Abstract
Endothelial progenitor cells (EPCs) play a fundamental role in tissue regeneration and vascular repair both by differentiating into endothelial cells and by secretion of vasoactive substances that promote angiogenesis and maintain vascular homeostasis. It has previously been shown that hyperglycaemia impairs early and late EPC functions, such as differentiation, proliferation and adhesion. However, its role in the regulation of the production of vasoactive substances in EPCs, especially in late EPCs, is less well defined. We investigated the effects of hyperglycaemia on the production of vasodilator, fibrinolytic and angiogenic growth factors, and also on the activity of superoxide dismutase (SOD) in late EPCs. For this purpose, late EPCs were incubated with different concentrations of D-glucose (5–40 mmol/L) for 24 hr. Levels of nitric oxide (NO), tissue plasminogen activator (t-PA), plasminogen activator inhibitor-1 (PAI-1), prostaglandin I2 (PGI2), vascular endothelial growth factor (VEGF) and the activity of SOD were measured by enzyme-linked immunosorbent assay (ELISA). Under high glucose stress conditions, late EPCs exhibited lower levels of NO, t-PA, PAI-1, PGI2 and VEGF compared to control medium (5 mmol/L glucose). Moreover, high glucose was also observed to decrease the activity of SOD in late EPCs. These results suggest that hyperglycaemia-induced impairment of late EPC secretion functions could contribute to the development of vascular disease in diabetes.
Introduction
Loss of endothelial integrity and impaired capacity for neovascularisation are thought to contribute to vascular complications of diabetes. 1 A growing body of evidence indicates that endothelial regeneration and repair do not exclusively rely on the proliferation of local endothelial cells but also involve bone marrow-derived endothelial progenitor cells (EPCs).2,3 It has been shown that the number of circulating EPCs is decreased, and that functions such as proliferation, adhesion, migration and incorporation into vascular structures are impaired, in patients with either type 1 or type 2 diabetes.4,5 Furthermore, the number of circulating EPCs is inversely correlated with the severity of peripheral vascular complications of patients with diabetes. 6 However, the mechanisms underlying EPC dysfunction in diabetes have not been fully elucidated.
It has long been recognised that good glucose control reduces the risk of microvascular complications of diabetes such as retinopathy, neuropathy and nephropathy. 7 Moreover, the long-term follow-up of participants in the Diabetes Control and Complication Trial (DCCT) has demonstrated that intensive insulin treatment and glycaemic control of type 1 diabetes patients decreases the incidence of acute coronary syndromes. 8 These results suggest that hyperglycaemia is a key pathogenic factor in the development of vascular diseases in diabetes mellitus. Recently, both in vitro and in vivo studies have demonstrated that glucose toxicity determined by hyperglycaemia is associated with impaired EPC functions. For example, Tepper et al. reported that EPCs from type 2 diabetes patients exhibit impaired proliferation, adhesion and incorporation into vascular structures, likely as a consequence of hyperglycaemia. 5 Moreover, Chen et al. demonstrated that hyperglycaemia enhances senescence and reduces the tube-forming ability of early and late EPCs from adult peripheral blood. 9
Growing evidence also suggests that EPCs contribute to the structural integrity of the vasculature, by promoting angiogenesis and maintaining vascular homeostasis through the secretion of matrix proteins, growth factors and cytokines. 10 Although many vasoactive substances can be secreted by both types of EPCs, most of the previous studies mainly focused on early, rather than late, EPCs.11,12 Actually, late EPCs seem more important in promoting vascular integrity since they express a variety of endothelial markers and functionally differentiate into mature endothelial cells. 13 The present study aims to evaluate the effects of hyperglycaemia on late EPC secretion by determining the release of vasoactive substances such as nitric oxide (NO), plasminogen activator inhibitor-1 (PAI-1), tissue plasminogen activator (t-PA), prostaglandin I2 (PGI2) and vascular endothelial growth factor (VEGF); and by measuring the activity of superoxide dismutase (SOD).
Experimental methods
Isolation of bone marrow mononuclear cells and cell culture 14
Whole bone marrow was isolated from both the femurs and tibias of Sprague-Dawley rats (150–175 g) (Weifang Medical University, China). Bone marrow mononuclear cells (MNCs) were fractionated by density gradient centrifugation (Histopaque®-1083, SIGMA, USA) and purity was evaluated by CD45 staining 15 (around 80%, Supplementary Figure S1). MNCs were plated on dishes precoated with fibronectin (ROCHE, Germany), and maintained in complete EGM-2 medium (supplemented with EGM-2 bullet kit, including 5% foetal calf serum, recombinant rat VEGF, recombinant human bFGF, Invitrogen, USA). After four days in culture, unattached cells were removed by a single washing step with phosphate buffer saline (PBS), after which fresh medium was added. Endothelial colonies subsequently appeared (on average one colony per 107 or 108 plated MNCs). Highly proliferative endothelial cells grew out from these colonies which formed a confluent monolayer. Cells in the third-fifth passage, namely late EPCs, were used for the experiments. 9 All animal protocols were approved by the local ethics committee at Weifang Medical University.
Identification of late EPCs
EPCs were characterised by the uptake of 1,1′-dioctadecyl- 3,3,3′,3′-tetramethylindo-carbo-cyanine-labelled acetylated low density lipoprotein (Dil-AcLDL, Molecular Probes, USA), and by fluorescein isothiocyanate labeled Anti-Ulex Europaeus Lectin 1/UEA1 (FITC-UEA-1, SIGMA, USA) staining. In short, the adherent cells were first incubated with 2 μg/ml Dil-AcLDL for 1 hr, after which they were fixed in 2% paraformaldehyde for 10 min, and counterstained with 10 μg/ml FITC-UEA-1 for 1 hr. After staining, samples were viewed with an inverted fluorescence microscope (Leica, Germany). Cells demonstrating double-positive fluorescence were identified as differential EPCs. Furthermore, the cellular expressions of vascular endothelial growth factor receptor 2(VEGFR2, eBioscience, USA, dilution 1:100), von Willebrand factor (Sigma, USA, dilution 1:200), vascular endothelial (VE)-cadherin (BD, USA, dilution 1:400) and platelet/endothelial cell adhesion molecule (PECAM)-1(eBioscience, USA, dilution 1:100) were analysed by Fluorescence Activating Cell Sorter (FACS).
Assay of secretion actions of late EPCs
To measure the secretory capacity actions of late EPCs, cultures were washed and re-fed with either basal medium, in which the D-glucose concentration was 5 mmol/L, or medium supplemented with additional glucose to reach the final concentration. To normalise for the resulting hyperosmosis, the parallel cultures were maintained in the presence of 5 mmol/L glucose and 35 mmol/L mannitol. 9 After 24 hr, the medium was collected and concentrated 10 × by centrifugation for 20 min at 5000 × g at 4°C using Ultrafree-4 centrifugal filter tubes with Biomax-5 membrane(Millipore, USA). Levels of NO, t-PA, PAI-1, PGI2 and VEGF, and the activity of SOD in the cell culture supernatants, were measured by sandwich enzyme-linked immunosorbent assay (ELISA, RD, USA) according to the manufacturer’s instructions. Briefly, a standard or testing sample (50 μl) was added to the 96-well plate containing immobilised monoclonal antibodies. After mixing by shaking gently, the plate was incubated at 37°C for 30 min. The wells were washed five times, and incubated with HRP-conjugate reagent at 37°C for 30 min. Afterwards chromogen solution A and chromogen solution B were added to each well. The reaction was terminated after 20 min by adding the stop solution. A blank well was taken as zero, and the optical density (OD) was measured at 450 nm with an ELISA reader (Bio-Rad Laboratories, USA).
Statistical analyses
Unless otherwise indicated, results are reported as means±standard error (SE) from 5–6 independent experiments (In each of the experiments, every group includes from 6 to 8 samples). The normality of sample distribution was verified by the Kolmogorov-Smirnov test with tests for skewness and kurtosis. Since all series of data were normally distributed, statistical analyses were performed by one-way analysis of variance (ANOVA), followed by Tukey’s or Tambane’s test between the groups, and p < 0.05 was considered to be statistically significant. All data were analyzed using SPSS software (version 15.0; SPSS, Chicago, USA).
Results
Characterisation of bone marrow-derived EPCs
The bone marrow-derived MNCs that initially seeded were round (Figure 1A). The distinct colonies formed after 48 hr reaching a peak on the seventh day. The colonies of early EPCs appeared with the round cells in the centres and the typical spindle cells at the periphery after seven days (Figure 1B). After three to four weeks, the third-fifth passage cells (late EPCs) showed characteristic homogeneity and cobblestone-like morphology similar to mature endothelial cells as previously reported (Figure 1C). Late EPCs were identified as double-positive for Dil-AcLDL uptake and lectin binding affinity (Figures 1D, 1E and 1F). Moreover, the majority of the cells expressed endothelial-specific markers: vWF, VEGFR-2, VE-cadherin and PECAM-1 (Figure 1G).
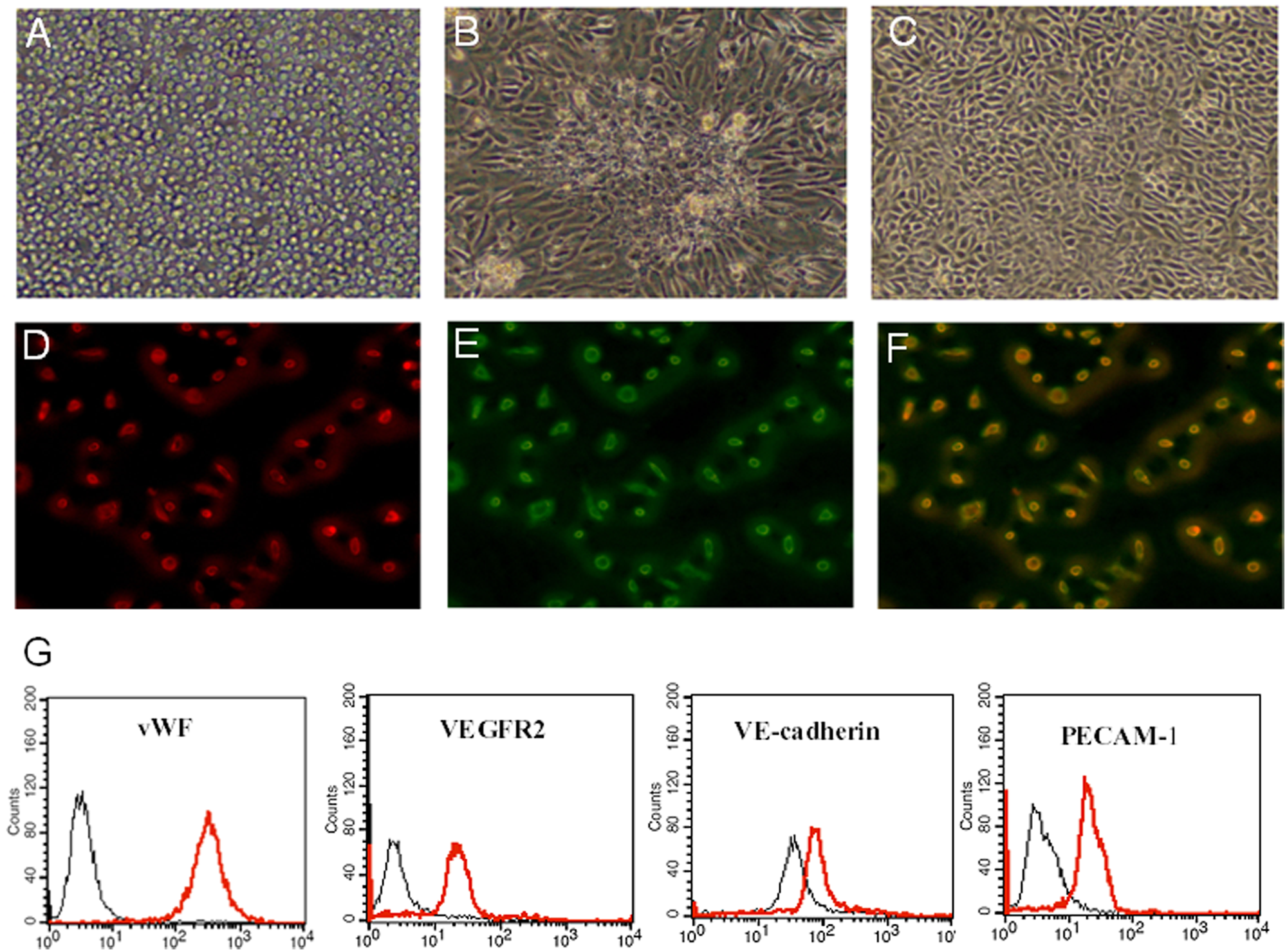
Characterisation of bone marrow-derived endothelial progenitor cells (EPCs).
Effects of high glucose on NO production in late EPCs
To investigate the effects of high glucose on NO production in late EPCs, cells were incubated for 24 hr in mediums containing different concentrations of glucose or mannitol. The cultured media were collected, and levels of NO determined by ELISA. The results show that glucose concentrations of 10 or 20 mmol/L glucose had no effects on the production of NO from EPCs. However, a glucose concentration of 40 mmol/L was observed to significantly decrease NO production compared to the control medium (5 mmol/L glucose), whereas the osmotic control did not influence the production of NO (Figure 2).
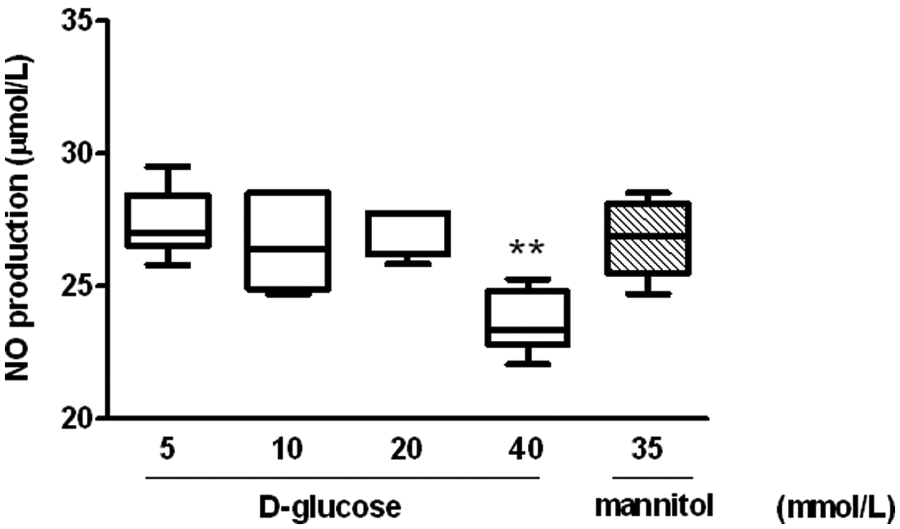
Effects of high glucose on nitric oxide (NO) production in late endothelial progenitor cells (EPCs).
Role of high glucose on the activity of SOD in late EPCs
The bioactivity of glucose on SOD was measured in late EPCs. As shown in Figure 3, the incubation of cells with glucose decreased SOD activity in late EPCs in a dose-dependent manner. As compared to the control medium (5 mmol/L glucose), the SOD activity of late EPCs assessed by ELISA was reduced by 14.9% and 20.3% in the high-glucose mediums of 20 and 40 mmol/L respectively. In contrast, the osmotic control did not change SOD activity in late EPCs (Figure 3).
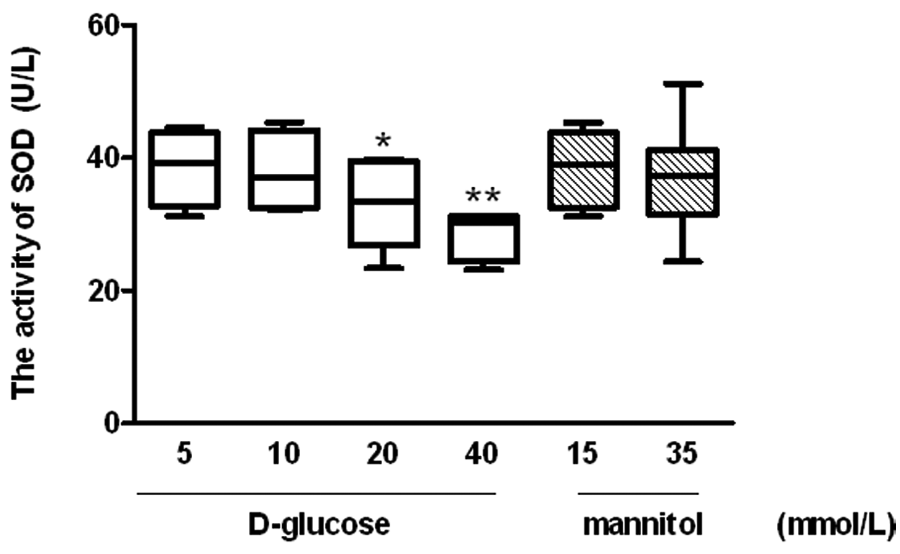
Role of high glucose on the activity of superoxide dismutase (SOD) in late endothelial progenitor cells (EPCs).
Influence of high glucose on PAI-1 and t-PA secretion from late EPCs
Since the balance between PAI-1 and t-PA determines fibrinolytic activity, we also investigated the effects of glucose on the production of PAI-1 and t-PA from late EPCs. It was observed that the incubation of late EPCs with high concentrations of glucose (40 mmol/L) led to a significant decrease in the production of t-PA and PAI-1. Interestingly, a glucose concentration of 20 mmol/L also produced a decrease in the production of t-PA, but did not alter PAI-1 production. Moreover, the PAI-1/ t-PA ratio showed a positive correlation with the level of glucose concentration (Figures 4A, 4B and 4C).
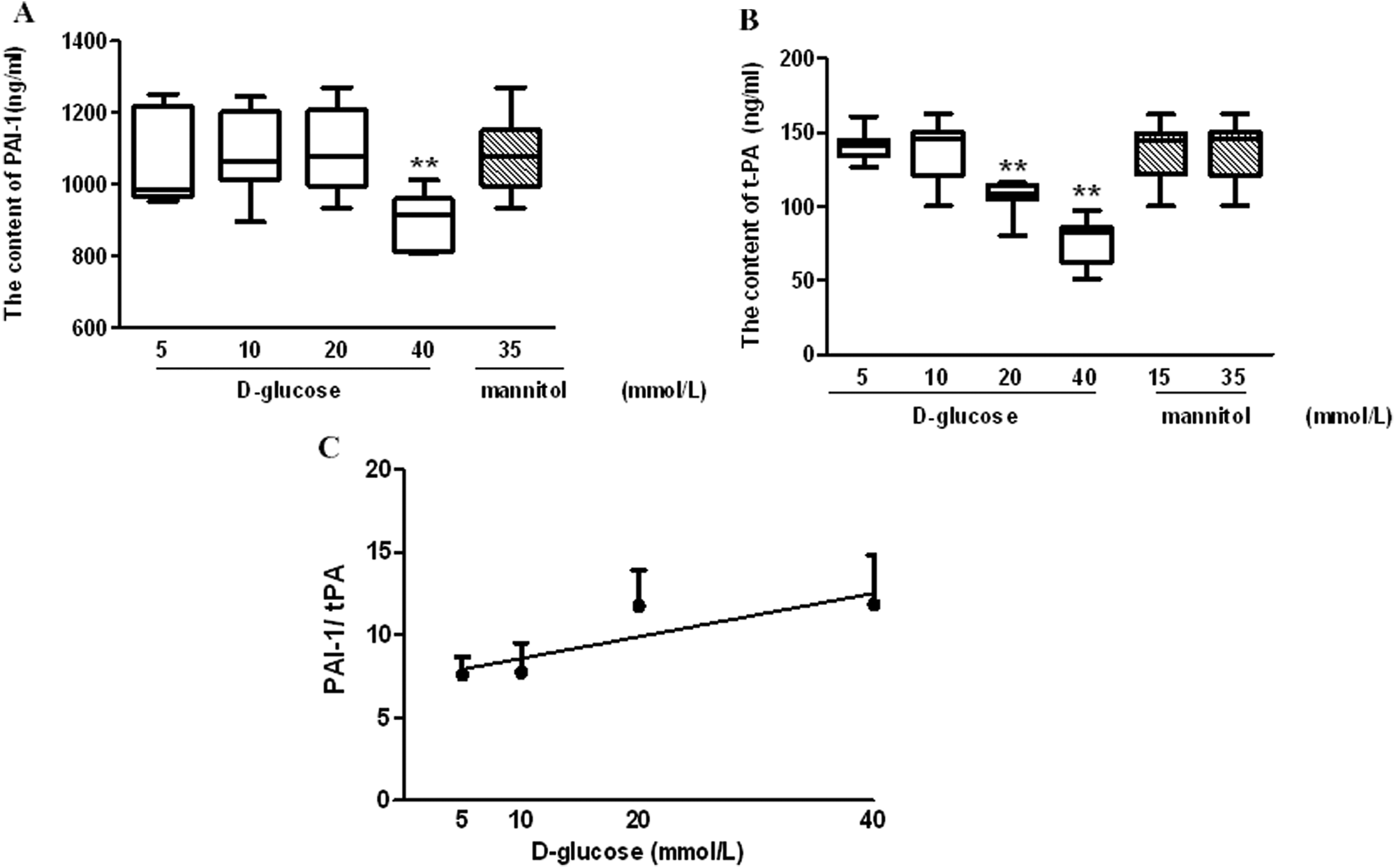
Influence of high glucose on plasminogen activator inhibitor-1 (PAI-1) and tissue plasminogen activator (t-PA) secretion from late endothelial progenitor cells (EPCs).
Impact of high glucose on PGI 2 secretion from late EPCs
Given that PGI2 has a short half-life (<20 min at physiological pH), levels of 6-keto-prostaglandin F1α (6-keto-PGF1α), the PGI2 stable degenerative product, were quantified to evaluate the impact of glucose on PGI2 secretion from late EPCs. The production of 6-keto-PGF1α by glucose-stressed EPCs was observed to be reduced in a dose-dependent manner. As compared to that in the control medium (5 mmol/L glucose), the 6-keto-PGF1α produced by late EPCs was reduced by 11.8%, 14.8% and 24.1% in glucose concentrations of 10, 20 and 40 mmol/L respectively (Figure 5).
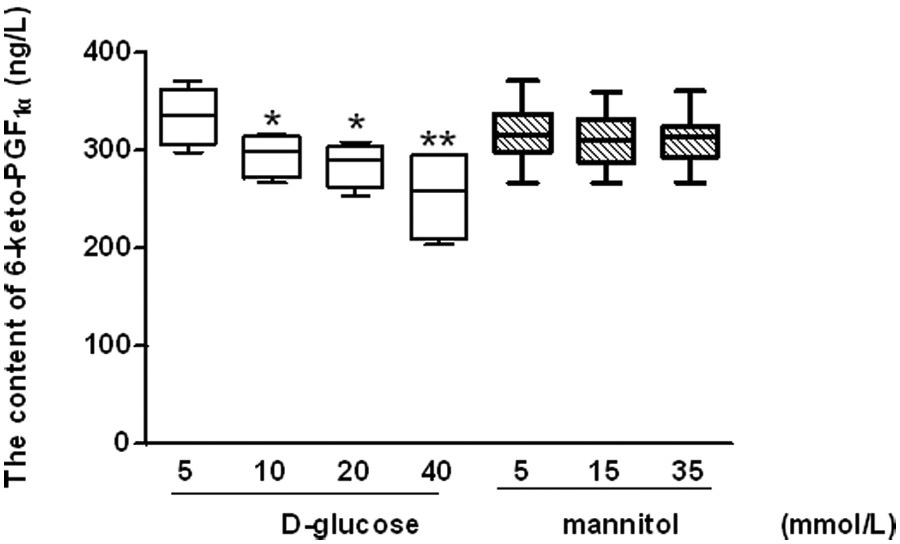
Impact of high glucose on prostaglandin I2 (PGI2)secretion from late endothelial progenitor cells (EPCs).
Effects of high glucose on growth factor release from late EPCs
Incubation of late EPCs with 20 or 40 mmol/L glucose led to a significant decrease in the release of VEGF from late EPCs (Figure 6).
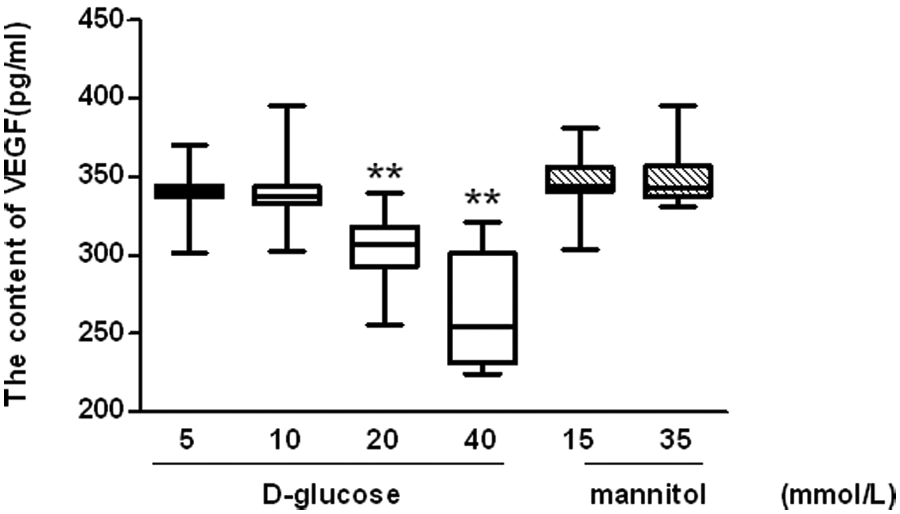
Effects of high glucose on vascular endothelial growth factor (VEGF) release from late endothelial progenitor cells (EPCs).
Discussion
Hyperglycaemia plays a key role in the development of vascular complications in diabetic patients. 16 This has been partially attributed to a reduction in EPC number and angiogenicity. 17 EPCs can be isolated from bone marrow, peripheral blood, and umbilical cord blood.14,18 In the present study, EPCs were cultured from bone marrow and Sprague-Dawley rats (used to generate diabetic rodent models19,20) served as a marrow source. Early EPCs appear within four to seven days of culture, are spindle-shaped and have a limited proliferation potential. Late EPCs develop after two to three weeks of culture and have characteristics of endothelium lineage, with a cobblestone shape and long-term proliferation and clonogenic potential. Yoon et al. 13 have reported that late EPCs appear to be a major underlying component contributing to vascular integrity and homeostasis, and may serve as a potential therapeutic target for vascular regeneration. Thus, in this study, a high glucose cell culture model was used to simulate clinical hyperglycaemia for the in vitro evaluation of the secretory functions of late EPCs. Our findings show that hyperglycaemia causes a reduction in NO, PGI2, t-PA, PAI-1 and VEGF in late EPCs. Furthermore, hyperglycaemia decreases the SOD activity of late EPCs.
NO is a major vasodilator and a key survival factor of the endothelium. Endothelial dysfunction is characterised by low bioavailability of endothelium-derived NO which itself is an independent predictor of future cardiovascular events. 21 The reduction in NO production is therefore ossibly contributory to the development of endothelial dysfunction and atherosclerosis in diabetes mellitus. 22 Ozuyaman et al. have demonstrated that NO can stimulate EPC mobilisation from bone marrow stem cell niches to the peripheral circulation, implying that they participate in neovascularisation. 23 Furthermore, transplantation of autologous EPCs overexpressing endothelial nitric oxide synthase (eNOS) in injured vessels enhances the vasculoprotective prosperities of reconstituted endothelium. 24 Therefore, it seems that NO produced by EPCs creates a favorable and optimal environment in regulating function. In the present study, we have shown that a 24 hr incubation with high glucose (40 mmol/L) significantly decreases bioavailable NO, which is in agreement with the findings of Chen et al. 9 Our findings are also similar to those observed in mature vascular endothelial cells. 25
It has become evident that O2- is important in the development of mature endothelial cell and EPC dysfunction.26,27 Increased vascular oxidative stress is closely associated with reduced extracellular SOD activity.28,29 It has previously been shown that EPCs have high intracellular expression levels of SOD which protects against oxidative stress. 30 Tao et al. 31 demonstrated that augmenting Cu/Zn SOD expression in human EPCs by shear stress accelerates O2- neutralisation. This leads to an increase of EPC repair potential in the vascular system. We have here shown that high glucose decreases SOD activity. Thus decreased SOD activity may not be sufficient to neutralise the high O2- caused by diabetes, which may contribute to increased risk of developing cardiovascular disease in diabetic patients. Besides NO and SOD, both clinical and experimental studies suggest that thrombus formation and plaque rupture are other major aspect of cardiovascular disease. 32 Thus, we studied the effects of high glucose on the fibrinolytic factors t-PA and PAI-1. In addition to endothelial cells, EPCs also produce t-PA, at amounts comparable to that secreted by mature endothelial cells. 33 As the primary inhibitor of t-PA, PAI-1 also plays a critical role in the regulation of fibrinolysis, and the balance between these proteins is thought to control thrombolysis. In the present study, although high glucose was found to significantly decrease the secretion of both t-PA and PAI-1 by EPCs, the reduction of t-PA was greater than that of PAI-1. Moreover, the PAI-1/ t-PA ratios showed a positive correlation with the glucose concentration. This indicates that high glucose, by disturbing the balance between PAI-1 and t-PA, induces EPCs to exhibit decreased fibrinolytic potential which may contribute to vascular risk.
EPCs have high anti-thrombogenic potentials, similar to that of mature endothelial cells, and they have approximately half the anti-platelet function as compared to mature endothelial cells. 33 In addition to NO, PGI2 is a potent inhibitor of platelet activation. Moreover, PGI2 is a potent vasodilator contributing to the protection and maintenance of homeostasis in the vasculature. 34 Suppressed levels of PGI2 have been reported in the early stages of diabetes. For example, the production of PGI2 by the blood vessels in patients with diabetes is depressed and urinary and circulating levels of 6-keto-PGF1α are reduced. 35 Furthermore, decreased PGI2 has been linked to platelet hyperaggregability, increased adhesiveness and increased release of PGH2 and TXA2 in diabetes patients. 36 Our findings show that high glucose decreases EPC production of PGI2 which may contribute to increased platelet activity, and partially explain why the platelet activity recovers with improved glycaemic control in patients with diabetes. 37
As one of the most important factors involved in angiogenesis as well as vasculogenesis, VEGF induces endothelial cell and EPC proliferation, migration and the recruitment of EPCs from the bone marrow. 38 Moreover, VEGF exerts strong cytoprotective and prosurvival activity by inducing expression of anti-apoptotic and antioxidant proteins.39,40 It has also been demonstrated that gene and protein levels of VEGF are higher in EPCs compared to mature endothelial cells. 10 In the present study, high glucose decreased production of VEGF from EPCs which could contribute to the impairment of EPCs and EPC-related vascular repair in clinical diabetes.
Taken together, the present study demonstrates that hyperglycaemia exerts deleterious effects on late EPC secretion actions. These findings provide further insight into the mechanisms by which EPCs exhibit impaired vascular repair and abnormal neovasculogenesis in diabetes patients.
Footnotes
Acknowledgements
We would like to thank Dr Emil Avsar for critical reading of the manuscript and Professor Yuguang Gao, the director of the stomatological laboratory in Weifang Medical College, for general support.
Funding
This work was supported by the National Natural Science Foundation of China (grant no. 30900290), the Natural Science Foundation of Shandong Province (grant no. ZR2009CQ027), Program for New Century Excellent Talents in University (grant no. NCET-10-0922) and the Foundation of Shandong Educational Committee (grant no. J09LF06).
Conflicts of interest statement
None declared.
