Abstract
Background:
Advanced glycation end product is an established risk marker in diabetic vascular disease, but its possible associations with atherosclerosis in a general population are yet to be investigated. We studied the degree of carotid atherosclerosis and its association with skin autofluorescence in an elderly population.
Methods:
Carotid ultrasound and skin autofluorescence measurements were performed in a subpopulation within the ‘Malmö Diet and Cancer Cardiovascular Cohort’ re-examination study (n = 523). Total plaque area including all prevalent plaques in the right carotid artery was calculated. Complete data on all variables were available for 496 subjects (mean age 72 years).
Results:
Each 1 standard deviation increment of skin autofluorescence was associated with increased risk of prevalent large plaques (odds ratio, 1.32; 95% confidence interval, 1.05–1.66; p = 0.018) independently of diabetes and cardiovascular risk factors. The top versus bottom tertile of the skin autofluorescence was associated with an approximately twofold risk of being in the population with the highest plaque burden [top quartile with total plaque area ⩾ 35 mm2 (odds ratio, 1.88; 95% confidence interval, 1.05–3.39; p for trend = 0.027)] in fully adjusted analysis.
Conclusion:
In an elderly population, skin autofluorescence was associated with increasing degree of carotid atherosclerosis measured as total plaque area, independently of diabetes and cardiovascular risk factors.
Background
Atherosclerotic cardiovascular disease (CVD) is a major cause of mortality and morbidity worldwide. 1 Atherosclerosis in the carotid arteries has been convincingly related to coronary atherosclerosis and cerebrovascular ischemia.2,3 In addition to traditional risk scores, measuring preclinical atherosclerosis using methods such as carotid intima–media thickness (cIMT) and carotid plaque area is used to predict cardiovascular events.4–6 Advanced glycation end product (AGE) is a marker of hyperglycaemia and reflects the normal ageing process. AGEs are also increased and involved in the development of renal disease and are considered to be contributors to micro- and macrovascular complications in diabetes. 7 AGE formation is a complex process. It involves several different mechanisms, and is a result of chronic hyperglycaemia and hyperlipidaemia. 8 There are multiple forms of AGEs and many of those have a characteristic fluorescence.
Skin autofluorescence (skin AF) is a well-validated, non-invasive method of measuring the level of tissue accumulation of AGEs, usually within the skin of the forearm.9,10 Earlier studies have shown that skin AF is correlated with the degree of diabetes complications and renal failure. 11 Skin AF is also shown to be a good predictor of cardiovascular mortality in type 2 diabetes, 12 and a recent meta-analysis confirmed that skin AF levels could be considered predictors of all-cause mortality and cardiovascular mortality in patients at high risk. 13 Furthermore, in a study on type 1 diabetes patients with no known cardiovascular events, skin accumulation of autofluorescent AGEs was associated with arterial stiffness. 14 Hence, the role of AGEs in diabetic vascular disease is well established, but there is also evidence of associations of AGE and atherosclerosis independently of diabetes. A study by Den Dekker et al. 15 demonstrated increased skin AF in documented subclinical and clinical atherosclerosis, independently of known risk factors.
Thus, we aimed to investigate the association of skin AF and degree of carotid atherosclerosis in a general, elderly population.
Methods
Between 1991 and 1996, a prospective, population-based ‘The Malmö Diet and Cancer Study’ (MDC) was conducted in the city of Malmö, Sweden. Men and women, aged 44–74 years and living in Malmö, were invited to participate. Baseline examination included questionnaires, blood sample donations and anthropometrical measurements in 30,447 subjects. A subsample of the study population (n = 6103) was randomly invited into a substudy ‘The Malmö Diet and Cancer Cardiovascular Cohort’ (MDC-CC), to study cardiovascular risk factors and early atherosclerosis by performing an ultrasound examination of the right carotid artery, collecting blood samples and data on anthropometry. During 2007–2012, a new clinical re-examination was performed within the MDC-CC cohort with addition of oral glucose tolerance test (OGTT) in 3734 subjects, 16 and skin AF measurements were performed successfully in a consecutive subsample comprising 523 subjects, now aged 72.3 (±5.0). After excluding subjects with missing data on covariates included in the multivariate analysis, 496 subjects remained eligible for the study (Figure 1). A written informed consent was obtained from all subjects. All studies were approved by the Ethics Review Board at Lund University (MDC baseline ID LU-5190, MDC follow-up ID 532–2006).
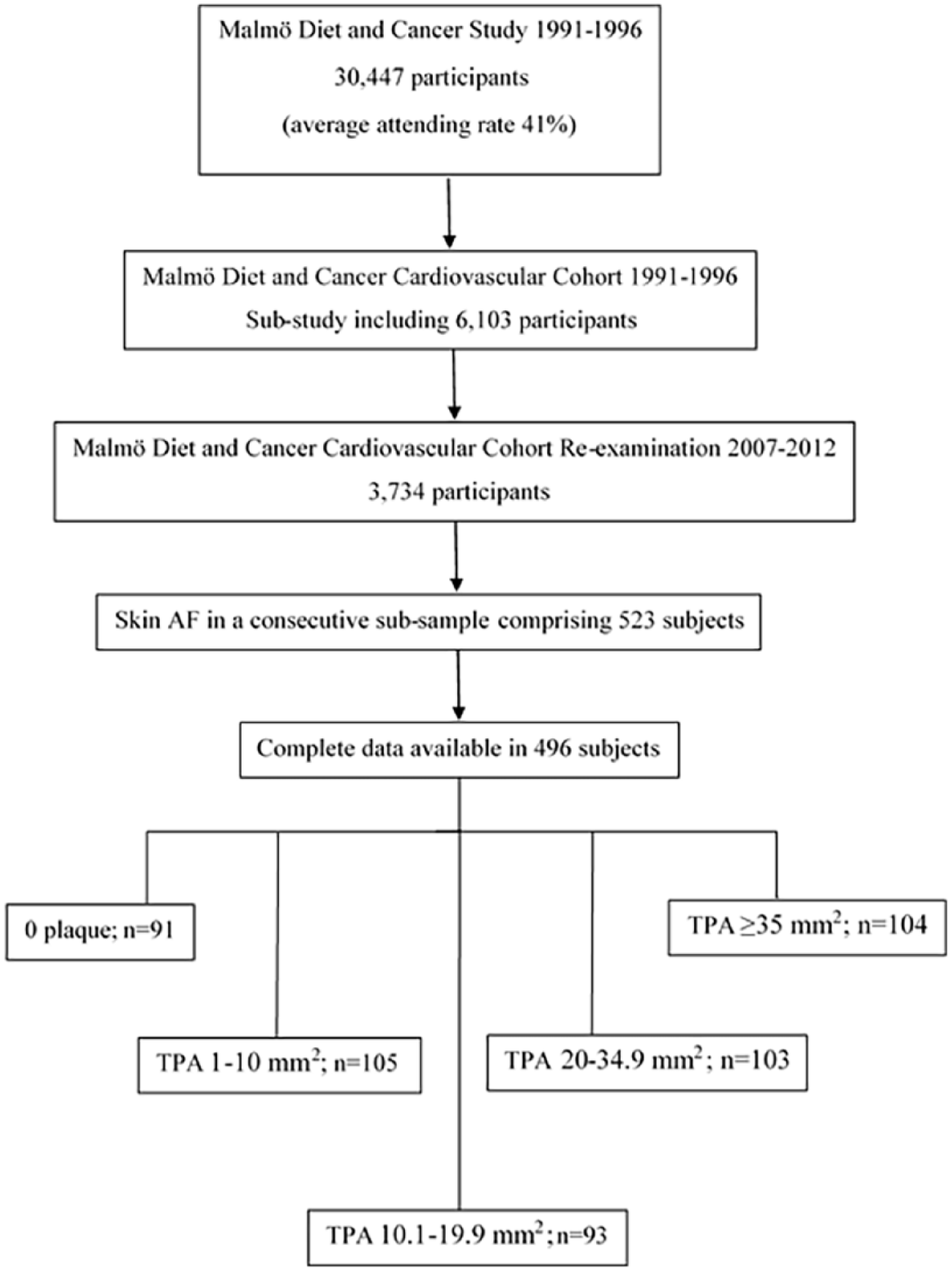
Flowchart of the selected study population.
Laboratory assays
Analyses of plasma lipids drawn after an overnight fast were carried out at the Department of Clinical Chemistry, Skåne University Hospital, Malmö, which is a part of a national standardization and quality control system. Triglycerides (TG) and total cholesterol were analysed using routine clinical methods on Cobas instruments (Roche Diagnostics Inc., Mannheim, Germany). Fasting plasma glucose (FPG) was analysed using the HemoCue Glucose System (HemoCue AB, Ängelholm, Sweden).
Clinical examination
Systolic blood pressure (SBP) was obtained from subject’s non-dominant forearm, following a 5-min rest in the supine position using an automatic device (Omron, Omron Healthcare Co, Ltd, Kyoto, Japan). Diabetes mellitus was defined as a history of physician-diagnosed diabetes, FPG of ⩾7 mmol/L, OGTT 2 h plasma glucose value of ⩾11.0 mmol/L or antidiabetic treatment at the time of the examination.
Ultrasound examination
Carotid ultrasound examinations were carried out using an Acuson Sequoia 512 (Acuson, Mountain View, CA, USA), with a 7 MHz linear array transducer. Images of all plaques within the right carotid artery were obtained. The machine settings were fixed throughout all examinations. Only overall gain and focus settings were used to optimize image quality. All sonographers were experienced. Plaques were defined as a focal thickening of intima–media greater than 1.2 mm when measured from the media–adventitia interface to the intima–lumen interface. 17 Plaque area was outlined and measured online in a longitudinal view in a projection showing the largest plaque area (intraobserver variability for offline plaque area measurements was 8.9%). Total plaque area (TPA) was calculated as a sum of all plaque areas within the right carotid artery (common carotid artery, internal carotid artery and the carotid bifurcation).
Skin AF
Skin AF was measured using an AGE Reader (DiagnOptics®, Groningen, The Netherlands) at room temperature. During measurement, a skin surface of approximately 2 cm2 is illuminated with an ultraviolet light source that ranges between 300 and 420 nm, whereas subject’s skin emits light in a different wavelength that is registered by a spectrometer. The major contribution in fluorescence comes from AGEs linked to collagen. A complete description of the measurement technique is available elsewhere. 9 An average of three readings was calculated and reported in arbitrary units (AU).
Statistical analysis
All variables that were skewed (TG, glucose and total cholesterol) were log transformed prior to analysis. TPA was modelled as the dependent variable. Subjects with prevalent plaques in the right carotid artery (n = 405) were ranked into quartiles. Plaque prevalence was scored as follows: 0 = no plaques in the right carotid artery, 1 = lowest quartile of TPA in the right carotid artery and 4 = highest quartile of TPA in the right carotid artery. The cut-off for the top quartile of TPA in subjects with prevalent plaques in the right carotid artery was used to define large plaques (n = 104) and was calculated to be ⩾35 mm2. Skin AF was normalized (Z-transformed) and entered in the model as an independent variable. Logistic regression models were used to calculate odds ratios (ORs) for TPA ⩾ 35 mm2 per 1 standard deviation (SD) increment of skin AF adjusted for Model 1 (age and sex) and Model 2 [age, sex, waist, SBP, smoking, total cholesterol, TG, FPG, antihypertensive treatment (AHT), lipid-lowering treatment and diabetes].
Furthermore, for illustration purposes, skin AF levels were divided into tertiles and related to the prevalence of large plaques (TPA ⩾ 35 mm2) after Model 1 and Model 2 adjustments, to explore if any relationship between skin AF and degree of atherosclerosis is linear.
In addition, we explored the relationships between skin AF and TPA in the right carotid artery using a linear regression model, with five categories of TPA as dependent variables. Multicollinearity diagnostics were performed for all variables. The interaction between sex and skin AF was explored using an interaction term in the logistic regression model. All analyses were performed using SPSS Windows version 22.0. A two-tailed p-value of <0.05 was considered statistically significant.
Results
Clinical characteristics of the study population are shown in Tables 1 and 2. Subjects with no plaques in the right carotid artery were assigned to Q0 (n = 91). Quartile (Q) cut-off points were based on TPA mm2 distribution and calculated as follows: Q1 (n = 105, 1–10 mm2), Q2 (n = 93; 10.1–19.9 mm2), Q3 (n = 103; 20–34.9 mm2) and Q4 (n = 104; ⩾ 35 mm2). Subjects without plaques in the right carotid artery and subjects divided into quartiles of plaque area were then combined into a quintile plaque score (Q0–Q4). The population was elderly [mean age 72.3 (±5.0)] and comprised 62.3% females (n = 309). The subjects were of mainly Swedish descent; however, no data on ethnicity were collected. A total of 34 subjects (6.9%) suffered a cardiovascular event (coronary event or stroke) prior to the re-examination study, and 10.1% had prevalent diabetes, either previously known or diagnosed within the study via OGTT values of FPG of ⩾7 mmol/L or OGTT 2 h plasma glucose value of ⩾11.0 mmol/L. The median TPA within the right carotid artery was 15 mm2.
Characteristics of the study population within tertiles of skin AF.
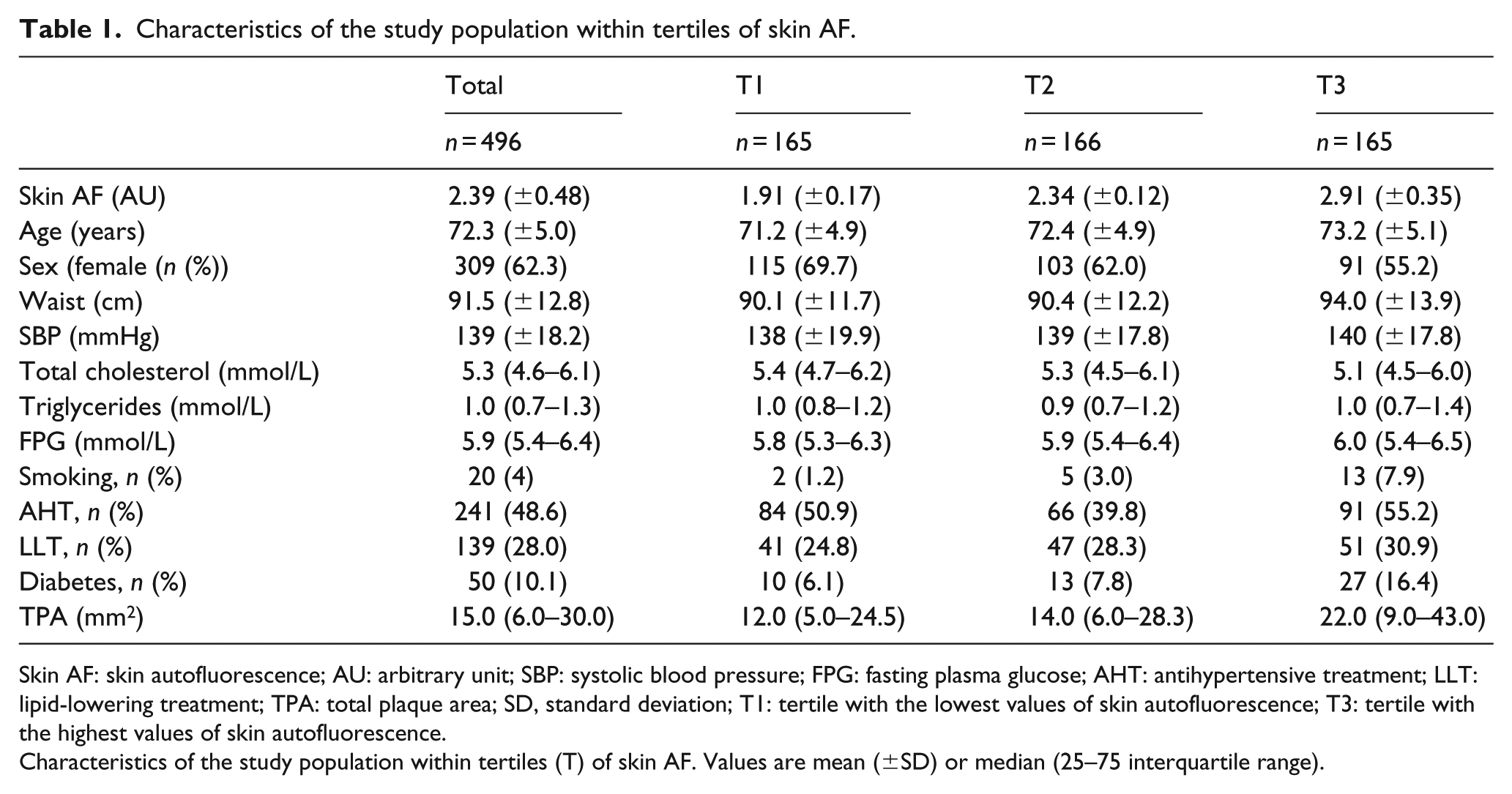
Skin AF: skin autofluorescence; AU: arbitrary unit; SBP: systolic blood pressure; FPG: fasting plasma glucose; AHT: antihypertensive treatment; LLT: lipid-lowering treatment; TPA: total plaque area; SD, standard deviation; T1: tertile with the lowest values of skin autofluorescence; T3: tertile with the highest values of skin autofluorescence.
Characteristics of the study population within tertiles (T) of skin AF. Values are mean (±SD) or median (25–75 interquartile range).
Characteristics of the study population within a plaque score based on total plaque area.
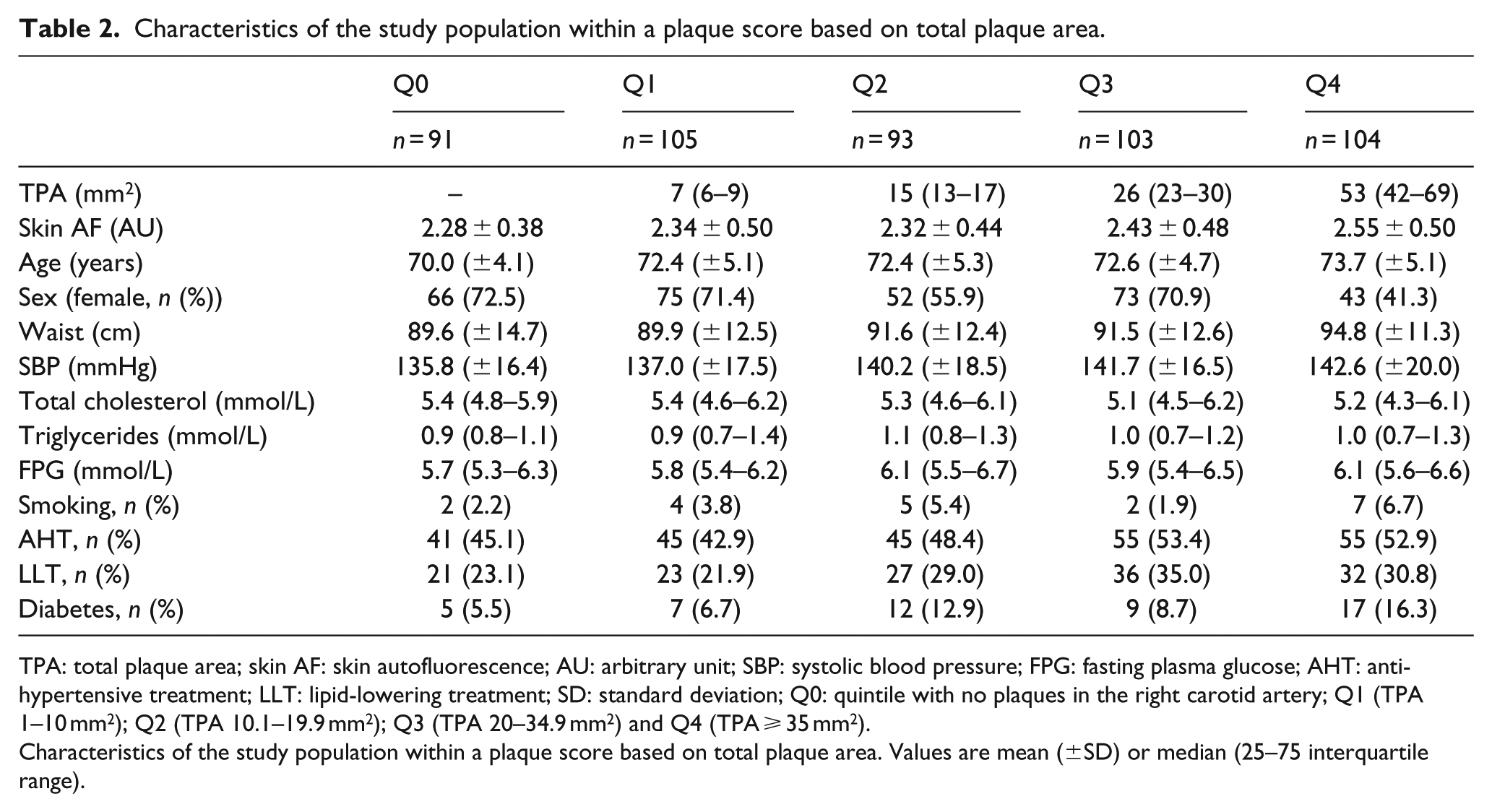
TPA: total plaque area; skin AF: skin autofluorescence; AU: arbitrary unit; SBP: systolic blood pressure; FPG: fasting plasma glucose; AHT: antihypertensive treatment; LLT: lipid-lowering treatment; SD: standard deviation; Q0: quintile with no plaques in the right carotid artery; Q1 (TPA 1–10 mm2); Q2 (TPA 10.1–19.9 mm2); Q3 (TPA 20–34.9 mm2) and Q4 (TPA ⩾ 35 mm2).
Characteristics of the study population within a plaque score based on total plaque area. Values are mean (±SD) or median (25–75 interquartile range).
Mean skin AF increased from 2.28 ± 0.38 in the group without plaque in the right carotid artery to 2.55 ± 0.50 in those with plaque area ⩾35 mm2 (Q4) (Table 2).
In a multivariable logistic regression, each 1 SD of increment in skin AF was associated with increased ORs of prevalent TPA 35 ⩾ mm2 in the right carotid artery in Model 1 adjusted for age and sex [OR = 1.93; 95% confidence interval (CI) = 1.21–3.06; p = 0.006]. The association remained significant after further adjustment according to Model 2 (OR, 1.32; 95% CI, 1.05–1.66; p = 0.018; Table 3). Further adjustment for diabetes medication (oral and insulin) on top of Model 2 resulted in no loss of significant association for skin AF and prevalence of large plaques (OR, 1.31; 95% CI, 1.04–1.66; p = 0.020).
ORs of large plaques in the right carotid artery (vs no plaque or small plaque in the right carotid artery).
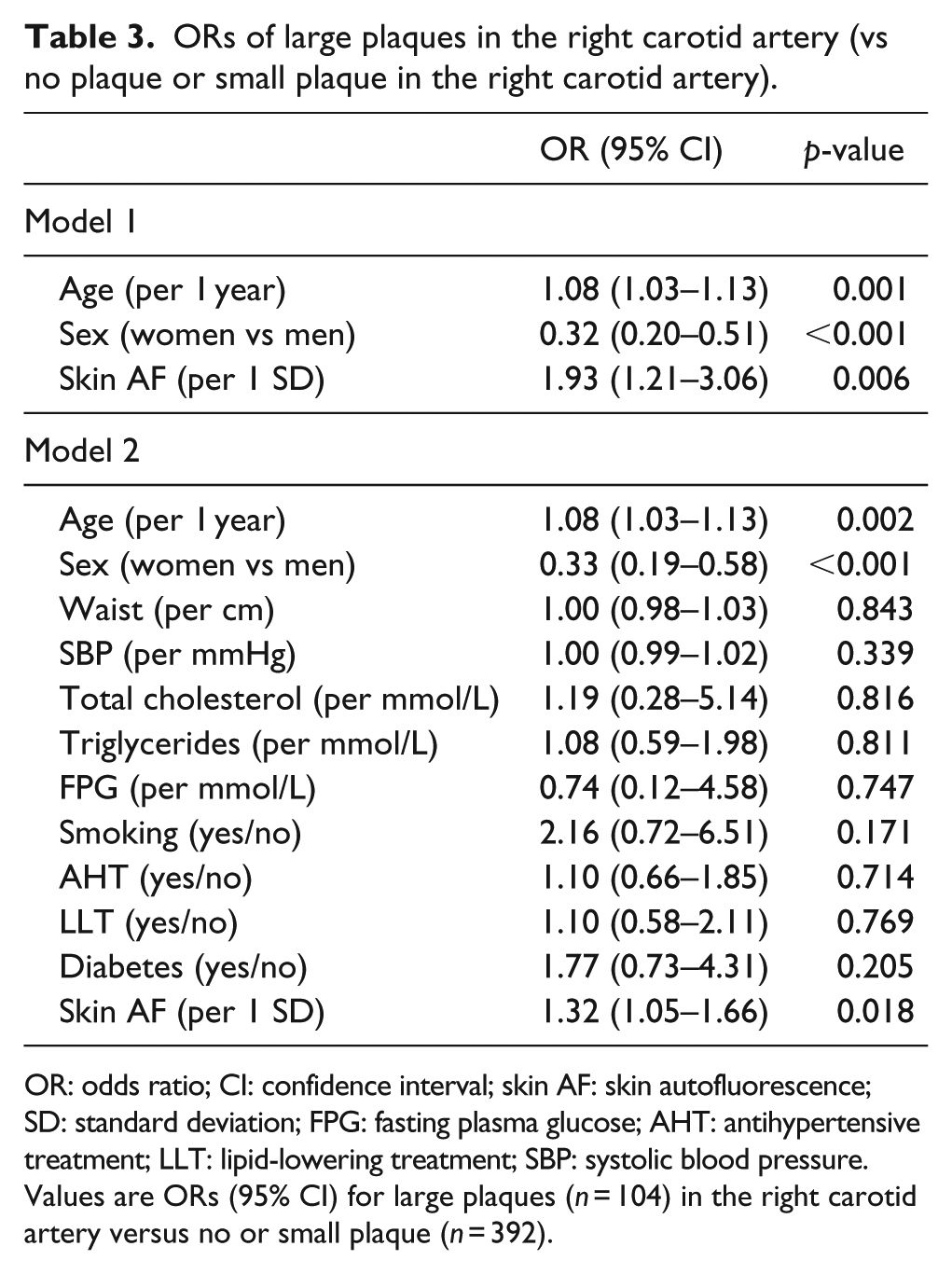
OR: odds ratio; CI: confidence interval; skin AF: skin autofluorescence; SD: standard deviation; FPG: fasting plasma glucose; AHT: antihypertensive treatment; LLT: lipid-lowering treatment; SBP: systolic blood pressure.
Values are ORs (95% CI) for large plaques (n = 104) in the right carotid artery versus no or small plaque (n = 392).
In order to examine whether the relationship between skin AF and TPA ⩾ 35 mm2 was equal across the entire distribution of skin AF, analyses of tertiles were performed (Table 4 and Figure 2). The linear trend with increased risk across tertiles was significant and revealed that the increased risk of large plaque mainly was observed in those with the highest values of skin AF (tertile 3; Table 4).
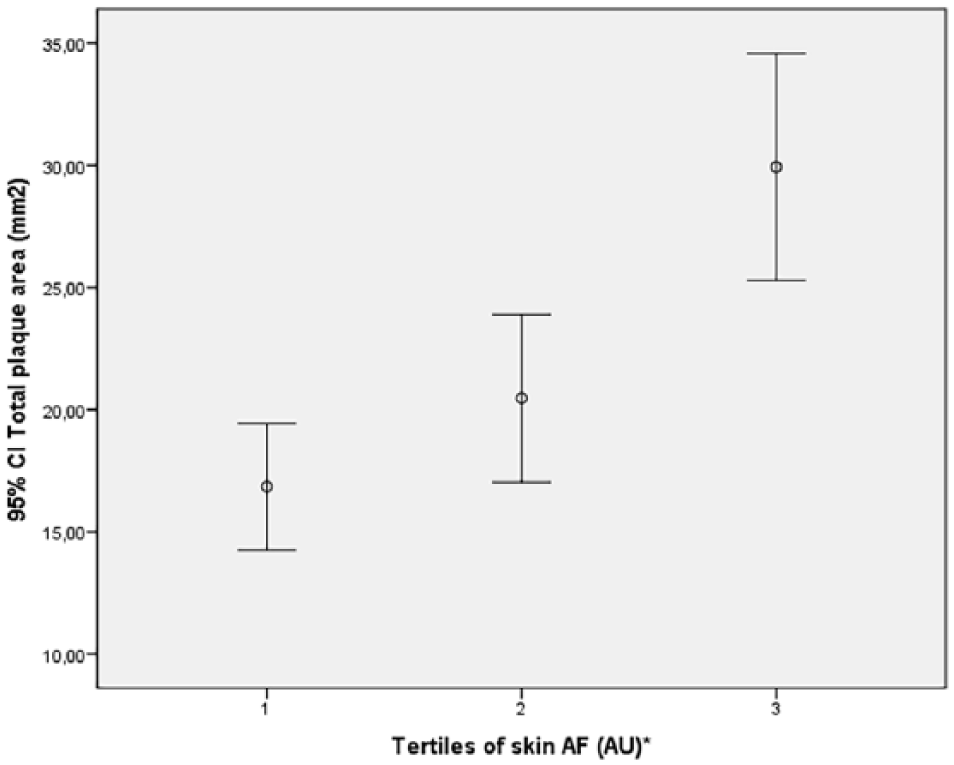
Tertiles of skin AF levels in relation to TPA in the right carotid artery.
ORs of TPA > 35 mm2 (vs small plaque or no plaque in the right carotid artery) in relation to tertiles of skin AF.

OR: odds ratio; TPA: total plaque area; skin AF: skin autofluorescence; CI: confidence interval; SD, standard deviation; TG: triglycerides; FPG: fasting plasma glucose; SBP: systolic blood pressure.
Values are ORs (95% CIs) for TPA ⩾ 35 mm2 (n = 104) versus no plaque or small plaque (n = 392) in the right carotid artery adjusted for age and sex (Model 1) and adjusted for age, sex, waist, SBP, smoking, total cholesterol, TG, FPG, antihypertensive treatment (AHT), lipid-lowering treatment (LLT) and diabetes. Values of skin AF within tertiles [mean (±SD)]: T1: 1.91 (±0.17), T2: 2.34 (±0.12) and T3: 2.92 (±0.35).
Furthermore, the association of skin AF with prevalent TPA ⩾ 35 mm2 in the right carotid artery remained significant (OR, 1.69; 95% CI, 1.02–2.82; p = 0.044) in sensitivity analyses excluding subjects with diabetes (n = 50) in fully adjusted model.
We also explored the relationships between skin AF and plaque area using a linear regression model, with categories of TPA (0–4) as dependent variables. Skin AF was significantly associated with increasing degree of TPA after adjustments in model 2 (β = 0.342; p = 0.010; Supplementary Table 1). The associations remained significant after further adjustment for diabetes medication on top of Model 2 (β = 0.328; p = 0.012).
There were no problems with collinearity between variables; variance inflation factor for skin AF was 1.091 in the full model (Model 2). Interaction analyses with respect to sex have been performed and were not significant (p = 0.588).
Discussion
The key finding of this study is that each 1 SD of increment in skin AF was associated with increased atherosclerotic burden measured as TPA in the right carotid artery independently of diabetes, as well as metabolic and CVD risk factors, in a general, elderly population. In addition, tertile analyses revealed that the most pronounced difference in risk burden for large plaques in the right carotid artery was due to the excess risk in the tertile with highest skin AF values.
TPA has been shown to be a good predictor of future cardiovascular events.5,6 In this study, tertile distribution of skin AF showed to be increasing with increasing TPA in the right carotid artery. These findings are in line with previous findings where AGEs are associated with atherosclerosis independently of diabetes and other risk factors. 15
AGEs
AGEs accelerate the oxidative damage to different types of cells by altering protein structure and function, mainly through activation of the receptor for AGE (RAGE). The oxidative stress that cells are exposed to during AGE binding to RAGE is believed to be a trigger in plaque formation. 18 There are also exogenous sources of AGEs such as tobacco smoking and food digestion. By adjusting for smoking, we tried to eliminate the confounding effects of exogenous AGE sources.
Several studies have shown that circulating levels of AGE are associated with cardiovascular events in high-risk subjects.19,20 The prevalence of AGE in atherosclerotic lesions in subjects with diabetes is previously established. 21 Hanssen et al. 22 demonstrated that higher levels of AGEs within human carotid plaques are associated with rupture-prone plaque phenotype independently of diabetes and glucose levels.
Skin AF
Over the recent decade, evidence of associations between skin AF and atherosclerosis have been accumulating. Skin AF has been shown to be increased in subjects with prevalent CVD and is also associated with future cardiovascular events.23,24 In addition, skin AF levels were recently shown to independently predict plaque instability such as plaque rupture and calcification in patients with CVD. 25
A study performed by Lutgers et al. 26 in a population without diabetes and clinically manifest CVD, where cIMT was positively related to skin AF in 59 subjects, proposed that increased skin AF may represent subclinical atherosclerosis. Furthermore, Den Dekker et al. 15 performed a study in subjects with documented subclinical and clinical atherosclerosis and found skin AF to be positively correlated with atherosclerosis measured as IMT in carotid and femoral arteries and coronary calcium score independently of diabetes. Despite the fact that associations found by Den Dekker et al. and within our study assess the same disease, namely atherosclerosis, cIMT and plaque area have different patterns of risk factors and different natural history and reflect the disease differently. While cIMT is mainly related to hypertension, 6 carotid plaque represents intimal thickening with lipid core, macrophages, foam cells, smooth muscle cells and a fibrous cap – depending on the degree of plaque progression. In addition, several observational studies have demonstrated the predictive power of presence of carotid plaque.27,28 Furthermore, the study by Den Dekker reports skin AF measurements in a younger population (mean age for controls 48 years, subjects with subclinical atherosclerosis 52 years and subjects with clinical atherosclerosis 64 years) than the data presented here. Since AGEs accumulate with age, and skin AF is a measurement of AGEs binding to long-lived proteins (15 years for collagen), 29 we believe that this study substantiates the knowledge of AGE accumulation and its associations with atherosclerosis in elderly subjects.
Consequently, there is reason to believe that accumulation of AGEs plays a significant role in inflammatory processes that result in CVD and hence skin AF may be an additional tool in risk assessment.
Strengths and limitations
Our study has both strengths and limitations. By studying a general, elderly population and adjusting for metabolic risk factors and diabetes, we believe that we illustrated that skin AF may have a role in, or reflect, atherosclerosis, beyond factors included in the metabolic syndrome and diabetes. By additional adjustment for smoking, AHT and lipid-lowering treatment, all of those being factors that are involved in, or reflect, either atherosclerosis development or AGE formation, we tried to eliminate other possible confounding factors. However, since atherosclerosis is a multifactorial disease, caution should be taken when drawing conclusions about associations. One limitation of this study is the fact that only the right carotid artery was examined. Examining several vascular sites would obviously improve the accuracy of the calculated plaque burden.
Furthermore, all ultrasound examinations and subsequent measurements of plaque area or cIMT are vitiated by subjectivity, resulting in interobserver variability. Unfortunately, we do not have data on interobserver variability for online measurements of TPA, but the same sonographers that did the online measurements tested their interobserver variability for offline measurements of plaque area with interobserver variability of 8.9%. In addition, all sonographers performing ultrasound examinations have continuous discussions regarding plaque length and area outlining, thereby minimizing the differences in area measurements.
As for skin AF measurements, those are affected by skin pigmentation. Unfortunately, we did not have data on participant ethnicity. However, we used a later version of the AGE-reader software (2.3) that is developed with an adjusted algorithm that allows usage within a broader group of subjects with darker skin colour. 30
Another limitation is that this study population consisted of mostly elderly subjects within a narrow age range (72.3 ± 5.1 years). It is not unlikely that subjects who had high accumulated AGE levels and high TPA at the baseline examination (1991–1996) deceased before the follow-up examination. However, this would reduce the observed associations, if anything. It is also possible that subjects who by the time of the re-examination were seriously ill chose not to participate in the MDC-CC. As this is a cross-sectional study, causality cannot be proven, a problem in common to all cross-sectional studies. Finally, the study was undertaken in individuals of mainly Swedish (Caucasian) descent, and the conclusions may not be generalizable to all ancestries.
Conclusion
In an elderly population, skin AF as a measure of AGE accumulation was independently associated with increased degree of atherosclerosis within the right carotid artery measured as TPA. Prospective studies examining the relation of AGE and atherosclerosis beyond diabetes are indeed warranted.
Supplemental Material
Supplementary_table_1(DVR) – Supplemental material for Skin autofluorescence as a measure of advanced glycation end product levels is associated with carotid atherosclerotic plaque burden in an elderly population
Supplemental material, Supplementary_table_1(DVR) for Skin autofluorescence as a measure of advanced glycation end product levels is associated with carotid atherosclerotic plaque burden in an elderly population by Amra Jujić, Gerd Östling, Margaretha Persson, Gunnar Engström, Peter M Nilsson, Olle Melander and Martin Magnusson in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank Cecilia Kennbäck for technical assistance during the acquisition of data.
Author contributions
A.J. and G.Ö. have participated in acquisition, analysis and interpretation of data and drafting the article. M.P., G.E., P.M.N. and O.M. have participated in acquisition of data, drafting the article and a critical revision. O.M. and M.M. have participated in the analysis and interpretation of data, drafting the article and a critical revision. All authors have given a final approval of the version to be submitted.
Availability of data and material
The data that support the findings of this study are available upon request from the data access group of Malmö Diet and Cancer study by contacting data manager Anders Dahlin (
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.M. and O.M. were supported by grants from the Swedish Medical Research Council, the Swedish Heart and Lung Foundation, the Medical Faculty of Lund University, Skåne University Hospital, the Albert Pahlsson Research Foundation, the Crafoord Foundation, the Ernhold Lundströms Research Foundation, the Region Skane, the Hulda and Conrad Mossfelt Foundation, the Southwest Skanes Diabetes Foundation, the King Gustaf V and Queen Victoria Foundation, the Lennart Hansson Memorial Fund, Knut and Alice Wallenberg Foundation and the Marianne and Marcus Wallenberg Foundation. P.M.N. was funded by Research Council of Sweden (K2008-65X-20752-01-3 and K2011-65X-20752-04-6), E. Lundström Foundation and ALF from Region Skåne. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. M.M. was also supported by the Kockska foundation and the Wallenberg Center for Molecular Medicine, Lund University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
