Abstract
Type 2 diabetes mellitus is a type of metabolic disorder characterized by hyperglycaemia with multiple serious complications, such as diabetic neuropathies, diabetic nephropathy, diabetic retinopathy, and diabetic foot. Platelet-derived growth factors are growth factors that regulate cell growth and division, playing a critical role in diabetes and its harmful complications. This review focused on the cellular mechanism of platelet-derived growth factors and their receptors on diabetes development. Furthermore, we raise some proper therapeutic molecular targets for the treatment of diabetes and its complications.
Keywords
Introduction
Platelet-derived growth factors (PDGFs) are growth factors that regulate cell growth and division. They are synthesized by platelets, smooth muscle cells, activated macrophages and endothelial cells upon activation. 1 Particularly, PDGFs play a significant role in blood vessel formation during embryogenesis, mesenchymal cells migration, proliferation and chemotaxis. As platelets are improperly activated and aggregated in type 2 diabetes mellitus (T2DM), PDGFs can largely affect the processes of diabetes mellitus and its complications via PKC Ө and PKCε, nuclear factor-kappa B (NF-κB), PI3K, PLCγ, Src/Smad1/Col4, JAK/STAT, PI3K/Akt/mTOR, p38 MAPK-SHP-1 and ERK/Akt pathways, especially the impairment of endothelial migration and proliferation by the inflammatory and angiogenic effects of PDGFs.2-4 This review focused on the cellular mechanism of PDGFs and their receptors on diabetes mellitus and its complications. Furthermore, we explored the potential therapeutic strategies based on the downstream signals regulating PDGF/PDGFR families.
T2DM is one of the metabolic disorders caused by impairment of insulin secretion and/or peripheral insulin resistance coupled with other risk factors such as obesity, genetic abnormality, leading to dysfunction on the utilization of glucose by the liver, skeletal muscle and adipose tissues. In 2017, around 4 million people aged from 20 to 79 died due to diabetes mellitus, and the top 3 countries with the largest number of patients are China, India and the United States, respectively. 5 In 2017, the global healthcare expenditure on diabetes between the ages of 20 and 79 was estimated to be US$727 billion. 5
The most prominent complication of T2DM is microangiopathy induced by oxidative stress and excessive production of reactive oxygen species (ROS) during hyperglycaemia, increasing the risk of cardiovascular disease by twofold to fourfold.6,7 Microangiopathy can be involved in all tissues and organs of the whole body, mainly in retina, kidneys, nerves and myocardium, causing further complications, such as diabetic neuropathies, diabetic nephropathy (DN), diabetic retinopathy (DR) and diabetic foot. Meanwhile, the non-hyperglycemic mechanism existing in hypertension and obesity also causes vascular injury. 8
Diabetic neuropathies, including distal symmetric polyneuropathy, autonomic neuropathies and atypical neuropathies, are nerve-damaging disorders due to diabetic microvascular injury. 9 The incidence of painful diabetic peripheral neuropathy (DPN) is 25% among diabetic patients. The reduced neuroprotection and angiogenesis, poor glucose control–induced oxidative stress, inflammation, smoking status and high body mass index (BMI) can exacerbate the diabetic neuropathies. 10 Sensory nerve loss and degeneration promoted by oxidative stress lead to sensation impairment. Recently, pre-diabetic neuropathy characterized by small neurofibropathy has been clinically proposed, and the pathogenesis was related to hypertension, dyslipidemia and oxidative-nitrosative stress. 11
DN, a kidney dysfunction with excessive accumulation of extracellular matrix (ECM) in the mesangial region, is mainly caused by hyperglycaemia-associated oxidative stress and ischaemia, inflammation, and abnormal renin–angiotensin–aldosterone system (RAAS).12,13 DN is traditionally diagnosed by increased albuminuria and decreased estimated glomerular filtration rate (eGFR). An increase in albumin excretion rate (AER) is thought to be correlated with microangiopathy-induced DN, while patients with T2DM can also progress to non-albuminuric DN characterized by macroangiopathy and a decrease in AER. 14 The abnormal kidney function of non-albuminuric DN is associated with hypertension, obesity or dyslipidemia and is not necessarily related to diabetes. 15
DR is a chronic microvascular degeneration and is the leading cause of retinal detachment and blindness. 16 A blood-retinal barrier is formed by endothelial cells of retina vessels, protecting nerve tissue against impairment. The retinal metabolic disorder will result in neurodegeneration and vasculopathy under improperly controlled glycaemia, blood pressure and lipid levels.16,17 DR includes non-proliferative diabetic retinopathy (non-PDR) that begins with slight degeneration of blood vessels and PDR in the late-stage characterized by fragile neovessels formation after ischaemia. The progression of DR depends on the duration, glucose levels, blood pressure, lipids levels and the presence of proteinuria. Another specific type is macular edema, a condition in which fluid in the blood continues to leak out of the retinal blood vessels. 15
Diabetic foot is attributed to vascular complications, neuropathies, opportunistic infections and metabolic abnormalities, such as hyperglycaemia, ROS generation and oxidative stress. 18 Minor injuries may be difficult to heal for a long time due to oxygen and nutrient deficiency, which eventually renders diabetic patients disabled. 19
Since T2DM and its complications are not solely related to high blood glucose levels, the diagnosis of diabetes and prediabetes based on hyperglycaemia is methodologically flawed. Instead, genetic biomarkers play an essential role in identifying individual risk factors and can help to improve drug development. 15
Characteristics of PDGF in diabetic complications
PDGFs and their receptors
PDGFs are inactive in their monomeric forms, including four polypeptide chains (PDGF-A, PDGF-B, PDGF-C and PDGF-D), while they are active by dimerization of these four polypeptides, forming homodimeric isoforms (PDGF-AA, PDGF-BB, PDGF-CC and PDGF-DD) and/or heterodimeric isoform (PDGF-AB).20,21 PDGFs apply their cellular effects by binding to tyrosine kinase transmembrane receptors (PDGFRs) which contain homodimeric and heterodimeric isoforms (PDGFR-αα, PDGFR-ββ and PDGFR-αβ) (Figure 1).21,22 PDGFRs comprise extracellular binding domains and intracellular tyrosine kinase domains. 23 PDGF-AA only binds to PDGFR-αα, while PDGF-BB binds to the others. PDGF-AB and PDGF-CC both signal to PDGFR-αα and PDGFR-αβ. Besides, PDGF-DD binds to PDGFR-ββ and PDGFR-αβ. 23 Binding to the extracellular domains leads to autophosphorylation and activation of tyrosine residues in the cytoplasm. 24 Downstream signals include Janus kinase (JAK)/signal transducer and activator of transcription (STAT), rapidly accelerated fibrosarcoma (Raf)/extracellular regulated protein kinases (ERK), phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PKB, Akt)/the mammalian target of rapamycin (mTOR), Src homology and Collagen (Shc)/Ras-mitogen-activated protein kinase (MAPK), growth factor receptor-bound protein 2 (Grb2)/Ras-MAPK, and phospholipase C-γ (PLC-γ)/protein kinase C (PKC) pathways. 25 Afterwards, a complex signalling cascade is initiated which acts as a transcription factor targeting specific PDGF genes (Figure 1). 25 PDGF is upregulated by external stimuli, such as hypoxia, hyperglycaemia thrombin and other cytokines. In addition, PDGF can be induced by autocrine stimulation of tumour cells. 26
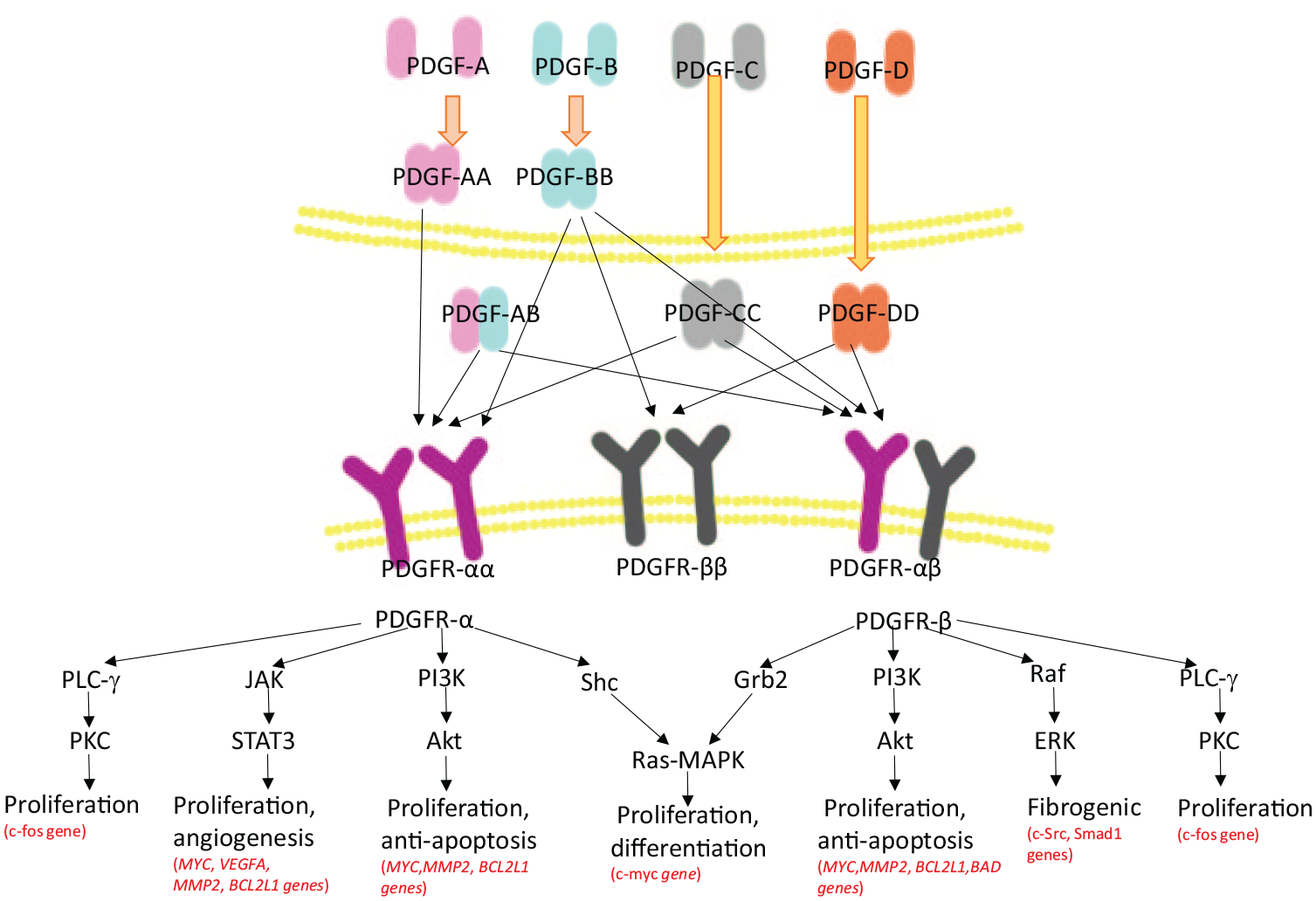
The families of PDGF/PDGFR and the downstream signalling pathways. PDGF-AA and PDGF-AB are secreted in active isoforms by cleaving their C-terminal retention motifs inside the cell. While PDGF-CC and PDGF-DD are inactive after secretion and then become active outside the cell, 27 PDGFs interact with PDGFRs and form a complex signalling cascade through several pathways. 25 Specifically, active PDGFR-α initiates PI3K/Akt/mTOR pathway, targeting at MYC, MMP2, BCL2L1 genes, which leads to increased proliferation and inhibition of apoptosis. Besides, it also activates the JAK/STAT signalling pathway, encoding MYC, VEGFA, MMP2, BCL2L1 genes, promoting proliferation and angiogenesis. Furthermore, PDGFR-α and PDGFR-β trigger the activation of the Ras-MAPK pathway by engaging Shc and Grb2, respectively. Shc and Grb2 activate the c-myc gene, stimulating proliferation, and differentiation. Activation of both PDGFR-α and PDGFR-β triggers the activation of PLC-γ/PKC pathway, accelerating cell growth via expressing the c-fos gene. Meanwhile, the interactions of PDGFR-β, Raf/ERK signalling pathways contribute to the fibrogenic process, with the increased expression of c-Src and Smad1. 25
PDGF in type 2 diabetes mellitus
PDGFs can largely affect the processes of diabetes mellitus and its complications via diverse signalling pathways. Details of this are shown in Figure 2.
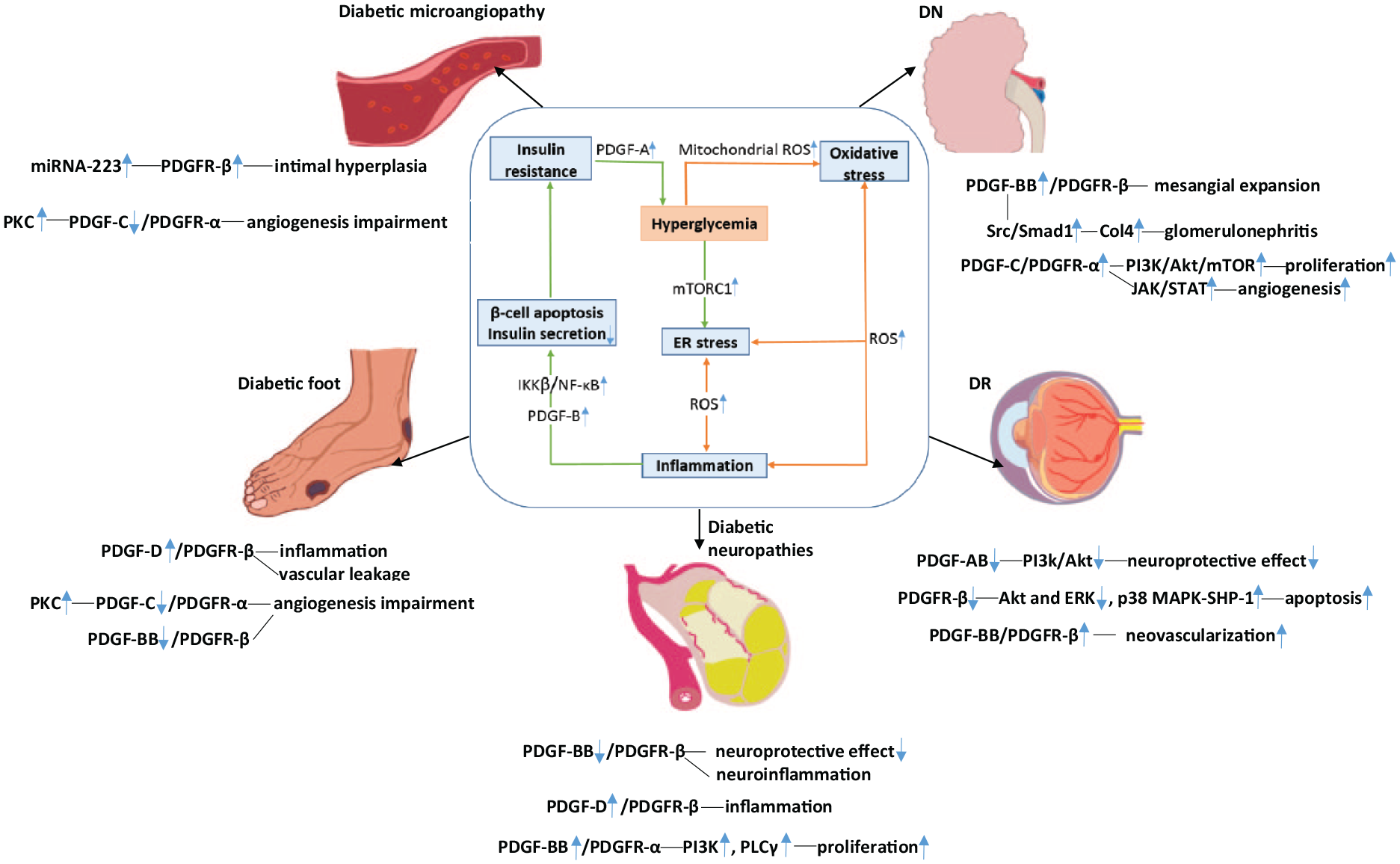
Diverse signalling pathways targeting PDGFs-related diabetes mellitus and its complications.
Glucotoxicity induces both endoplasmic reticulum (ER) stress and oxidative stress and is associated with inflammation, the main factor of impaired insulin secretion in pancreatic islets and insulin resistance in myocytes, hepatocytes and adipocytes. 28
ROS are free radicals produced during carbohydrate metabolism in the mitochondria. Excess ROS cause oxidative stress and activate pro-inflammation cytokines. 29 ER stress is promoted by glucolipotoxicity via the upregulation of the mammalian target of rapamycin complex 1 (mTORC1). Insulin resistance in muscle and adipocytes caused by ER stress is involved in the activation of IκB kinase-β (IKKβ)/NF-κB, IRE1/c-Jun N-terminal kinase (JNK), MAPK pathways and the inhibition of the PI3K pathway. 30 In obese animals with diabetes, inflammatory mediators lead to pancreatic β cell apoptosis and antioxidants decrease by activating the IKKβ/NF-κB pathway in adipose tissue, thus reducing the insulin secretion. 31
Recently, it was reported that the levels of PDGF-BB increased in gestational diabetes mellitus patients and showed a negative correlation with the β-cell-specific genes expression, leading to insulin resistance. 32 Moreover, in diabetes mellitus patients with obesity, studies have shown that inhibition of glycogen synthesis and insulin resistance was highly implicated in hyperinsulinemia with elevated PDGFA gene expression induced by hypomethylation. The growing secretion of PDGF-AA levels activates PKC Ө and PKCε, which decreases the expression of insulin receptor substrate 1 (IRS1) and the insulin receptor (INSR), thus blocking insulin signalling via inactivation of the insulin-induced Akt pathway.33,34
Diabetic vascular complications
Diabetic microangiopathy in the retina, kidneys, nerves and feet is induced by hyperglycaemia associated with activation of the polyol pathway, increased formation of advanced glycation end products (AGEs), activation of the PKC pathway and activation of the hexosamines pathway, which could contribute to the production of ROS. 7
The activated platelets can release agents that induce vascular smooth muscle cell (VSMC) proliferation and dedifferentiation to initiate repair after vascular injury. Then miRNA-223 is produced by activated VSMCs to reduce the expression of PDGFR-β, inhibiting the over intimal hyperplasia. The impaired angiogenesis potential in patients with hyperglycaemia is the biggest obstacle for promoting angiogenesis. Moriya and Ferrara 19 showed that phosphorylation of ERK and Akt induced by PDGF-C was substantially downregulated in vascular endothelial cells from diabetic mice, and the effect of PDGF-C/PDGFR-α was inhibited by increased PKC, which lead to angiogenesis impairment and impaired blood flow recovery after hyperglycaemia-induced ischaemia. Moreover, the levels of miRNA-223 are substantially decreased in patients with T2DM, leading to higher expression of PDGFR-β and excessive intimal hyperplasia of the injured vessel. 35 Excessive expression of PDGF-D results in the inactivation of NOTCH1 and NF-κB signalling, suppressing the target genes such as VEGF and MMP-9. This leads to excessive intimal hyperplasia by promoting migration and angiogenesis of endothelial cells as well as tumour cells.36,37
Hyperglycaemia is a high-risk factor of vascular remodelling, and results showed that microRNA-24 could inhibit PDGF-BB/PDGFR-β signalling pathway and reduce vascular remodelling. 38 Vasodilation is impaired by the bone morphogenic protein 4 (BMP4)-stimulated upregulation of PDGF-AA through the increased phosphorylation of p-Smad1/5. 4
Diabetic neuropathies
Expressed by many small non-cleaved cells, PDGF exhibits potentialities of neuroprotection, wound healing enhancement and calcium metabolism adjustment. 39
PDGF-BB has been shown to have neuroprotective effects on ischaemia tissue. PDGF-BB/PDGFR-α axis in the rat brain was reported to control the proliferation of oligodendrocytes through activating PI3K and PLCγ pathways. 40 In humans, the recruitment of the blood vessels, proliferation and migration of pericytes stimulated by PDGF-BB/PDGFR-β signalling pathway can reduce neuroinflammation and promote the neurogenesis after nerve damage. Meanwhile, pericytes also release the brain-derived neurotrophic factor (BDNF) and β-nerve growth factor (β-NGF), which contribute to neuroregeneration by supporting the growth and differentiation of neurons. 41 However, the neuroprotective agents are insufficient under hyperglycaemia, resulting in ischaemia and axonal degeneration in peripheral nerves. 42 Besides, brain injury–triggered PDGF-D/PDGFR-β pathway contributes to inflammation with the recruitment and infiltration of macrophages and pericytes in intracerebral haemorrhage (ICH) mice model. 43
DN
DN exhibits clinical-pathological characteristics including a thickening of glomerular basement membranes and mesangial expansion. It has been demonstrated that the PDGF-BB/PDGFR-β pathway in kidneys could induce excessive accumulation of ECM and promote the proliferation and dedifferentiation of mesangial cells and fibroblasts, leading to the progression of glomerulosclerosis. The underlying mechanism of the PDGF-BB/PDGFR-β pathway is based on the upregulation of type 4 collagen (Col4) via the activation of angiotensin II–induced Src/Smad1 pathway, contributing to the development of glomerulonephritis. 44
Langham et al. 45 reported that PDGF-BB expression was significantly increased in a renal biopsy from patients with hyperglycaemia, indicating that urinary excretion of PDGF-BB could be an early diagnosis marker for diabetic kidney disease (DKD).
Moreover, the contribution of PDGF-C/PDGFR-α can increase ECM accumulation as well. 46 The signalling pathway was illustrated to stimulate PI3K/Akt/mTOR and JAK/STAT pathways, promoting cell proliferation. Furthermore, the PI3K/Akt/mTOR pathway also inhibited cell apoptosis, and the JAK/STAT pathway promoted cell angiogenesis. 47
DR
It was observed that the expressions of PDGF-AA and PDGF-BB were increased during the development of DR in the Goto-Kakizaki rat model with T2DM. 48 Increased PDGF-BB levels, found in diabetic retinal cells, such as vascular endothelial cells, VSMCs and pericytes, can contribute to retinal angiogenesis, which is the main pathological characteristic of DR. Accordingly, anti-PDGF-BB drugs may be applied to DR treatment by reducing retinal neovascularization. 49
Interestingly, PDGF-BB can also prevent pericyte cellular apoptosis through the activation of PDGF-BB/PDGFR-β-induced Akt or ERK phosphorylation, which increases DNA synthesis under normal glucose concentration in the retina. Pericyte apoptosis in DR caused by hyperglycaemia is related to the inhibition of survival effects of PDGF-BB/PDGFR-β downstream signalling via the activation of PKC δ-p38 MAPK-SHP-1 pathway. 50 PDGF-CC, activating both PDGFR-α and PDGFR-β, was able to prevent apoptosis of retinal neurons without inducing neovascularization via increasing phosphorylation of glycogen synthase kinase 3β (GSK3β) at Ser9 and promote the production of anti-apoptotic agents.42,51 In addition, it was demonstrated that PDGF-AB showed a neuroprotective effect on retinal ganglion cells by activating the PI3k/Akt signalling pathway. 52 Whereas, the neuroprotective agents are insufficient under hyperglycaemia, resulting in ischaemia and hypoxia-induced neovascularization in the retina. 42
Diabetic foot
Patients with diabetic foot suffer low recovery rates of dermal wound healing. 53 These patients are associated with insufficient angiogenic response, lower limb ischaemia and hypoxic conditions. Up to 50% of >1 year chronic wounds show resistance to medical treatment. 54 Okonkwo et al. indicated that PDGF-BB/PDGFR-β axis promoted wound healing by producing strengthened chemotactic migration, mitosis and proliferation of inflammatory cells and fibroblasts to promote the formation of granulation tissue at wounds. Besides, neovascularization also plays a vital role in wound healing, as adequate blood supply is essential for wound healing. They also showed that vascular endothelial cells migrating to the wound under the stimulation of the PDGF-BB/PDGFR-β pathway ultimately promoted neovascularization. 55 In a collagen type I transgenic reporter mouse model, the drug imatinib, an inhibitor of the tyrosine kinase activity of PDGFR-β, was capable of inhibiting pericyte recruitment and migration by decreasing the expression of glial antigen 2, thus inhibiting the angiogenesis in wound tissue. 55
PDGF-D/PDGFR-β is also involved in the angiogenic response of wound healing by recruiting macrophages and decreasing vascular permeability. 56 Besides, PDGF-C has been reported to promote angiogenesis independent of vascular endothelial growth factor-A (VEGF-A). The underlying mechanisms still need further investigation.
Although enhanced neovascularization is critical to the repair of microangiopathy by improving blood supply, it also has adverse effects on macroangiopathy, such as accelerating plaque progression in atherosclerosis. The newly formed fragile vessels correlated with inflammation can lead to intra-plaque haemorrhage, resulting in acute coronary events. 57 Based on this, the proliferative response of PDGF-BB/PDGFR-β and PDGF-D/PDGFR-β should be inhibited in the macroangiopathy.
Therapeutic ways
Existing therapeutic plans
T2DM treatment mainly aims to maintain blood glucose levels, including dietary management, exercise, oral medications and, sometimes, insulin use via injections or pumps. Nowadays, metformin is the initial therapy in treating T2DM, which effectively decreases hepatic glucose production and increases insulin sensitivity. Besides, anti-diabetic drugs such as sulfonylureas, glinides and GLP1 analogues are effective in stimulating insulin secretion. 58 Although diabetic symptoms can be reduced, these therapies are only temporary solutions. Thus, further investigations are needed to understand the underlying mechanisms and explore new targets for the therapeutics of diabetes mellitus.
Platelets are improperly activated and aggregated in T2DM, characterized by hyperglycaemia, insufficient insulin secretion, insulin resistance, ROS overproduction, inflammation and abnormal lipid metabolism. 59 Therefore, antiplatelet drugs became more popular because they could prevent diabetic complications multifactorially by initiating the repair process, preventing excessive repair and reducing the incidence of ischaemia in wounded tissue. For example, aspirin, a non-steroidal anti-inflammatory drug (NSAID), performs the antiplatelet effect by activating cyclo-oxygenase-1 (COX-1) in platelets, causing the decreased synthesis of the powerful agonist of platelets, thromboxane A2 (TXA2). Clopidogrel and prasugrel have proven to block the P2Y12 receptor, and thereby inhibit platelet aggregation by restricting adenosine diphosphate–regulated downstream signalling. 60 However, due to the resistance of antiplatelet drugs correlated with medication non-compliance during the treatment, antiplatelet drugs are limited in clinical use. 59
Future directions
T2DM
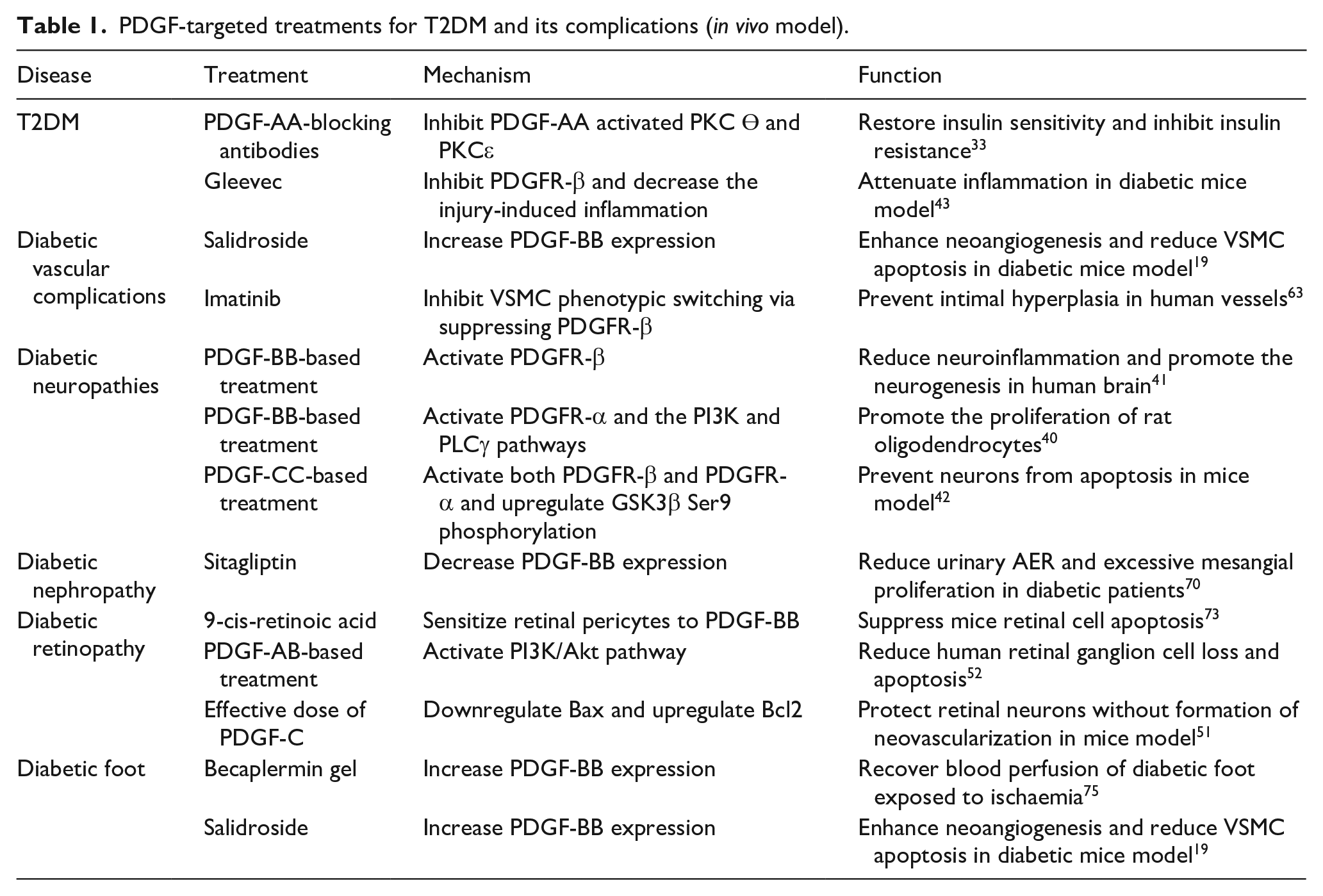
PDGF-targeted treatments (in vivo and in vitro) can be utilized as potential therapeutics for both T2DM and its complications via various downstream signalling pathways (Tables 1 and 2).
PDGF-targeted treatments for T2DM and its complications (in vivo model).
PDGF-targeted treatments for T2DM and its complications (in vitro model).

Gleevec, an anti-tumour drug, was reported to inhibit PDGFR-β with high affinity and decrease injury-induced inflammation in the ICH mice model. Thus, Gleevec could be applied to the treatment of diabetes by attenuating inflammation. 43 Chicoric acid (CA) exerted effective cardiovascular protective effects via impeding PDGF-BB-induced over intimal hyperplasia in rat models of carotid artery ligation. Meanwhile, CA and chlorogenic acid were reported to enhance immune function and exert an anti-inflammatory effect. Furthermore, they have recently been implicated in decreased glucose production via inhibition of glucose-6-phosphatase (G6P), an increase in insulin secretion through upregulation of ATP, and an increase in insulin sensitivity induced by the activation of AMP-activated protein kinase (AMPK).61,62 Abderrahmani et al. 33 demonstrated that anti-PDGF-AA agents could inhibit insulin resistance and improve insulin sensitivity.
Diabetic vascular complications
Salidroside, which could increase PDGF-BB expression, was demonstrated to enhance neoangiogenesis and reduce VSMC apoptosis, and thus recover blood perfusion in the hind limbs of diabetic mouse model. 63 PDGF-C-based therapies can be used in the treatment of diabetic cardiovascular diseases by stimulating angiogenesis and revascularization of ischemic tissues. 19 Han et al. 64 showed that PDGF-C could promote proliferation and be suppressed by retinoic acid. Thus, regulating PKC to increase PDGF-C expression may be a therapeutic way for the treatment of diabetic vascular complications.
However, aberrant neoangiogenesis could contribute to blood vessel damage. Imatinib could be used to inhibit VSMC phenotypic switching via suppressing PDGFR-β expression in the balloon-injured artery model, preventing over intimal hyperplasia. 63 Compensatory miRNA-223 after injury in diabetes mellitus reduced the risk of intimal hyperplasia by also suppressing PDGFR-β expression. 35 PDGF-D-blocking antibodies may prevent over intimal hyperplasia by reducing migration and angiogenesis of endothelial cells as well as tumour cells.
Diabetic neuropathies
Metabolic control of glucose levels is the basis of DPN therapy, while treatments should address different aspects of the pathogenesis as well, aiming at inhibiting oxidation, improving ischaemia, inactivating aldose reductase and upregulating nerve repair agent.
Kubo et al. 65 showed that PDGF-BB could indicate the neurological adverse effects of patients suffering from subarachnoid haemorrhage and neurological deficit.
PDGF-based treatments are now utilized for neuron-related diseases by suppressing neurodegeneration. 66 This work has provided evidence that PDGF-CC-based treatments were neuroprotective by upregulating of GSK3β Ser9 phosphorylation, which reduced neuron loss and apoptosis following neuron lesion in mice model. 42 Some evidence proved that an effective dose of PDGF-C was sufficient to protect retinal neurons without the formation of neovascularization. 51
DN
It was demonstrated that PDGF-BB/PDGFR-β pathway could accelerate mesangial cell and epithelial cell proliferation from the initial processing in both human and rat models. PDGF-BB/PDGFR-β exhibited a more powerful effect on the mesangial expansion of DN than PDGF-A/PDGFR-α. 67 In this case, therapy targeting PDGFR-β, such as the RAAS inhibitor, can potentially inhibit excessive proliferation. 68 PP2, an Src inhibitor, was reported to have a predictive value of the suppression of Col4 synthesis associated with the downregulation of Smad1 in vitro, which could inhibit the development of glomerulonephritis through PDGF-BB/PDGFR-β pathway. Also, small interfering RNA (siRNA) was applied to inhibit c-Src, which blocked the phosphorylation of Smad1, thereby preventing mesangial proliferation in cultured mesangial cells. 44 The potential to decrease mesangial proliferation was observed in imatinib, the inhibitor of PDGFR-β, providing the therapeutic mechanism for reversing renal microvascular damage in vitro. 69 Recently, sitagliptin, a dipeptidyl peptidase-4 (DPP-4) inhibitor, which has been widely used in the treatment of T2DM for the hypoglycemic effects, was shown to reduce urinary AER and delay the progression of DN correlated with the decrease in PDGF-BB expression. 70
DR
Measurements of PDGF-BB in the urine of diabetic patients play a critical role in the early diagnosis of diabetic retinal dysfunction, as the expressions of PDGF-BB are increased during the development of DR in the diabetic rat model. 48
Given the importance of PDGF-BB in retinal angiogenesis, Sadiq et al. demonstrated that intraocular injections of anti-PDGF-BB agents exhibited strong inhibition of angiogenesis, thus inhibiting DR progression. 71 Chen et al. 72 showed that AKT2-targeted PDGF-BB treatment could inhibit VSMCs proliferation and migration via reducing expression of miR-612, providing a potential treatment strategy for neointimal formation in DR. Besides, imatinib, an inhibitor targeting PDGFR-α and PDGFR-β, was proved to reduce retinal neovascularization in the oxygen-induced retinopathy (OIR) mouse model. 49
However, PDGF-BB is needed for the prevention of pericyte cellular apoptosis. This protective function is inhibited by the high concentration of blood glucose. This study has demonstrated that 9-cis-retinoic acid could sensitize mice retinal pericytes to PDGF-BB and thus suppress cell apoptosis, which suggested new DR therapeutics. 73 This work provided evidence that PDGF-AB-based treatments could be neuroprotective to the human retina by reducing human retinal ganglion cell loss and apoptosis following axotomy. 52 However, mesenchymal stem cells producing PDGF-AB resulted in reactive gliosis and inflammation which further developed into ablation retinae. 74
Wang et al. 51 reported that an effective dose of PDGF-C could protect retinal neurons without the formation of neovascularization by downregulation of pro-apoptotic Bcl2-associated X (Bax) protein and upregulation of anti-apoptotic B-cell CLL/lymphoma-2 (Bcl2) protein in a focal retinal degeneration mouse model.
Therefore, the therapy targeting PDGFs signalling seems to be an attractive treatment for DR via different pathophysiology. Nevertheless, the application is restricted by the interference of PDGF/PDGFR pathways for diverse therapeutic objectives.
Diabetic foot
Increasing VEGF-A and PDGF-BB secretion induces a regenerative process in tissue repair, providing a potential molecule drug for diabetes with foot ulcers. 63 In particular, PDGF-BB (becaplermin gel), approved by the United States Food and Drug Administration (FDA), is a drug to recover blood perfusion to diabetic foot exposed to ischaemia by stimulating neovascularization. 75 Salidroside, which increased PDGF-BB expression, could enhance wound healing, through elevated neovascularization, cell proliferation and epithelial formation. This resulted in thicker, more highly organized collagen fibres deposited in the wound. 63 Zhang et al. 76 found that oxygen-ozone treatment on diabetic foot ulcers showed more effective curative effects compared to standard wound dressing therapy. It also showed that the expression levels of PDGFs and VEGF were higher in the oxygen-ozone treatment group, promoting the healing of diabetic foot ulcers (Tables 1 and 2).
Nonetheless, the clinical efficiency of PDGF-BB-based treatment in diabetic foot wounds is sometimes not ideal, so the theoretical mechanisms under the pathological conditions require a more in-depth study.
Platelet-derived growth factors (PDGFs) play a critical role in blood vessel proliferation, angiogenesis, inflammation, anti-apoptosis and the fibrogenic process. Therefore, unbalanced PDGFs levels will affect the processes of diabetes mellitus and its complications. Treatments targeting PDGF/PDGFR signalling pathways might be potential therapeutic ways for diabetes mellitus and its complications.
Given the complexities of PDGF therapies used for clinical application via PKC Ө and PKCε, NF-κB, PI3K, PLCγ, Src/Smad1/Col4, JAK/STAT, PI3K/Akt/mTOR, p38 MAPK-SHP-1 and ERK/Akt pathways, the underlying mechanisms need further investigation.
Conclusion
T2DM is a chronic metabolic disorder with impaired insulin sensitivity. T2DM and its complications are largely related to the pathogenesis of microvascular disease in multiple organs, such as the eyes, the kidneys, peripheral nervous systems and feet. The underlying cellular mechanisms in vessels include hyperglycaemia, oxidative stress, inflammation, ER stress, VSMC proliferation, apoptosis, neoangiogenesis and neovascularization. 3 As PDGFs play a significant role in blood vessel proliferation, migration, angiogenesis and fibrogenic process, unbalanced PDGF levels can largely contribute to diabetes mellitus and its complications. Currently, T2DM treatment mostly relies on maintaining blood glucose levels, including dietary management, doing exercises, oral medications and insulin use. However, current therapies such as metformin are temporary solutions. Although antiplatelet drugs such as aspirin, clopidogrel, and prasugrel can inhibit the effects of blood vessel proliferation, migration and angiogenesis induced by platelets, they exert resistance effects due to medication non-compliance during the treatment. Instead, treatments based on PDGF/PDGFR through PKC Ө/PKC, NF-κB, PI3K, PLCγ, Src/Smad1/Col4, JAK/STAT, PI3K/Akt/mTOR, p38 MAPK-SHP-1 and ERK/Akt pathways may be potential therapies for complications associated with diabetes. For example, insulin sensitivity can be restored by PDGF-AA inhibitors by attenuating PKC Ө and PKCε activity. 33 PDGF-D-blocking antibodies may prevent over intimal hyperplasia by reduced inactivation of NOTCH1 and NF-κB signalling. 36 The binding of PDGF-BB to PDGFR-α in the brain promoting the proliferation of oligodendrocytes can reverse nerve injury through activating PI3K and PLCγ pathways. 40 Besides, the downregulation of the PDGF-BB/PDGFR-β pathway can inhibit the development of glomerulonephritis via the suppression of Col4 synthesis, which is associated with the downregulation of Src/Smad1 pathway in vitro. 44 Moreover, the PDGF-α signalling pathway contributes to DN due to the stimulation of both PI3K/Akt/mTOR and JAK/STAT pathways. 25 Survival effects of the PDGFR-β signalling pathway in the retina can be blunted by the inhibition of Akt or ERK phosphorylation as well as the activation of the p38 MAPK-SHP-1 pathway. 50 Furthermore, PDGF-CC exhibits a neuroprotective effect on both the brain and retina via the upregulation of GSK3β Ser9 phosphorylation. 42 However, the underlying mechanisms remain unclear. Therefore, further investigations are needed to explore and improve potential therapeutic strategies for diabetes mellitus and its complications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81700436, 81501421).
