Abstract
Aim:
The aim of the present study was to investigate the effect of the mobilization of EPCs by AMD3100 combined with G-CSF on wound healing in diabetic mice.
Methods:
The full-thickness excisional wounds model of diabetic mice (db/db) was examined by hematoxylin and eosin staining, immunohistochemical staining, and western blotting to compare the wound healing and neovascularization among the combination, AMD3100 alone, G-CSF alone, and control groups.
Results:
The wounds reached the complete closure in the combination, AMD3100 alone, G-CSF alone, and control groups on days 17, 20, 21, 21 after surgery, respectively. In addition, the combination group promoted the inflammatory cell recruitment and glandular formation. On day 10 from injury, the protein expression of CD31 in the combination group was significantly higher compared with the other three groups (p < 0.001). The level of SDF-1 protein remained high in the combined group until on day 10 after surgery (p < 0.001).
Conclusion:
The mobilization of endogenous EPCs by AMD3100 combine with G-CSF is able to enhance the complete healing of full-thickness wounds and neovascularization in db/db mice may by SDF-1/CXCR4 axis. These findings provided a novel method and indication of duration of mobilization on diabetic wound healing and tissue regeneration.
Keywords
Introduction
Refractory wound is a difficult point in clinical diagnosis and treatment. Diabetic ulcers are a representative example of refractory wounds, accounting for 1/3 of all wounds. Following the advancements in molecular biology, cell biology, biomaterials, and tissue engineering, certain progress has been made in the theoretical knowledge and treatment of wounds; however, the clinical effects of refractory wounds are not sufficient and further research is required.
Angiogenesis is the core step of wound repair. However, vascular disease is the pathological basis of several chronic complications of diabetes. It has been shown that endothelial progenitor cells (EPCs) are the precursors of endothelial cells and play an important role in angiogenesis, which was first discovered by Asahara et al. in the peripheral blood using immunomagnetic beads. EPCs are mostly derived from the bone marrow, embryonic liver, umbilical cord, and peripheral blood. In adulthood, EPCs are mainly derived from the bone marrow, which can replace damaged vascular endothelial cells and maintain their integrity. 1 It has been confirmed that EPCs transplantation can promote the angiogenesis of ischemic hind limbs in diabetic mice. 2 However, the number of EPCs and their ability to migrate to the target organs has been shown to be affected in high-sugar environments. 3 Therefore, the regulation of the number and function of EPCs in patients with diabetes, as well as EPCs transplantation, have become fundamental in the treatment of refractory diabetic wounds.
In recent years, the main function of EPCs has been shown to be promoting angiogenesis in the treatment of diabetes-caused wounds, which are hard to heal, including mononuclear cell transplantation, endogenous EPC mobilization, and EPC gene modification. 4 Certain endogenous factors (ischemia, injury and stress, amongst others) and exogenous factors (such as cytokines and drugs) can stimulate the migration of bone marrow EPCs to the peripheral blood. Granulocyte colony stimulating factor (G-CSF) has previously been reported to increase the number of circulating CD34+ cells and promote arteriogenesis in patients with coronary artery disease. 5 However, in hyperglycemic environments, the use of G-CSF is not effective in mobilizing EPCs, due to the abnormal sympathetic nerve-induced low mRNA and protein level of stromal cell-derived factor 1 (SDF-1), also known CXCL12. 6 AMD3100, a synthetic stromal cell-derived factor receptor antagonist, can change this. It was reported by Nishimura et al. 7 that the application of AMD3100 can effectively mobilize EPCs, which can rapidly, safely, and effectively mobilize CD34+ hematopoietic cells in mice, dogs, and humans. 8 In addition, some studies also found that wound healing was significantly delayed in GM-CSF KO mice, 9 and G-CSF promoted contraction of wound in WT mice. 10 It was shoardium through EPwn by our previous studies that AMD3100 combined with G-CSF had an enhanced effect on the mobilization of bone marrow EPCs compared with AMD3100 alone. In non-diabetic conditions, G-CSF followed by the addition of AMD3100 had a better mobilization effect and peaked on day 7, but in diabetic conditions, AMD3100 followed by the addition of G-CSF had a better mobilization effect and peaked on day 10. 11
Based on this finding, in the present study, a diabetic wound model was established using full-thickness excisional skin from diabetic mice and the optimal mobilization method of AMD3100 combined with G-CSF from our previous study was adopted to observe the effect of EPCs mobilization on wound healing and angiogenesis. The findings of the present study can provide a novel theoretical basis for the use of autologous bone marrow EPCs in the treatment of chronic refractory wounds caused by diabetes mellitus, which could help avoid the short-term effect of allogeneic transplantation due to rejection reactions, overcome the limitation of collecting cells for in vitro culture and amplification, and reduce the pain suffered by patients due to bone marrow aspiration.
Materials and methods
Animals
A total of 60 male C57BLKS/J diabetic mice (db/db, 9–10 weeks, 36.0–39.0 g) were purchased from Changzhou Cavens Lab Animal Co.Ltd [license no, SCXK (su) 2016-0010]. Mice were housed in a well-ventilated holding room with a 12-h light-dark cycle at an ambient temperature of 23 ± 2°C and 70% humidity, with free access to food and water. All animal experiments were in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. The present study was approved by the Animal Care and Ethics Committee of Kunming Medical University (Kunming, China).
In vivo excisional wound model
Two full-thickness excisional wounds were made on the dorsal skin of anesthetized [1% pentobarbital sodium, 50 mg/kg, intraperitoneally (i.p)] mice using a sterile 10-mm-diameter dermal punch.
Then, wounded mice were divided randomly into four experimental groups and received subcutaneous injection of saline or drugs [AMD3100 (Selleck Chemicals, Cat NoS8030) or G-CSF (Qilu Pharmaceutical Co., Ltd, S20063065) in left lower extremity near the wound once a day immediately after wounding: (i) The control group was treated with saline for 6 days; (ii) the AMD3100 group was treated with a reference dose [6 mg/(kg days) 7 ] on the first day, and treated with saline on the other 5 days; (iii) the G-CSF group was treated with a reference dose [100 μg/(kg days) 12 ] for 5 days, and on the last day it was treated with saline; and (iv) The combination group was treated with AMD3100 on the first day, and G-CSF for the 5 days. 11 There are 15 male db/db mice in each group.
Wound dressing was performed using iodopor for all db/db mice on days 3, 7, 10, 14, and 17 from surgery. The percentage of the initial wound that remained open was quantified at different time points (on days 0, 3, 7, 10, and 14 after surgery), until the wound was completely covered by epidermal cells in one group. The healing time of the unhealed group continued to be observed, but histological analysis was not performed. In addition, on days 3, 7, 10, and 14 after surgery and at the time that the wound had healed, three db/db mice were randomly selected from each group for a total of six wounds. About 1% pentobarbital sodium (200 mg/kg, i.p) was used for euthanasia. After death, the part of the skin wounds were fixed with 4% paraformaldehyde for 48 h at 4°C, and then embedded with paraffin for histopathological analysis [hematoxylin and eosin (H&E))], immunohistochemical staining (IHC). The other part of the skin wound tissue was directly frozen at −80°C for western blotting (WB).
Wound healing rate
In each group, the area of unhealed wound was drawn on transparent wound dressing on days 3, 7, 10, and 14 after surgery, and then the wounds were analyzed by Image Pro Plus 6.0. The wound healing rate of mice in different phases was calculated using the following formula: Wound healing rate = (initial wound areas − unhealed wound areas)/initial wound areas × 100%
H&E staining and histological evaluation
H&E staining was conducted according to routine protocols. Briefly, wounds were harvested from db/db mice on days 3, 7, 10, 14, and 17 following after surgery. Next, after deparaffinization and rehydration, 5-μm longitudinal sections were stained with hematoxylin solution for 5 min followed by five dips in 1% acid ethanol (1% HCl in 70% ethanol) and then rinsed in distilled water. The sections were then stained with eosin solution for 3 min, followed by dehydration with graded alcohol and clearing in xylene. As part of the histological evaluation, all slides were examined by a pathologist who had no knowledge of the previous treatment. Six slides in total, the parameters of epidermal and dermal regeneration, granulation tissue thickness in each slide was scored separately, and the scores are presented as the mean ± standard error. The parameters measured were epidermal and dermal regeneration, granulation tissue thickness, and angiogenesis. The margins of the wound in each of the sections, as well in normal control wounds, were used as comparisons for scoring (Table 1). 13 With regards to angiogenesis, CD31 by IHC staining was used to evaluate angiogenesis. The mounted slides were photographed using an Olympus CX31 microscope (Olympus Corporation).
Histological scores of wound.

IHC staining
Wounds were harvested from db/db mice on days 3, 7, 10, 14, and 17 after surgery from mobilization. The harvested tissue including skin wound and the normal skin tissue 0.5 cm from the edge of the wound. CD31 staining was used to evaluate angiogenesis. Longitudinal sections (5-μm thick) of the paraffin-embedded skin wound were kept at 60°C for 2 h in the oven, followed by deparaffinization with xylene and rehydration with an ethanol gradient (100-70%). Following treated with 0.01 M citrate buffer (pH 6.0) at 110°C for 15 min (Beijing Solarbio Science & Technology Co., Ltd., Cat No. C1031), endogenous hydrogen peroxidase was quenched with 3% H2O2 at 37°C for 10 min and 5% Bovine Serum Albumin at 37°C for 40 min to prevent non-specific antibody binding. Then, the slides were rinsed with water and incubated with the primary antibody (CD31, dilution, 1:300; Cell Signaling Technology, Inc. Cat No. 77699S) overnight at 4°C. For negative controls, the primary antibody was replaced by non-immunized serum. The next day, the slides were rinsed and incubated with the corresponding secondary antibody (according to the manufacturer’s instructions, Zhongshan Jinqiao Biotechnology Company. Cat No. PV-900) at 37°C for 30 min, followed by 3,3′-diaminobenzidine (DAB) at 37°C for 5–8 min and terminated color reaction with distilled water, hematoxylin staining at 37°C for 5 min. The slides were then examined and photographed using an Olympus CX31 microscope with magnification of ×200. The DAB staining was analyzed using Image-Pro Plus 6.0 software (Media Cybernetics, Inc.).
WB
Equal amounts (30 mg) of protein extracts were harvested from the wounds of mice on days 3, 7, 10, 14, and 17 after surgery. The protein was extracted using RIPA lysis buffer (Beyotime Institute of Biotechnology), and the protein concentration was measured using a BCA protein concentration assay kit (Beyotime Institute of Biotechnology). The samples were then separated via 10% SDS-PAGE. Proteins were transferred onto nitrocellulose membranes, which were then incubated with the primary antibodies, CD31 (1:2,000; Cell Signaling Technology, Inc. Cat No. 77699S), SDF-1(1:1,000, GeneTex, Inc. Cat No. GTX116092) and β-actin (dilution, 1:500; Abcam. Cat No. AB6276), overnight at 4°C, followed by incubation with the secondary antibody, HRP-conjugated anti-rabbit IgG (Merck KGaA), for 40 min at room temperature. Immunoreactive bands were visualized by Immobilon Western HRP Substrate Luminol reagent (EMD Millipore) which was then exposed to an X-Ray film in a dark room. Densitometric analysis was performed using Image J software (National Institutes of Health).
Statistical analysis
Data are presented as the mean ± standard error of the mean. SPSS v.24.0 (IBM Corp.) was used to perform the statistical analyses. A one-way ANOVA with a Bonferroni’s post-hoc test was used to compare wound closure rates among the studied groups. The p < 0.05 was considered to indicate a statistically significant difference. GraphPad Prism v.6.0 (GraphPad Software, Inc.) software was used to plot the statistics.
Results
AMD3100 plus G-CSF accelerates wound healing following full-thickness skin excision
Changes in the wound areas over time are presented as the percentage of the initial wound areas in (Figure 1(a)). All wound evaluations were double-blinded. The results showed that wound repair was accelerated in the wounds treated with AMD3100 plus G-CSF compared with treatment with the control, AMD3100 alone and G-CSF alone. Wounds reached complete closure on day 21 from surgery in the control group and G-CSF alone groups. The wounds of the db/db mice,which treated with AMD3100 alone, reached complete closure on day 20 from surgery. The healing time was reduced to 17 days in mice from the AMD3100 plus G-CSF group (Figure 1(b)). Following statistical analysis at each time point after injury, the wound closure in the combined group was significantly improved compared with that in the control group (p < 0.001). In addition, the wound closure rate in the combination group was superior to that in the other three groups on days 10 and 17 after surgery (p < 0.001). The wound closure rate in the AMD3100 alone group was superior to that in the control group on day 7, and the difference was statistically significant compared with the G-CSF alone group on days 14 (p = 0.031) and 17 (p = 0.014) from injury. In addition, there was no significant differences between the G-CSF alone and the control groups at each time point following injury (Figure 1(b)).
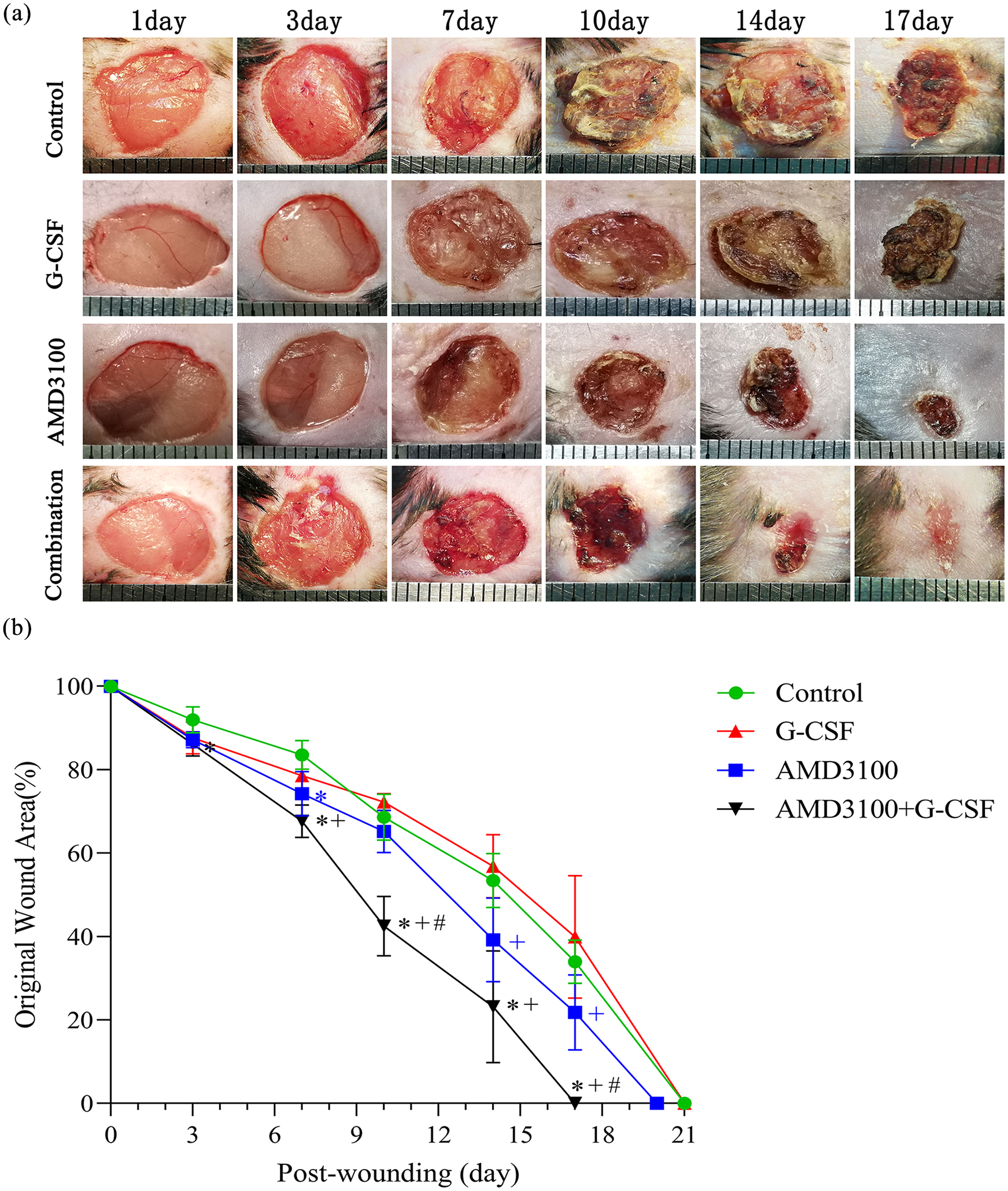
Accelerated wound healing in mice treated with combination of AMD3100 and G-CSF: (a) wound healing. Representative photographs of wounds in mice (n = 6). And the distance between the vertical intervals in the ruler is 1 mm and (b) Quantitative analysis of wound closure in mice (n = 6).
AMD3100 plus G-CSF promotes cell recruitment and glandular formation
Histologically, on day 3 after surgery (the inflammatory period) more cells, particularly inflammatory cells, were recruited into the wound area in the combination group compared with the AMD3100 or G-CSF alone and control groups, especially inflammatory cells. On day 7, granulation tissue was initially formed at the wound margin, and there was more in the combination group compared with the other three groups. On day 10, granulation tissue was further formed, and part of it was transformed into collagen fibers, in which the capillaries and the initial formation of basal layer could be observed in the combination and AMD3100 alone group. On day 14, the epidermal cells covered the wound surface with good continuity in the combination group. In the other groups, further formation of collagen fibers and the epidermal layer was recorded, and a small amount of scabs could still be observed under the microscope. On day 17, the epidermis was of a suitable thickness, with good keratinization, and a small number of new glands was observed in the combination group. By contrast, in the other groups, the epidermal cells essentially covered the wound surface, with disorganized collagen fibers observed and no glands present (Figure 2).
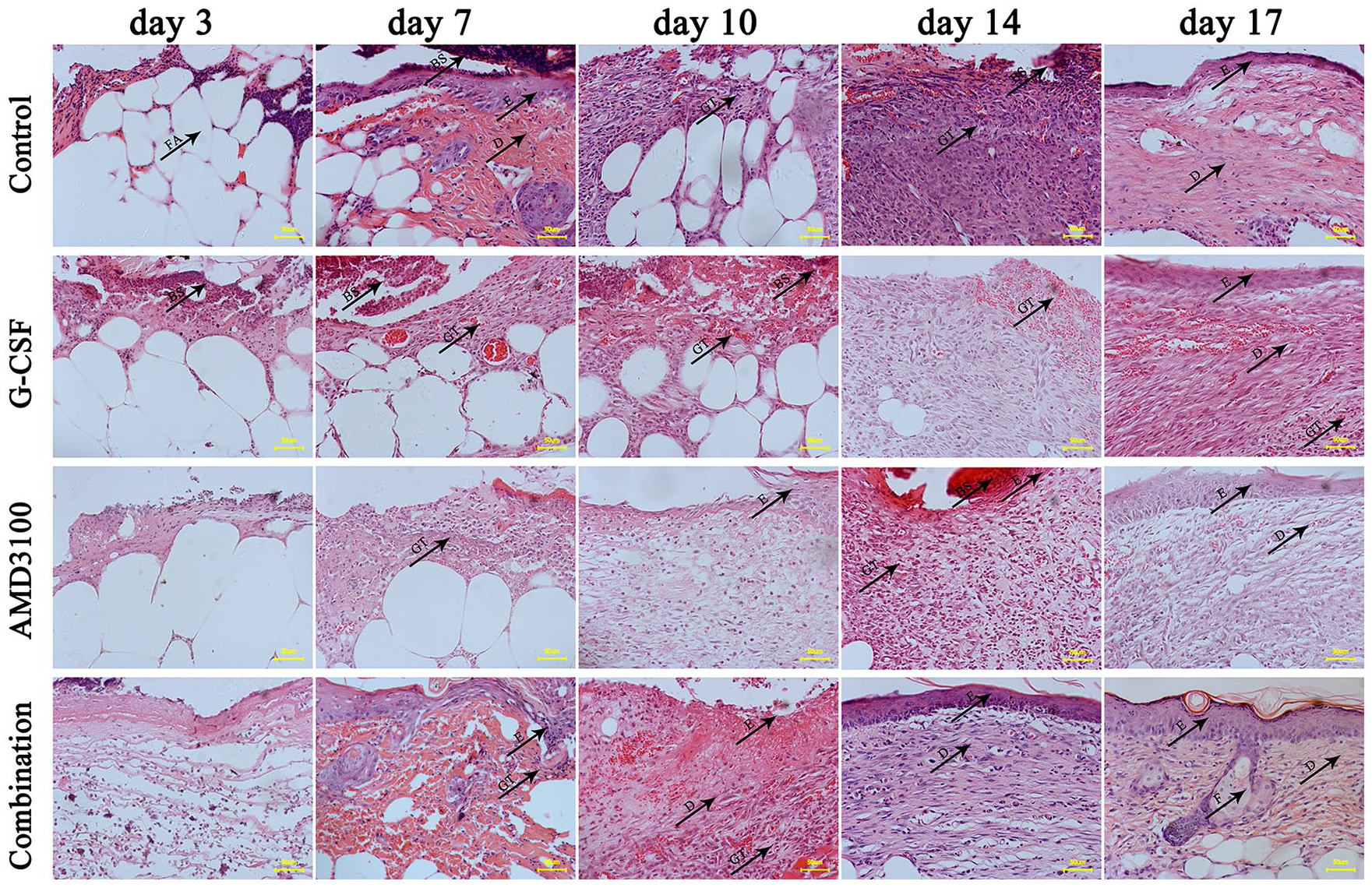
AMD3100 plus G-CSF promoted the recruitment of cells and glandular formation.
Figure 3 shows the histological scores of the wounds throughout the experiment according to the criteria in Table 1. In addition, the number of inflammatory cells in the wound area was measured at each time point of wound healing, and the epidermis thickness was measured on day 17 after surgery in each group using Image Pro Plus 6.0 under a magnification of ×40. The results showed that the number of inflammatory cells peaked on day 10 after surgery in the combination group (Figure 3(a)), which higher than that in other three groups (p < 0.001), and under a magnification of ×40, the average epidermis in the combination group was thicker (22.14 ± 1.80 μm) compared with the other three groups (p < 0.001).
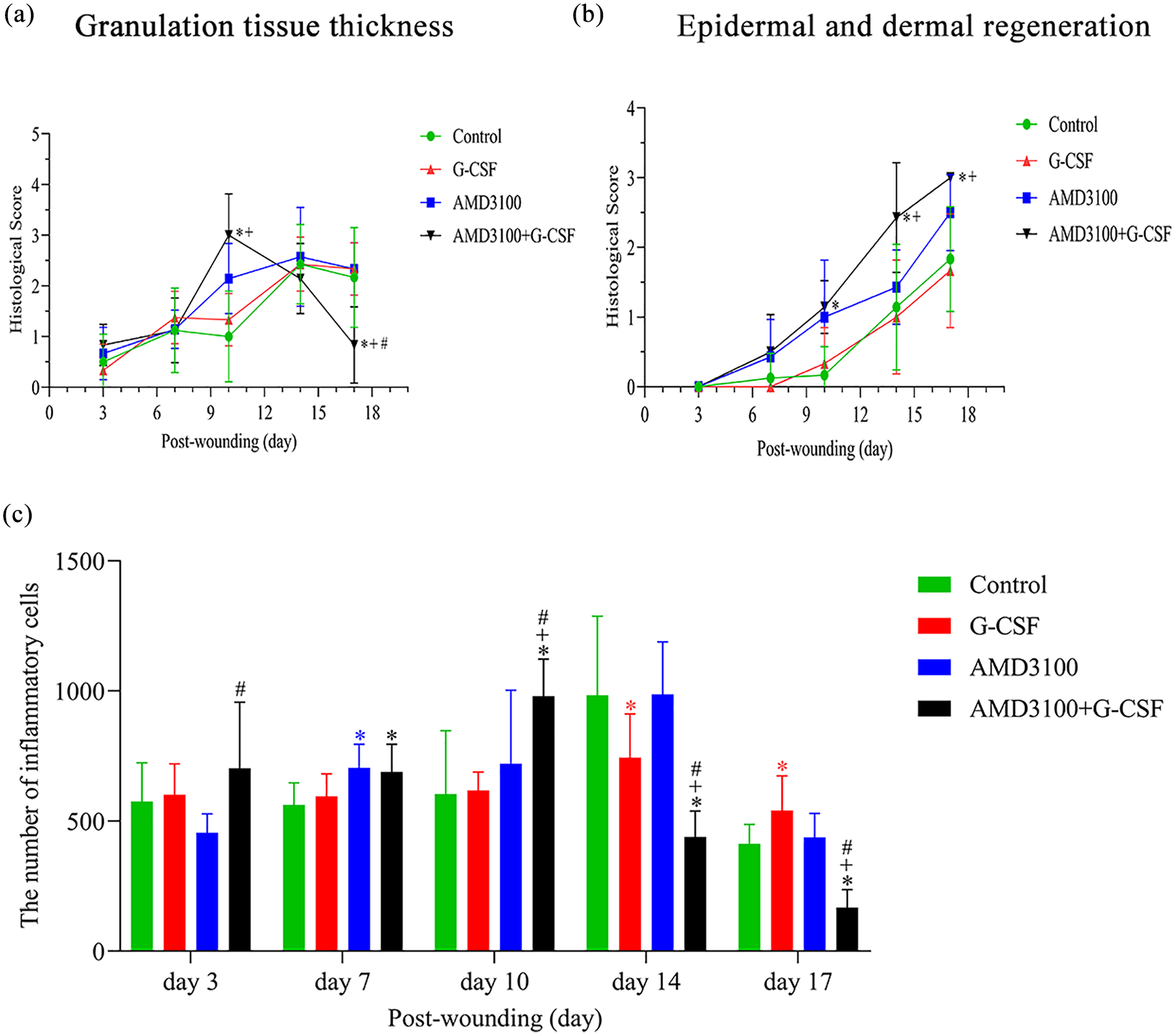
Time course of histological score in db/db mice wounds throughout the experiment: the histological score of granulation tissue thickness (a) and epidermal and dermal regeneration (b) and the number of inflammatory cells (c) Granulation tissue thickness.
AMD3100 plus G-CSF increases the expression of SDF-1 and CD31
The expression of SDF-1 protein in the wound tissue were evaluated using WB (Figure 4). The results showed that the level of SDF-1 protein remained high in the combined group until on day 10 after surgery (p < 0.001). In addition, on day 7 after surgery, SDF-1 protein reached peak in AMD3100 alone group (p < 0.001), G-CSF alone group (p < 0.001) and combined group (p < 0.001), and there were significantly higher than those in control group.
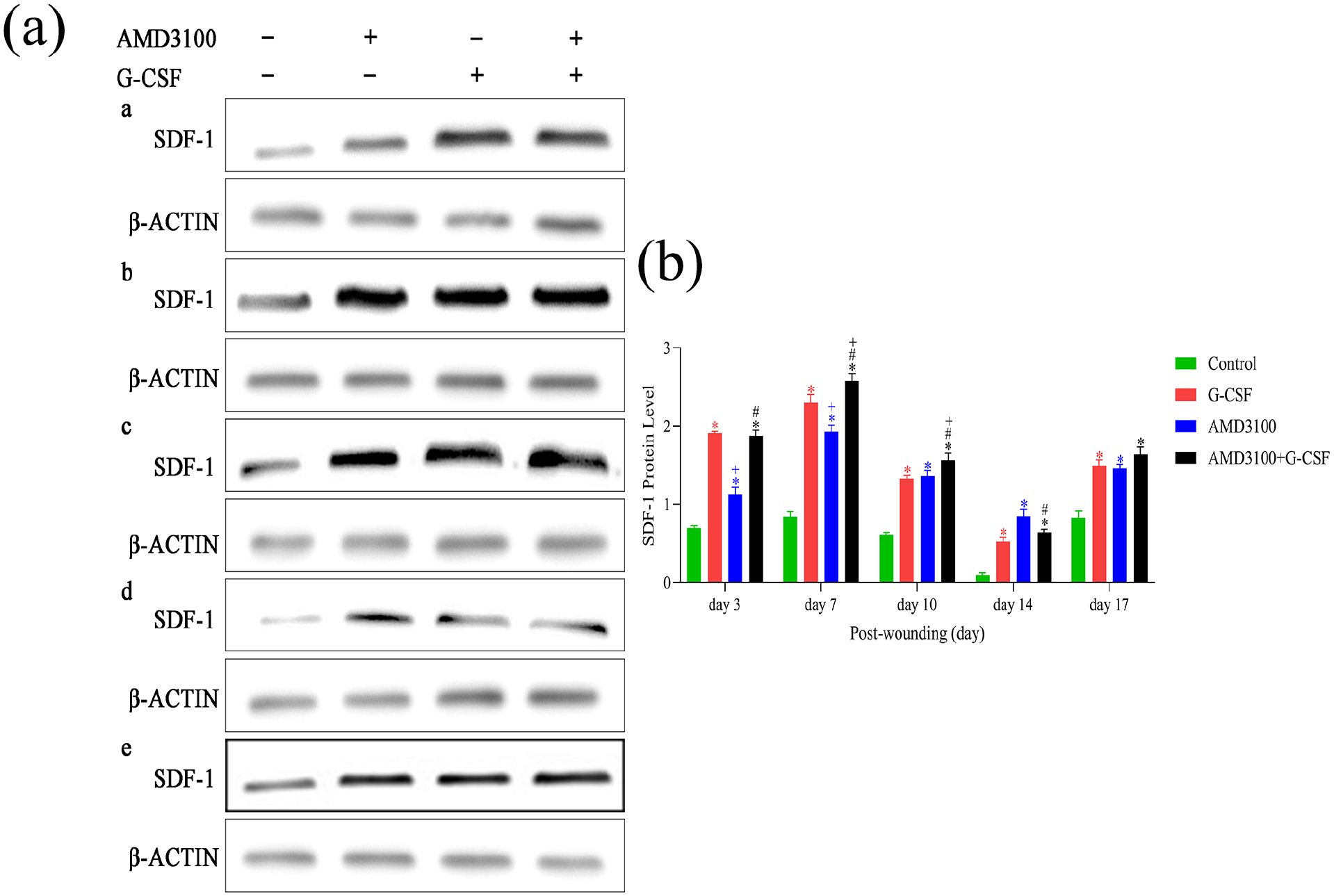
AMD3100 plus G-CSF increased the expression of SDF-1.
CD31+ cells were defined as isolated brown-yellow endothelial cells or clusters exhibiting a single microvessel, which expressed in both differentiated and undifferentiated endothelial cells (Figure 5(a)). CD31+ cells were expressed in the wound tissues of db/db mice from all groups on days 3, 7, 10, 14, and 17 after surgery. The WB results showed that the protein level of CD31 in the combination group was significantly higher compared with the other three groups (p < 0.001) on days 3, 10, and 17 after surgery. And there was also a significant difference in the expression of CD31 between the AMD3100 alone group and G-CSF alone group on day 10 after surgery. In addition, there were significant differences between the combined and control groups on day 7 (p < 0.001) and 14 after surgery (p < 0.001) (Figure 5(b) and (c)).
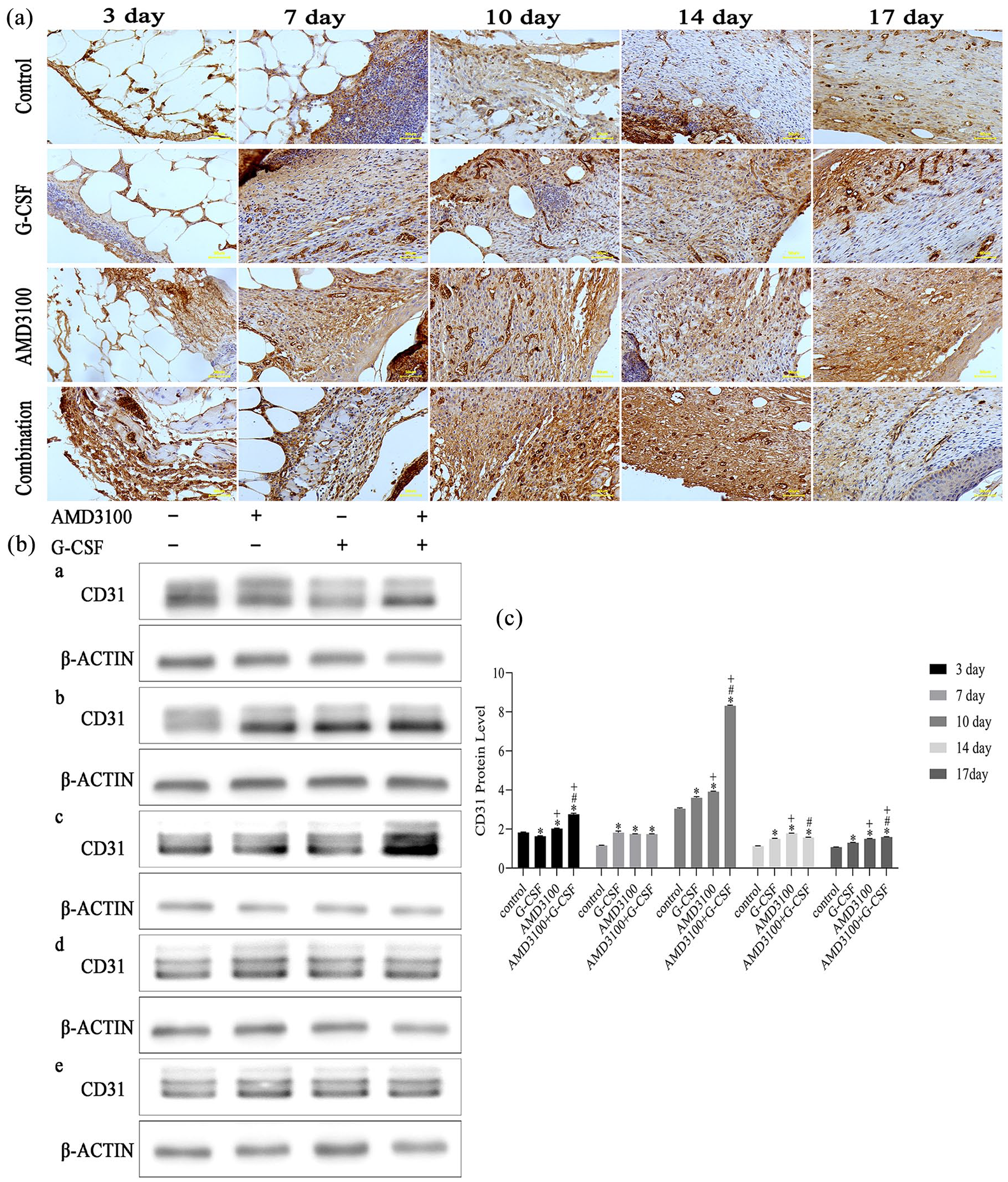
AMD3100 plus G-CSF increased the expression of CD31: (a) the CD31 positive cells in the wound area on day 3, 7, 10, 14, and 17 post-mobilization by IHC (200 X). Scale bar: 50 µm and (b-c) CD31 proteins were evaluated using WB (a–e represent the day 3, 7, 10, 14, and 17 post-wounding, n = 6 in each group).
Discussion
Patients with diabetes suffer from long-term hyperglycemia with angiopathy, and the number and function of EPCs involved in angiogenesis are impaired, rendering the wounds formed in this environment challenging to heal. 14 Therefore, regulating the number and function of EPCs in patients with diabetes has become key to treating diabetic wounds.
Mobilization is defined as with the cytokines and chemokines, the hematopoietic stem/progenitor cells are released into the peripheral blood following the destruction of adhesion molecules, which mediate the anchoring of hematopoietic stem/progenitor cells to bone marrow stromal cells. Mobilizing bone marrow EPCs to the peripheral circulation by G-CSF alone in patients with diabetes is challenging. Studies have shown that the G-CSF mobilization effect is indirect and includes the sympathetic nerve, regulation of CXCL12-producing cells (macrophages, stromal cells, and osteoblasts) and CXCL12 lyase CD26/dipeptidyl peptidase-4 (DPP-4).6,15 However, a high glucose environment can damage the bone marrow microenvironment, leading to sympathetic neuropathy, hematopoietic stem cell number imbalance, and CXCL12 lyase CD26/ DPP-4 adaptive regulation.16,17 Therefore, this condition is called “diabetes stem cell mobilization disease.” 18 In addition, Fadini et al. 19 also found that diabetes limits EPCs mobilization through G-CSF, but not through AMD3100, which was confirmed by our previous study. 11 In that study it was found that the mobilization EPCs by AMD3100 + G-CSF was superior to that by G-CSF + AMD3100 before peaking. 11 Similarly, in the present study, it was also observed that healing in the combined group was superior to that in the other three groups, with the AMD3100 alone group performing slightly better than the G-CSF alone group.
The inflammatory phase is the early stage of wound repair and is mainly characterized by the recruitment inflammatory cells into the damaged area. However, diabetic wounds are trapped in a persistent inflammatory state with a high expression of pro-inflammatory cytokines. Therefore, prolonged inflammatory responses in wounds are associated with impaired healing. 20 AMD3100 or G-CSF can inhibit the recruitment of inflammatory cells to the inflammatory sites of the colon, reduce the expression of inflammatory cytokines, and alleviate submucosal edema of the intestine.21,22 According to the H&E, the thin scab covering the wound was observed in the combination group on day 3 after surgery, while this phenomenon occurred in the other three groups ~7 days after mobilization; however, it shortens the time for a large number of inflammatory cells to gather on the wound. Therefore, AMD3100 and G-CSF not only recruit cells in the injured area, but also regulate the inflammatory response in that area, thus promoting healing; however, further verification of this process required.
Diabetic wounds are challenging to heal, due to vascular lesions and abnormal angiogenesis. In recent years, the advantages of activating “endogenous” stem cells are more prominent, as compared with the transplantation of “exogenous” stem cells. Our results suggested that the mobilization effect of AMD3100 may not be affected and may be slightly enhanced compared with G-CSF in db/db mice. And when it combined with G-CSF could significantly accelerate wound healing in diabetic wound by promoting angiogenesis. In addition, in this experiment, the expression of SDF-1 in the wound tissue were detected, which showed that it in the AMD3100 alone group was higher than that in the control group on day 3 and 7 after mobilization, and the SDF-1 protein level remained high in the combined group after mobilization. Noticeably, SDF-1 significantly increased in G-CSF alone group on day 7 after mobilization, which suggested that in diabetes, G-CSF might mobilize SDF-1+ cells from bone marrow to peripheral blood through the SDF-1/CXCR4 axis. Similar results confirming the G-CSF might have activated CXCR4/SDF-1 axis and contributed to the homing of CD34+/CXCR4+ cells into the ischemic border area and to the reduction in the myocardial infarction size. 23 Therefore, AMD3100 plus G-CSF may through SDF-1/CXCR4 axis, so that it can improve EPCs mobilization from the bone marrow to the peripheral blood, promoting EPC participation in angiogenesis at the ischemic site, thereby promoting diabetic wound healing. Another researchers study has shown that both G-CSF and AMD3100 can mobilize EPCs through the phosphatidylinositol 3-kinase/protein kinase B pathway. 24 Therefore, further experiments are required to explore whether AMD3100 and/or G-CSF are associated with the aforementioned two signal pathways and other EPC mobilization-related molecular mechanisms.
In addition, there is no obvious side effects such as weight loss, hair loss, and anorexia irritability were observed in this animal experiment. An early clinical trial has demonstrated that AMD3100 with G-CSF in mobilizing hematopoietic stem cells in patients with multiple myeloma, with transient and mild side effects. The most of these adverse events were injection site pain, erythema, headache, paresthesia, bone pain, and gastrointestinal disorders. 25 Therefore, the adverse event in AMD3100 with G-CSF in mobilizing EPCs in patients with diabetic wound is needed to further clinical observe.
In conclusion, this experiment confirmed that, in diabetes, AMD3100 + G-CSF can promote wound healing by mainly mobilizing endogenous EPCs and promoting angiogenesis, as well as recruiting more cells into the wound area through the SDF-1/CXCR4 axis. This finding provides novel insight for the improvement of the “diabetes stem cell mobilization disease,” while the interaction between AMD3100 and G-CSF and the related signaling mechanism requires further study. It is expected to provide a safe, accurate and effective mobilization protocol for patients with diabetic wounds, and to improve autologous EPCs and promote the healing of diabetes-caused refractory wounds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant. no.81660321), the special project of Yunnan Science and Technology Department combined with the Kunming Medical University Project of Basic Applied Research [grant. no.2017FE468(-177)], and the Health Science and Technology plan project of Yunnan province (grant. no. 2017NS295).
