Abstract
Early control of glycaemia is key to reduce vascular complications in individuals with Type 1 diabetes. Therefore, encouraging children and adolescents with T1DM to take responsibility for controlling glucose levels is an important yet a challenging task. The rapid expansion of continuous glucose monitoring (CGM) systems has allowed for more comprehensive analysis of glycaemia in T1D. Moreover, CGM devices have the ability to calculate rate of change in glucose levels and display the information as trend arrows. In turn, this can help to take evasive actions to return glucose levels to near physiological glycaemia, which can be highly motivating for young people with T1DM. In the absence of standardised, evidence-based guidance, this consensus document, generated by experts from the Arab Society of Paediatric Endocrinology and Diabetes and international advisors, summarises recent literature on the use of trend arrows in young people with T1DM. The use of trend arrows in different CGM systems is reviewed and their clinical significance is highlighted. Adjusting insulin doses according to trend arrows is discussed while also addressing special situations, such as exercise, fasting, nocturnal hypoglycaemia and menstruation. Adequate understanding of trend arrows should facilitate optimisation of glycaemic control in the T1D population.
Introduction
It is well established that lowering average glucose levels, measured as HbA1c, is key for the reduction of early and late microvascular complications as well as longer term macrovascular disease in individuals with type 1 diabetes.1–3 However, good glycaemic control requires more than just a reduction in HbA1c, given that both hypoglycaemia and glycaemic variability (GV) are associated with adverse clinical outcome,4–7 which may be modifiable. 8 This explains recent guidelines indicating the need to focus on a number of glycaemic parameters for the management of type 1 diabetes individuals. 9 Taken together, early optimisation of glycaemic control is important in diabetes, which can be a challenge, particularly in the younger age group.
The increased use of continuous glucose monitoring (CGM) systems has allowed for a more focused management of glycaemia by addressing the three main glycaemic markers (time in range, hypoglycaemia and GV), and these parameters form an important part of the modern diabetes consultation. However, there is another aspect that is more relevant to patients than diabetes specialists but is seldom addressed in clinical guidelines: use of trend arrows with CGM devices. Previously published review articles giving advice on the use of trend arrows have been useful to guide both patients and health care professionals, but these have often focused on the adult population or addressed a specific CGM system.10–14 Given the importance of early glycaemic control after the diagnosis of type 1 diabetes, there is a gap in the literature related to the use of trend arrows in the younger population, while also addressing different CGM systems.
In this work, we provide a consensus statement on employing the trend arrows for optimising glycaemia in younger individuals with diabetes, based on expert opinion from a recent Arab Society for Paediatric Endocrinology and Diabetes (ASPED) meeting coupled with an extensive review of literature.
Optimising glycaemic control in the younger population with type 1 diabetes
There are particular challenges in maintaining consistent glucose levels in young children and adolescents with diabetes.15,16 While young children are generally characterised by high insulin sensitivity, Dawn phenomenon is a prominent feature in adolescence, demonstrating early morning hyperglycaemia coupled with low levels of insulin growth factor-1 (IGF-1).17,18 Insulin resistance increases as children with T1DM approach puberty, and this coincides with the time when greater self-determination is desired, including whether or not they choose to comply with the need to manage their diabetes. This is reflected by increasing difficulty in achieving good glycaemic control, which persists until their mid-20s. 19
In its 2018 position statement on the management of T1DM in children and adolescents, 20 the American Diabetes Association (ADA) reminded clinicians that ‘children are not little adults’ and that their treatment ‘must not be extrapolated from adult diabetes care’. 20 Furthermore, research studies have shown that CGM helps clinicians, parents and people with diabetes to optimise glycaemic control.15,21
Continuous glucose monitoring
CGM devices consist of an embedded sensor having a minimally invasive subcutaneous probe, attached to the arm or abdomen. The probe measures glucose concentrations every few minutes in the interstitial fluid, using different technologies and transmit the data to a receiver (e.g. a cell phone or other reader device).22,23 Systems may display measurements in real time or intermittently. Real-time sensors (Dexcom [Dexcom Inc; San Diego, CA]; GlucoMen® Day CGM [A. Menarini Diagnostics srl; Florence, Italy]; Guardian™ and Enlite™ [Medtronic Inc; Minnesota, MN]; Eversense CGM system [Senseonics Inc; Germantown, MA]) continuously send glucose and temperature measurements to the receiver, whereas intermittent scanning CGM (isCGM) requires patients to scan the sensor with a handheld receiver, that can be a cell phone, to access glucose measurements (Freestyle Libre [Abbott Diabetes Care; Alameda, CA]).21,24 In the Freestyle Libre 2 device, an alarm function has been incorporated to alert of low or high glucose levels; however, the actual value is only displayed when a scan with the device reader, or smart phone, is performed. A later version of glucose sensor, Libre 3, does not require scanning; thus, behaving similarly to a traditional CGM, but still not widely available.
CGM can improve glycaemic control by providing insight into the direction, magnitude, duration, frequency, rate of change; thus, addressing possible causes of glucose fluctuations in response to meals, insulin injections, hypoglycaemic episodes and exercise.22,25,26 The advantages include convenience, reduced pain from repeated SMBG and a more comprehensive glucose profile27–30; thus, helping to improve time in range (TiR; 70─180 mg/dL [3.9─10.0 mmol/L]), decrease hypoglycaemic exposure and limit glucose variability.30–33 Moreover, it has a key role in reducing nocturnal hypoglycaemia, which is reassuring to patients and parents/caregivers. 34
The large quantity of raw data collected by the receiver is transformed into the ambulatory glucose profile (AGP) – an efficient, standardised and meaningful outline of glucose variability, post-prandial glucose excursions and hypoglycaemic trends amongst other information.25,35–41
In the last 10 years, there has been an increase in uptake of CGM by children and adolescents with T1DM. 42 In 2009, JDRF (formerly known as the ‘Juvenile Diabetes Research Foundation’) study found only 50% of 8–14-year-olds and 30% of 15–24-year-olds used CGM at least 6 days per week. 43 In 2020, another study found that 68% of 14–25-year-olds used a CGM device at least 5 days per week, but this group still monitored less frequently than adults. 16 Reasons for this may include physical pain associated with sensor insertion, embarrassment linked to wearing the device, anxiety, a sense of being overwhelmed by the quantity of information, alarm fatigue and the potential for conflict with parents who have access to data. 44 Nevertheless, many children and adolescents with T1DM and their parents report high satisfaction with CGM and particularly appreciate the ease of use and greater awareness of hypoglycaemia.44–46 and improve in glycaemic control.16,42,47
Trend arrows with CGM systems
Trend arrows and their clinical significance.

aValues for FreeStyle Libre 1 and 2 (Abbot Diabetes Care, Witney, UK).
bValues for Dexcom G5, G6 and 670 (Dexcom Inc, San Diego, CA).
cValues for Medtronic Veo and Medtronic 640G (Medtronic Inc, Minneapolis, MN).
dValues for Eversense Smart Transmitter (Senseonics Inc, Germantown, MA).
A person in otherwise steady-state conditions (between meals, not exercising and not under any stress) with an RoC of 2 mg/dL/min (0.1 mmol/L/min) will expect to see sensor glucose increase by about 60 mg/dL (3.3 mmol/L) in the following 30 minutes. 11 Upright up or down trend arrows for pre-meal glucose checks have been shown to be associated with a greater than two-fold increase in the risk of postprandial hyper- or hypoglycaemia, respectively 49 ; thus, representing a useful tool for improving postprandial glucose levels.
This information helps people with T1DM to accurately define mealtime bolus insulin doses, make correction doses or decide to eat a starchy snack or perform some exercise to return their glucose levels close to physiological glycaemia.12,14,41,49,50 Currently, however, there is no standardised protocol for interpreting trend arrows across different CGM systems (see Table 1). 11 Therefore, this work presents a simple approach to managing trend arrows in the younger population with T1D, under different conditions, in order to optimise glycaemia based on recent literature in the field. Moreover, we highlight the importance of adequate interpretation of trend arrows which offers a powerful tool in increasing self-management decisions thus helping with patient/carer engagement.
Trend arrows add a layer of sophistication to CGM systems. It is essential that people with T1DM, and their caregivers, are at first educated about CGM, and understand the effects of meals, day-to-day activities and different types of medication (including different forms of insulin) before starting to use trend arrows and sensor glucose levels to fine tune their insulin dose.10–12,51 People with diabetes will need support from HCPs to first establish individual glucose targets and then to learn to interpret arrows, adopt preventive actions and evaluate the impact of those actions through a new glucose reading. 52 Children and adolescents with T1DM and/or their parent or caregiver should be able to understand the potential impact of previous insulin doses, food intake, time since last meal, exercise levels, stress levels, etc., before making decisions about their next dose. 52 It is important to be aware that these arrows represent a trend, not a definitive numerical glucose value, and to take into account the 4–15-minute time lag between the interstitial fluid and the actual blood glucose concentrations, especially when glucose levels are rising rapidly (≥3 mg/dL/min or ≥0.2 mmol/L/min).11,53,54 Moreover, it should be acknowledged that the ‘action’ needed for trend arrows indicating a rise or fall in glucose levels cannot be standardised as it is likely differ between patients. To add to the complexity, managing trend arrows may need different approaches in the same patient according to circumstances (exercise, nocturnal changes, stress-related…etc.,).
Day-to-day use of trend arrows
Insulin dose adjustments based on correction factors and trend arrows for Freestyle Libre and Dexcom devices.
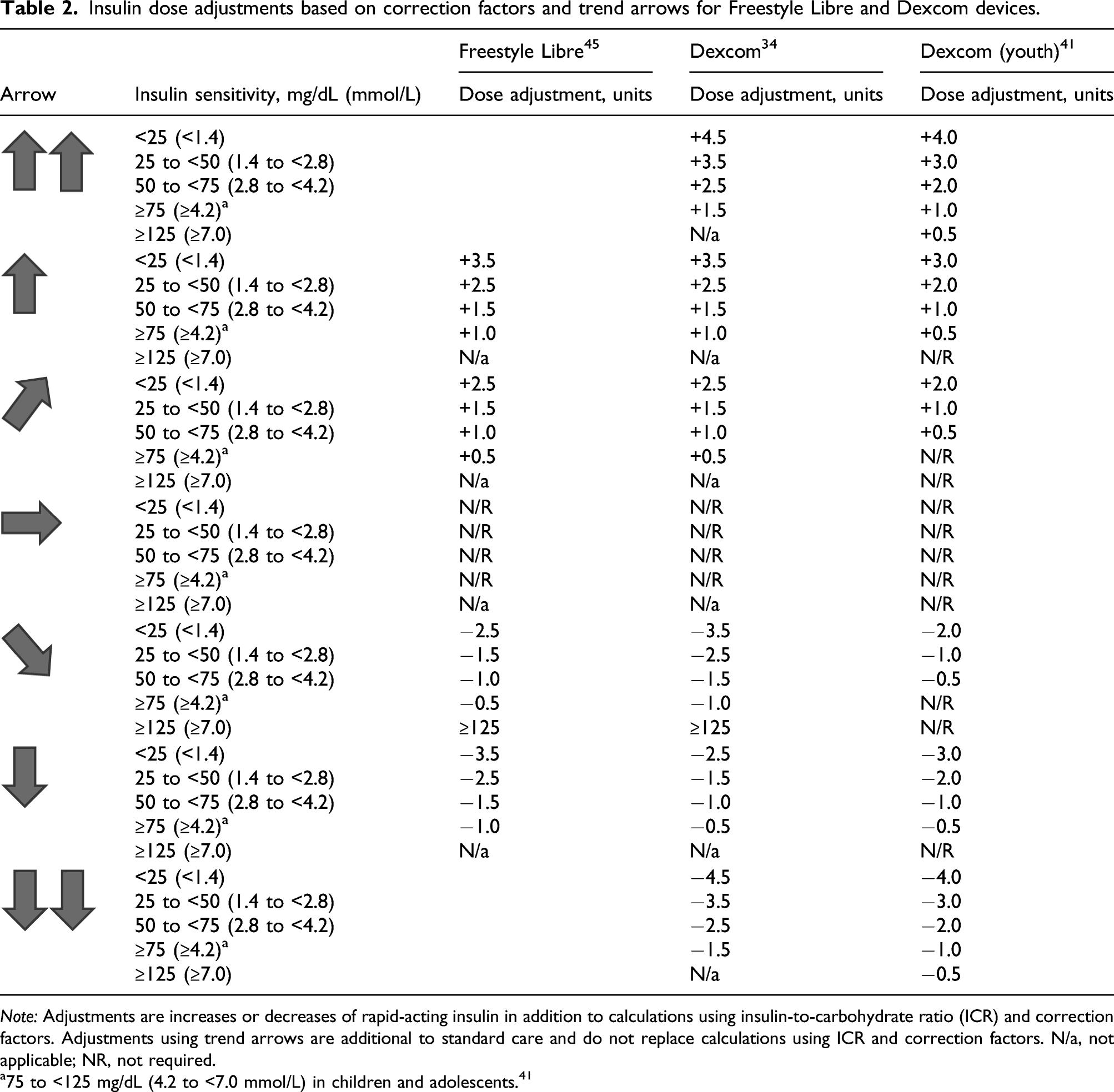
Note: Adjustments are increases or decreases of rapid-acting insulin in addition to calculations using insulin-to-carbohydrate ratio (ICR) and correction factors. Adjustments using trend arrows are additional to standard care and do not replace calculations using ICR and correction factors. N/a, not applicable; NR, not required.
a75 to <125 mg/dL (4.2 to <7.0 mmol/L) in children and adolescents. 41
Recommendations for the Dexcom device have been amended for younger people with T1DM. 51 To account for the greater variation in insulin sensitivity in this population, the authors included an additional insulin sensitivity band of 75 to <125 mg/dL (4.3 to <6.9 mmol/L) (Table 2). 14 In general, the bands relate to paediatric developmental stages: ≥125 mg/dL (≥6.9 mmol/L) is generally appropriate for toddlers/pre-schoolers, 75 to ≤125 mg/dL (4.3 to ≤6.9 mmol/L) for young school-age children, 50 to ≤75 mg/dL (2.8 to ≤4.2 mmol/L) for older school-age children, 25 to ≤50 mg/dL (1.4 to ≤2.8 mmol/L) for early pubertal teens and <25 mg/dL (<1.4 mmol/L) for pubertal teens. 51 While comprehensive, this can be daunting for some and therefore it is important to have an individualised approach to educating patients and caregivers on trend arrows.
Ziegler and colleagues have prepared a series of tables combining the aforementioned information plus glucose measurements to suggest suitable dose adjustments. One of these tables is for children and adolescents including trend information for CGM devices from all manufacturers, and recommendations for people in the hypo- and hyperglycaemic ranges. 11
For those injecting multiple daily insulin (MDI) doses, bolus doses should ideally keep postprandial glucose levels ≤162 mg/dL (≤9.0 mmol/L), with levels returning to their target level within the following 2–4 hours. 38 Upward trend arrows after 2–4 hours suggest the need for a higher bolus insulin dose, while downward may suggest a need for a lower dose or additional carbohydrate ingestion to avoid hypoglycaemia,10,13,51 which is based on typical insulin sensitivity ranges for paediatric patients.
Before making the decision to alter the bolus insulin dose, consideration should be given to the size and timing of last insulin dose, recent food intake, exercise and time of day any other factors that might influence the rate at which insulin is exhausted.12,13 Children and adolescents are advised to wait at least 3 hours after the prandial insulin dose before administering a subsequent correction dose to avoid insulin ‘stacking’ and potential hypoglycaemia. 14 Children may require an extra snack at bedtime if glucose levels are in the lower target range and are accompanied by downward arrows.
All children and adolescents with T1DM should check glucose more regularly after an insulin dose modification: those who increased their insulin dose should check hourly and those who decreased their insulin dose/ingested carbohydrate should review glucose readings more frequently to reduce the risk of hypoglycaemia. 13
Use of trend arrows under specific circumstances
The considerations listed above that influence the decision to alter a dose of bolus insulin in response to a trend arrow are brought into sharper focus on sick days, around exercise, during menstruation and at bedtime.10,14,51
Nocturnal hypoglycaemia
Nocturnal hypoglycaemia is common in children with T1DM and is potentially dangerous as they sleep deeply, are more likely than older people to participate in spontaneous exercise/physical play, have blunted nocturnal counter-regulatory hormone responses and are more likely to experience large fluctuations in glucose levels. 51 For some parents, fear of hypoglycaemia may discourage from trying to achieve tighter glycaemic control. 46 Laffel and colleagues 51 recommend a bedtime glucose target of ∼130 mg/dL (∼7.2 mmol/L) with a flat or up-angled trend arrow. Extreme caution should be exercised when adding extra bolus doses or correction doses close to bedtime. In contrast, a downward arrow before bed should be taken seriously, and an early recheck in glucose is required or alternatively carbohydrate can be given to avoid a further fall in glucose (depending on actual levels).
Sick days
The effects of illness and any accompanying medications can result in unstable glucose levels and a more intense monitoring regimen, and trend arrows can be of particular value in such a scenario. As a minimum, in accordance with international guidelines, 55 monitoring should be more frequent on sick days and patients may consider more frequent insulin corrections (e.g. every 2–3 hours), 13 guided by glucose levels and trend arrows. Patients are recommended to do more frequent ketone testing, especially if they are on agents that can interfere with CGM readings (acetaminophen with some devices and/or high doses of ascorbic acid), which may impair some sensor glucose readings; thus, running the risk of hypoglycaemia.10,13,51,55
Exercise
International diabetes organisations recognise that exercise and physical activity in general are essential for children’s well-being and can have a positive effect on glycaemic control, blood lipid profiles, physical fitness and quality of life and can decrease the total daily dose of insulin.
56
However, intense physical activity can result in dysglycaemia as a result of increased insulin sensitivity.
57
Type, duration, intensity and timing of exercise, composition and timing of meals prior to, during and after exercise, pre-exercise glucose levels and injection sites influence glucose levels during and after exercise.56,57 Low to moderate intensity aerobic exercise mainly increases glucose uptake and insulin sensitivity, hence increasing the risk of hypoglycaemia for 24 hours (or even longer) after exercise, while anaerobic moderate to high intensity exercise may lead to acute hyperglycaemia but can also increase the risk for nocturnal hypoglycaemia.10,57,58 Exercise intensities can be assessed via the following question and answers: ‘Can you speak comfortably during a prolonged exercise session?’ ‘Yes, easy’ = low intensity aerobic exercise. ‘Yes, but it is somewhat difficult to carry on a full conversation’ = moderate intensity aerobic exercise. ‘No, too difficult’ = Intense (an)aerobic exercise.
Trend arrows (and alerts where available) are particularly beneficial before, during and after exercise and other physical activity as the onset of hypoglycaemia may be less noticeable due to the masking effect of exertion.14,51,56 In general, people using exogenous insulin are recommended to plan their exercise; however, this is generally not practical for young children due to the spontaneous nature of play.51,56 International guidelines recommend re-setting alerts to more sensitive levels before, during and after exercise, taking into account the expected glucose response to exercise (increase, stabilising or decrease) and the patient’s general risk of hypoglycaemia (Figure 1).
56
Glucose checks should be performed as frequently as practical to increase awareness of changes in glucose levels. Exercise should be discontinued at least temporarily if sensor glucose levels drop below 90 mg/dL (5.0 mmol/L),
56
particularly in the presence of a downward trend arrow. Therapy recommendations during exercise for children and adolescents with low risk of hypoglycaemia and exercise experience. (a) Elevated blood ketone levels should lead to repeated controls after exercise to avoid ketosis or diabetic ketoacidosis. If sensor glucose is >270 mg/dL (>15.0 mmol/L) and blood ketones are >1.5 mmol/L, then only mild aerobic exercise may be continued. (b) 50% of regular insulin correction factor. (c) Restart exercise when reaching sensor glucose levels of ≥90 mg/dL (≥5.0 mmol/L) and upward trend arrows. (d) Check sensor glucose at least 30 min after carbohydrate consumption and repeat treatment if required. Adapted from Moser et al. Pediatric Diabetes, 2020.
56
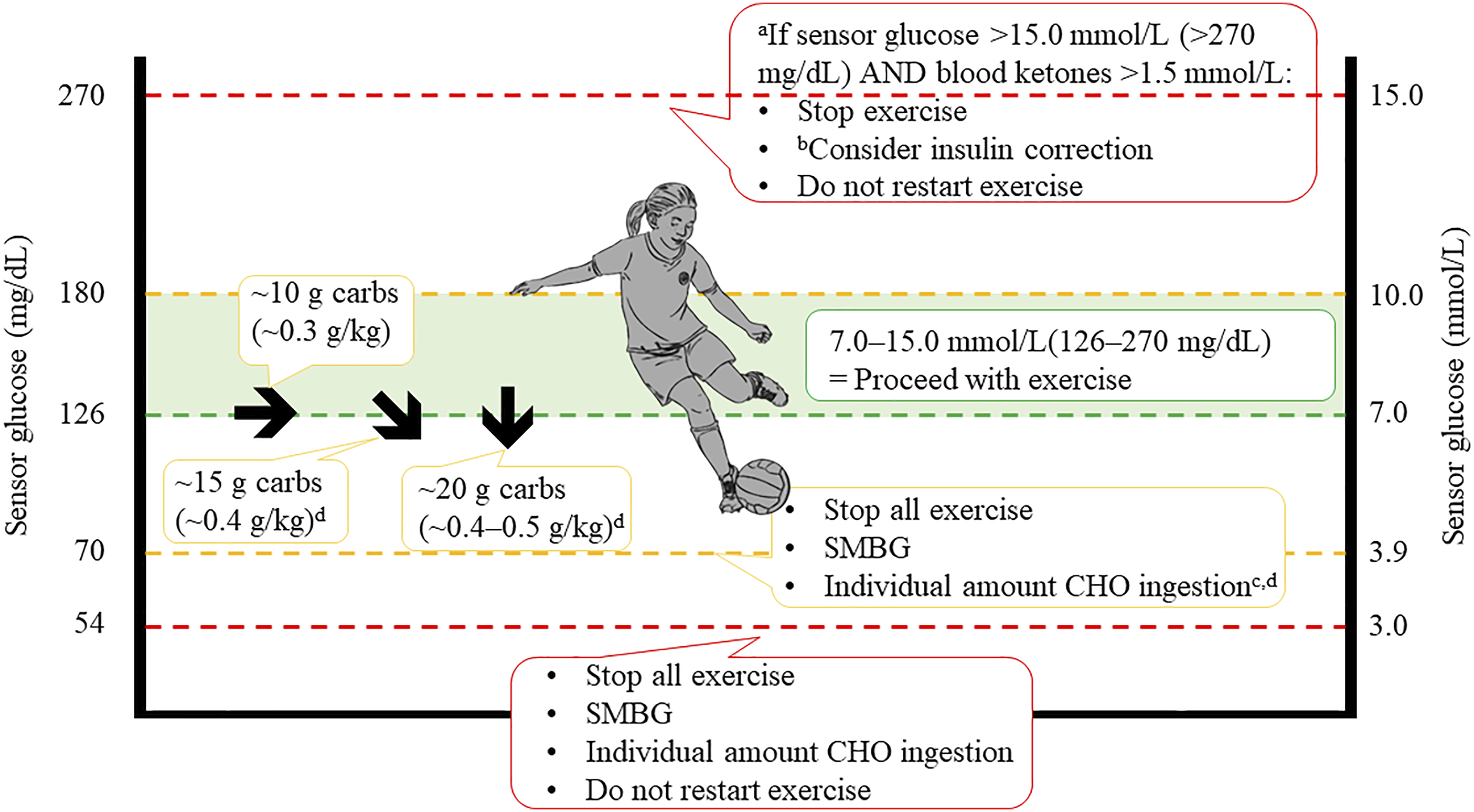
Frequent monitoring should continue for at least 2 hours after exercise and for 6–8 hours after prolonged aerobic exercise,10,13,51,56 with special attention to trend arrows. Starchy snacks should be given in response to sensor glucose levels at or below 80 mg/dL (4.4 mmol/L) and small bolus insulin corrections can be considered (50% of the regular insulin correction dose) if sensor glucose is increasing rapidly and/or at very high sensor glucose levels. 56 Especially after high intensity (interval) training and resistance training, acute hyperglycaemia can occur due to the catecholamine effect on hepatic glycogenolysis and gluconeogenesis.
In real life, some patients manage glucose levels below 90 mg/dl during exercise by having a high glucose snack and continue their exercise programme. However, this should be discouraged, and a break is advised together with carbohydrate supplement with exercise resuming once glucose levels and trend arrows stabilise and show no further drop.
Insulin dose corrections close to bedtime should be avoided as it may increase the risk of post-exercise nocturnal hypoglycaemia. During the nocturnal phase, the CGM hypoglycaemia alert should be set at a higher level than on resting days and those that are using isCGM should perform one or two scans (e.g. at 1 a.m. and 3 a.m.).
Ramadan
Data from CGM downloads have indicated increased risks of hypoglycaemia among young people with diabetes who fast during Ramadan, especially during daylight hours. 59 ISPAD guidelines for young people with T1DM provide guidance on the use of CGM and isCGM during Ramadan fasting, 60 and ASPED has recently published recommendations for the use of CGM in the Middle East and North Africa region which include advice for managing glucose levels in Ramadan. 61 Furthermore, several studies have demonstrated that CGM with trend arrows can help adults and children to fast without the risk of life-threatening episodes of severe hypoglycaemia or diabetic ketoacidosis (DKA), provided that they are well trained on the use of the technology.62,63 In a prospective pilot study 51 children with T1DM (mean age 14 years), all participants were taught how to use isCGM with trend arrows to manage their glucose levels at home prior to Ramadan. Participants were able to fast on 67% of days during Ramadan: the main reason not to fast was hypoglycaemia, but no child experienced severe hypoglycaemia or DKA during this period. 63
Menstruation
Changes in oestradiol and progesterone levels during the menstrual cycle have been shown to influence insulin sensitivity. CGM shows that the risk of hyperglycaemia increases significantly throughout the first half of the cycle while the risk of hypoglycaemia increases in the second half, albeit not significantly. 55 These changes are highly variable, and post-pubertal girls and women need to pay closer attention to glucose levels in the second half of their cycle. 64
Differences in the calculation of rate of change of glucose
Studies have found the time-lag in glucose readings from CGMs compared to plasma glucose concentrations to be of approximately 5–10 minutes during hyperglycaemic excursions.65,66
Also, using model simulations, it has been suggested that CGM devices may overestimate low glucose values and underestimate high glucose values, which can complicate patient management. 66 However, lag time of newer CGM sensors has been cut down to an average of less than 2.5 minutes across a wide range of glucose levels with superior accuracy documented in low and high glucose ranges, emphasising the progress made with optimising glucose algorithms of the device. 67
Glucose fluctuations can also be high following hypoglycaemic events, which can lead to a period of glycaemic instability. Such glucose swings might be even more pronounced after severe hypoglycaemia events; however, to a certain extent, they can also occur after mild or moderate hypoglycaemia. 68
While a large number of studies have investigated accuracy of CGM devices, caution should be exercised when interpreting the data. Some used capillary glucose monitoring devices for comparison, which themselves have accuracy issues even with the use of devices that undergo regular quality control checks. 69 Therefore, for testing accuracy, glucose levels should be checked using a reliable method, and the FDA developed clear guidance on the standards required. 70 Mean average relative difference (MARD) in glucose of a device compared with a reliable standard has been used as a simple measure to assess accuracy, although this is not without flaws. 71 In general, it is currently accepted that a CGM device is clinically safe to use once MARD drops below 10%, provided a wide range of glucose levels are tested.
Real-time continuous glucose monitoring devices help to detect glycaemic excursions associated with exercise, meals and insulin dosing in patients with type 1 diabetes. However, the delay between interstitial and blood glucose may result in CGM underestimating the true change in glycaemia during activity. The accuracy of newer CGM technology can be impacted during prolonged aerobic exercise, and patients need to be aware of this potential CGM ‘time-delay’. In some devices (using Dexcom G4 Platinum with 505 algorithm and G5 technology), lag time was over 10 minutes behind SMBG readings during exercise with a bias of 7–19 mg/dL/min (0.4–1.1 mmol/L/min), coupled with MARD increasing to 13% (6–22%). Due to this clinically important delay in CGM versus SMBG, patients should increase vigilance and perform more frequent capillary glucose tests around exercise if hypoglycaemia is suspected. 72
The role of education
With the increasing use of CGM, there is a greater opportunity for patient engagement and motivation. However, using these devices to optimal benefit requires adequate and thorough patient education. This includes explaining AGP, interpreting glucose data and trend arrows. Equally, it is important to ensure sufficient training for HCPs in device use and data interpretation, both in primary and secondary care settings. 73 Therefore, education programs should be developed for patients and caregivers in order to maximise the benefits of such devices.
To benefit from trend arrows, patients must be instructed on how to correctly read and act on these cues. Education should include actively showing patients the trend arrow icons on their CGM reader, followed by appropriate discussion of their importance. Next, patients should be instructed on how to react to the trend arrows, such as through medication adjustment, effects of diet, change in activity level, or actions to prevent hypoglycaemia and optimise their time in range.10,13,51
AGP can serve as a powerful tool for education about diabetes self-management and teach families a dynamic approach to diabetes management. 74
CGM data also give HCPs insight into patients’ behaviours and glycaemic patterns and may reveal previously undetected issues such as hypoglycaemia.75,76 Retrospective review of CGM data provides opportunities for patient education and encourages communication with the health care professional to make shared decisions in order to optimise glycaemic control.
The hidden benefit of trend arrows
Physiological, educational and emotional development during childhood and increasing levels of self-determination call for regular reviews of individual target levels and ways of engaging children and adolescents with T1DM to ensure that targets are met. Recent evidence suggests that using trend arrows frequently improves diabetes management and increases patient engagement. 51 Moreover, recent work has shown that appropriate use of trend arrows with adequate education does not only improve glycaemia but also reduces total insulin dose. 77
Understanding trend arrows is likely to increase understanding of patients and caregivers of diabetes; thus, offering further motivation. Patients are motivated by the ability to make in-the-moment adjustments and decisions about fine-tuning of insulin doses, correction factors and carbohydrate content of meals. Education on the use of trend arrows is essential, and when done well can enable young people to navigate difficult circumstances with reduced fear of hypoglycaemia, particularly in special situations such as exercise or fasting during Ramadan without the risk of life-threatening episodes of severe hypoglycaemia or DKA.61,64
Conclusions
This narrative review summarises the use of trend arrows by young people with T1DM and offers some recommendations by expert diabetologists. Correct use of trend arrows with adequate education has the potential to optimise glycaemic control, by lowering glucose, minimising hypoglycaemia and limiting glucose variability, while also improving engagement with self-management of diabetes. While currently there are no standardised, evidence-based guidance for the use of trend arrows in decision making and therapy adjustment in the younger people with diabetes, evidence for the use of trend arrows is increasing, and a simple approach to the management of this population is presented in Figure 1. More clinical trials on the use of CGM trend arrows in children and adolescents with T1DM and analyses of real-world data are needed to provide evidence on which such guidance could be based. It should be emphasised that education around the use of trend arrows should be individualised as patients/caregivers differ in the ability to acquire information. Also, it is best to have a step-by-step approach with focus on the CGM system used by the patient, given differences in trend arrow display comparing various CGM devices.
Footnotes
Acknowledgments
The authors wish to express their sincere appreciation to the additional international guest advisors, Fiona Campbell and Iain Cranston.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been funded by an unrestricted medical education grant from Abbott Laboratories GmbH.
Ethical approval
ASPED ethics committee has approved the study, and the study conforms to recognized standards.
Authorship
All authors participated in the decision to publish this manuscript and all were involved in drafting, reviewing and revising the outline and subsequent full draft of the manuscript.
