Abstract
Aim:
To investigate risk factors associated with kidney disorders in patients with type 2 diabetes (T2D) at high cardiovascular (CV) risk.
Methods:
In DEVOTE, a cardiovascular outcomes trial, 7637 patients were randomised to insulin degludec (degludec) or insulin glargine 100 units/mL (glargine U100), with standard of care. In these exploratory post hoc analyses, serious adverse event reports were searched using Standardised MedDRA® Queries related to chronic kidney disease (CKD) or acute kidney injury (AKI). Baseline predictors of CKD, AKI and change in estimated glomerular filtration rate (eGFR) were identified using stepwise selection and Cox or linear regression.
Results:
Over 2 years, eGFR (mL/min/1.73 m2) decline was small and similar between treatments (degludec: 2.70; glargine U100: 2.92). Overall, 97 and 208 patients experienced CKD and AKI events, respectively. A history of heart failure was a risk factor for CKD (hazard ratio [HR] 1.97 [95% confidence interval [CI] 1.41; 2.75]) and AKI (HR 2.28 [95% CI 1.64; 3.17]). A history of hepatic impairment was a significant predictor of CKD (HR 3.28 [95% CI 2.12; 5.07]) and change in eGFR (estimate: −8.59 [95% CI −10.20; −7.00]).
Conclusion:
Our findings indicate that traditional, non-modifiable risk factors for kidney disorders apply to insulin-treated patients with T2D at high CV risk.
Trial registration:
NCT01959529 (ClinicalTrials.gov).
Keywords
Introduction
Chronic kidney disease (CKD) is a common comorbidity in type 2 diabetes (T2D), co-occurring in approximately 50% of patients. 1 The presence of CKD is associated with a higher risk of cardiovascular disease (CVD), including atherosclerotic CVD and heart failure, 2 which is a leading cause of morbidity and mortality in people with diabetes. 3 CKD is a dynamic disease that progresses at a variable rate; prior published literature has demonstrated that the rate of estimated glomerular filtration rate (eGFR) decline is associated with hospitalisation 4 and mortality. 5 In patients with T2D and CKD, the natural history of CKD appears to be heterogeneous. 6 Although there are several well-established risk factors for CKD, such as hyperglycaemia and hypertension, many are complex and not well-defined. 6
Acute kidney injury (AKI) encompasses a group of syndromes defined by a sudden reduction in glomerular filtration rate (GFR) 7 that affect as many as one in five hospitalised patients. 8 The development of AKI is associated with a wide range of poor health outcomes and increased healthcare costs.7,9 There is increasing recognition that CKD and AKI are closely linked and likely to promote one another,10,11 and patients with diabetes are at higher risk of these complications than those without diabetes.12,13 The prompt identification of patients at highest risk of CKD, AKI and their negative sequelae will assist in the development of strategies to optimise their care.
In DEVOTE, a cardiovascular outcomes trial (CVOT), the cardiovascular (CV) safety of insulin degludec (degludec) was compared with that of insulin glargine 100 units/mL (glargine U100) in patients with T2D at high CV risk. 14 In DEVOTE, degludec was non-inferior to glargine U100 with respect to the incidence of CV events (hazard ratio [HR] 0.91 [95% confidence interval [CI] 0.78–1.06]), whilst patients experienced significantly fewer severe hypoglycaemic events (rate ratio: 0.60 [95% CI 0.48–0.76]), at similar levels of glycaemic control with degludec versus glargine U100. 14 As DEVOTE enrolled patients with moderate CKD and included a small cohort with severe CKD, 14 it provides additional opportunities to investigate risk factors for kidney disorders in this patient population. This is notable as, previously, patients with kidney disease (particularly severe disease) have often been excluded from randomised clinical trials investigating CV outcomes, despite strong associations between kidney disease, CV events and death in patients with CVD. 15
The objective of these post hoc exploratory analyses of DEVOTE data was to investigate the factors associated with the risk of first kidney disorders (CKD and AKI) and the change in eGFR for patients with T2D at high CV risk. An additional objective was to explore bidirectional temporal associations between severe hypoglycaemia and the risk of first kidney disorders.
Research design and methods
DEVOTE trial design
The design and primary results of DEVOTE have been published. 14 In brief, DEVOTE was a double-blind, randomised, treat-to-target, active comparator-controlled CVOT. Patients with T2D at high CV risk were randomised 1:1 to degludec (100 units/mL) or glargine U100, administered once daily, in addition to standard care. Eligible patients were treated with ⩾1 oral or injectable antihyperglycaemic agent and with glycated haemoglobin (HbA1c) ⩾7.0% (53 mmol/mol) or <7.0% and treated with ⩾20 units/day of basal insulin. Patients were ⩾50 years of age with ⩾1 CV or kidney condition (moderate CKD: eGFR of 30–59 mL/min/1.73 m2 per the Chronic Kidney Disease Epidemiology Collaboration [CKD-EPI] equation), or ⩾60 years of age with ⩾1 pre-specified CV risk factors. Patients receiving haemodialysis, peritoneal dialysis or with an eGFR <30 mL/min/1.73 m2 at screening were ineligible. DEVOTE included 214 patients with severe CKD (defined as an eGFR of <30 mL/min/1.73 m2 per the CKD-EPI equation) at baseline who met inclusion/exclusion criteria at screening. 16 The trial protocol was approved by the independent ethics committee or institutional review board at each trial centre. Written informed consent was obtained from each patient before any trial-related activities. Separate ethics approval was not required for these post hoc analyses, as there were no new studies involving animals or humans.
Outcomes
CKD and AKI were identified in investigators’ reports of serious adverse events (SAEs; defined in Supplemental Methods S1) using narrow Standardised Medical Dictionary for Regulatory Activities Queries (SMQ; version 19.0) adverse event (AE) reporting terms related to CKD and AKI (detailed in Supplemental Methods S2). eGFR was estimated from serum creatinine using the CKD-EPI equation. 17 Demographic and clinical factors predicting the following outcomes were assessed: time to first CKD SAE, time to first AKI SAE and change in eGFR. Any patients that experienced a CKD or AKI SAE in DEVOTE were considered in the analyses (including those defined as having either moderate or severe CKD at baseline). It is worth noting that SAEs could include the worsening of a pre-existing medical condition if the defining criteria were met (Supplemental Methods S1). Patients that experienced both CKD and AKI SAEs were included in the analyses of both endpoints, while only the first SAE reports (e.g. first CKD SAE and first AKI SAE in the trial) for each patient were considered and any further CKD or AKI SAEs in DEVOTE were excluded. Severe hypoglycaemia (requiring assistance from another person) 18 was self-reported in DEVOTE and adjudicated by an independent Event Adjudication Committee. Bidirectional temporal associations between positively-adjudicated severe hypoglycaemia and time to first kidney SAE were also investigated. In the present article, the term ‘kidney disorder’ has been used to encompass both CKD and AKI SAEs; however, these endpoints were analysed separately.
Statistical analysis
Significant predictors of time to first kidney disorder were identified using stepwise selection in SAS PHREG with p-value thresholds of 0.1 and 0.05 determining whether a single predictor should be added or removed from the model. Candidate covariates were available baseline demographic and clinical data (listed in Supplemental Figure S1). Randomised treatment was forced to be part of the final model to account for stratification. Final estimates were based on a Cox proportional hazard model that included randomised treatment and all significant predictors simultaneously. The relative importance of model predictors was their Chi-square contribution.
Change in eGFR from baseline to 24 months was analysed using a mixed model for repeated measures (MMRM) within patients using an unstructured covariance matrix among visits at 12 and 24 months. Interactions between visit and treatment and between visit and baseline eGFR were included as fixed effects. Baseline predictors of change in eGFR were identified by stepwise selection using a generalised linear model (SAS GLMSELECT) that was forced to include baseline eGFR, visit and randomised treatment. The relative importance of model predictors was calculated based on the F-value contribution of each variable.
Time to first kidney disorder was analysed by Cox regression with severe hypoglycaemia (Yes/No) as a time-varying covariate to compare the risk of an event (CKD or AKI) with or without having a prior severe hypoglycaemic event in different time periods. 19 Analyses were repeated adjusting for baseline covariates including treatment, sex, age, smoking status, geographic region, diabetes duration, insulin naïve, CV risk and eGFR, in alignment with pre-specified sensitivity analyses of the DEVOTE primary endpoint. 14 Time-to-event analyses were also conducted for severe hypoglycaemia using Cox regression with first kidney disorder (Yes/No) as a time-varying covariate to compare the risk of severe hypoglycaemia with or without having experienced a prior kidney disorder event in different time periods.
In all analyses, a two-sided p-value <0.05 was considered statistically significant, no adjustment was made for multiplicity and transformations were performed by natural log (using base e = 2.718).
Results
Patient demographics and baseline characteristics of the overall trial population (N = 7637) have been described previously. 14 Median observation time was 2.0 years. Treatment arms were well matched with respect to baseline characteristics including the use of renin-angiotensin system inhibitors. 14
Demographic and clinical predictors of kidney disorders
Overall, CKD occurred in 97 patients: 45 patients (1.2%) with degludec and 52 patients (1.4%) treated with glargine U100 (Supplemental Table S2). AKI events were more frequent than CKD events, occurring in 208 patients: 87 patients (2.3%) treated with degludec and 121 patients (3.2%) treated with glargine U100 (Supplemental Table S2). In total, 43 patients experienced both CKD and AKI events. Baseline demographic and clinical characteristics for patients stratified by kidney SAE status in DEVOTE are presented in Supplemental Table S3. Baseline eGFR was identified as the most important predictor for time to first CKD and time to first AKI (Table 1). Other predictors of first CKD (in order of relative importance) included a history of hepatic impairment (defined as a modified Child-Pugh score [bilirubin and albumin values only] of ⩾2), a history of heart failure, higher systolic blood pressure (BP), a history of amputation, diabetes duration ⩾15 years, male sex, and higher HbA1c and age (a lower risk in older patients) (Table 1). For first AKI, baseline predictors (in order of relative importance) included a history of heart failure, prior insulin regimen (a higher risk in patients with more complex baseline insulin regimens) and patients treated in the US (Table 1). Furthermore, our analyses identified a lower risk of first AKI with degludec versus glargine U100 (adjusted HR 0.73 [95% CI 0.53; 1.00] p = 0.049).
Baseline demographic and clinical predictors of time to first kidney disorder in patients with T2D at high CV risk.
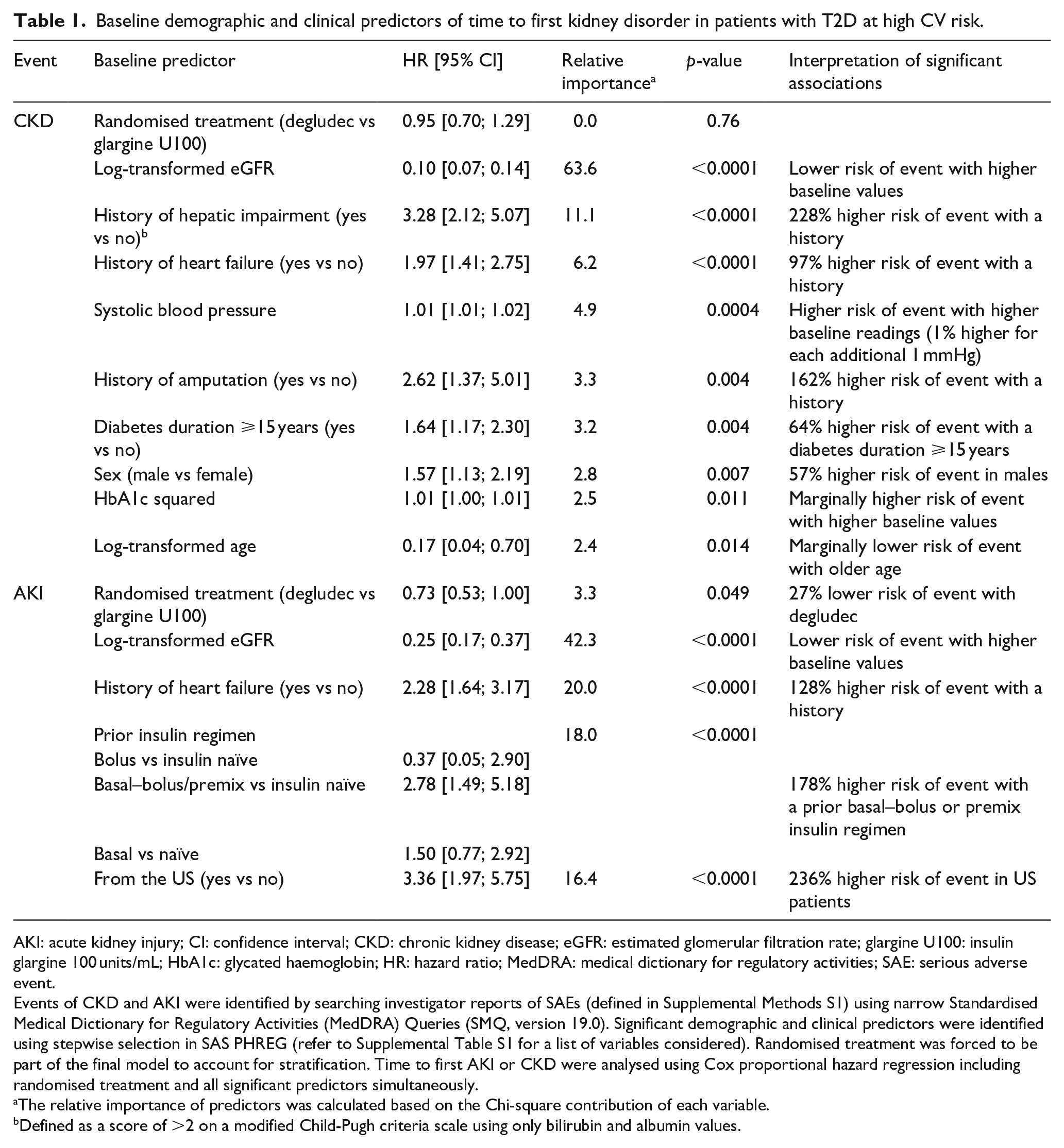
AKI: acute kidney injury; CI: confidence interval; CKD: chronic kidney disease; eGFR: estimated glomerular filtration rate; glargine U100: insulin glargine 100 units/mL; HbA1c: glycated haemoglobin; HR: hazard ratio; MedDRA: medical dictionary for regulatory activities; SAE: serious adverse event.
Events of CKD and AKI were identified by searching investigator reports of SAEs (defined in Supplemental Methods S1) using narrow Standardised Medical Dictionary for Regulatory Activities (MedDRA) Queries (SMQ, version 19.0). Significant demographic and clinical predictors were identified using stepwise selection in SAS PHREG (refer to Supplemental Table S1 for a list of variables considered). Randomised treatment was forced to be part of the final model to account for stratification. Time to first AKI or CKD were analysed using Cox proportional hazard regression including randomised treatment and all significant predictors simultaneously.
The relative importance of predictors was calculated based on the Chi-square contribution of each variable.
Defined as a score of >2 on a modified Child-Pugh criteria scale using only bilirubin and albumin values.
Demographic and clinical predictors of change in eGFR
In the overall trial population, mean (SE) eGFR (mL/min/1.73 m2) was 67.96 (0.25) at baseline, decreasing to 65.07 (0.32) after 24 months (a mean [SE] change of −2.51 [0.18] over 2 years; Figure 1(a)). In both treatment groups, eGFR declined at a similar rate, with no significant difference between randomised treatments (estimated treatment difference: 0.22, [95% CI −0.43; 0.87] p = 0.50) (Figure 1(b)). Predictors of eGFR decline overall were similar to those identified for CKD alongside additional baseline demographic and clinical factors with lower relative importance (Table 2). These additional factors included a more rapid decline in eGFR with a history of proteinuria, high CV risk, higher triglyceride levels, a history of peripheral artery disease and no history of stroke or angina.
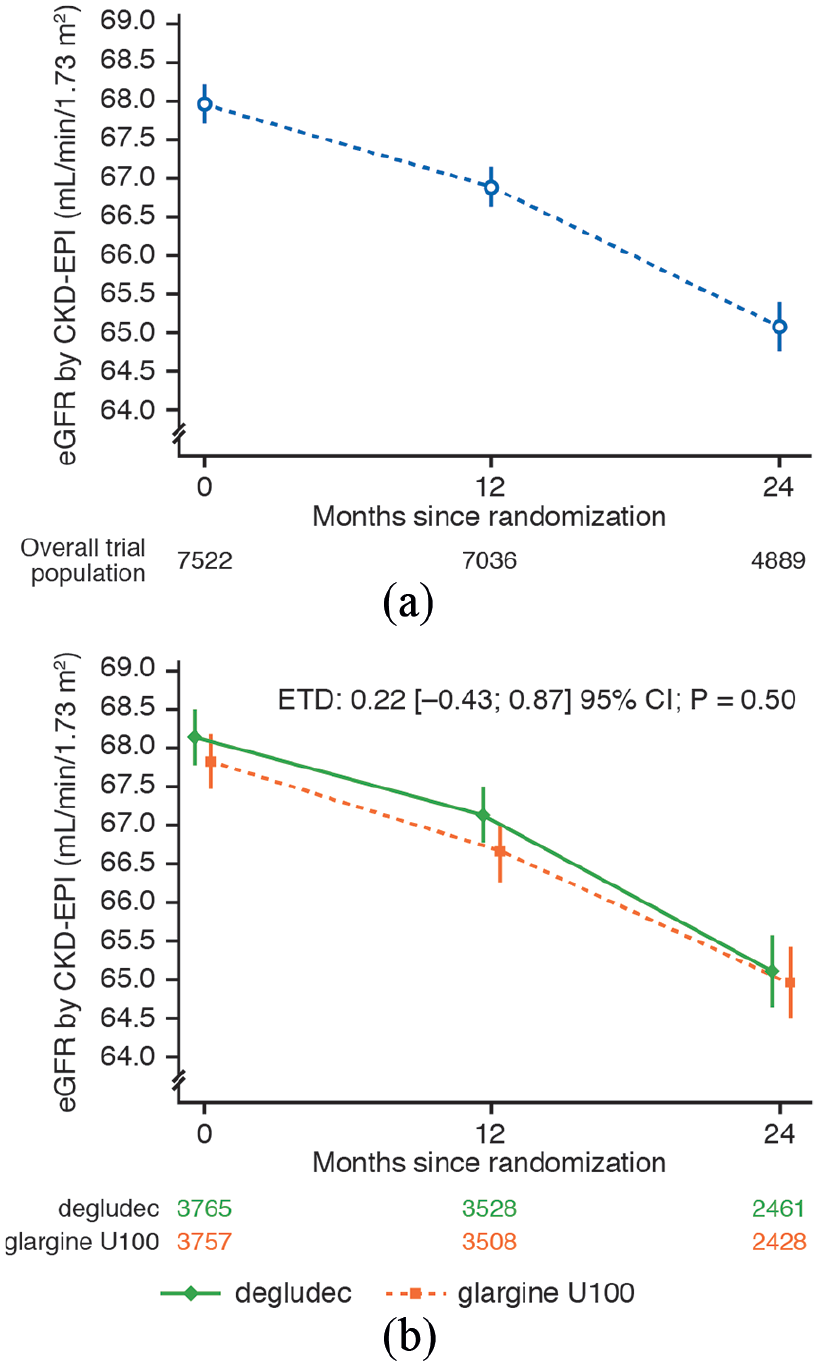
eGFR: (a) In the overall trial population, (b) by randomised treatment in patients with type 2 diabetes at high cardiovascular risk. Plot shows mean ± standard error eGFR (by CKD-EPI equation) in the overall trial population (a) and in patients randomised to degludec or glargine U100 (b) in DEVOTE. ETD (degludec/glargine U100) for change in eGFR from baseline to the 24-month visit was analysed using a MMRM within patients using an unstructured covariance matrix among visits at 12 and 24 months of the trial. Interactions between visit and treatment and between visit and baseline eGFR were included as fixed effects.
Baseline demographic and clinical predictors of change in eGFR in patients with T2D at high CV risk.
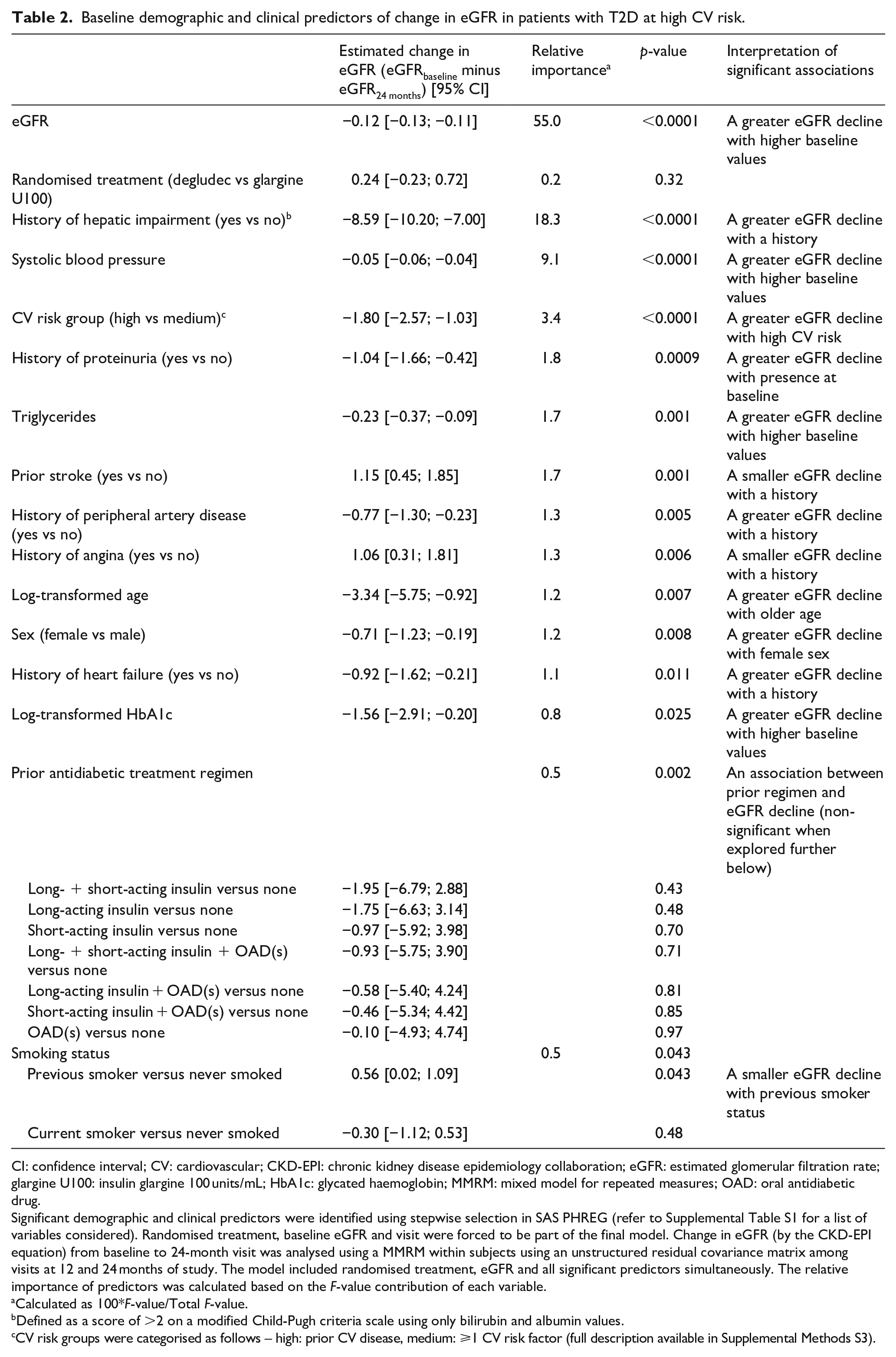
CI: confidence interval; CV: cardiovascular; CKD-EPI: chronic kidney disease epidemiology collaboration; eGFR: estimated glomerular filtration rate; glargine U100: insulin glargine 100 units/mL; HbA1c: glycated haemoglobin; MMRM: mixed model for repeated measures; OAD: oral antidiabetic drug.
Significant demographic and clinical predictors were identified using stepwise selection in SAS PHREG (refer to Supplemental Table S1 for a list of variables considered). Randomised treatment, baseline eGFR and visit were forced to be part of the final model. Change in eGFR (by the CKD-EPI equation) from baseline to 24-month visit was analysed using a MMRM within subjects using an unstructured residual covariance matrix among visits at 12 and 24 months of study. The model included randomised treatment, eGFR and all significant predictors simultaneously. The relative importance of predictors was calculated based on the F-value contribution of each variable.
Calculated as 100*F-value/Total F-value.
Defined as a score of >2 on a modified Child-Pugh criteria scale using only bilirubin and albumin values.
CV risk groups were categorised as follows – high: prior CV disease, medium: ⩾1 CV risk factor (full description available in Supplemental Methods S3).
Bidirectional temporal associations between severe hypoglycaemia and kidney disorders
Experiencing severe hypoglycaemia at any time during the trial was a significant predictor of subsequent first kidney disorders (CKD and AKI; both p < 0.05) (Table 3). In the 7 days following a severe hypoglycaemic event, patients were at a higher risk of first CKD (HR 23.0 [95% CI 5.7; 93.6] p < 0.0001) and first AKI (HR 16.3 [95% CI 5.2; 51.1] p < 0.0001) than those without severe hypoglycaemia. Thereafter, between 8 days and the end of the trial, patients were at a higher risk of first AKI following a severe hypoglycaemic episode than patients without severe hypoglycaemia (HR 2.3 [95% CI 1.5; 3.6] p = 0.0002), while the risk of first CKD was no longer significantly higher after day 8 (HR 1.9 [95% CI 0.9; 4.0] p = 0.079). When adjusting for baseline variables associated with general frailty in this population (e.g. age, CV risk, diabetes duration and baseline eGFR), temporal associations were modified, but significantly higher risks were still apparent following a severe hypoglycaemic event (Supplemental Table S4). The converse temporal association between first kidney disorders and subsequent severe hypoglycaemic events were also significant (Supplemental Table S5). There were significantly higher risks of severe hypoglycaemia after experiencing first CKD or AKI, particularly in the 7 days following a first kidney disorder (CKD HR 15.6 [95% CI 2.2; 110.8] p = 0.006; AKI HR 8.0 [95% CI 1.1; 57.1] p = 0.038).
Temporal association between severe hypoglycaemia and time to first kidney disorder in DEVOTE.
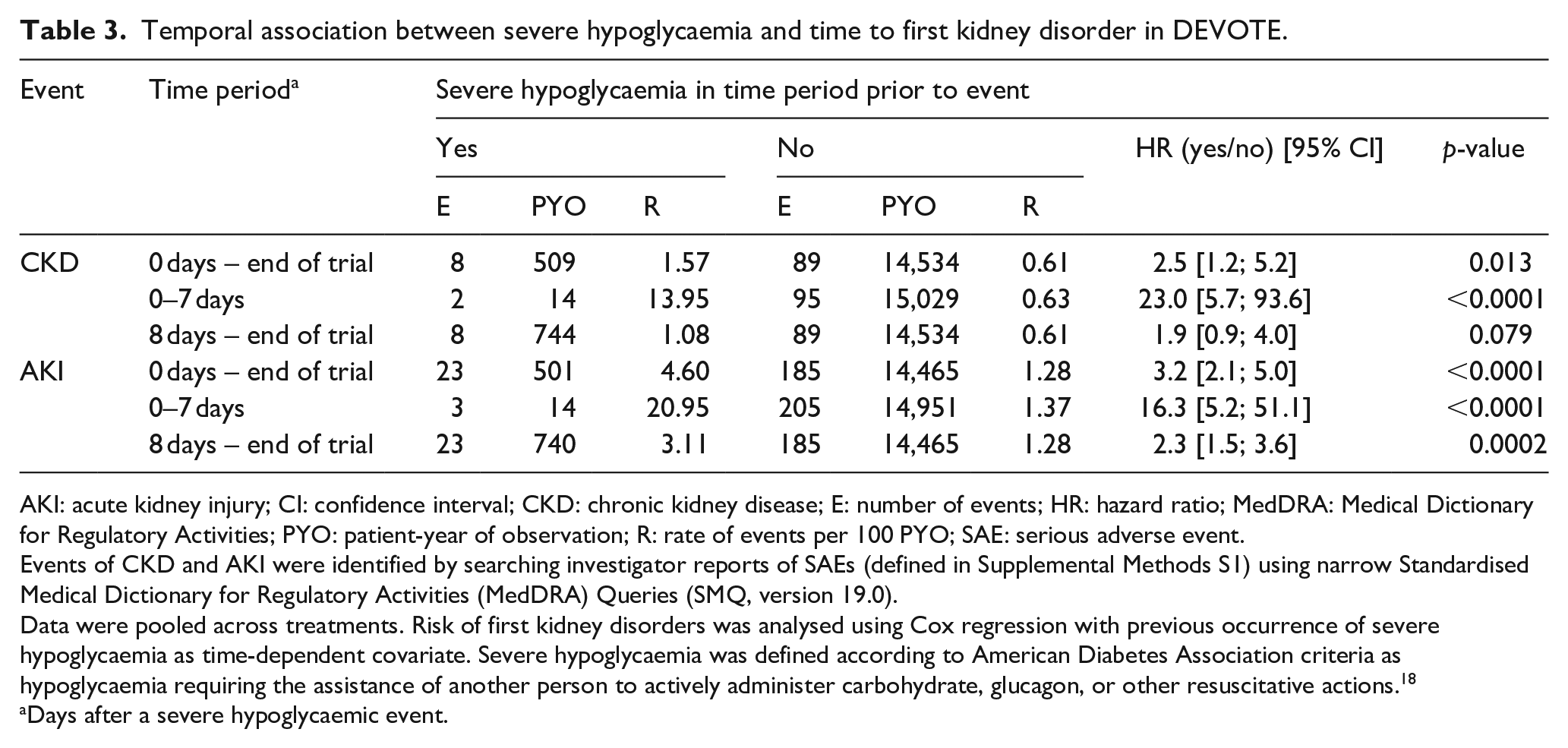
AKI: acute kidney injury; CI: confidence interval; CKD: chronic kidney disease; E: number of events; HR: hazard ratio; MedDRA: Medical Dictionary for Regulatory Activities; PYO: patient-year of observation; R: rate of events per 100 PYO; SAE: serious adverse event.
Events of CKD and AKI were identified by searching investigator reports of SAEs (defined in Supplemental Methods S1) using narrow Standardised Medical Dictionary for Regulatory Activities (MedDRA) Queries (SMQ, version 19.0).
Data were pooled across treatments. Risk of first kidney disorders was analysed using Cox regression with previous occurrence of severe hypoglycaemia as time-dependent covariate. Severe hypoglycaemia was defined according to American Diabetes Association criteria as hypoglycaemia requiring the assistance of another person to actively administer carbohydrate, glucagon, or other resuscitative actions. 18
Days after a severe hypoglycaemic event.
Discussion
These exploratory post hoc analyses of DEVOTE data identified a history of heart failure as a shared risk factor for CKD and AKI, while both a history of hepatic impairment and raised systolic BP predicted CKD risk and eGFR decline. Furthermore, our findings indicate substantially higher risks of first CKD and AKI following a severe hypoglycaemic event and vice versa, that is, there were also markedly higher risks of severe hypoglycaemia following a first kidney disorder event.
There were modest declines in eGFR over 2 years in DEVOTE that were smaller than expected in this high-risk patient population in a clinical setting (e.g. eGFR declined twice as fast in the community-based Atherosclerosis Risk in Communities [ARIC] study). 20 This may reflect the well-controlled and closely monitored clinical trial setting, but also supports the maintenance of eGFR over 2 years in the DEVOTE patient population using long-acting basal insulin analogues in addition to standard of care. Of course, there may be other differences between the DEVOTE and ARIC study cohorts that are associated with the differential rates of eGFR decline such as HbA1c, BP or pharmacotherapies. 21
Most demographic or clinical parameters found to be associated with the risk of first kidney disorders or eGFR decline in DEVOTE were consistent with the literature on the progression of kidney disease. 22 Baseline eGFR was the strongest predictor of CKD and AKI, while data-driven selection of the log-transformed variable indicated that risk increased exponentially with poorer baseline rates. By contrast, lower baseline eGFR was associated with a smaller decline during the trial (i.e. a smaller absolute change), which may be partly due to regression toward the mean. Another strong predictor of kidney disorders in DEVOTE was a history of heart failure, which was associated with a two-fold higher risk of both first CKD and AKI. This is not surprising as heart failure and CKD share several common risk factors (e.g. age, diabetes, hypertension, obesity, dyslipidaemia and smoking) and often coexist. 23 The pathophysiology between the heart and the kidneys is complex, bidirectional and currently not fully understood. There are several postulated mechanisms to explain this bidirectional association, including hemodynamic changes leading to increased renal venous congestion and poor forward flow, which may compromise GFR, and adverse effects of some therapies (e.g. diuretics) that may cause AKI and subsequent CKD progression. Several other hormonal or inflammatory pathways have also been implicated. 23 A history of hepatic impairment at baseline was associated with a three-fold higher first CKD risk and was also associated with eGFR decline, in agreement with the complex inter-relationship between the liver and kidneys described in the literature, 24 although it was not associated with an increased risk of first AKI in our study.
Patients with more advanced T2D were at greater risk of first kidney disorders, as a long diabetes duration and more complex prior insulin regimens were associated with first CKD and AKI risk, respectively. Likewise, a history of amputation was associated with first CKD risk; this might be considered an indicator of T2D progression, concomitant renovascular disease, smoking history and/or poor overall management. Notably, baseline glycaemic control was not identified as an important predictor of first AKI risk and was only a weak predictor of change in eGFR or first CKD risk in the present analyses that considered the influence of numerous demographic and clinical variables simultaneously. Baseline triglyceride levels were not identified as an important predictor of first CKD or AKI risk, and only a weak predictor of eGFR decline, contrasting with the findings of a retrospective database study of German patients with T2D. 25 A history of stroke or angina was associated with a smaller eGFR decline during the trial (i.e. a smaller absolute change). While CKD is a major risk factor for coronary artery disease and reduced eGFR is associated with a higher risk of stroke (approximately 40% higher), less is known about the potential for the reverse associations.26,2,27 This finding may be stochastic or it is possible that there were associations between prior stroke or angina with impaired kidney function on entry to DEVOTE, with less potential for absolute change in eGFR during the trial as a consequence.
Patients from the US were at higher risk of developing first AKI than those from other countries. While the factors underlying this association are unclear and may be stochastic, there are other lifestyle factors (e.g. diet or activity levels), lower compliance, or concomitant medications (e.g. diuretics or nonsteroidal anti-inflammatory drugs) that may not have been captured in our analysis, but may have increased the risk of first AKI in US patients. In contrast with previous reports, 28 there was a lower first CKD risk in older patients in our analyses. This is likely to be an artefact reflecting the DEVOTE eligibility criteria whereby patients aged ⩾50 years were required to have ⩾1 coexisting CV or kidney condition, but those aged ⩾60 years only had to have ⩾1 CV risk factor. 14 We report a 27% lower risk of first AKI with degludec versus glargine U100. While the mechanism for this is unclear, previous studies have reported associations between sustained glycaemic variability and kidney dysfunction in Asian patients with diabetes.29,30 Degludec has a more stable pharmacological profile and lower hypoglycaemia risk than glargine U100,14,31,32 but whether this contributed to the lower AKI risk observed in the present study is unknown and may be stochastic, while this difference may not be clinically relevant. The potential for differential risks of kidney disorders between diabetes therapies warrants consideration in future clinical trials. A number of traditional risk factors for AKI have been identified that include age, common comorbid diseases (e.g. CVD and CKD) and specific exposures (e.g. sepsis and some nephrotoxic agents), 7 although many of these traditional risk factors are not readily modifiable. Perhaps of greater interest from an interventional viewpoint are several novel risk factors for AKI that are potentially modifiable by pharmacotherapy or lifestyle changes, including obesity, anaemia and hyperglycaemia. 7 However, as to which of these are useful targets for interventions requires further exploration.
We report substantially higher risks of subsequent first kidney disorders after experiencing severe hypoglycaemia at any time during the trial. These findings are in agreement with the results of supplementary analyses that indicate a temporal association between severe hypoglycaemia and the adjudicated first nephropathy endpoint in LEADER 33 (Supplemental Table S6, data not published). Severe hypoglycaemia is a predictor of macrovascular events, adverse clinical outcomes and mortality in patients with T2D.34–36 Large randomised trials have reported adjusted HRs of 1.74 to 3.27 for associations with all-cause mortality34,35 and a meta-analysis of cohort studies reported an overall relative risk of CV outcomes of 2.05 [1.74; 2.42] 95% CI for patients with T2D experiencing at least one severe hypoglycaemic event versus those without. 37 Previous studies suggest the potential for links between hypoglycaemia and kidney outcomes.38,39 For instance, in a population-based study of T2D patients, hypoglycaemia was associated with higher risks of CKD and all-cause mortality over 10 years. 38 Moreover, AKI is a risk factor for hypoglycaemia during critical illness 40 and this risk extends beyond hospitalisation in patients with diabetes. 41
It is unclear whether there are direct pathophysiological links between severe hypoglycaemia and CV or kidney outcomes, or whether severe hypoglycaemia is primarily a marker of vulnerability. We found that significant associations persisted after adjusting for baseline variables, which are expected to partly describe variation in vulnerability among patients. However, temporal associations were bidirectional, and there were similar elevated risks of severe hypoglycaemia after experiencing either first CKD or AKI; hence, our findings provide support for a shared vulnerability in this patient population. When interpreting our findings, it is important to note that they were post hoc and reported without adjustment for multiple comparisons. Kidney disorders were not adjudicated in DEVOTE, which limits results interpretation to hypothesis generation. However, all available baseline factors were considered simultaneously and a data-driven approach employed to identify relevant predictors. Strengths of the present analyses include the double-blind, active comparator-controlled trial design, characterisation of kidney status at baseline and the independent adjudication of severe hypoglycaemia.
In summary, most predictors were consistent with the literature on the progression of kidney disease and, thus, indicate that traditional, non-modifiable risk factors for kidney disorders are relevant in patients with T2D at high CV risk. Furthermore, bidirectional temporal associations identified between severe hypoglycaemia and first kidney disorders highlight that these patients may warrant additional clinical attention, particularly shortly after the AE.
Supplemental Material
sj-pdf-1-dvr-10.1177_1479164120970933 – Supplemental material for Risk factors for kidney disorders in patients with type 2 diabetes at high cardiovascular risk: An exploratory analysis (DEVOTE 12)
Supplemental material, sj-pdf-1-dvr-10.1177_1479164120970933 for Risk factors for kidney disorders in patients with type 2 diabetes at high cardiovascular risk: An exploratory analysis (DEVOTE 12) by Aslam Amod, John B Buse, Darren K McGuire, Thomas R Pieber, Rodica Pop-Busui, Richard E Pratley, Bernard Zinman, Marco Bo Hansen, Ting Jia, Thomas Mark and Neil R Poulter in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
We thank the trial investigators, trial staff and trial participants for their participation. DEVOTE was conducted at 438 sites and the protocol was approved by the independent ethics committee or institutional review board at each site. Medical writing support was provided by Anna Campbell and editorial assistance provided by Clare Lowe, of Watermeadow Medical, an Ashfield company, part of UDG Healthcare plc, funded by Novo Nordisk. DEVOTE research activities were supported at numerous US centres by Clinical and Translational Science Awards from the National Institute of Health’s National Center for Advancing Translational Science.
Authors’ contributions
A.A. is the guarantor of this work and, as such, had full access to all the data in the study, and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors confirm that they meet the International Committee of Medical Journal Editors (ICMJE) uniform requirements for authorship and that they have contributed to the following tasks. Conception: all authors; Data analysis: T.M.; Data interpretation: all authors; Manuscript drafting and critical revision: all authors. All authors share in the final responsibility for the content of the manuscript, as well as the decision to submit it for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.A. has received personal fees related to advisory boards and lectures from Novo Nordisk, Sanofi (South Africa), AstraZeneca, Merck Sharp and Dohme Corp., Lilly South Africa, Boehringer Ingelheim, Merck Biopharma, Servier Laboratories and Novartis South Africa. J.B.B.’s contracted consulting fees (including travel support) are paid to the University of North Carolina by Adocia, AstraZeneca, Dance Biopharm, Dexcom, Eli Lilly, Fractyl, GI Dynamics, Intarcia Therapeutics, Lexicon, MannKind, Metavention, NovaTarg, Novo Nordisk, Orexigen, PhaseBio, Sanofi, Senseonics, vTv Therapeutics and Zafgen; he reports research support – in the form of grants, travel and non-financial support – that is paid to the University of North Carolina by AstraZeneca, Eli Lilly, Intarcia Therapeutics, Johnson & Johnson, Lexicon, Medtronic, NovaTarg, Novo Nordisk, Sanofi, Theracos, Tolerion and vTv Therapeutics; he is a consultant to Cirius Therapeutics Inc, CSL Behring, Mellitus Health, Neurimmune AG, Pendulum Therapeutics and Stability Health and reports travel support from all companies as well as personal consulting fees from Cirius Therapeutics Inc and CSL Behring; he holds stock/options in Mellitus Health, Pendulum Therapeutics, PhaseBio and Stability Health; and he is supported by grants from the National Institutes of Health (UL1TR002489, R01HL110380, U01DK098246, UC4DK108612, U54DK118612, R01DK119913, R01DK112939, R61HL142680, P30DK124723), JDRFI, PCORI and the American Diabetes Association. D.K.M. has received personal fees from Boehringer Ingelheim, Janssen Research and Development LLC, Sanofi US, Merck Sharp and Dohme Corp., Eli Lilly USA, Novo Nordisk, GlaxoSmithKline, AstraZeneca, Lexicon Pharmaceuticals, Eisai, Pfizer, Metavant, Applied Therapeutics, Afimmune and Esperion. T.R.P. has received research support from Novo Nordisk and AstraZeneca (paid directly to the Medical University of Graz); and personal fees as a consultant from Adocia, Arecor, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Novo Nordisk and Roche Diabetes Care. T.R.P. is also the Chief Scientific Officer of CBmed (Center for Biomarker Research in Medicine), a public-funded biomarker research company. R.P.B. has received research support to University of Michigan from AstraZeneca; is supported by grants from the National Institutes of Health (NIDDK-1-R01-DK-107956-01, UC4 DK101108); and has received personal fees for consultancy from Novo Nordisk and Bayer. R.E.P. reports grants from Hanmi Pharmaceutical Co.; grants from Janssen; consulting fees from Merck; grants, speaker fees and consulting fees from Novo Nordisk; consulting fees from Pfizer; grants from Poxel SA; grants and consulting fees from Sanofi; consulting fees from Scohia Pharma Inc.; and consulting fees from Sun Pharmaceutical Industries. All fees and honoraria for services were paid directly to AdventHealth (formerly Florida Hospital), a non-profit organisation. B.Z. has received grant support from Boehringer-Ingelheim, AstraZeneca and Novo Nordisk; and consulting fees from AstraZeneca, Boehringer-Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk and Sanofi. T.J. is a full-time employee of, and holds stock in, Novo Nordisk A/S. At the time of the study, T.M. and M.B.H. were full-time employees of, and held stock in, Novo Nordisk A/S; T.M. and M.B.H. continue to hold stock in, but are no longer employed by, Novo Nordisk A/S. N.R.P. has received personal fees from Servier, Takeda, Novo Nordisk and AstraZeneca in relation to speakers’ fees and advisory board activities (concerning diabetes mellitus); and research grants for his research group (relating to type 2 diabetes mellitus) from Diabetes UK, National Institute for Health Research Efficacy and Mechanism Evaluation (NIHR EME), Julius Clinical and the British Heart Foundation. Additionally, N.R.P. was a recipient of NIHR Senior Investigator Awards and has been supported by the NIHR Imperial Biomedical Research Centre.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial and these secondary analyses were funded by Novo Nordisk.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
