. 11 At the same time, in the bioinformatics analysis of lncRNA expression related to inflammation in type 2 diabetes, Huang Ling et al. found that DBH-AS1 was also one of the differentially expressed lncRNAs. 12 However, the levels of DBH-AS1 expression in individuals with T2DM combined with CHD, as well as its influence on the risk of cardiovascular outcomes in this patient population, remain unelucidated.
This investigation represented the first evidence of the variable expression of DBH-AS1 in T2DM combined with CHD patients and investigated its predictive value and mechanism for cardiovascular events.
Methods and materials
Research object and follow-up
A total of 182 T2DM combined with CHD patients admitted to Qinhuangdao Hospital of Traditional Chinese Medicine were selected as study objects. In addition, 60 T2DM patients without CHD were included as a control group.
The included patients met the following criteria: (1) The determination of T2DM conforms to the American Diabetes Association criteria 13 ; (2) The ascertainment of CHD aligns with the diagnostic standards for CHD established by the Cardiovascular Division of the Chinese Medical Association in the year 2018 14 ; (3) Individuals who are 18 years of age or above.
Exclusion criteria: (1) Combined with type 1 diabetes mellitus, specific type of diabetes (diabetes caused by endocrine diseases, such as Cushing’s syndrome, acromegaly, and hyperthyroidism; Drug- or chemical-induced diabetes, such as antiretroviral drugs, diuretics, etc.) or pregnancy diabetes mellitus; (2) Acute metabolic disorders associated with diabetes mellitus; (3) Combined with malignant tumors or blood diseases; (4) Taking drugs such as glucocorticoids and aspirin that may affect the results of the HbA1c test; (5) congenital heart disease.
Comparison of basic data between non-cardiovascular events and cardiovascular events group.
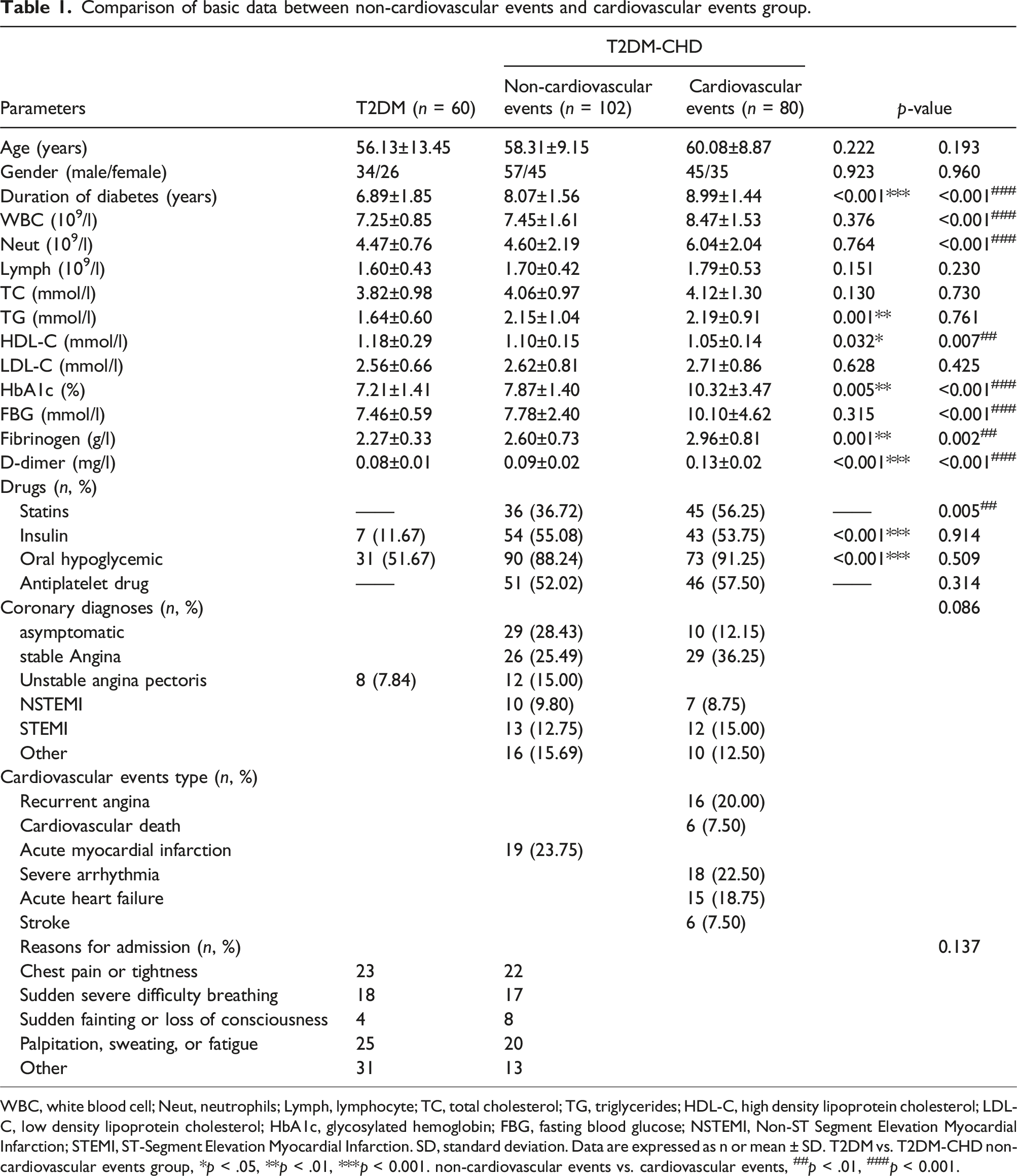
WBC, white blood cell; Neut, neutrophils; Lymph, lymphocyte; TC, total cholesterol; TG, triglycerides; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HbA1c, glycosylated hemoglobin; FBG, fasting blood glucose; NSTEMI, Non-ST Segment Elevation Myocardial Infarction; STEMI, ST-Segment Elevation Myocardial Infarction. SD, standard deviation. Data are expressed as n or mean ± SD. T2DM vs. T2DM-CHD non-cardiovascular events group, *p < .05, **p < .01, ***p < 0.001. non-cardiovascular events vs. cardiovascular events, ##p < .01, ###p < 0.001.
All subjects in this study signed informed consent and were approved by the Medical Ethics Committee of Qinhuangdao Hospital of Traditional Chinese Medicine.
Cell line and cell culture
The human coronary artery endothelial cells (HCAECs) were acquired from the American Type Culture Collection (ATCC). These cells were cultivated in Dulbecco’s Modified Eagle’s Medium (DMEM) (Bioind, Haemek, Israel) supplemented with 10% Fetal Bovine Serum (FBS) (Gibco, MD, USA) and 1% antibiotic/antimycotic solution (Sangon, Shanghai, China) within an atmosphere of 5% CO2 at a temperature of 37°C.
Cell transfection
Cell cultures were maintained in a medium supplemented with 25 mM glucose to develop high-glucose-exposed cells (HG), in alignment with previously documented procedures. 15 To generate a plasmid for the overexpression of DBH-AS1, the gene sequence of DBH-AS1 was ligated into the pcDNA3.1 vector (obtained from Invitrogen). MiR-483-5p mimics, miR-483-5p inhibitors and their corresponding negative controls (mimic NC) were sourced from GenePharma (Shanghai, China). To facilitate in vitro transfection, the cells were treated with pcDBH-AS1 or miR-483-5p mimics and their respective NCs, utilizing Lipofectamine 3000 (Invitrogen, CA, USA) for a duration of 24 h, following the instructions provided in the product protocol.
qRT-PCR
The separated serum was thawed over ice initially. Total RNA was isolated using TRIzol reagent (Life Technologies, Carlsbad, CA), in compliance with the supplier’s recommended protocol. The quality and quantity of the extracted samples were evaluated with a Nanodrop 2000 instrument (Thermo Fisher Scientific, USA). The RNA samples were initially converted into cDNA through reverse transcription using HiScript II Q RT SuperMix (Vazyme, Nanjing, China). Subsequently, the cDNA was amplified and detected using ChamQ SYBR qPCR Master Mix (Vazyme, Nanjing, China). The thermal cycling conditions included an initial denaturation at 95°C for 30 s, followed by 40 amplification cycles of 95°C for 10 s and 60°C for 60 s. The expression levels were quantified utilizing the 2−ΔΔct approach. The primer sequence is as follows:
DBH-AS1 forward: 5′-CGTCCACTCGTCTGTTCACT-3′.
DBH-AS1 reverse: 5′-TAACACCCCATCCGCTTGT-3′.
U6 forward: 5′-TTGAGCGGGCAAATCG-3′.
U6reverse: 5′-GCCCTATCGATGCAAGCA-3′.
miR-483-5p forward: 5′-GCCGAGAAGACGGGAGGAAA-3′.
miR-483-5p reverse: 5′-CTCAACTGGTGTCGTGGA-3′.
Cell proliferation experiment
Cells were inoculated into 96-well culture dishes. Subsequently, a volume of 10 μL of CCK-8 reagent (Invitrogen, CA, USA) was added to each well, and the plates were incubated at a temperature of 37°C for a duration of 2.5 h. The optical density of the cultures was then determined at a wavelength of 450 nm utilizing a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA).
Cell apoptosis assay
After 48 h of transfection, cells were subjected to a hypoxic environment for an additional 48 h. Subsequently, the HCAECs were gently rinsed with PBS at room temperature. Cells were then incubated in the dark with propidium iodide (PI) and fluorescein isothiocyanate (FITC)-labeled annexin V (Abcam) for a 20-min period. Flow cytometry was utilized to analyze and differentiate apoptotic cells from the population.
Enzyme-linked immunosorbent assay (ELISA)
Following various experimental interventions, the levels of interleukin-1β (IL-1β, product code: KE1127) and interleukin-18 (IL-18, product code: KE1383) of cells were quantified employing enzyme-linked immunosorbent assay (ELISA) kits. The absorbance at a wavelength of 450 nm was recorded using a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA).
Dual-luciferase assay
A construct containing the DBH-AS1 3′UTR and its different variants was engineered and inserted into the pmirGLO luciferase expression vector (Promega, Madison, WI, USA). This resulted in the creation of two distinct constructs, named DBH-AS1-WT and DBH-AS1-MUT. These plasmids were then introduced into the HCAECs together with either miR-483-5p mimics or miR-483-5p inhibitors. The relative luciferase activity was determined using the Dual Luciferase Assay Kit (Promega, USA).
Statistical analyses
Values are presented as the mean ± standard deviation (SD) and analyzed via an independent t test, one-way ANOVA, or two-way ANOVA. Statistical analyses were conducted using GraphPad Prism version 9.0 and SPSS 26.0. ROC curve was used to evaluate the diagnostic potential of DBH-AS1 in patients with T2DM combined with CHD and the predictive value of cardiovascular events in these patients. Logistic regression analysis was used to evaluate the risk factors of cardiovascular events in patients with T2DM complicated combined with CHD. A p-value of less than 0.05 was deemed statistically significant.
Result
Differential expression of DBH-AS1 and its clinical predictive value
The expression level of DBH-AS1 in the serum was substantially reduced in T2DM combined with CHD patients compared to those with T2DM alone (Figure 1(a)). Moreover, this down-regulation was even more pronounced in patients who experienced cardiovascular events (Figure 1(b)). Figure 1(c) illustrated the potential of serum DBH-AS1 as a diagnostic indicator for differentiating between T2DM and T2DM combined with CHD. The graph revealed that the Area Under the Curve (AUC) of the ROC curve was 0.928, with a diagnostic sensitivity of 92.9% and a specificity of 81.7%. It was proposed that DBH-AS1 may effectively categorize patients with isolated T2DM combined with CHD patients from those with T2DM. The role of serum DBH-AS1 in predicting the occurrence of cardiovascular events in T2DM combined with CHD patients was shown in Figure 1(d). The AUC of ROC curve was 0.848, with a sensitivity of 80.0% and a specificity of 87.2%. The AUC of DBH-AS1 for predicting each event in cardiovascular events, including recurrent angina, cardiovascular death, acute myocardial infarction, severe arrhythmia, acute heart failure and stroke were 0.837 (Figure 2(a)), 0.752 (Figure 2(b)), 0.894 (Figure 2(c)), 0.819 (Figure 2(d)), 0.850 (Figure 2(e)), and 0.846 (Figure 2(f)). Expression and diagnostic role of DBH-AS1. (a)-(b) The expression of DBH-AS1 was detected by qRT-PCR. (c) ROC curve of DBH-AS1 for the diagnosis of T2DM complicated with CHD. (d) ROC curve of the predictive value of DBH-AS1 for cardiovascular events in T2DM complicated with CHD patients. ***p < .001. ROC curve of DBH-AS1 predicting each cardiovascular event. ROC curve of the predictive value of DBH-AS1 for (a) recurrent angina, (b) cardiovascular death, (c) acute myocardial infarction, (d) severe arrhythmia, (e) acute heart failure and (f) stroke.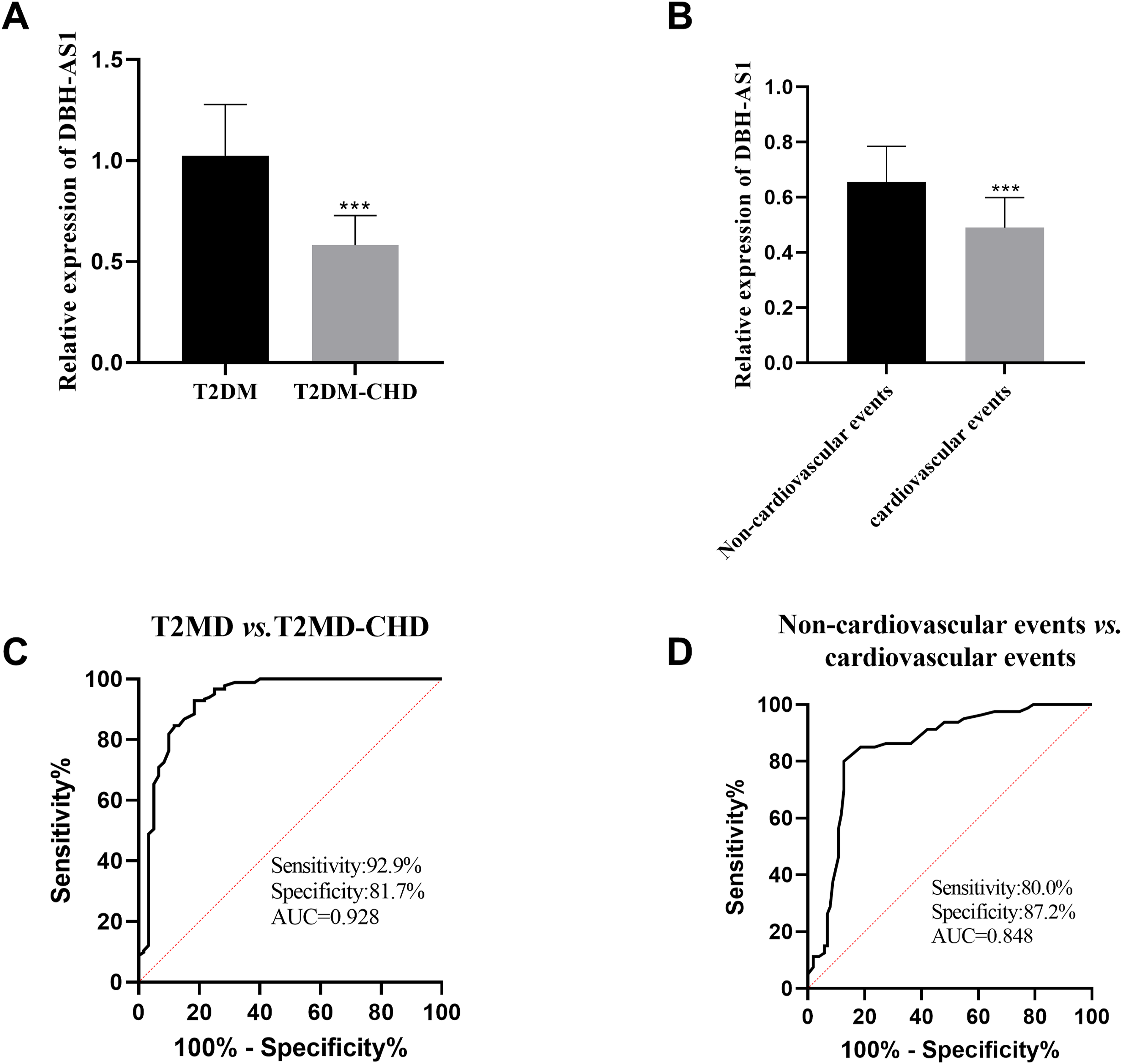
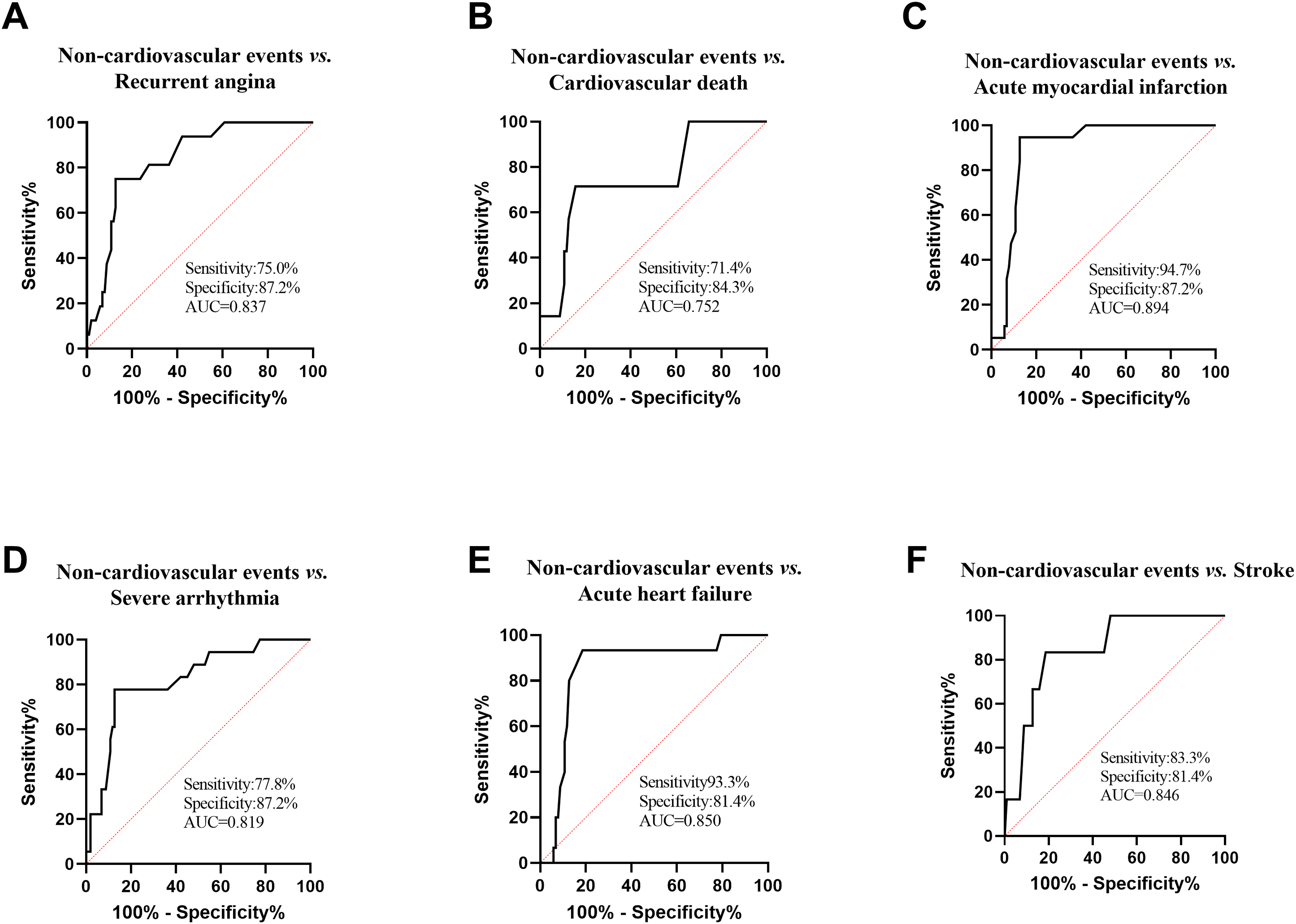
Association of DBH-AS1 with pathological features of patients with cardiovascular events
Correlation between DBH-AS1 and clinicopathological features in patients with cardiovascular events.
WBC, white blood cell; Neut, neutrophils; Lymph, lymphocyte; TC, total cholesterol; TG, triglycerides; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HbA1c, glycosylated hemoglobin; FBG, fasting blood glucose; *p < .05, **p < .01.
Logistic regression analysis of risk factors for cardiovascular events in T2DM patients with CHD
Logistic regression analysis of risk factors for cardiovascular events.
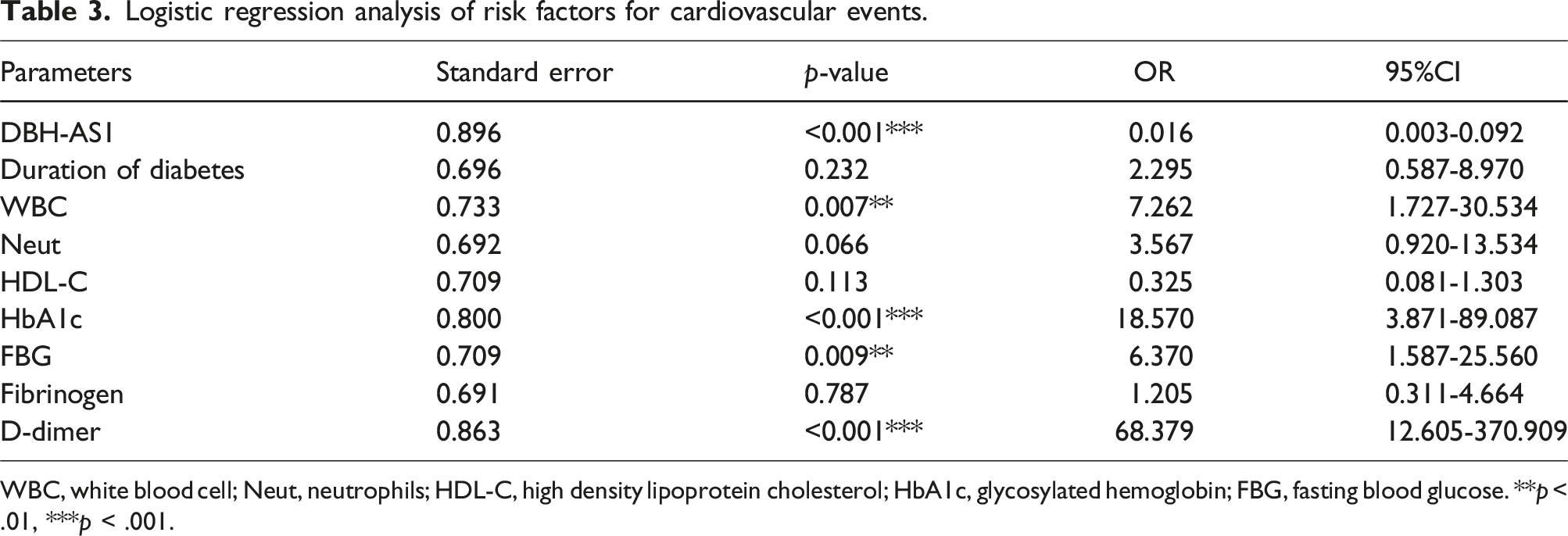
WBC, white blood cell; Neut, neutrophils; HDL-C, high density lipoprotein cholesterol; HbA1c, glycosylated hemoglobin; FBG, fasting blood glucose. **p < .01, ***p < .001.
Effect of DBH-AS1 overexpression on HCAECs induced by high glucose
Following the transfection of a vector encoding DBH-AS1 into HCAECs exposed to high glucose, there was an increase in the expression of DBH-AS1, as depicted in Figure 3(a). Subsequent to exposure to high glucose levels, the ability of HCAECs to proliferate was diminished, and there was a concomitant increase in the apoptotic frequency. However, the promotion of DBH-AS1 expression markedly enhanced the proliferative potential of the cells. Concurrently, the elevated apoptosis rate was reversed (Figure 3(b), and 3(c)). Furthermore, the secretion of pro-inflammatory cytokines (IL-6 and IL-8) by HCAECs was elevated following exposure to high glucose levels. However, the expression of DBH-AS1 to a higher level mitigated this effect, leading to reduced levels of both IL-6 and IL-8 (Figure 3(d), and 3(e)). Effect of overexpression of DBH-AS1 on high glucose-induced HCAECs. (a) The expression of DBH-AS1 in high glucose-induced HCAECs was detected by qRT-PCR. (b) Cell proliferation was detected by CCK-8. (c) Apoptosis was detected by flow cytometry. (d)-(e) The ELIASA kit was used to detect IL-6 and IL-8 concentrations. ***p < .001, **p < .01 vs. control; ###p < .001, ##p < .01, #p < .05 vs. HG + pcDNA3.1.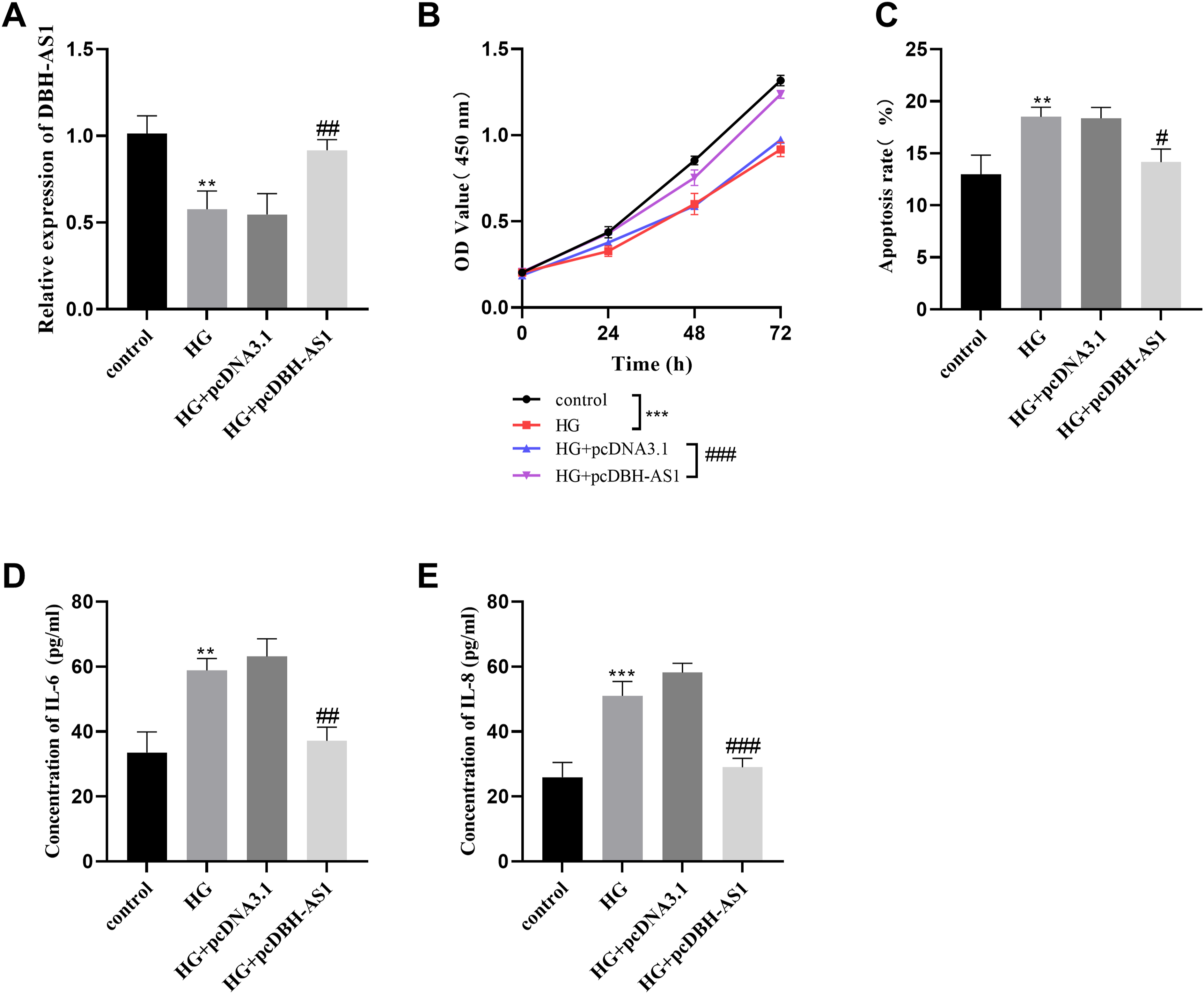
DBH-AS1 has the potential to interact with miR-483-5p
The serum levels of miR-483-5p were markedly higher in T2DM combined with CHD compared to those patients with T2DM (Figure 4(a)). Furthermore, among T2DM patients with CHD, the expression of DBH-AS1 in serum was higher in patients experiencing cardiovascular events than in those non-cardiovascular events (Figure 4(b)). Results from Pearson’s correlation coefficient test revealed a significant inverse relationship between the levels of DBH-AS1 and miR-483-5p expression in T2DM accompanied by CHD patients (Figure 4(c)). Simultaneously, there was a significant increase in the expression of miR-483-5p in HCAECs induced by high glucose (Figure 4(d)). Through lncRNASNP v3 database (lncRNASNP v3 (hzau.edu.cn)) and found a potential binding site between DBH-AS1 and miR-483-5p (Figure 4(e)). Following treatment with miR-483-5p mimics, a reduction in luciferase activity was observed in the DBH-AS1-WT construct, whereas an enhancement of luciferase activity was detected in the DBH-AS1-WT construct upon treatment with miR-483-5p inhibitors. Conversely, the DBH-AS1-MUT construct exhibited minimal changes in luciferase activity (Figure 4(f)). Collectively, these findings suggested that DBH-AS1 may serve as a target for miR-483-5p and represses its expression. DBH-AS1 negatively regulated miR-483-5p. (a)-(b) The expression of miR-483-5p in serum was detected by qRT-PCR. (c) DBH-AS1 was negatively correlated with the expression of miR-483-5p.(d) The expression of miR-483-5p in high glucose-induced HCAECs was detected by qRT-PCR. (e) The binding site sequence between DBH-AS1 and miR-483-5p was forecasted utilizing the lncRNASNP3 database. (f) The interaction between DBH-AS1 and miR-483-5p was verified by dual luciferase reporter gene assay. ***p < .001, **p < .01.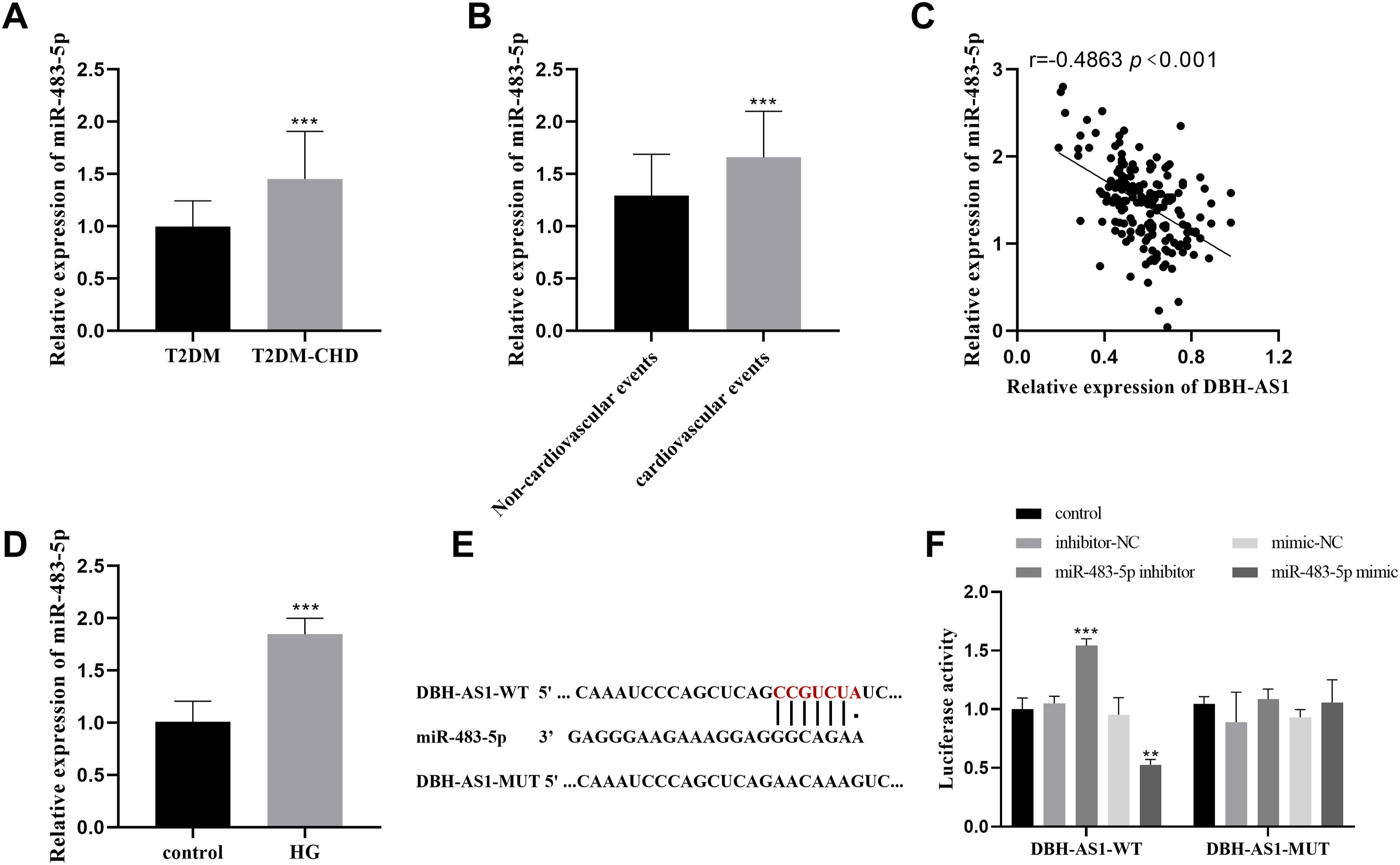
Overexpression of miR-483-5p reversed the effect of DBH-AS1 upregulation on HCAECs
To explore the impact of miR-483-5p on the biological activities mediated by DBH-AS1 in vitro, we conducted transfections using pcDBH-AS1 and miR-483-5p mimics in high glucose-stimulated HCAECs. The findings revealed that the levels of miR-483-5p were reduced following transfection with pcDBH-AS1 and increased upon transfection with miR-483-5p mimics (Figure 5(a)). CCK-8 assays demonstrated that the overproduction of miR-483-5p abrogated the promotional effect of pcDBH-AS1 on cell proliferation, as shown in Figure 5(b). Furthermore, miR-483-5p mimics were capable of counteracting the anti-apoptotic influence of pcDBH-AS1 overexpression (Figure 5(c)). Simultaneously, miR-483-5p also mitigated the reduction in IL-6 and IL-8 levels induced by pcDBH-AS1 (Figure 5(d), and 5(e)). miR-483-5p mimics partially counteracts the effects of pcDBH-AS1 on high glucose-induced HCAECs. (a) miR-483-5p level was quantified via qRT-PCR. (b) Cell proliferation was assessed using the CCK-8 assay. (c) Apoptosis was detected by flow cytometry. (d)-(e) The ELISA kit was used to detect IL-6 and IL-8 concentrations. ***p < .001, **p < .01 vs. HG + pcDNA3.1; ###p < .001, ##p < .01, #p < .05 vs. HG + pcDBH-AS1 + mimic-NC.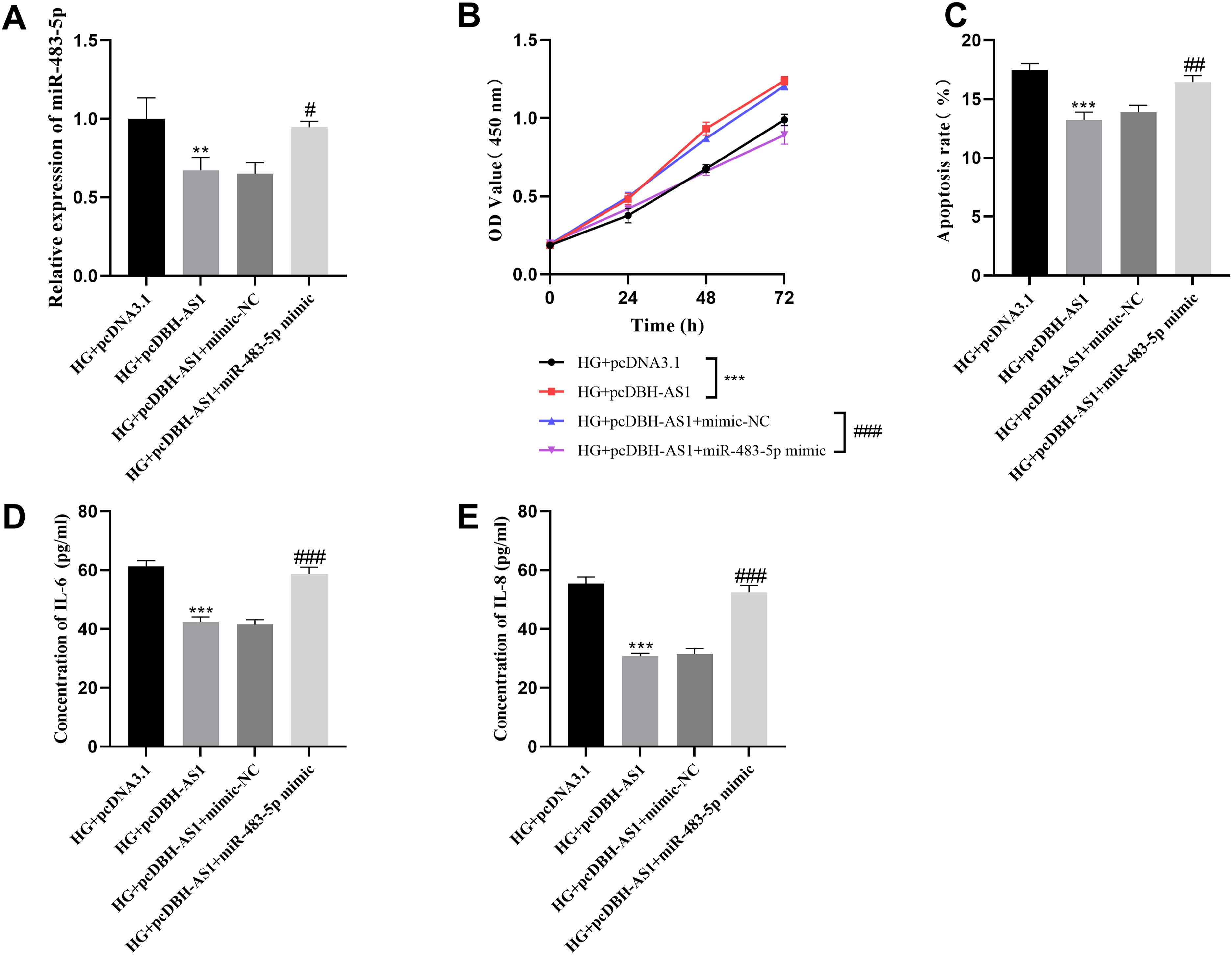
Discussion
According to multiple research efforts, lncRNA has been proposed as a potential biomarker for the biomarker of T2DM and is believed to play a role in the underlying mechanisms of the disease. For instance, overexpression of LINC01018 functions as a promising biomarker for the detection of T2DM and ameliorates beta-cell dysfunction induced by high glucose levels. 16 Various lncRNAs have important significance in various pathological changes and dysfunction changes in different stages of T2DM. 17 At the same time, multiple lncRNAs are also involved in the progression of cardiovascular disease. LncRNA-FA2H-2 is considered a promising candidate as an inflammatory indicator for evaluating the risk of CHD onset. 18 The upregulation of lncRNA PVT.1 has been shown to suppress apoptosis and enhance proliferation in HCAECs, indicating that PVT.1 holds significant promise as a therapeutic candidate for the treatment of coronary artery disease. 19
In this study, we selected T2DM combined with CHD patients as research subjects and explored the expression of DBH-AS1 in such patients and the prediction of cardiovascular events in them. In addition, this study explored the predictive value of DBH-AS1 for the occurrence of individual cardiovascular events. DBH-AS1 has the ability to distinguish between patients with simple T2DM and T2DM combined with CHD patients and has a good predictive effect on the occurrence of cardiovascular events in these patients. Interestingly, logistic regression analysis showed that WBC was a risk factor for the development of cardiovascular events, whereas neutrophils were not, which may indicate that neutrophils do not play a key role in the development of cardiovascular events, and perhaps other types of cells in the leukocytes play a key role in cardiovascular events. It is also possible that the limited number of patients included in this study may have contributed to the different results. At the same time, logistic regression analysis indicated that DBH-AS1 expression was an independent factor affecting the occurrence of cardiovascular events in T2DM complicated with CHD patients. The findings suggested a strong association between DBH-AS1 and the pathological characteristics of patients.
In previous studies, lncRNA DBH-AS1 has been reported to be down-regulated in patients with inflammation-related T2DM. 12 The results of this study suggested that lncRNA DBH-AS1 expression in T2DM combined with CHD patients was lower than that in patients with T2DM alone. And it was lowest in patients who developed cardiovascular events. This may be due to the interaction between DBH-AS1 and inflammatory factors. In order to further validate this observation, we conducted a series of in vitro assays. Following the upregulation of DBH-AS1 in high glucose stimulated HCAECs, an increase in cellular proliferation and a reduction in the apoptosis ratio were detected. Most notably, the levels of pro-inflammatory cytokines IL-6 and IL-8 were suppressed by DBH-AS1, corroborating our prior conjectures.
Research has demonstrated that inflammation, aging, and oxidative stress each play a significant role in the development of T2DM. 20 T2DM is inherently characterized by a pro-inflammatory condition, and atherosclerotic cardiovascular disease serves as the principal culprit for mortality in these patients. The role of inflammation in the onset of atherosclerotic cardiovascular disease appears to be critical. 21 The inflammatory response is thought to contribute to the development of T2DM by inducing insulin resistance, and this response is further exacerbated under conditions of hyperglycemia, facilitating the onset of long-term complications associated with diabetes. 22 Focusing on inflammatory pathways may represent a component of effective strategies for the prevention and management of diabetes and its related complications.
Moreover, we investigated the influence of DBH-AS1 on the cellular function by modulating the miR-483-5p. Increasing evidence indicates that miR-483-5p is engaged in safeguarding cellular function and survival by targeting particular transcripts. 23 With significance, increased concentrations of miR-483-5p in the circulatory system are firmly associated with metabolic risk factors, acting as encouraging indicators for diagnosis. As such, miR-483-5p emerges as a promising biomarker for assessing the likelihood of incurring diabetes and cardiovascular diseases, and it also presents potential as a target for therapeutic interventions. 23 The experimental results, as determined through rigorous dual luciferase reporter assays, indicate a precise binding interaction between DBH-AS1 and miR-483-5p, leading to a suppression in the expression levels of miR-483-5p. Furthermore, the enhancement of DBH-AS1 expression on cells was counteracted by the elevated expression of miR-483-5p. It was suggested that DBH-AS1 is involved in the progression of the disease by regulating the expression of miR-483-5p.
Additionally, our investigation faces certain constraints, including the possibility that DBH-AS1 could be implicated in the progression of the disease via alternative mechanisms, such as senescence, oxidative stress, or the existence of additional genes that interact with DBH-AS1. Some of the patients included in this study were treated with antiplatelet drugs such as ticagrelor and clopidogrel, and it is uncertain whether these drugs affect the levels of non-coding RNA and thus affect the comparison effect between the groups. Consequently, further studies are essential to solve these questions. Another key limitation is that samples from T2DM-CHD group were taken during acute admission, whereas samples from the control group were collected in a stable outpatient setting. This might have led to confounders which render the findings regarding prediction of CHD hypothesis-generating. Overall, detection of DBH-AS1 expression provides a non-invasive and fast way to predict T2DM-CHD, so DBH-AS1 may be a potential biomarker for T2DM-CHD.
Conclusion
Generally, our results indicated that DBH-AS1 is reduced in individuals suffering from T2DM complicated with CHD, and it exhibits a favorable predictive influence on the incidence of cardiovascular events. Moreover, DBH-AS1 appears to contribute to the progression of the illness by modulating the expression of miR-483-5p as a target. Consequently, DBH-AS1 holds potential as a biomarker for the prediction of cardiovascular events.
Supplemental Material
Supplemental Material - Predictive value of lncRNA DBH-AS1 for cardiovascular events in patients with type 2 diabetes mellitus with coronary heart disease
Supplemental Material for Predictive value of lncRNA DBH-AS1 for cardiovascular events in patients with type 2 diabetes mellitus with coronary heart disease by Xintong Wang, Yan Li, and Jiaoding Tian in Diabetes & Vascular Disease Research.
Footnotes
Author contributions
Conceptualization, X.W. and J.T.; Data curation, Y.L.; Formal analysis, X.W. and Y.L.; Funding acquisition, J.T.; Investigation, Y.L.; Methodology, X.W. and Y.L.; Project administration, J.T.; Resources, Y.L.; Software, Y.L.; Supervision, J.T.; Validation, X.W. and J.T.; Visualization, X.W.; Writing - original draft, X.W.; Writing - review & editing, J.T.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper is funded by Scientific Research project of Hebei Provincial Administration of Traditional Chinese Medicine. The project number is 2022535.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
