Abstract
Aims:
With the intensification of antidiabetic treatment, there is an increasing risk of hypoglycaemia. We aimed to determine incidence, characteristics and outcomes.
Methods:
Prospective, observational, multicenter cohort study. The included 3810 patients with type 2 diabetes had their treatment intensified at baseline.
Results:
The incidence of hypoglycaemia was 11.4% per year with 4.2 ± 4.4 episodes per patient. Hypoglycaemia was more frequent in patients with high blood glucose variability. Predictors were heart failure (odds ratio: 1.66; 95% confidence interval: 1.20–2.29) and insulin use (odds ratio: 4.03; 95% confidence interval: 3.05–5.33), with dipeptidyl peptidase–4 inhibitors being associated with reduced risk (odds ratio: 0.69; 95% confidence interval: 0.53–0.89). Macrovascular events were more frequent among patients reporting severe episodes of hypoglycaemia (odds ratio: 3.39; 95% confidence interval: 1.32–8.73). Microvascular events were more frequent in patients with non-severe episodes (odds ratio: 1.92; 95% confidence interval: 1.49–2.49).
Conclusion:
Case-by-case evaluation of patients as well as appropriate selection of antidiabetic pharmacotherapy and blood glucose treatment goals could maximize the benefits while reducing the risks of antidiabetic treatment.
Introduction
Cardiovascular (CV) disease is the leading cause of death in patients with type 2 diabetes mellitus (T2DM). Because T2DM manifests as a result of glucose intolerance, the influence of hyperglycaemia on the development of CV complications has been thoroughly investigated. However, over the past decade, the focus on intensive glucose-lowering regimens1,2 has demonstrated that treatment-mediated hypoglycaemic episodes may be associated with an increased risk of adverse events.3,4 Reported rates of hypoglycaemia in patients with T2DM vary considerably, which may be due to differences in setting (e.g. clinical practice vs clinical trials 1 ), differences in definitions and laboratory thresholds, 5 as well as the impact of patient characteristics. However, data from several epidemiological studies have revealed that the prevalence of hypoglycaemic events is high, and may be increasing as a result of the emphasis on stringent glycaemic control.1,6,7
In the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial, increased prevalence of hypoglycaemia was postulated as a potential cause of the higher mortality rate in the patients undergoing intensive glucose reduction treatment. 8 Although subsequent analysis found no evidence of this, a link with asymptomatic hypoglycaemia was not ruled out. In contrast, the Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) study found a clear association between incidence of severe hypoglycaemia and both mortality and CV events. 4 However, it was noted that the effects of less severe episodes or those that may have gone unnoticed may have confounded the results. In both studies, no direct causal relationship between hypoglycaemia and mortality or CV complications was identified.
It is clear that the association of hypoglycaemia and incident comorbidities in patients with T2DM remains to be clarified. With this in mind, the observational, prospective, multicenter DiaRegis cohort study was conducted. DiaRegis involved patients with T2DM who required treatment intensification at baseline due to inadequate glycaemic control with either single- or dual-agent oral antidiabetic pharmacotherapy. The present analysis constitutes the 2-year follow-up of DiaRegis, in which we have analysed the relationship between hypoglycaemia and diagnosis of new onset comorbidities with the goal of contributing valuable information regarding the effect of hypoglycaemia in real-world patients. In addition, we determined specific predictors of both hypoglycaemia and achievement of optimal glucose control.
Methods
DiaRegis is a prospective, observational, multicenter cohort study including 3810 patients with type 2 diabetes. It was conducted in accordance with Good Epidemiological Practice (GEP) and applicable regulatory requirements. The protocol was approved by the ethics committee of the State Chamber of Physicians Thuringia (Jena, Germany) on 4 March 2009. Details regarding the study design and baseline patient characteristics have been previously published. 9 All patients enrolled in the DiaRegis registry provided written informed consent and were followed for a total of 24 months. DiaRegis was endorsed by the ‘Diabetics with Heart Failure Foundation’, a subsidiary of the ‘German Diabetes Foundation’.
Patients
DiaRegis enrolled consecutive patients with T2DM who were receiving single- or dual-agent treatment with oral antidiabetic drugs, and in whom the treating physician deemed a change of therapy to be necessary. This may have been due to inadequate glycaemic control or due to medication being unsafe or poorly tolerated. Modification of patients’ treatment regimens was achieved by increasing the doses of drugs prescribed at baseline and/or using different drugs and/or additional medications (i.e. a second and/or a third drug). According to the study design, changes in treatment regimen were at the discretion of the physician and were not dependent on protocol-defined criteria.
Patients who did not require treatment intensification or were receiving injectable antidiabetic therapy prior to baseline were not eligible. Other exclusion criteria were: patients who were not under regular supervision of the treating physician for the duration of the study; patients with type 1 diabetes mellitus (T1DM); pregnancy; diabetes mellitus secondary to malnutrition; infection; surgery; patients with hereditary forms of diabetes mellitus (maturity onset diabetes of the young); cancer; limited life expectancy; acute emergencies; participation in a clinical trial; or other reasons that precluded attendance to follow-up visits. Patients were followed for a total of 2 years.
Physicians
Using a database of physicians, approximately 9350 office-based physicians (general physicians, internists, practitioners and diabetologists) were contacted. To ensure a random distribution of physicians from all regions of Germany, a conditional random sampling method was used. This resulted in 313 participating physicians, which represented 3.3% of those who were initially contacted.
Data collection
For DiaRegis, data were collected at baseline, as well as at 6, 12 and 24 months. At each of these time points, the following information was recorded: glycated haemoglobin (HbA1c), fasting plasma glucose (FPG), postprandial plasma glucose (PPG) 2 h after the last meal, body weight, hypoglycaemia, comorbidities, medication, and EuroQuol-5 dimensional (EQ-5D) and Diabetes Treatment Satisfaction Questionnaire (DTSQ) scores. All of this information was documented using a secure electronic database at the Institut für Herzinfarktforschung (Ludwigshafen, Germany).
Hypoglycaemic episodes were prospectively documented in a patient diary, which provided a more realistic picture of the incidence of hypoglycaemia in clinical practice as compared with retrospective data. Documentation of hypoglycaemia was based on whether help was required (from a medical or non-medical person, or leading to hospitalization) and whether a particular episode of hypoglycaemia was symptomatic or not.
An automatic check was performed to verify that the data entry was accurate and complete. All data sets were included in the statistical analyses. Additionally, data on comorbidities and other risk factors were based on each patient’s medical history, as recorded by the treating physician, but diagnoses were not independently adjudicated. Rates of hypoglycaemia were based on recollection of historical events (any hypoglycaemic episode that occurred during the year prior to enrolment) and new events (hypoglycaemic episodes that occurred during the 2 years of follow-up). Vascular disease included coronary heart disease (CHD), stroke, and peripheral arterial disease (PAD). Macrovascular complications were defined as new myocardial infarction (MI), stroke, or PAD (requiring any peripheral intervention). Microvascular complications included a new diagnosis of retinopathy, nephropathy, neuropathy or amputation.
Endpoints
The primary endpoints were the incidence and characteristics of hypoglycaemia within the 2-year follow-up. Secondary endpoints included evaluation of the number of glycaemic events per patient, association between glycaemic control and incidence of hypoglycaemia, identification of factors predictive of hypoglycaemic episodes, and assessment of the association of hypoglycaemia with new comorbidities. A further endpoint was the composite of the achievement of HbA1c < 7% with no hypoglycaemia and no weight gain.
Statistical analysis
Distributions of numeric variables are described by medians and quartiles or by the mean ± standard deviation (SD). All descriptive statistics are based on the available case numbers. Changes in patient characteristics, laboratory values, and treatment regimens from baseline to the end of the 2-year follow-up were evaluated using Wilcoxon’s signed rank test or the McNemar test. Logistic regression analyses were used to identify independent predictors of hypoglycaemic events and of achieving the recommended target level of glycaemic control. Odds ratios (ORs) and 95% confidence intervals (CIs) were used to express the strength of the association between each variable and the two outcomes. Statistical analysis was performed using SAS version 9.2 (SAS Institute, Cary, NC, USA).
Results
A total of 3810 patients with T2DM were enrolled, of whom 3058 (80.3%) had a known status at 2-year follow-up. Among these 3058 patients, 2983 (97.5%) were alive, and 75 (2.5%) were dead at the end of the follow-up period. Baseline patient characteristics for those with a known status at 2-year follow-up are presented in Table 1. From baseline to year 2, antidiabetic drug use, glycaemic control and body weight were all significantly altered. The use of sulfonylureas and glitazones declined by 4.4% and 6.7%, respectively, whereas the use of insulin increased by 7.6% (all p < 0.0001). With respect to glycaemic control, improvements were observed in HbA1c (7.7% vs 7.1%) and FPG levels (150.3 vs 132.2 mg/dl), but a deterioration was observed in PPG control (201.6 vs 239.7 mg/dl; all p < 0.0001).
Baseline characteristics of patients with a known status at the end of the 2-year follow-up.
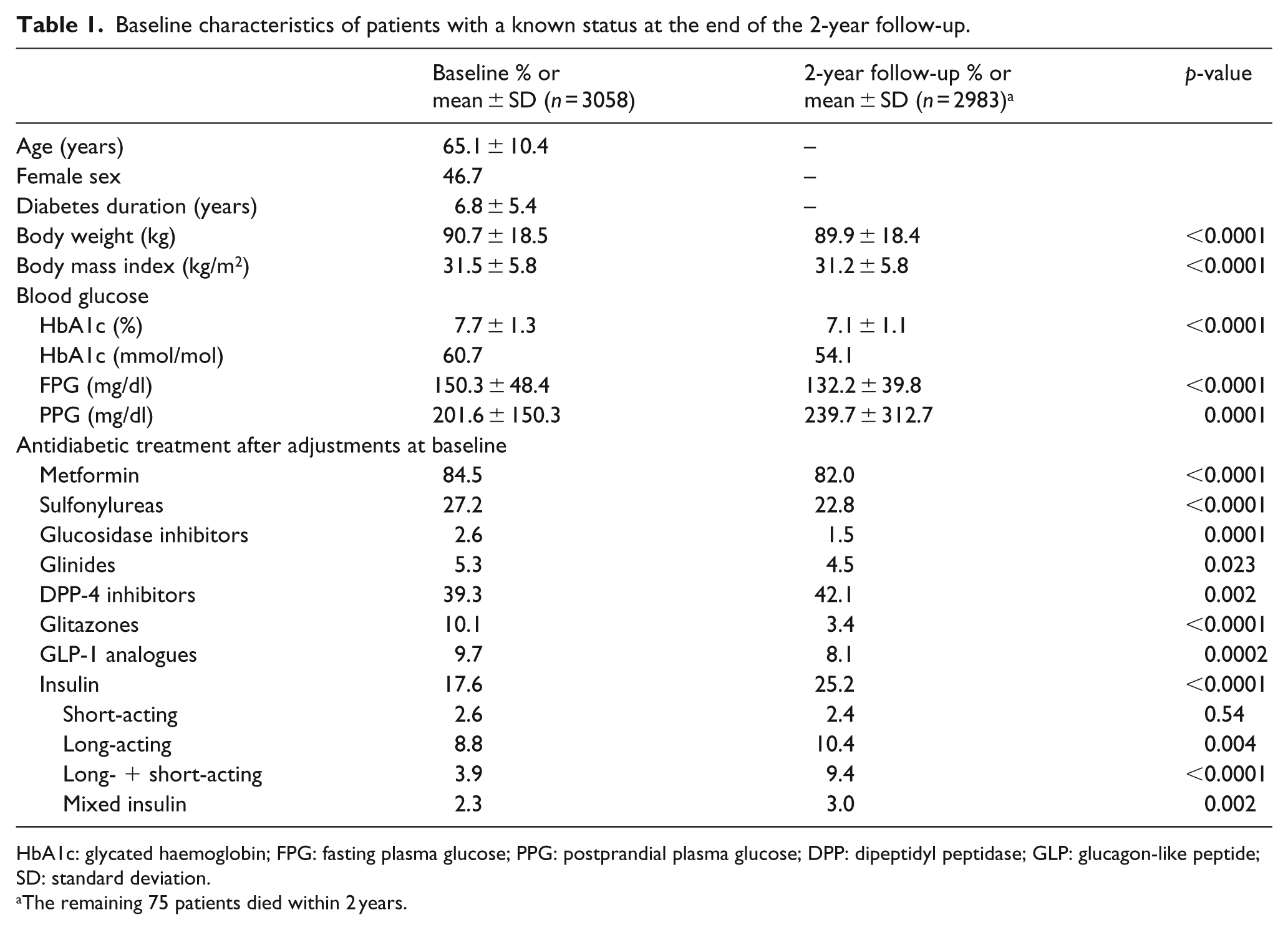
HbA1c: glycated haemoglobin; FPG: fasting plasma glucose; PPG: postprandial plasma glucose; DPP: dipeptidyl peptidase; GLP: glucagon-like peptide; SD: standard deviation.
The remaining 75 patients died within 2 years.
Incidence, characteristics and predictors of hypoglycaemia
The incidence of hypoglycaemia among the 3058 patients with a known status at 2-year follow-up was 17.8%, with a mean of 5.4 (±5.8) episodes per patient (Table 2, upper panel). Most hypoglycaemic episodes were either asymptomatic (11.1%) or symptomatic without the need for help (12.6%). Rates of hypoglycaemia during the follow-up period were substantially higher in patients with a history of hypoglycaemia during the year prior to enrolment (40.3%) relative to those without a prior history of hypoglycaemia (14.9%) (Table 2, lower panel).
Incidence of hypoglycaemic events.
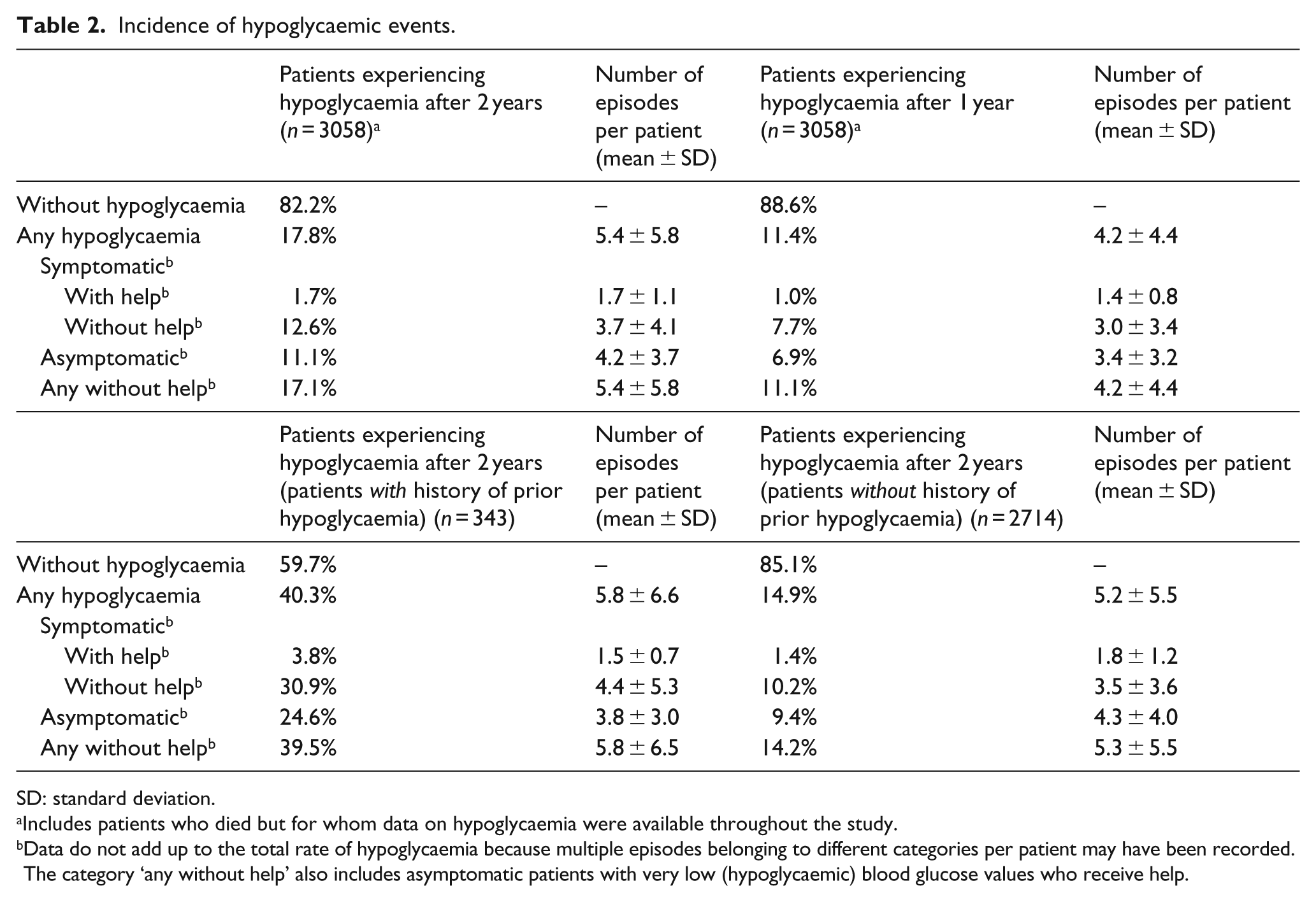
SD: standard deviation.
Includes patients who died but for whom data on hypoglycaemia were available throughout the study.
Data do not add up to the total rate of hypoglycaemia because multiple episodes belonging to different categories per patient may have been recorded. The category ‘any without help’ also includes asymptomatic patients with very low (hypoglycaemic) blood glucose values who receive help.
Blood glucose control (HbA1c, FPG and PPG) was similar among patients with and without new hypoglycaemic episodes (Table 3, upper panel). Hypoglycaemia occurred more frequently in patients with a medium (22.5%) or high (20.9%) degree of self-reported blood glucose variability (Table 3, lower panel) than in those reporting a low degree of blood glucose variability (5.1%).
Blood glucose control and variability in blood glucose control in patients with and without hypoglycaemia at the end of the 2-year follow-up.
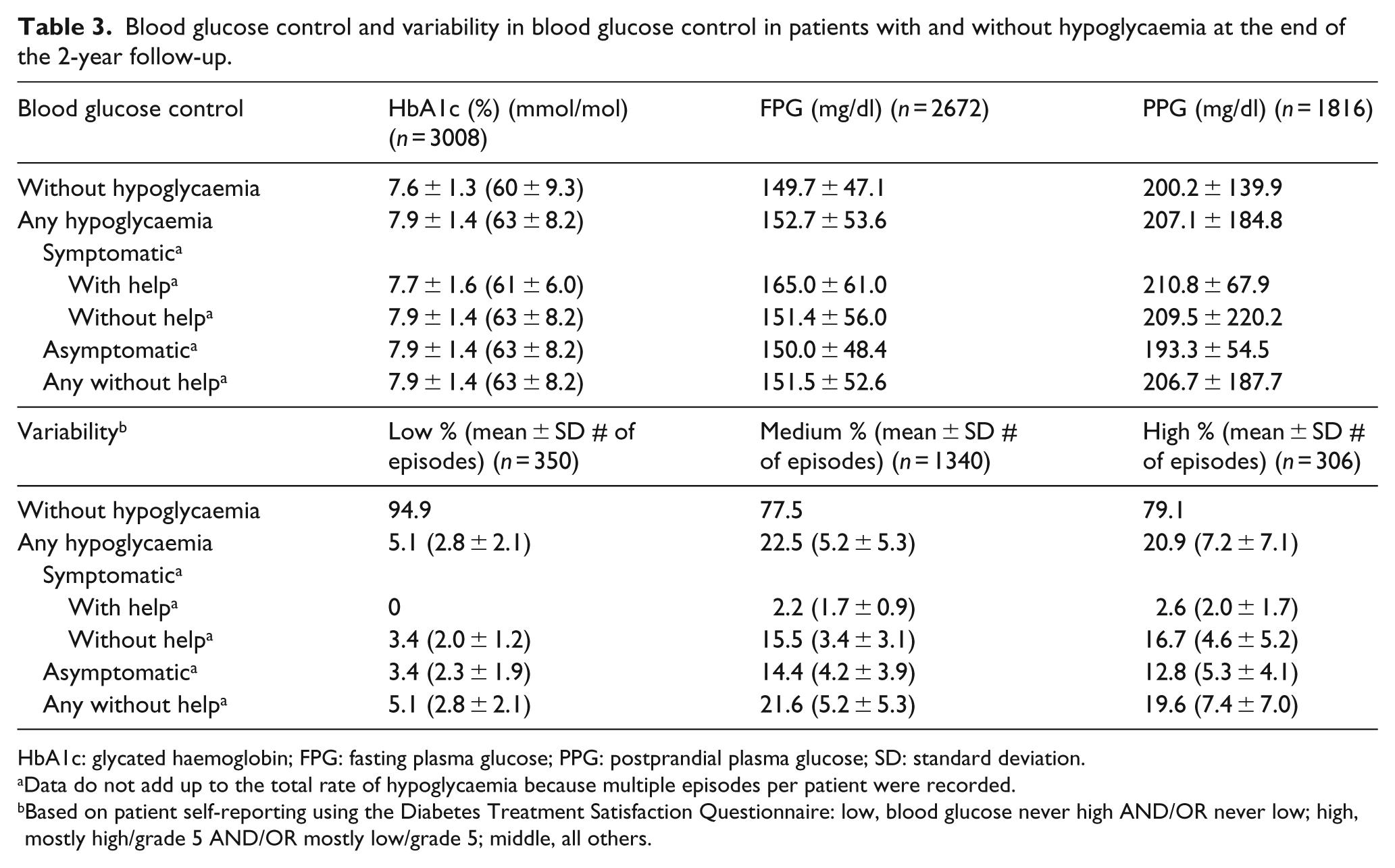
HbA1c: glycated haemoglobin; FPG: fasting plasma glucose; PPG: postprandial plasma glucose; SD: standard deviation.
Data do not add up to the total rate of hypoglycaemia because multiple episodes per patient were recorded.
Based on patient self-reporting using the Diabetes Treatment Satisfaction Questionnaire: low, blood glucose never high AND/OR never low; high, mostly high/grade 5 AND/OR mostly low/grade 5; middle, all others.
As illustrated in Figure 1, multivariate analysis revealed the following two independent predictors of hypoglycaemic episodes: heart failure at baseline (OR: 1.66; 95% CI: 1.20–2.29) and insulin use (OR: 4.03; 95% CI: 3.05–5.33). Conversely, the use of dipeptidyl peptidase (DPP)-4 inhibitors was associated with a 31% relative risk reduction in hypoglycaemia (OR: 0.69; 95% CI: 0.53–0.89).
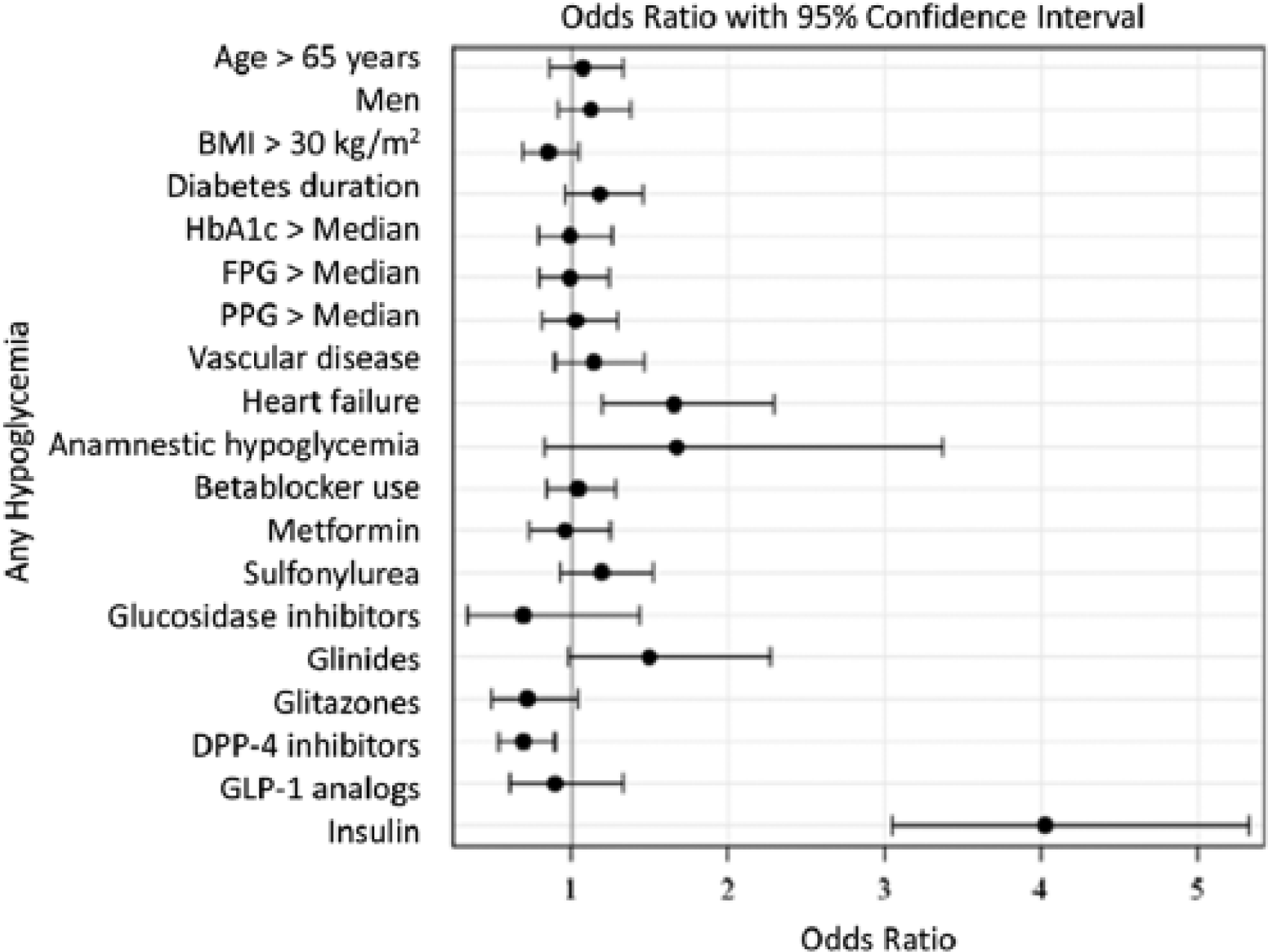
Multivariable predictors of new episodes of hypoglycaemia during the 2-year follow-up period.
Association of hypoglycaemia with new comorbidities in DiaRegis
During the 2-year follow-up, the incidence of newly diagnosed concomitant diseases was recorded (Figure 2). Relative to patients without hypoglycaemia, the frequency of the following comorbidities was significantly higher among those who experienced a hypoglycaemic episode: peripheral neuropathy (12.8% vs 8.6%; p < 0.05), clinically relevant depression as assessed by the treating physician (10.7% vs 4.5%; p < 0.0001), heart failure (7.5% vs 3.6%; p < 0.0001), non-proliferative retinopathy (6.6% vs 1.8%; p < 0.0001), autonomous neuropathy (4.0% vs 2.1%; p < 0.05), stable angina (3.8% vs 1.8%; p < 0.01), and amputation (0.8% vs 0.2%; p < 0.05).
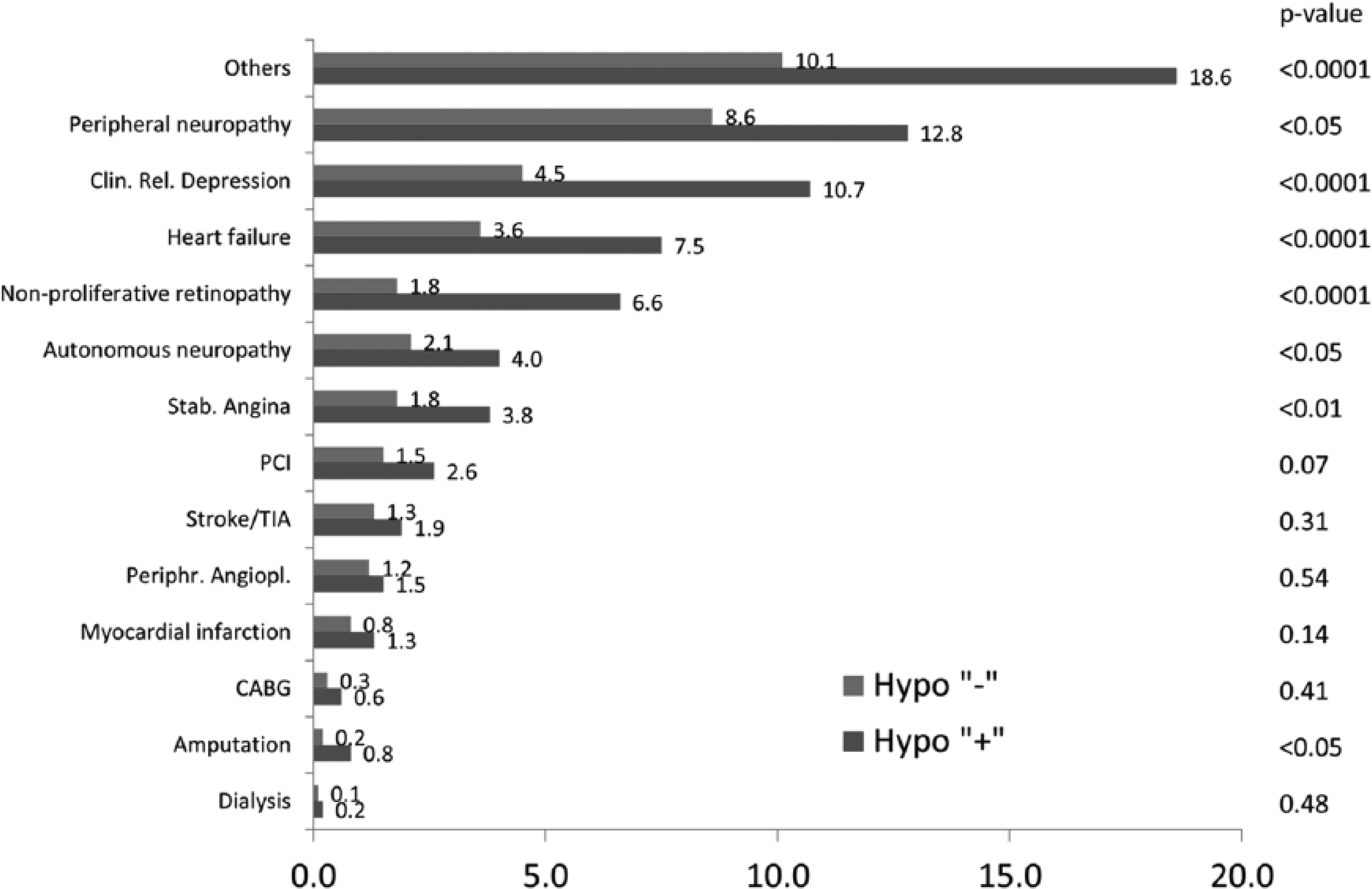
Newly diagnosed comorbidities in patients with or without hypoglycaemia during the 2-year follow-up.
Since adverse event rates/newly diagnosed concomitant diseases were relatively low, further analysis was conducted according to micro- and macrovascular events. Table 4 displays the associations between severe, non-severe and any hypoglycaemic episode with the occurrence of micro- and macrovascular events. There was an increase in the risk of any type of vascular event for all three categories of hypoglycaemic episode (ORs between 2.20 and 2.60). Macrovascular events (MI, stroke or PAD) were more frequent in patients reporting severe episodes of hypoglycaemia (OR: 3.39), as were MI (OR: 5.28) and stroke (OR: 4.53) when analysed as individual categories. Microvascular events (retinopathy, nephropathy, neuropathy or amputation) were more frequent in patients with non-severe episodes of hypoglycaemia (OR: 1.92; 95% CI: 1.49–2.49).
Association between hypoglycaemia and vascular events during the 2-year follow-up.
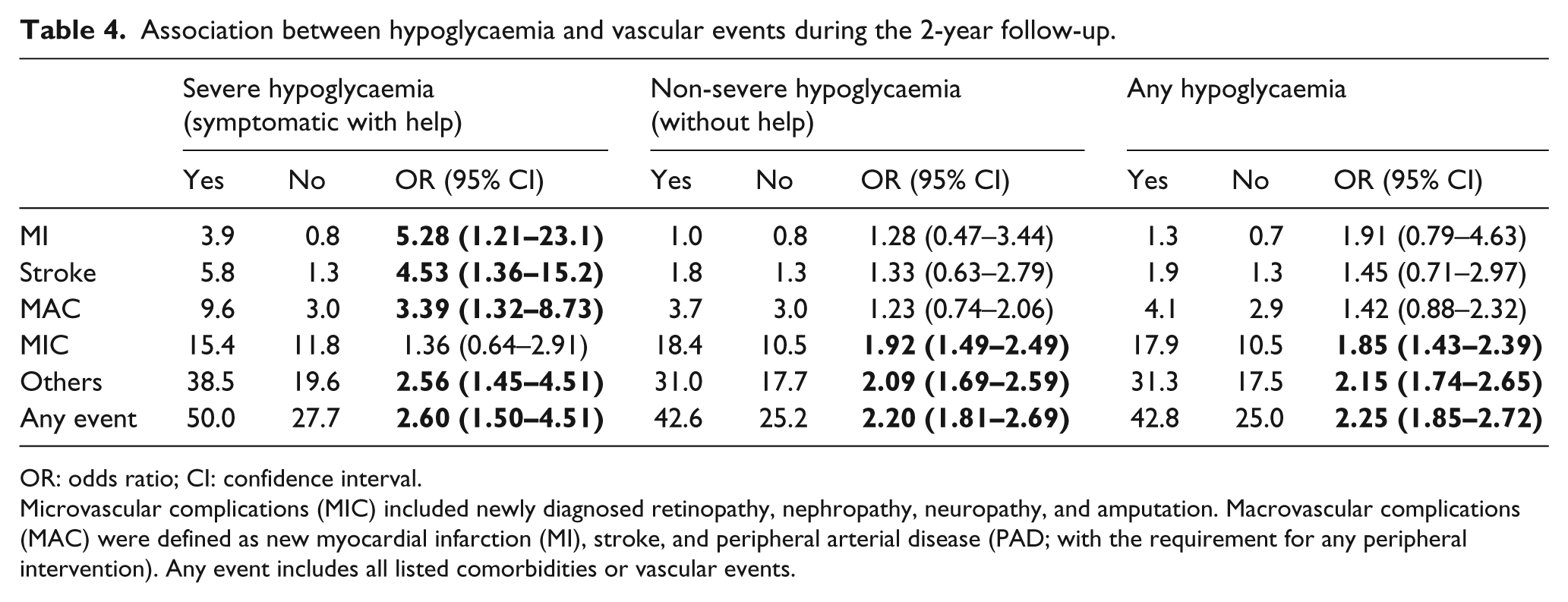
OR: odds ratio; CI: confidence interval.
Microvascular complications (MIC) included newly diagnosed retinopathy, nephropathy, neuropathy, and amputation. Macrovascular complications (MAC) were defined as new myocardial infarction (MI), stroke, and peripheral arterial disease (PAD; with the requirement for any peripheral intervention). Any event includes all listed comorbidities or vascular events.
Blood glucose target achievement (HbA1c < 7%) without hypoglycaemia or body weight gain
With regard to the composite endpoint of attaining target HbA1c levels (<7%) without hypoglycaemia or body weight gain, negative predictors were: a duration of T2DM of >5.6 years (OR: 0.64; 95% CI: 0.54–0.76), heart failure (OR: 0.76; 95% CI: 0.49–0.92), and treatment with a sulfonylurea (OR: 0.74; 95% CI: 0.60–0.91) or insulin (OR: 0.50; 95% CI: 0.37–0.67; Figure 3).
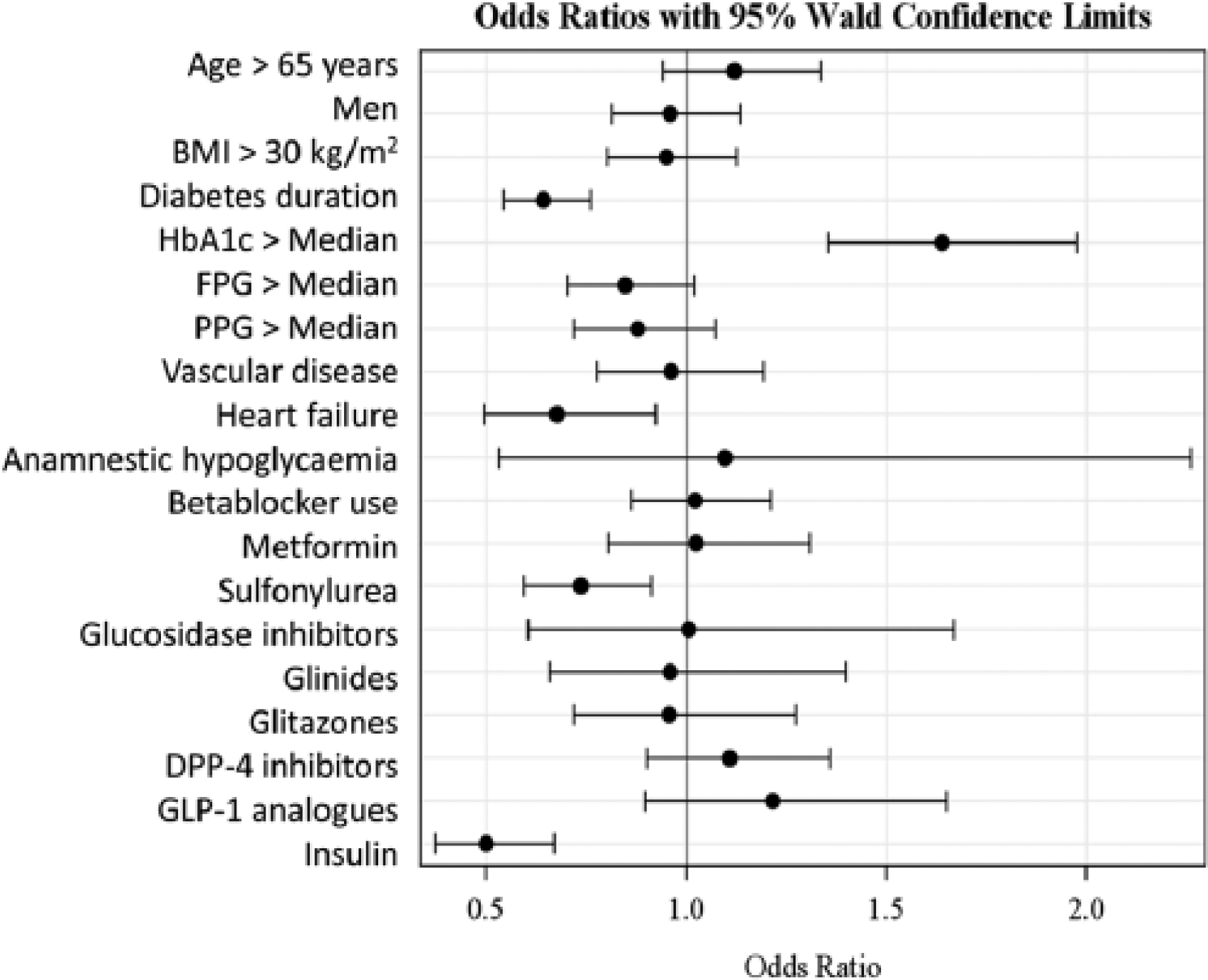
Multivariable adjusted predictors of the composite endpoint of HbA1c < 7% with no hypoglycaemia and no weight gain (n = 3058).
Discussion
For the cohort of patients with T2DM who were included in the DiaRegis study, both the retrospective rate of hypoglycaemia during the year prior to enrolment 10 as well as the prospective rate during the first year of follow-up have been previously reported. 11 In the present analysis, the relationship between incident hypoglycaemia and the diagnosis of new comorbidities during the 2-year follow-up period was explored. Hypoglycaemic episodes were reported by 17.8% of the patients (11.4% per year), and hypoglycaemia was associated with an increased risk of micro- and macrovascular events. Multivariable logistic regression analysis indicated that a high degree of intra-individual variability in blood glucose levels, heart failure at baseline, and treatment with insulin were independent predictors of hypoglycaemia in patients with T2DM. Attainment of glycaemic goals without hypoglycaemia or body weight gain was less common in patients with heart failure, longer duration of T2DM, and treated with a sulfonylurea and/or insulin.
Incidence, characteristics and predictors of hypoglycaemia
Hypoglycaemia is a frequent adverse effect of antidiabetic treatment. Although it is more common in patients with T1DM, it is regularly experienced by patients with T2DM. In general, hypoglycaemia is more frequently reported in patients with a longer duration of the disease, as well as in patients treated with either exogenous insulin or drugs that amplify endogenous insulin levels (e.g. sulfonylureas). Considering all types of hypoglycaemia (i.e. asymptomatic or symptomatic; with or without the need for help), we observed that 17.8% of all patients had at least one episode of hypoglycaemia during the 2-year follow-up, and the mean yearly rate was 11.4%. Among these episodes, 1.7% were symptomatic and required help.
Hypoglycaemia was found to be more frequent in patients with a high degree of blood glucose variability. Although these data were self-reported and not confirmed by continuous glucose monitoring or a blood glucose diary, these findings are in accordance with previous studies predicting hypoglycaemia based on self-monitoring data. 12 Patients who recorded variable and frequently very low blood glucose readings were at a higher risk of subsequent severe hypoglycaemia. In contrast, patients with lower HbA1c levels did not display an increased risk of severe hypoglycaemic episodes. 12 These findings were replicated in later studies. In fact, Engler et al. demonstrated that while an average HbA1c level of >7% did not confer a reduced risk of hypoglycaemia compared with an average value of <7%, it was associated with higher glycaemic variability. 13 Monnier et al. 14 reported that the risk of asymptomatic hypoglycaemia was increased in patients with greater blood glucose variability, also highlighting the need to minimize glucose fluctuations to reduce the risk of hypoglycaemia. Finally, Qu et al. found that intra-day and inter-day glycaemic variability was significantly associated with the risk of hypoglycaemia in insulin-treated patients with T2DM, even after adjusting for mean glucose and HbA1c levels. 15 Thus, it would appear that both treatment- and patient-related factors influence the relationship between hypoglycaemia and blood glucose variability. These data suggest that patient-recognized variability in glycaemic control is more valuable than focusing on the risks of hypoglycaemia with strict blood glucose control.
Results from the DiaRegis study have indicated that elderly patients (>65 years old) are at increased risk of experiencing severe episodes of hypoglycaemia. 16 Similar findings were reported in a recent analysis of data from the US National Electronic Injury Surveillance System project as well as a national household survey of insulin use. Compared with insulin-treated patients aged between 45 and 64 years, the probability of being hospitalized for insulin-related hypoglycaemia was fivefold higher in insulin-treated patients aged ⩾80 years old. Furthermore, these studies confirmed previous findings. 17 The increased risk of hypoglycaemia among elderly patients may be attributed to the longer duration of diabetes, frequency of sulfonylurea/insulin use, and unawareness related to hypoglycaemia. 18 In fact, patients with impaired awareness of hypoglycaemia have a sixfold greater risk of experiencing severe hypoglycaemia, 19 and attenuated physiologic responses to critically low glucose levels can be fatal. 20 Therefore, instead of raising HbA1c goals, an optimal strategy for elderly patients may involve placing a greater emphasis on self-monitoring of blood glucose. In addition, drugs that increase the risk of hypoglycaemia, such as insulin and sulfonylureas, should be used with caution in the elderly, and potentially substituted for drugs that have reduced likelihood of inducing hypoglycaemia (e.g. DPP-4 inhibitors and glucagon-like peptide (GLP)-1 analogues).
To date, few reports have explored the occurrence of hypoglycaemia in patients with T2DM and heart failure, despite a high proportion of diabetic patients suffering from this latter condition.21,22 The cause of heart failure in this patient population is not entirely clear, with endothelial dysfunction, counter-regulatory hormone secretion, impaired cardiomyocyte function, and catecholamine release all being stated as potential contributory factors.23–26 In a retrospective analysis of 748 patients hospitalized with congestive heart failure, episodes of hypoglycaemia (defined as a glucose level of <70 mg/dl) were detected in approximately one-third of the cohort. 25 Furthermore, the patients with T2DM experienced a higher percentage of days with a hypoglycaemic event in comparison to those without the condition. Rapid drops in blood glucose levels have been linked to episodes of cardiac ischaemia, 24 which would result in the accumulation of myocardial damage over time if these glucose fluctuations persisted. The higher incidence of hypoglycaemia in patients with heart failure in our study may reflect a prolonged period of blood glucose variability. This is corroborated by the increased risk of hypoglycaemia in patients with a prior history of it. Further studies to elucidate the link between heart failure and hypoglycaemia in patients with T2DM are required.
It might be speculated that hypoglycaemia would also be more prevalent in patients with vascular disease, as many of the factors that could cause heart failure would be likely to negatively affect the vasculature. 27 However, in the present study, the presence of vascular disease was not found to be predictive of the occurrence of hypoglycaemia.
Numerous epidemiological studies have evaluated the incidence of severe hypoglycaemia in patients receiving insulin for T2DM. Indeed, a Scottish study reported an annual rate of 7.3% in insulin-treated patients, compared with 0.8% in those treated with sulfonylurea, 28 and the UK Hypoglycaemia Study Group found a similar incidence (7.0%) in patients receiving less than 2 years of insulin or sulfonylurea therapy. 6 Furthermore, another investigation indicated that patients with T2DM who had been treated with insulin for more than 5 years had an incidence of 15%, confirming that the risk of severe hypoglycaemia in insulin-dependent patients is directly related to treatment duration. 29 In addition, patients with T2DM were more likely to require hospitalization for severe hypoglycaemia than patients with T1DM. 30 In a multi-ethnic cohort of Northern Californian patients with T2DM, at least one episode of severe hypoglycaemia was reported by 11% of the study population during the preceding year, with 59% and 23% of the patients on insulin or a combination of oral antidiabetic drugs, respectively. 7 Notably, these rates are substantially higher than those reported in the ACCORD and ADVANCE clinical trials (i.e. 5% and 0.7%, respectively).1,2,7,31
Other studies have reported that the use of a sulfonylurea and/or insulin was associated with higher rates of hypoglycaemia than the use of DPP-4 inhibitors. In the UK Prospective Diabetes Study (UKPDS), the rate of hypoglycaemic episodes was similar for sulfonylureas and insulin (each 7%). 32 A meta-analysis of 21 randomized clinical trials found that glibenclamide (the most widely used sulfonylurea) was associated with an 83% greater relative risk of hypoglycaemia compared with other sulfonylureas, and a 52% greater risk when compared with other insulin secretagogs. 33 Another study found that in elderly patients, glimepiride induced fewer episodes of hypoglycaemia than did glibenclamide (6% vs 38%) as well as a reduced incidence of severe hypoglycaemia (0.86 vs 5.6 cases per 1000 person-years, respectively). 34 Moreover, it is known that the efficacy of insulin treatment is often compromised by the risk of hypoglycaemia. Therefore, it has been suggested that each patient’s risk profile should be considered when using insulin. 35 Notably, a regimen involving the combination of once-daily basal insulin (usually at bedtime) with oral antidiabetic drugs has been demonstrated to reduce hypoglycaemic episodes, while maintaining a similar level of glycaemic control. 36 However, this treatment combination often becomes ineffective over time, and many patients require a fixed-dose combination of twice-daily insulin or prandial insulin with bolus basal insulin. Importantly, whichever insulin regimen is favoured, the risk of hypoglycaemia has been found to increase longitudinally. 6 Incretin-based drugs have a low intrinsic risk of hypoglycaemia because of their glucose-dependent action. DPP-4 inhibitors and GLP-1 analogues are also associated with a low rate of hypoglycaemia when added to metformin, making them attractive second- or third-line therapeutic options. 35
Association of hypoglycaemia with newly diagnosed comorbidities
This study revealed a strong correlation between hypoglycaemia and the development of micro- or macrovascular disease. This might not be surprising, given that CV disease is the most common cause of death in patients with T2DM, 37 and there is broad evidence indicating that hypoglycaemia is a serious CV risk factor. 3 Hypoglycaemia is known to exert a variety of effects on the vasculature and blood, and these are likely to be amplified in patients with T2DM, many of whom display micro- and macrovascular complications. 27 Indeed, Wright et al. postulated that hypoglycaemia may have a greater detrimental effect on blood vessels in patients with more established vascular disease. 27 High incidence of hypoglycaemia is reflective of glucose variability, a factor that has also been explicitly linked to the development of comorbidities.24,38 While it is difficult to separate the effects of hypoglycaemia and glucose variability, the former has been shown to have an effect on mortality in patients with T2DM, even after adjustment for glycaemic lability index. 25
A number of clinical trials have examined CV outcomes while comparing intensive glucose-lowering treatment with conventional therapy. The UKPDS, which enrolled 5102 patients with newly diagnosed T2DM, found higher rates of severe hypoglycaemia alongside no significant reduction in CV complications with intensive therapy.32,39 However, a 10-year follow-up identified modest post-trial CV risk reductions, suggesting a delayed benefit. 39 More recently, the ACCORD trial studied 10,252 patients with T2DM and pre-existing CV disease and/or CV risk factors; however, the trial was terminated due to excess mortality in the intensive treatment arm. 1 While the rate of hypoglycaemia was elevated in the intensive therapy arm, a post-hoc analysis concluded that hypoglycaemia did not account for the increased mortality rate.8,40 Nevertheless, it is difficult to exclude the contribution of hypoglycaemia. 41 In contrast, in the ADVANCE study, which analysed 11,140 patients with T2DM, intensive therapy was not associated with increased mortality. 2 However, intensive glucose-lowering therapy led to severe hypoglycaemia, which was linked to vascular events and CV-related death. 4 The Veterans Affairs Diabetes Trial (VADT) clinical trial, which involved 1791 military veterans with poorly controlled T2DM, did not demonstrate any improvement in CV outcomes in patients receiving intensive glucose-lowering treatment, but again showed a higher rate of hypoglycaemia. 31
Limitations
This study presented certain limitations. Indeed, data on hypoglycaemic events were self-recorded, which may have allowed for patient-associated bias in our results. Moreover, complete documentation of hypoglycaemic events was difficult when considering asymptomatic episodes or nocturnal hypoglycaemia, which can go undetected.42,43 Nevertheless, this study examined real-life, day-to-day patient experiences with hypoglycaemia and how they relate to comorbidities and treatment. Considering comorbidities, diagnoses were physician-based but not independently adjudicated, which could have caused some error; however, these diagnoses were recorded from patients’ charts and represent findings from real clinical management of patients. Also, with regard to antidiabetic pharmacotherapy, there were key differences noted between baseline and the 2-year follow-up. In this respect, the association between insulin treatment, which increased from 17.6% to 25.2% from the first to the last visit, and the risk of hypoglycaemia or adverse events may have been underestimated. In contrast, the relationship between glitazones and risk may have been overestimated, as they were used less at the end of the study. In addition, because data on specific compounds were not documented, differences in risk within each drug class could not be determined. Furthermore, we did not analyse the association between hypoglycaemia, CV events, and individual classes of antidiabetic medications. In addition, the fact that treatment intensification did not occur in a randomized way might have produced bias. Nevertheless, the treatments were decided by physicians in true patient settings. Therefore, although the possibility exists that this study population did not fully represent patients with T2DM, these results should be typical of patients in real-life clinical practice.
Conclusion
Hypoglycaemia is a frequent complication of intensified glucose control in patients with T2DM. It is associated with an increased burden of comorbidities as well as micro- and macrovascular complications. Consideration of individual patient characteristics, along with the appropriate selection of antidiabetic pharmacotherapy and careful selection of treatment goals, may have the potential to maximize the benefits of individualized, target-oriented, blood glucose control as well as reducing the adverse effects of hypoglycaemia.
Key messages
Treatment-mediated hypoglycaemic episodes may be associated with an increased risk of adverse events. Hypoglycaemia is often asymptomatic and a causal connection with mortality has not been established.
In the present real-world survey, the yearly incidence of hypoglycaemia was 11.4% per year with 4.2 ± 4.4 episodes per patient in those with treatment intensification at baseline.
Incident hypoglycaemia was associated with an increased risk of micro- and macrovascular events.
Multivariable logistic regression analysis indicated that a high degree of intra-individual variability in blood glucose levels, heart failure at baseline, and treatment with insulin were independent predictors of hypoglycaemia
Footnotes
Acknowledgements
The authors wish to acknowledge the excellent support of Mirjam Strasser (Winicker Norimed GmbH) and Sonja Frey/Benjamin Weidig (Stiftung Institut für Herzinfarktforschung) during the preparation and completion of the registry. In addition, they wish to acknowledge the support of all participating physicians and patients, without whom this registry would not have been possible.
Declaration of conflicting interests
Steffen Schneider (S. S.) has no conflicts of interest to disclose.
Funding
Funding was provided by Bristol-Myers Squibb and AstraZeneca. Diethelm Tschöpe (D. T.), Peter Bramlage (P. B.) and Anselm K Gitt (A. K. G.) have received research support and honoraria for lectures from Bristol-Myers Squibb and AstraZeneca.
