Abstract
Continuous glucose monitoring and flash glucose monitoring systems are increasingly used by people with diabetes on multiple daily injections of insulin and continuous subcutaneous insulin infusion. Along with real-time updates on current glucose levels, these technologies also use trend arrows to provide information on the direction and rate of change of glucose. Two systems, the Dexcom G5 and the FreeStyle Libre, have recently been approved for use without the need for adjunct capillary blood glucose, and there is a need for practical guidance for insulin dosing which incorporates rate of change in the insulin dosing algorithm. Here, we review the integration of rate of change trend arrow information into daily glucose management, including rapid-acting insulin dosing decisions. Based on the FreeStyle Libre flash glucose monitoring system, we also review a practical decision-support tool for actions to take when using trend arrows in conjunction with current glucose readings.
Keywords
Introduction
Continuous glucose monitoring (CGM) and flash glucose monitoring systems measure real-time glucose in the subcutaneous interstitial fluid (ISF) of users, rather than in capillary blood. A number of studies indicate that CGM and flash glucose monitoring devices improve glycaemia and reduce the risk of hypoglycaemia, particularly in those with type 1 diabetes.1–4 A noted advantage of such ISF sensor devices is that, alongside the current glucose reading, they typically provide a trend arrow that indicates the direction and rate of change (RoC) of glucose. Such trend arrows can be an important aid to assist decision-making for people with diabetes who use either CGM or flash glucose monitoring devices.
An important observation is that a rising pre-meal glucose confers a twofold increase in the risk of postprandial hyperglycaemia, whereas a falling pre-meal glucose is associated with a twofold increase in the risk of postprandial hypoglycaemia. 5 Therefore, the pre-meal RoC may be used to alter insulin dosing decisions and ultimately improve postprandial glycaemic control. 5 In this article, we review the available tools that can help with insulin decisions and explore their advantages and limitations.
Interpreting RoC trend arrows requires the user to understand how this information may be used within the context of their diabetes day. Although subjects in clinical trials have been provided with treatment algorithms for managing their diabetes in line with CGM data,6–8 real-world guidance for use of glucose RoC information alongside current glucose readings provided by CGM or flash glucose monitoring systems is limited. The UK Association of Children’s Diabetes Clinicians guidance on flash glucose monitoring is an established tool in this respect. 9
Recent surveys by Pettus and colleagues10,11 have suggested that people with diabetes using CGM rely on RoC trend arrows to make insulin dosing adjustments, both when faced with making correction doses in response to incidental high glucose and when calculating a mealtime bolus. Importantly, in response to RoC trend arrows, respondents using CGM made significantly larger dose adjustments than would be recommended by published algorithms for using RoC trend arrows for insulin dose management. 12
In this context, a discussion of best use of RoC trend arrows is warranted, which outlines the different scenarios in which RoC trend arrows may be interpreted. An important outcome of this discussion is to identify the actions that might be taken by the user, including how this impacts dose adjustments for mealtime insulin and also for correction doses for incident hyperglycaemia between meals. Just as important is that these actions should be simple to understand and follow.
What do trend arrows show?
Trend arrows indicate both a direction and a RoC for glucose that help the user project what their glucose levels will be over the next 15–30 min. Consequently, they provide important guidance for making diabetes self-management decisions, not possible with fingerprick capillary blood glucose (BG) testing.
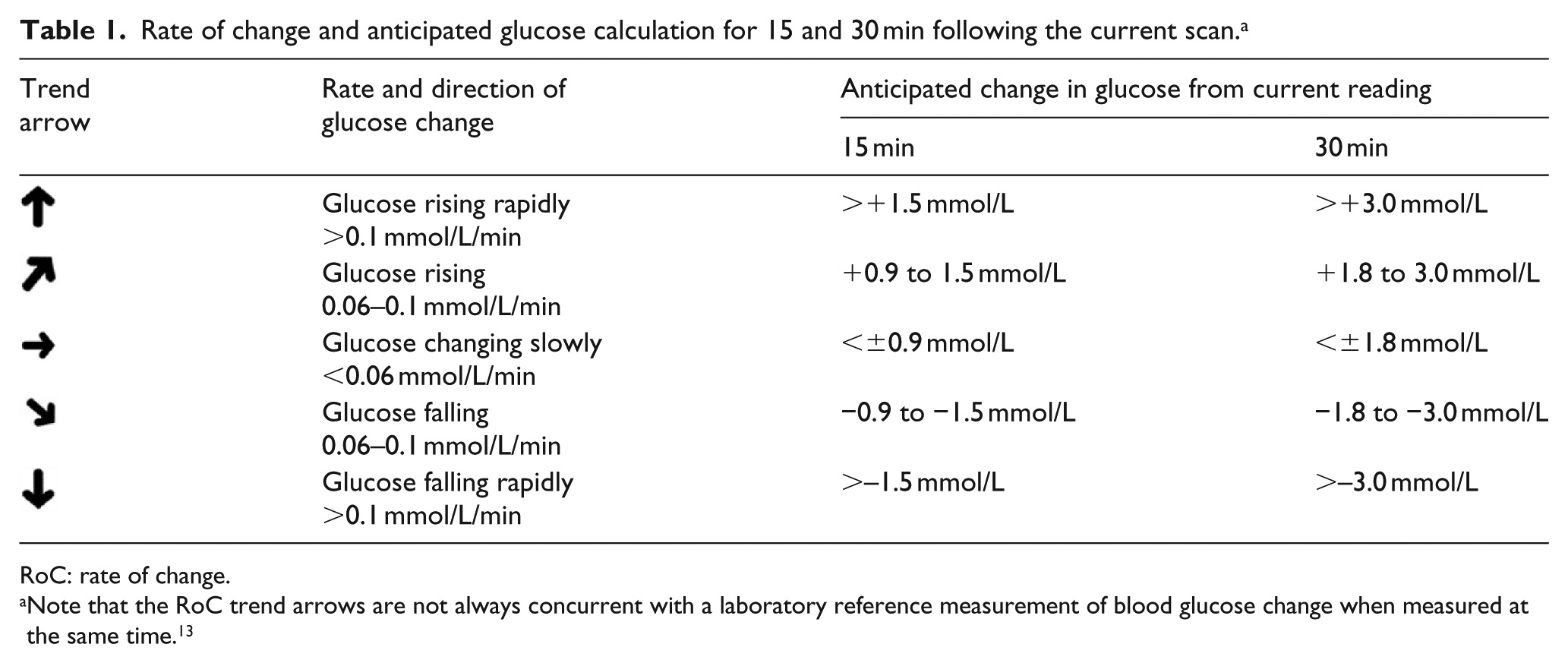
The FreeStyle Libre flash glucose monitoring system quantifies the RoC of glucose based on five trend arrow orientations. These are shown in Table 1, along with the calculated anticipated change in glucose for 15 and 30 min.
Thus, depending on their current glucose reading, each of the five trend arrows provides the user of the FreeStyle Libre system with important information about the need to make a treatment choice now, or to wait and keep scanning. It is important to note that the RoC trend arrows are not always concurrent with a laboratory reference measurement of changing BG, 13 and users should always follow the manufacturer’s guidance as to when a capillary glucose measurement may be required. This includes when low glucose is indicated and also when glucose is changing rapidly as indicated by trend arrows that are pointing vertically up or down.
Bolus insulin and glucose trends
When confronted by a glucose reading and a rising trend arrow, a key consideration is whether insulin from the most recent mealtime bolus is still active or not. This is illustrated in Figure 1.
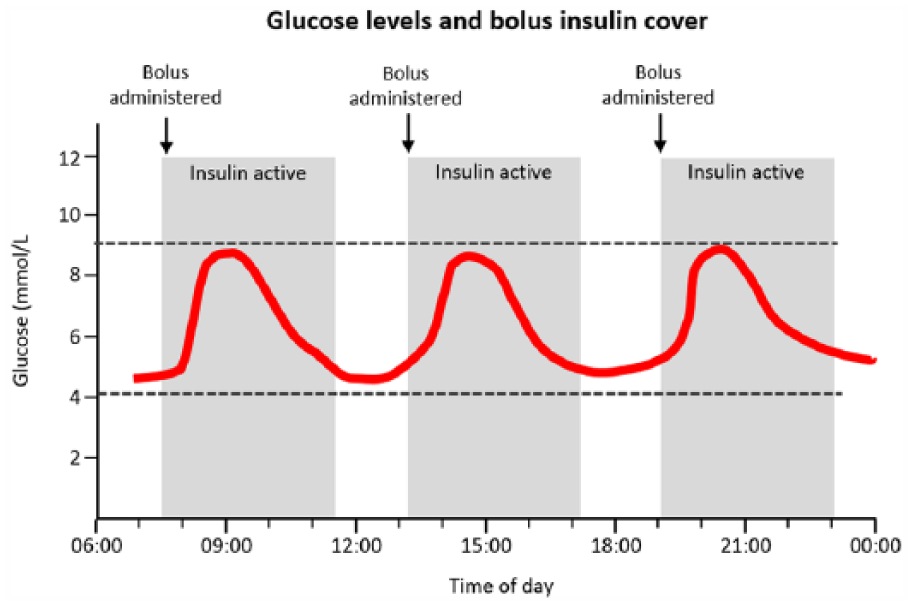
Idealised scenario for bolus insulin action and mealtime glucose control.
Under ideal circumstances, a pre-meal rapid-acting insulin bolus results in a postprandial glucose excursion of no more than 9.0 mmol/L, 14 which returns to target over an approximate 4-h period. As the insulin bolus expires, glucose is held stable by basal insulin.
A person with diabetes using CGM or flash glucose monitoring in this scenario will see their current glucose rising and falling, alongside a trend arrow that reflects this idealised dynamic. To achieve a postprandial glucose excursion <9.0 mmol/L, most users will usually need to deliver the meal bolus >15 min pre-meal, accurately count carbohydrates and apply an insulin:carbohydrate ratio which they know meets their individual requirements.
Using glucose readings and trend arrows in real life
In reality, and due to inherent variability in insulin requirements in type 1 diabetes, many people using CGM or flash glucose monitoring will find themselves in situations that will not match the ideal scenario described. They may check their glucose and note that it is rising above their target glucose range and will need to decide on what action to take, if any. Similarly, if they see their glucose falling ahead of a scheduled mealtime bolus, they will potentially need to recalculate the timing or dose of the insulin injection.
A range of scenarios is described in Figure 2. The ideal situation is shown in Figure 2(a), with an acceptable excursion of up to 9.0 mmol/L that subsequently returns to pre-meal levels. Figure 2(b) to (d) represents situations where glucose management potentially requires adjustment.
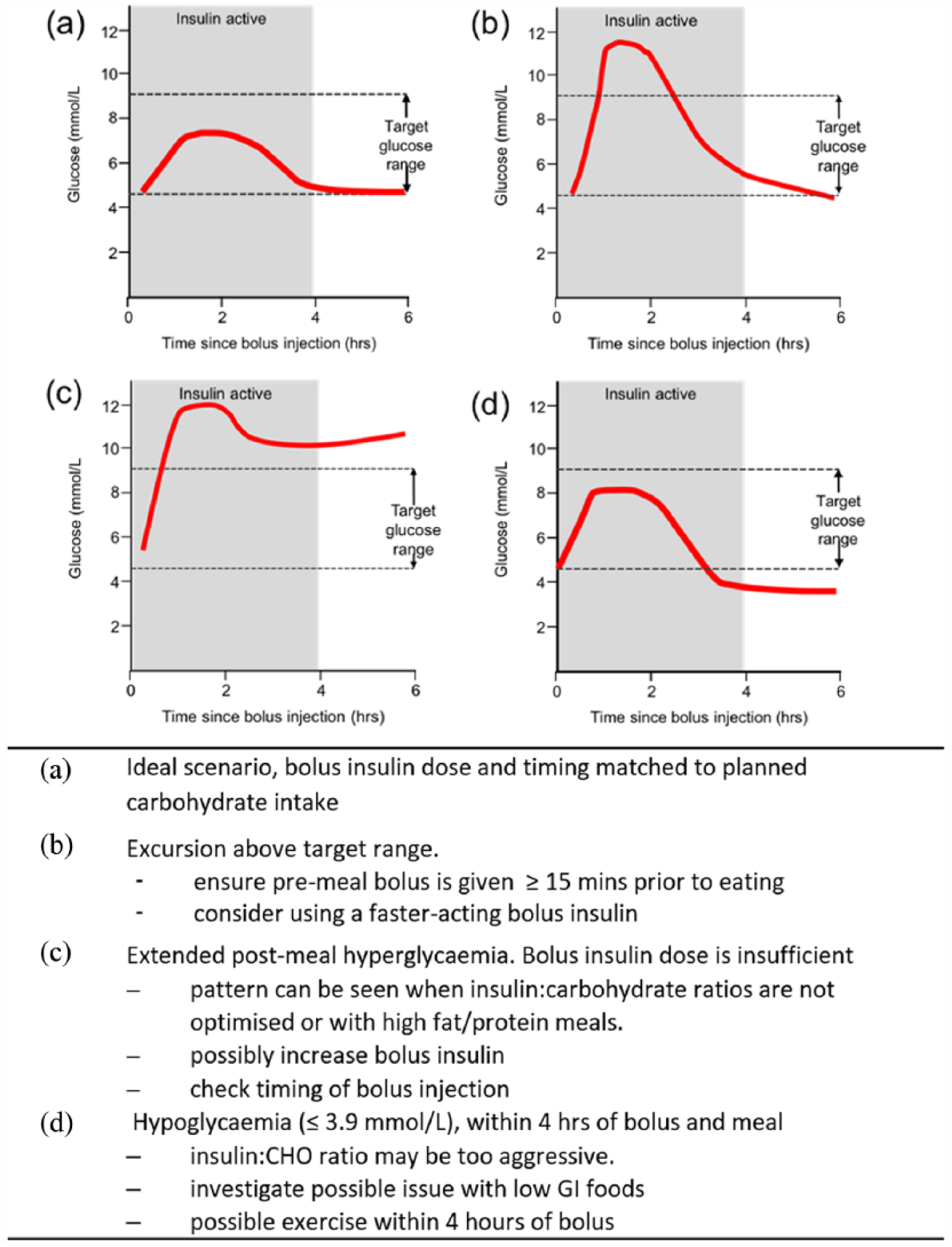
Real-world scenarios for bolus insulin and glucose control. (a) Ideal scenario (b) Excursion above target range. (c) Extended post-meal hyperglycaemia. (d) Hypoglycaemia (<3.9 mmol/L), within 4 h of bolus and meals.
In each case, whenever a person using CGM or flash glucose monitoring systems checks their glucose and sees the associated trend arrow, they will need to consider:
When was their last bolus of insulin administered?
How much insulin did they take and is insulin still active in their system – that is, was it within the last 4 h?
What is the impact of recent meals or snacks?
Have they undertaken any recent exercise, or do they expect to in the immediate future?
Are there any other glucose modulating factors that need to be taken into account, for example, stress, sickness or menstruation.
Similarly, overnight trends need to be considered as part of the overall picture of glycaemic control. Figure 3 shows a number of scenarios that represent possible evening and overnight glucose patterns and their interpretation in context of basal and bolus insulin.
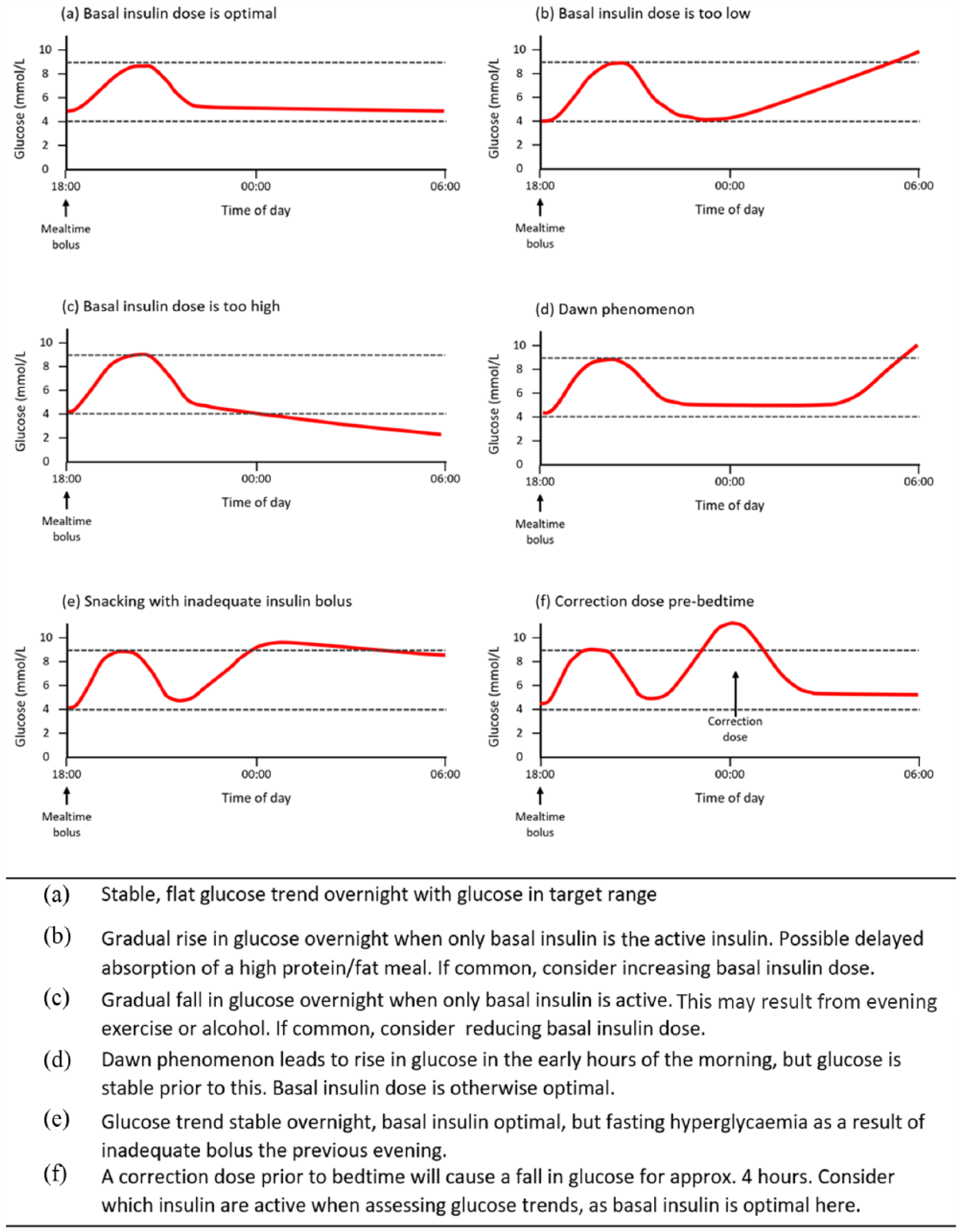
Patterns in overnight glucose trends. (a) Stable, flat glucose trend overnight with glucose in target range. (b) Gradual rise in glucose overnight. (c) Gradual fall in glucose overnight when only basal insulin is active. (d) Dawn phenomenon. (e) Glucose trend stable overnight. (f) Effects of a correction dose prior to bedtime.
Evidence base for trend arrow adjustment tools and bolus insulin calculations
DirectNet/JDRF study method
In two notable studies in type 1 diabetes, the Juvenile Diabetes Research Foundation Continuous Glucose Monitoring (JDRF CGM) study 7 and the DirecNet Applied Treatment Algorithm (DATA), 8 participants were provided with a bolus insulin adjustment tool for use alongside their current glucose readings and the associated trend arrows. The DATA study investigated trend arrow guided insulin adjustments to pre-meal boluses, whereas the JDRF study analysed trend arrow guided adjustments both to pre-meal boluses and between-meal correction doses. These adjustment algorithms are summarised in Table 2, and have helped promote a ‘10%/20% rule’ for correction insulin dosing, which accommodates the information provided by trend arrows alongside a current glucose reading (Table 3).
Trend arrow guided bolus insulin adjustment guidance.

It should be noted that this is only a guide and individualised adjustments in insulin dose may be required according to the needs of each patient.
Elevated glucose in the post-meal period.

It is worth noting that all the study subjects in the DATA trial indicated that the 10%/20% treatment algorithm gave good, clear directions for insulin dosing, as measured by an algorithm satisfaction questionnaire. 4
CGM trend arrow adjustment tools
To combat potentially confusing and discouraging mathematical calculations to manage mealtime insulin dosing decisions, two groups have proposed trend arrow adjustment tools (TAATs), in which each trend arrow orientation is associated with a fixed, pre-calculated dose-change to the regular mealtime insulin bolus.15,16 The first of these was developed and tested on children on continuous subcutaneous insulin infusion (CSII) therapy and directed them to add or subtract either 0.5 or 1.0 unit of mealtime insulin depending on the trend arrow orientation and receiver operating characteristic curve (ROC). 15 This study showed that this was as effective as using the 10%/20% method and seen as preferable among a pool of 20 children and adolescents, some as young as 5 years old. 15 Similarly, an as-yet untested TAAT for insulin-treated adults has recently been proposed, directing a fixed 1.0, 1.5 or 2.0 unit insulin adjustment depending on the direction and RoC shown by the trend arrows. 16
Both TAATs assume a standardised insulin sensitivity factor for insulin users, and the second one also assumes that the rate and direction of change in glucose for each trend arrow will be consistent for 45 min following a pre-meal reading.
Overall, one of the two methods discussed above can be used by individuals using CGM/flash glucose monitoring to make adjustments to their insulin dosing, taking the trend arrow direction and RoC into account. It should be emphasised that the evidence base underpinning TAATs is limited and currently derived from the paediatric setting, which restricts the generalisability. Furthermore, Pleus et al. 17 have highlighted the limitations of trend arrows for dosage adjustment as the actual BG trend often deviates from the CGM trend, in 10% of Dexcom cases and 8% of Freestyle Libre cases.
Using trend arrows between meals, when last bolus insulin is still active
An important real-world scenario not addressed in these study algorithms is when using trend arrows to adjust between-meal bolus doses in the approximate 4-h period following a mealtime bolus, during which time the mealtime insulin bolus will still be active. It is not uncommon during this period that a user will see their current glucose reading above target and may consider a correction dose of insulin to bring their glucose back towards target.
Under these circumstances, a person using CGM or flash glucose monitoring will need to consider several factors: (1) their current glucose reading; (2) the RoC in glucose as indicated by the trend arrows; (3) any ongoing action of their most-recent bolus, as discussed below; and (4) any planned or recent physical activity.
In scenarios where high glucose is present within 4 h of a mealtime bolus injection (e.g. Figure 2(b) and (c)), the trend arrows can assist the user to decide on a correction dose, based on their projected glucose levels and how recently mealtime insulin was injected. Since there will still be significant insulin activity within 2 h of the mealtime bolus, the most likely action will be to continue to scan and not administer a correction dose.
Using trend arrows to calculate mealtime insulin doses
Diabetes self-management includes taking a glucose reading before a meal, such that the user can calculate and deliver the necessary bolus insulin dose usually >15 min before eating with most short-acting insulin preparations. 18 The trend arrows provided by CGM or flash glucose monitoring add important information that impacts the mealtime insulin calculation, as illustrated in Figure 4.
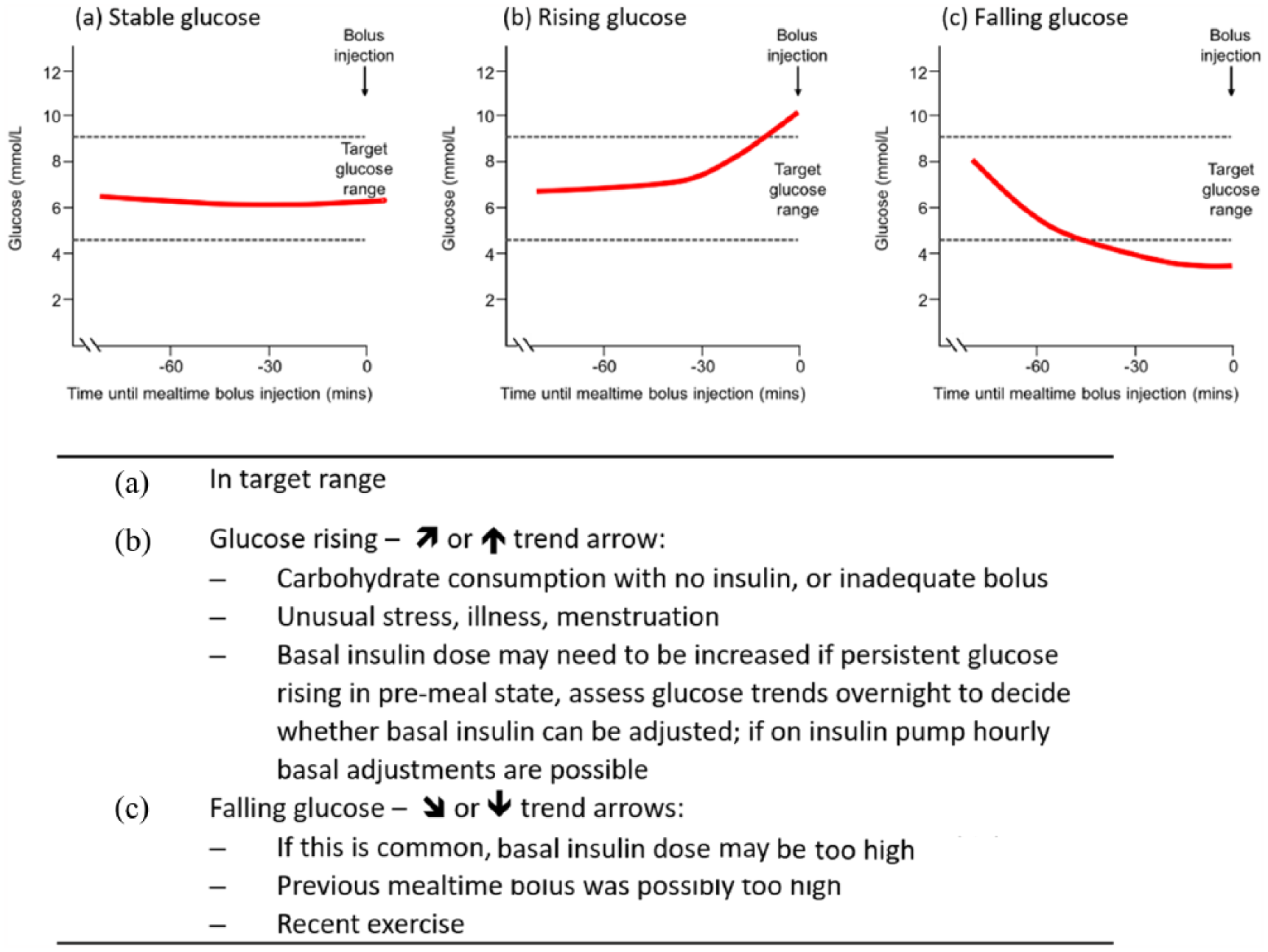
Pre-meal glucose scenarios (assuming last bolus > 4 h ago). (a) In target range. (b) Glucose rising. (c) Glucose falling.
The trend arrows that accompany CGM and flash glucose monitoring readings can help the user to calculate a correction to bolus dose, appropriate to their current glucose, factoring in where it is heading, and also accommodating the speed of that change. Under these circumstances, the 10%/20% guidance provided in the JDRF CGM study 7 and the DATA study 8 regarding changes to the bolus insulin calculation are appropriate (Table 2).
Using trend arrows between meals, with no bolus insulin active
In situations where a person with diabetes is between meals and has not had a bolus injection within the last 4 h (Figure 2, 4–6 h), their response to an incident of high glucose reading (Figure 2(c)) can be guided by the trend arrow that appears with their glucose reading, whether rising, falling or stable. The trend arrow will allow them to project where their current glucose reading is heading in 15–30 min, and thus indicate the size of the necessary correction dose can be increased or decreased by 10% or 20%, as indicated in the DATA and JDRF algorithms.7,8
Putting all this information together can help a person with diabetes make better-informed decisions about when and what treatment is needed. However, the additional complexity can also be daunting. Thus, there is a need to provide supportive guidance that maximises the benefit of the technology.
Decision support using the FreeStyle Libre flash glucose monitoring system
In general, when considering both the current glucose reading and the associated trend arrow, there are two actions that need to be addressed: (1) taking carbohydrate to avoid hypoglycaemia and (2) taking a correction dose of insulin to address a high glucose reading.
Taking action against hypoglycaemia
Flash glucose monitoring helps users avoid hypoglycaemia, as proven in the IMPACT and REPLACE studies in type 1 and type 2 diabetes.1,2 The time that users of the FreeStyle Libre flash glucose monitoring system spent below 3.9 mmol/L was reduced by 38% (IMPACT) and 43% (REPLACE), compared to subjects using Self-monitoring of blood glucose (SMBG). HbA1c was similar in the FreeStyle Libre and SMBG study arms, so overall glucose control was not changed. Both studies support the safe use of the Freestyle Libre system for non-adjunctive use. Moreover, both IMPACT and REPLACE showed significant improvement in quality of life measures and treatment satisfaction in the Libre arm compared with SMBG, indicating that the new sensor technology improves patient well-being in general.
These observations have been further supported in two randomised clinical trials in adults with type 1 diabetes treated with multiple daily injections, and using the Dexcom G4 Platinum CGM system.3,4 In the GOLD study, 3 CGM users spent only 2.79% of each day with glucose levels below 70 mg/dL (3.9 mmol/L) compared to 4.79% for patients using SMBG. In the DIAMOND study, 4 the CGM users spent 2.99% of each day with glucose below 70 mg/dL compared to 5.55% for SMBG users.
Thus, based on a current glucose reading at the lower end of their target glucose range, and seeing a falling trend arrow, the user can make a judgement about their risk of becoming hypoglycaemic, taking into account active insulin on board and recent physical activity.
If they scan and read current glucose is <4 mmol/L, they must check BG and if hypoglycaemia is confirmed, it must be treated. When using CGM/flash monitoring after treating a hypo, the user needs to be acutely aware of the 5–10 min lag which means the CGM/flash monitor will display hypoglycaemia for a further 5 min despite the BG having risen into the target range. During this time of rapid change in values, blood testing is advocated as the most accurate measure of glucose levels.
Correction doses to manage hyperglycaemia
The decision to take a correction dose of insulin to address a high glucose reading or adjusting a pre-meal bolus to accommodate a reading above target must take into account: (1) current glucose reading, (2) the RoC in glucose as indicated by the trend arrows, (3) any ongoing action of most-recent bolus and (4) planned or unplanned physical activity.
Bolus dose decision making is complex and the 10/20% rules may be a step too far for many insulin users. Using a bolus calculator, such as the Roche Expert meter, which allows bolus dose adjustment by ±10 or 20%, can assist users. However, the correction dose calculation usually requires a BG measurement, which limits its application and prevents non-adjunctive use of the Freestyle Libre and Dexcom systems.
We have therefore summarised the interplay between current glucose, RoC as indicated by trend arrows, and insulin dosing needs into a simple decision-support tool (Table 4). For any current glucose scan, a user can identify the glucose range in question, the direction and RoC of glucose using the trend arrow displayed and access brief guidance on a course of action. A traffic-light code is used to identify when there is a need for: immediate urgent attention (red); possible action, but without urgency (amber); and no immediate need for action (green).
Decision-support tool for matching current glucose and rate of change (as indicated by trend arrows), with treatment needs. a
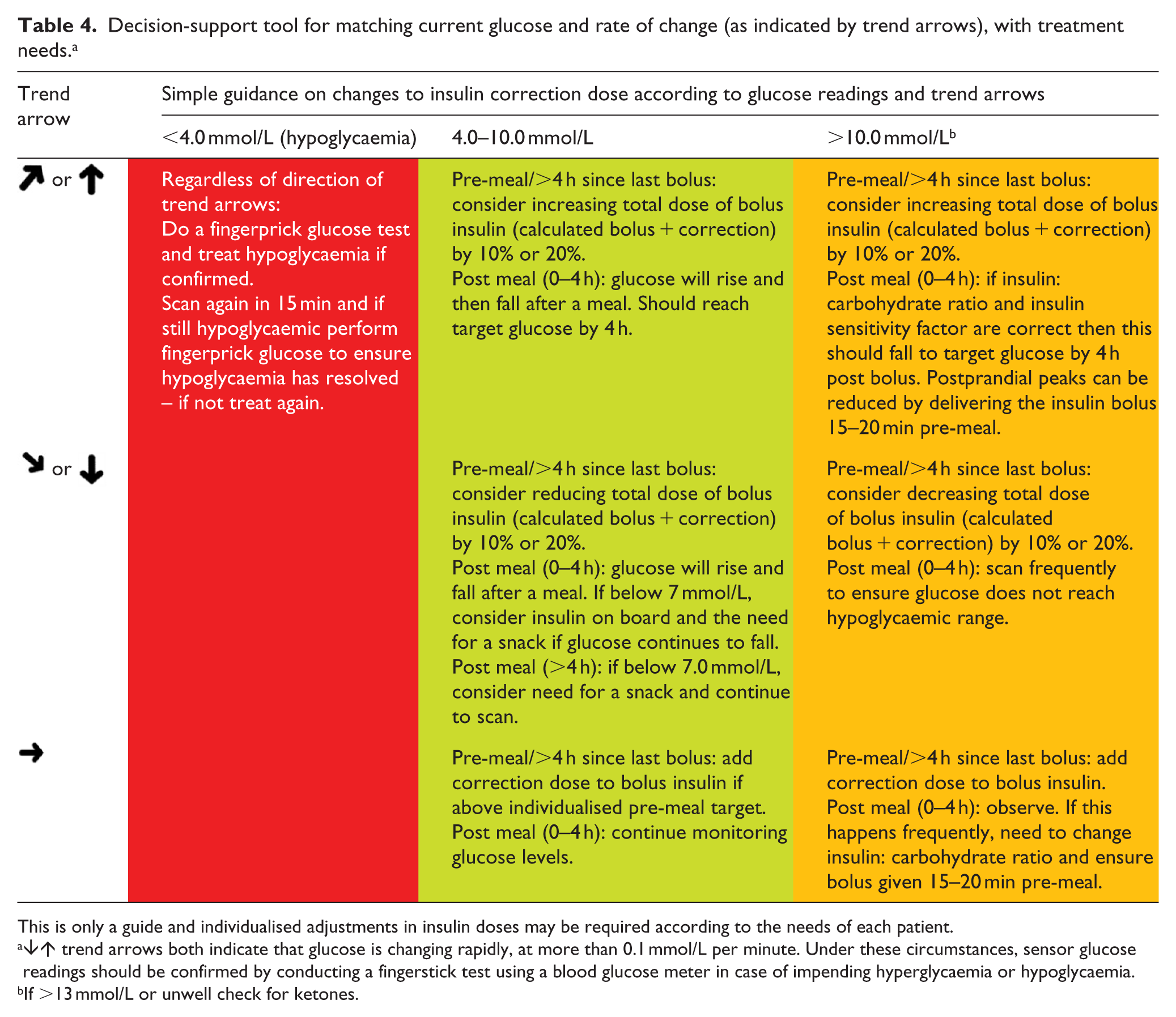
This is only a guide and individualised adjustments in insulin doses may be required according to the needs of each patient.
↓↑ trend arrows both indicate that glucose is changing rapidly, at more than 0.1 mmol/L per minute. Under these circumstances, sensor glucose readings should be confirmed by conducting a fingerstick test using a blood glucose meter in case of impending hyperglycaemia or hypoglycaemia.
If >13 mmol/L or unwell check for ketones.
Conclusion and future directions
The performance of CGM and flash glucose monitoring systems has attained a level of accuracy such that the Abbott FreeStyle Libre system and the Dexcom G5 system are now both approved in Europe and the United States for insulin dosing decisions, without the need for users to perform an adjunct SMBG test, except in defined situations.
Much of the value of these systems is in their use of trend arrows to indicate the direction and RoC of glucose. For people who manage their diabetes with daily insulin injections or CSII, trend arrows can be used in conjunction with their current glucose to modify treatment decisions as compared to snapshot SMBG readings. In this context, real-world guidance on the use of trend arrows in such day-to-day diabetes management decisions is only now emerging.
The JDRF and DATA clinical studies produced provided participants with a bolus insulin adjustment tool that takes into account both current glucose readings and associated trend arrows.2,3 This has given rise to a 10%/20% adjustment rule-of-thumb that has been used to compare ‘recommended’ bolus adjustment with patient-reported behaviours, which reveal more aggressive up or down insulin adjustments in response to trend arrows. 4 A simpler approach using TAATs has been proposed,15,16 and tested in a small number of users, 15 in which the need for a calculation has been replaced with a standard insulin dose adjustment that is associated with each trend arrow.
We want to extend the discussion of the best use of trend arrows to aid insulin bolusing decisions. As well as the current glucose reading and the associated trend arrow, it is important to consider whether the user has any insulin still active in their system from their most recent bolus. If so, this should moderate the calculation of the necessary correction dose.
Elevated glucose above target within 2 h of a prandial or corrective bolus ideally should not be treated with a further bolus due to the risk of ‘insulin stacking’ of active insulin which increases the risk of hypoglycaemia. Rather, continued scanning is recommended and timing of meal bolus reviewed.
For people with diabetes on multiple daily injections of insulin or on CSII, maintaining glucose within their target range involves juggling diet and exercise, alongside the stresses and strains of everyday life. Keeping on track will involve making frequent insulin bolusing decisions. By combining feedback on their current glucose levels, in conjunction with information on the direction and RoC of their glucose, users of ISF glucose sensing technologies, such as the FreeStyle Libre system, can make better informed choices about the timing and degree of correction doses of insulin that better fit with the glycaemic realities of their diabetes day.
While creating a general guidance on managing glucose levels and trend arrows is helpful in the majority of patients, it should be acknowledged that some individuals will require a ‘bespoke’ management plan, necessitating deviation from our recommendations.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.A.A. declares Institutional Research Grants from Abbott Diabetes Care, Bayer, Eli Lilly, Novo Nordisk, Roche and Takeda; honoraria/education support and consultancy fees from Abbott Diabetes Care, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Merck Sharp & Dohme, Novo Nordisk and Takeda. M.H.C. declares honoraria for advisory work for Abbott Diabetes Care. P.J. declares personal fees from Abbott Diabetes Care outside the submitted work. L.L. declares speaker honoraria from Minimed Medtronic, Animas, Roche, Sanofi, Insulet and Novo Nordisk; advisory panel activities for Abbott Diabetes Care, Roche, Sanofi, Minimed Medtronic, Animas and Novo Nordisk; grants to attend educational meetings from Sanofi, Novo Nordisk and Takeda. G.R. declares personal fees from Abbott Diabetes Care outside the submitted work. E.G.W. declares speaker honoraria from Abbott Diabetes Care, Diasend, Dexcom, Eli Lilly, Minimed Medtronic, Novo Nordisk and Sanofi Aventis and has served on advisory panels for Abbott Diabetes Care, Eli Lilly, Medtronic and Sanofi Aventis, and received grants to attend educational meetings from Boehringer Ingelheim, Diasend, Novo Nordisk, Roche and Sanofi Aventis.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.