Abstract
Objective
Accumulation of advanced glycation end products (AGEs) occurs during normal aging but markedly accelerates in people with diabetes. AGEs may play a role in various age-related disorders. Several studies have demonstrated that skin autofluorescence (SAF) reflects accumulated tissue levels of AGEs. However, very few studies have investigated SAF in the general population. The purpose of the present study was to more thoroughly evaluate the potential association among SAF, chronological age, and lifestyle habits in the general population.
Methods
A large cross-sectional survey of 10,946 Japanese volunteers aged 20 to 79 years was conducted. Volunteers completed a self-administered questionnaire and underwent SAF measurement on their dominant forearms. The associations of SAF with age and lifestyle habits were analyzed using a multiple stepwise regression analysis.
Results
Age was independently correlated with SAF. Lifestyle habits such as physical activity, nonsmoking, adequate sleep, low mental stress level, eating breakfast, and abstaining from sugary food were each independently associated with lower SAF.
Conclusions
SAF was associated with age and healthy lifestyle habits in this general Japanese population. The present study suggests that SAF measurement is a convenient tool for evaluating habitual lifestyle behaviors and may have potential for preventative health education.
Keywords
Introduction
Advanced glycation end products (AGEs) are formed by nonenzymatic reactions between sugars and amino groups of macromolecules, such as proteins, lipids, and nucleic acids.1–3 Because glycation occurs constantly during normal aging and the rate of degradation of glycated macromolecule derivatives is very slow, AGEs accumulate with increasing age.1–3
The pathogenicity of AGEs involves their cross-linking ability, which alters the functions and tertiary structures of proteins and lipids, as well as their proinflammatory interactions with AGE cellular receptors. Accumulating evidence shows that AGEs play important roles in the development of age-related disorders including diabetic vascular complications, osteoporosis, Alzheimer’s disease, cancer, and nonalcoholic steatohepatitis.3–10
Pentosidine and crossline have structural properties that cause them to emit fluorescent light across a specific range of wavelengths upon excitation by ultraviolet light.11,12 This unique characteristic has been used to develop technology that quantifies accumulated AGEs within the human skin. 13 Although several confounding factors exist, such as other fluorophores, skincare cream use, and skin pigmentation, skin autofluorescence (SAF) is a prominent biomarker that may reflect tissue accumulation of AGEs.14–17 Indeed, a positive correlation between SAF and AGE levels in human skin has been found, even with nonfluorescent AGEs.13–17 Moreover, SAF has been shown to predict future cardiovascular events and death among patients with diabetes.16,17 SAF values are also associated with the severity of chronic kidney disease and are correlated with an increased risk of death from cardiovascular disease in patients with chronic kidney disease.15,16 These observations suggest that SAF measurement is a convenient, noninvasive tool with which to evaluate the prevalence and severity of various age- and/or diabetes-related disorders.
The formation and accumulation of AGEs progress under inflammatory and oxidative stress conditions. 3 Furthermore, food- or tobacco-derived AGEs are absorbed by the human body and may have causative roles in the pathogenesis of chronic lifestyle-related diseases.18–21 Therefore, it is conceivable that lifestyle behaviors, including dietary patterns, physical activity, and amount of sleep, can affect AGE accumulation and resultant SAF values. However, very few studies have investigated SAF in the general population, with most involving a limited population size or number of lifestyle habits evaluated.22–25 Therefore, we conducted a survey among an exceptionally large number of apparently healthy adult volunteers in Japan to more thoroughly analyze the correlation among age, lifestyle habits, and SAF of the dominant volar forearm.
Methods and study design
Study population
From April to June 2013, public health education and promotion events were held in 10 urban areas in Japan (Sapporo, Sendai, Tokyo, Kanazawa, Nagoya, Osaka, Hiroshima, Fukuoka, Kagoshima, and Okinawa). During these events, participants were recruited for the present study, a questionnaire was conducted, and SAF was measured. From 2015 to 2016, additional monthly meetings that were open to the public were held in Tokyo, Japan, and walk-ins were welcomed. Apparently healthy volunteers were also recruited at these events and asked to complete a self-administered questionnaire before undergoing SAF measurement. Written informed consent was obtained from all participants. This study was approved by the research ethics committee of Kyoritsu Women’s College, Tokyo, Japan.
Questionnaire
The questionnaire comprised 12 multiple-choice questions regarding age, sex, and health-related lifestyle habits such as physical activity, smoking history, alcohol consumption, sleeping time, level of mental stress, and eating behaviors (Table 1). Each question was evaluated using a 5-point scale, where 1 was the unhealthiest behavior and 5 was the healthiest. For example, the question, “Do you eat a lot of vegetables?” was scored by the following answers: 5 = strongly agree, 4 = agree, 3 = undecided, 2 = disagree, and 1 = strongly disagree.
Questionnaire on lifestyle habits

SAF measurement
SAF was measured on the dominant volar forearm using the TruAge Scanner (Morinda Inc., American Fork, UT, USA), a consumer version of the AGE Reader mu (Diagnoptics Technologies B.V., Groningen, Netherlands), as previously described. 26 Certain types of AGEs autofluoresce when exposed to ultraviolet light.11,12 The AGE Reader illuminates skin with ultraviolet light (excitation range = 300–420 nm) and then detects the resulting fluorescent light (emission range = 420–600 nm), while simultaneously detecting light reflected from the skin in the 300- to 420-nm range. SAF, reported in arbitrary units, is defined as the ratio of the intensity of the emitted fluorescent light to that of the reflected light. 17 For comparison with SAF data from earlier studies, the AGE score from the TruAge Scanner was divided by 100 because the AGE score is simply a multiple (100×) of the SAF arbitrary unit.
Statistical analysis
Summary statistics, such as mean and standard deviation, were calculated for SAF by age group. Correlations among SAF, age, and lifestyle habits were examined by linear regression analysis in BellCurve™ for Excel (Social Survey Research Information Co., Ltd., Tokyo, Japan). Multiple stepwise regression analysis was performed to evaluate the influence of each lifestyle habit, described in Table 1, on the measured SAF values. This stepwise regression analysis was performed with the use of IBM SPSS for Windows, version 21.0.0 (IBM Corp., Armonk, NY, USA), and p < 0.05 was considered significant.
Results
In total, 10,946 apparent healthy Japanese adults (age of 20–70 years) were enrolled in the present study (male, n = 2493; female, n = 8453). The numbers of volunteers, by age strata, were 810 (20–29 years), 1403 (30–39 years), 2522 (40–49 years), 2875 (50–59 years), 2247 (60–69 years), and 1089 (70–79 years). As shown in Table 2, SAF significantly increased with the volunteers’ calendar age (p < 0.001).
Comparisons of skin autofluorescence values of the volar forearm, by age group, of three population studies including the present study

Data are presented as mean ± standard deviation.
The stepwise regression analysis showed that eight independent lifestyle factors were significantly associated with SAF. Physical activity (p < 0.001), nonsmoking status (p < 0.001), adequate sleep (p < 0.005), lower mental stress (p < 0.001), eating breakfast (p < 0.001), and avoiding sugary food (p < 0.005) were correlated with lower SAF levels, whereas eating moderately and eating vegetables first were associated with higher SAF values (Table 3). Among all factors, calendar age had the strongest association (R2 = 0.92 and y = 1.38x + 132.23, where x = calendar age and y = average SAF) (Figure 1).
Multivariate analyses of associations with skin autofluorescence
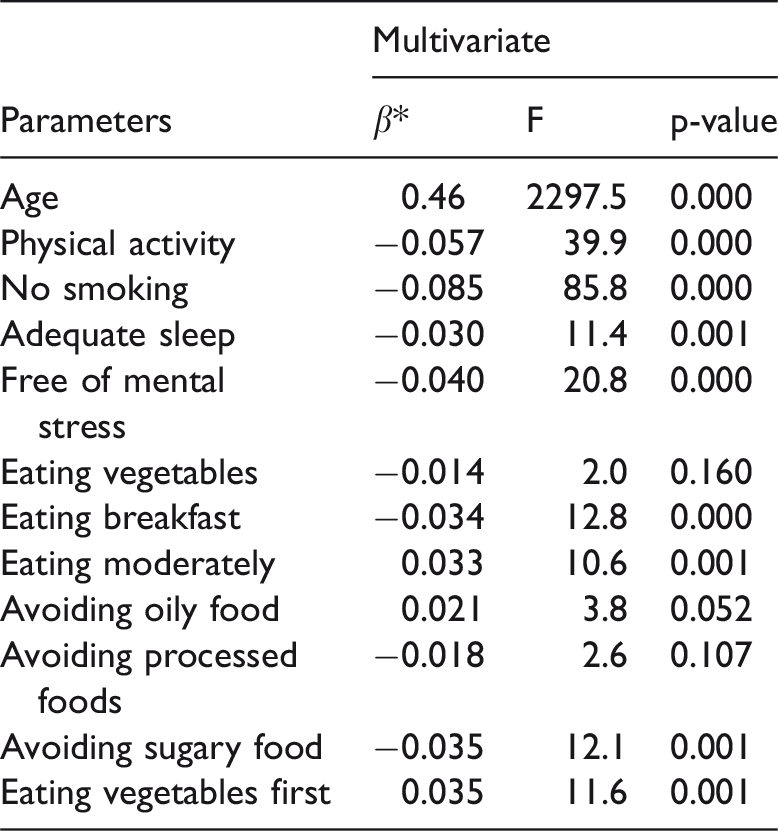
*β is the regression coefficient.
R2 = 0.20

Regression analysis of average skin autofluorescence and age, where x = age (years) and y = average SAF (arbitrary units). SAF, skin autofluorescence.
Discussion
AGE accumulation is part of the normal aging process.1–3 Indeed, several previous studies have demonstrated that calendar age is associated with increased AGE levels in humans.2,27–30 A comparison of eye lens samples from children younger than 4 years and adults aged 25 to 89 years revealed age-related increases in the AGE content of both water-soluble and alkaline-extracted fractions of lens proteins. 28 Gas chromatography–mass spectrometry analysis revealed that levels of a nonfluorescent AGE, carboxymethyl lysine (CML), and a fluorescent AGE, pentosidine, increased with age in the skin collagen of both patients with type 1 diabetes and individuals without diabetes. 29 Furthermore, while fetal and juvenile tissue samples contained no detectable CML-modified proteins, CML was present in adult tissues, where higher concentrations occurred with increased calendar age. 27 Age-related increases in CML, pentosidine, and carboxyethyl lysine were also observed in human cartilage and skin collagen samples. 30
In this study, we found that SAF increased with calendar age in apparently healthy Japanese adults. The present results extend previous findings from studies in the Netherlands and China that had relatively small numbers of subjects.22,23 However, the age-specific mean SAF values were moderately different among those studies (Table 2). Among individuals aged <60 years, the mean SAF values tended to be higher in our study than among those previously reported. However, among individuals aged 60 to 79 years, the mean SAF values of our subjects ranged between those of China and the Netherlands. Whether these differences are due to demographic and/or ethnic differences among the groups remains unclear. However, Yue et al. 23 reported that there was no statically significant difference in SAF between Dutch and Chinese populations in the 20- to 60-year age range. Thus, the middle-aged segment of the Japanese general population (40–59 years old) might have higher SAF values than the middle-aged segment of the Chinese or Dutch populations.
AGEs may reflect cumulative glycemic exposure and are less sensitive to enzymatic degradation and proteolysis. 31 Moreover, aging may reduce the expression and activities of antioxidant enzymes, such as glyoxalase, a crucial enzyme for detoxification of methylglyoxal. 32 AGEs may also suppress antioxidant enzyme expression and activity. 33 Therefore, crosstalk exists between aging and AGEs; physiological aging creates numerous conditions that stimulate the accumulation of AGEs, which then accelerate the aging process in target organs.
In the present study, we also found that SAF values were correlated with unhealthy lifestyle habits. Cigarette smoke is one of the major sources of exogenous AGEs. 34 In previous studies, SAF levels were shown to be elevated among cigarette smokers, although in a small number of subjects.22,23,35 Additionally, a smoking history may have an impact on SAF, with ex-smokers having higher levels than those who have never smoked.36,37 Consistent with these previous findings, we found that a longer smoking history was correlated with increases in SAF. Moreover, SAF was recently found to be correlated with pack-years of smoking in a large number of subjects, and smoking cessation resulted in a gradual decrease in SAF values, 21 supporting our observations.
Physical inactivity is a modifiable risk factor for obesity and diabetes, both of which can accelerate the formation and accumulation of AGEs. 38 Moreover, Japanese adult men with higher SAF had lower muscle strength and power. 39 Therefore, physical inactivity may have a pathological role in the elevation of SAF. However, very few studies have been conducted to address the causal link between physical inactivity and increased SAF values. It would be interesting to examine whether regular exercise or fitness may help control the accumulation of AGEs during the normal aging process.
Another interesting finding of the present study was the inverse association between sleeping time and SAF. In a previous study with a small number of Japanese subjects (n = 244), a trend toward increased SAF was observed among those receiving <6 hours of sleep per night, with a more significant relationship occurring in those aged 20 to 29 years. 40 Indeed, our larger sample size (n = 10,946) showed that adequate sleeping time was significantly correlated with lower SAF values, regardless of age. Sleep deprivation may evoke oxidative stress and increase the risk of obesity and diabetes,41,42 which may stimulate the formation and accumulation of AGEs. Furthermore, emotional and psychological stress not only disturbs adequate sleep but also activates the sympathetic nervous system, thereby impairing glucose metabolism. 43 This may partly explain the link between mental stress and increased SAF in our subjects.
Eating more vegetables may limit oxidative stress and AGE formation, 44 whereas high intake of processed foods rich in sugar and fats could augment the accumulation of AGEs. 20 Consumption of food-derived AGEs, occurring in high levels in many processed foods, may also have a causative role in some chronic diseases. 45 Moreover, skipping breakfast is associated with obesity and diabetes, and men who skipped breakfast had a higher risk of cardiovascular disease than men who did not skip breakfast. 46 Consistent with these observations, avoidance of sugary foods and eating breakfast every day were associated with lower SAF values in our subjects, while there were trends toward lower SAF values with avoidance of processed foods and eating more vegetables. Therefore, the present findings suggest that unhealthy lifestyle habits may accelerate the formation and accumulation of AGEs in apparently healthy adults. However, the seemingly healthy lifestyle habits of not eating too much and eating vegetables first were positively rather than inversely associated with SAF. Although the reason for the association between these specific habits and elevated SAF remains unknown, it is possible that these two lifestyle patterns were not adequately evaluated by our questionnaire.
Conclusions
The present study, which employed a cross-sectional survey, revealed associations between SAF and lifestyle habits but was unable to clarify any causal relationships. However, our findings suggest that SAF measurement may be useful for evaluating lifestyle habits in healthy adults and motivating them to make improvements.
Footnotes
Declaration of conflicting interest
FI and BJW are employees of Morinda Worldwide Inc. in Japan and the US, respectively.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
