Abstract
Objectives:
The aim of this study was to investigate the association between serum carbohydrate antigen 125 concentrations and incident type 2 diabetes mellitus risk in a prospective cohort.
Methods:
We included 18,983 eligible participants aged 63.1 years derived from the Dongfeng–Tongji cohort at baseline from September 2008 to June 2010, and they were followed until October 2013. Cox proportional-hazards models were used to estimate the hazard ratios and 95% confidence interval of type 2 diabetes mellitus incidence in relation to carbohydrate antigen 125 concentrations.
Results:
In all, 1594 incident cases of type 2 diabetes mellitus were observed after a median follow-up of 4.6 years. Carbohydrate antigen 125 concentrations were categorized into four groups according to the quartiles of distribution: <1.1, 1.1–5.6, 5.6–10.0 and ⩾10 U/mL. Compared with participants in the lowest quartile, the hazard ratio (95% confidence interval) of type 2 diabetes mellitus was 0.97 (0.81–1.15), 1.23 (1.05–1.45) and 1.48 (1.27–1.74) for quartile 2–quartile 4 of carbohydrate antigen 125 concentrations after adjustment for potential confounders (p for trend < 0.001). With per–standard deviation increase in carbohydrate antigen 125 levels, the hazard ratio of type 2 diabetes mellitus increased 12% (95% confidence interval, 8–16).
Conclusion:
Findings from this study indicated that serum carbohydrate antigen 125 concentrations were positively correlated with incident type 2 diabetes mellitus risk among a middle-aged and elderly Chinese population.
Introduction
In recent years, the prevalence of type 2 diabetes mellitus (T2DM) has been increasing significantly worldwide, particularly in developing countries. 1 In China, the prevalence of T2DM was 2.5% in 1994, but increased to 10.9% in 2013.2,3 T2DM is the major type of diabetes mellitus (DM) (more than 90%) 4 and is caused by a relative insulin deficiency, which is due to peripheral insulin resistance coupled with an inability of pancreatic beta cells to compensate adequately. 5 Furthermore, T2DM is often implicated with oxidative stress and inflammation.6,7
Carbohydrate antigen 125 (CA-125) is known as mucin 16 or MUC16 and served as a tumour marker of ovarian cancer. 8 However, CA-125 is not specific for ovarian cancer, and it is elevated in some non-ovarian cancers such as pancreatic cancer9,10 and relatively benign conditions including heart failure, coronary heart disease (CHD) and atrial fibrillation.8,11,12 Other studies also found that CA-125 levels were correlated with serum levels of tumour necrosis factor alpha (TNF-α), interleukin (IL)-6 and IL-10, closely related to cytokine system.13,14 Moreover, CA-125 levels increased under several inflammatory conditions such as pancreatitis and inflammatory bowel disease.10,15 Therefore, serum CA-125 might be a potential clinical biomarker for risk assessment of T2DM. However, the relationship between serum CA-125 and T2DM was inconsistent. Previous two case-control studies observed that serum CA-125 decreased 16 or increased 17 in patients with T2DM. Another cross-sectional study conducted in a Korean population indicated that individuals with impaired fasting glucose (IFG) tended to have increased CA-125 level, but no significant association between CA-125 levels and IFG was observed. 18 Moreover, no prospective studies investigated the association between serum CA-125 levels and risk of T2DM. Therefore, the relationships between them are remained to be further studied.
In this study, using data derived from the Dongfeng–Tongji (DFTJ) cohort study, we conducted a prospective study to explore the association of serum CA-125 levels with incident T2DM risk in middle-aged and elderly Chinese population.
Methods
Study population
The DFTJ cohort was an observational longitudinal study on chronic diseases in middle-aged and elderly retirees from the Dongfeng Motor Corporation in Shiyan, Hubei, China. A total of 27,009 retired employees were included in the baseline survey between September 2008 and June 2010, and 25,978 individuals (96.2% of those at baseline) completed the first follow-up from April to October 2013. More detailed information on the design, fundamentals and method of DFTJ cohort study has been previously reported elsewhere. 19 Individuals with available data of CA-125 concentration and DM status at baseline were included in this study (n = 25,720). After exclusion of participants who had a history of T2DM (n = 4953), tumour (n = 1376) and liver disease (n = 950), a total of 18,983 eligible participants (10,619 female and 8364 male participants) were included in the final analysis.
Ethics statement
The study was approved by the Medical Ethics Committee of the School of Public Health, Tongji Medical College, Huazhong University of Science and Technology and Dongfeng General Hospital, Dongfeng Motor Corporation. Each participant provided written informed consent.
Laboratory measurements
Peripheral venous blood samples were collected with a vacuum coagulation tube in the morning after overnight fasting. Serum CA-125 concentrations were determined by the Architect Ci8200 automatic analyser (Abbott Laboratories, Abbott Park, IL, USA). The Abbott Diagnostics reagents were used for the detection according to the manufacturer’s instructions, and the intra-assay coefficients of variation were 5.50% for CA-125.
Fasting blood glucose concentrations were determined through glucose oxidase method by Aeroset Automatic analyser (Abbott Laboratories). Blood lipids [triglyceride (TG), total cholesterol (TC), low-density lipoprotein (LDL) cholesterol and high-density lipoprotein (HDL) cholesterol] and hepatic function [alanine aminotransferase (ALT) and aspartate aminotransferase (AST)] were measured at the hospital’s laboratory with Architect Ci8200 automatic analyser (Abbott Laboratories). Blood cell analysis [total leukocyte count (TLC) and neutrophil percentage (NP)] was done using a fully automated analyser CELL-DYN 3700 (Abbott Laboratories). Blood glucose concentrations, blood lipids and hepatic function were measured at baseline and follow-up. Glycated haemoglobin (HbA1c) concentrations were assayed with high-performance liquid chromatography (D-10 System; Bio-Rad Laboratories, Hercules, CA, USA) at follow-up.
Assessment of DM
DM was diagnosed according to American Diabetes Association (ADA) criteria: 20 self-reported physician-diagnosed DM, or fasting plasma glucose (FPG) ⩾ 7.0 mmol/L, or use of anti-diabetic therapies or HbA1c ⩾ 6.5%.
Assessment of covariates
Baseline CA-125 concentrations were categorized into four groups according to the quartile of distribution: <1.1, 1.1–5.6, 5.6–10.0 and ⩾10.0 U/mL. Trained interviewers used a questionnaire to collect baseline and follow-up data including sociodemographic characteristics, medical history, drug use and lifestyle during face-to-face interviews. Information including smoking status (current, former and never), drinking status (current, former and never) and self-reported education attainment (primary school or below, junior high school, senior high school, and college or above) was also obtained during the interviews. Physical activity was defined as those who exercised more than 20 min per time regularly over half of a year. Trained nurses, technicians and physicians performed physical examinations including standing height, body weight, waist circumference (WC) and blood pressure. Body mass index (BMI) was calculated as weight in kilogrammes divided by the square of height in metres. Individuals who had a BMI ⩾ 24 kg/m2 were categorized into overweight/obesity group, and others were categorized into non-overweight/obesity group.
Statistical analysis
The analysis of variance (ANOVA) was conducted to test difference in means among quartile 1–quartile 4 of baseline CA-125 concentrations, and chi-square tests were conducted for categorical variables. Generalized linear model was used to explore the associations between FPG, ALT, AST, TC, TG, HDL, BMI, WC, TLC, NP and CA-125 concentrations. Cox proportional-hazards model was performed to estimate the hazard ratios (HRs) and 95% confidence intervals (CIs) of diabetes for different concentrations of CA-125. In the full-adjusted model, we included age; gender; education; smoking status; drinking status; BMI; physical activity; family history of diabetes; HDL and TG concentration; and past history of CHD, stroke and hypertension. To test the linear trend across CA-125 quartiles, we used the median value of CA-125 for each quartile and treated it as ordinal variable in the model. Stratified analyses were also performed according to gender (male and female), BMI (⩾24, <24 kg/m2), drinking status (ever drinker and never drinker) and smoking status (ever smoker and never smoker). The interactions of CA-125 concentrations and other covariates were examined by likelihood-ratio tests. In addition, sensitivity analyses were conducted by the exclusion of participants with incident T2DM during the first year of follow-up or exclusion of participants with prevalent CHD and stroke at baseline. The World Health Organization criteria 21 were also used to diagnose T2DM at follow-up for sensitivity analysis. All statistical analyses were carried out using SAS version 9.4 (SAS Institute, Cary, NC, USA). Two-sided statistical tests considered to be significant at p-values below 0.05.
Results
Baseline characteristics of participants by quartiles of CA-125 concentrations are presented in Table 1. In total, 18,983 eligible participants (10,619 female and 8364 male participants) were included in this study. The average age of the study individuals at baseline was 63.1 ± 7.8 years. The median (interquartile range) of CA-125 concentrations was 5.6 (1.1–10.0) U/mL in the total population, 6.0 (1.2–10.4) U/mL in male participants and 5.3 (1.0–9.7) U/mL in female participants. With increase in the CA-125 concentrations, the diabetes incidence increased (Q1: 8.0%, Q2: 8.0%, Q3: 8.1% and Q4: 9.6%; p < 0.001). Individuals with higher CA-125 concentrations were more likely to be male, older and tended to have lower levels of systolic blood pressure, diastolic blood pressure and higher levels of TC and FPG. The proportions of smoking and drinking were higher in participants with higher levels of CA-125. The prevalence of overweight/obesity was lower among individuals with higher CA-125 concentrations.
Baseline characteristics of study population according to serum CA-125 quartiles.
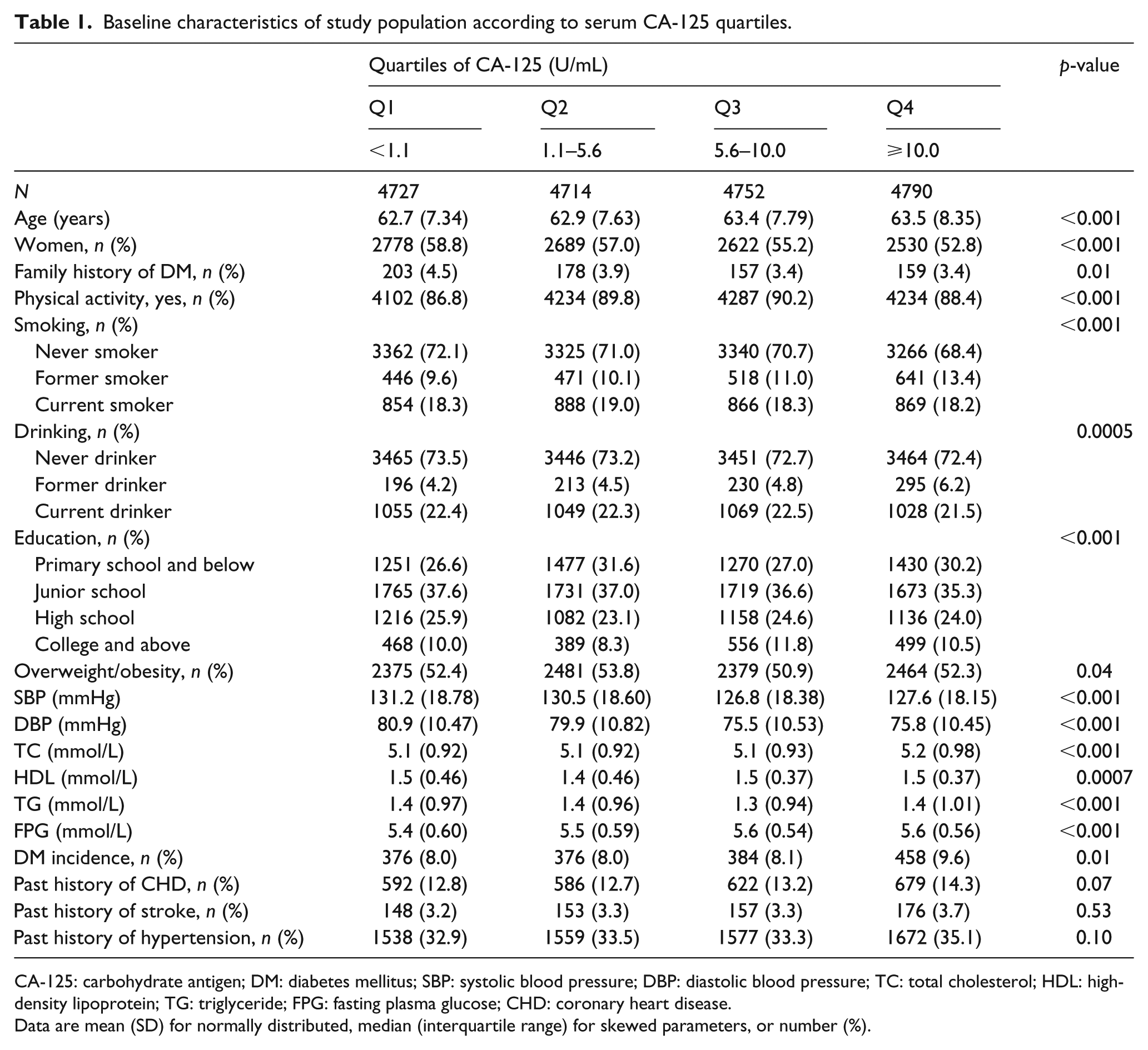
CA-125: carbohydrate antigen; DM: diabetes mellitus; SBP: systolic blood pressure; DBP: diastolic blood pressure; TC: total cholesterol; HDL: high-density lipoprotein; TG: triglyceride; FPG: fasting plasma glucose; CHD: coronary heart disease.
Data are mean (SD) for normally distributed, median (interquartile range) for skewed parameters, or number (%).
As shown in Table 2, FPG, ALT, AST, TC, TLC and NP were positively associated with CA-125 concentrations (β = 1.39, 0.02, 0.04, 0.22, 0.13 and 0.03, respectively; all p < 0.01); in contrast, BMI (β = –0.06, p = 0.001) and WC (β = –0.15, p < 0.001) were negatively associated with CA-125 concentrations after adjustment for potential confounders including age; gender; education; smoking status; drinking status; physical activity; BMI; family history of DM; and past history of CHD, stroke and hypertension.
Adjusted regression coefficients for the associations between CA-125 and other biomarkers at baseline. a
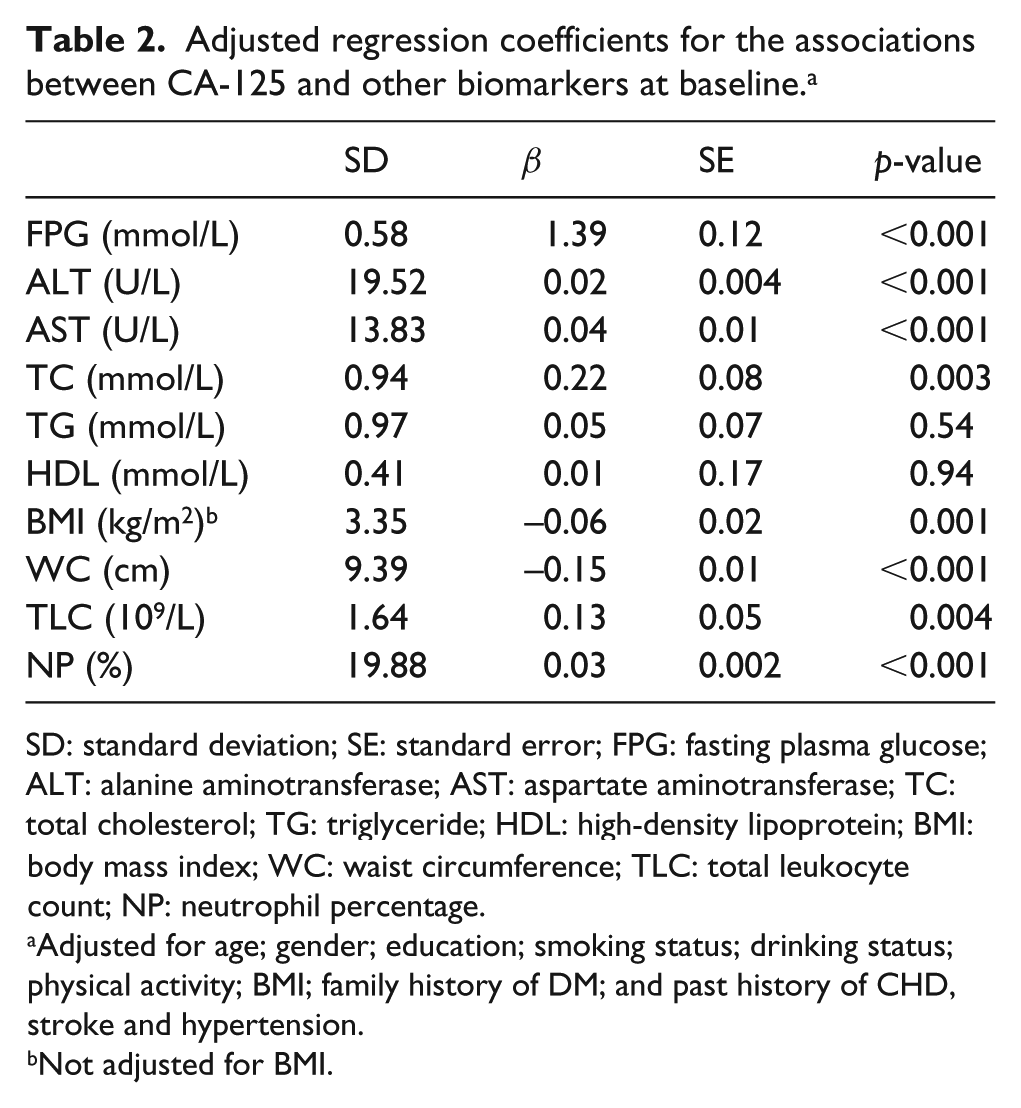
SD: standard deviation; SE: standard error; FPG: fasting plasma glucose; ALT: alanine aminotransferase; AST: aspartate aminotransferase; TC: total cholesterol; TG: triglyceride; HDL: high-density lipoprotein; BMI: body mass index; WC: waist circumference; TLC: total leukocyte count; NP: neutrophil percentage.
Adjusted for age; gender; education; smoking status; drinking status; physical activity; BMI; family history of DM; and past history of CHD, stroke and hypertension.
Not adjusted for BMI.
A total of 1594 incident cases of T2DM were observed after a median follow-up of 4.6 years. The association of serum CA-125 levels with risk of T2DM is shown in Table 3. Compared with individuals in the lowest quartile, the HR (95% CI) of T2DM were 1.17 (1.02–1.35), 1.27 (1.10–1.47) and 1.54 (1.34–1.77) for quartile 2–quartile 4 of CA-125 concentrations after adjustment for age and gender (p for trend < 0.001). Additional adjustment for family history of diabetes, physical activity, education, drinking status, smoking status and BMI did not substantially change the association [Q2 vs Q1: HR = 1.10, 95% CI (0.94–1.27); Q3 vs Q1: HR = 1.21, 95% CI (1.04–1.40); Q4 vs Q1: HR = 1.45, 95% CI (1.26–1.67); p for trend < 0.001; Table 2]. Further adjustment for TG; HDL; past history of CHD, stroke and hypertension obtained similar results. Per-SD increment of CA-125 concentrations was associated with a 12% increased risk of incident T2DM after adjustment for potential confounders [HR = 1.12, 95% CI (1.08–1.16)].
Association of serum CA-125 levels and risk of type 2 diabetes incidence.
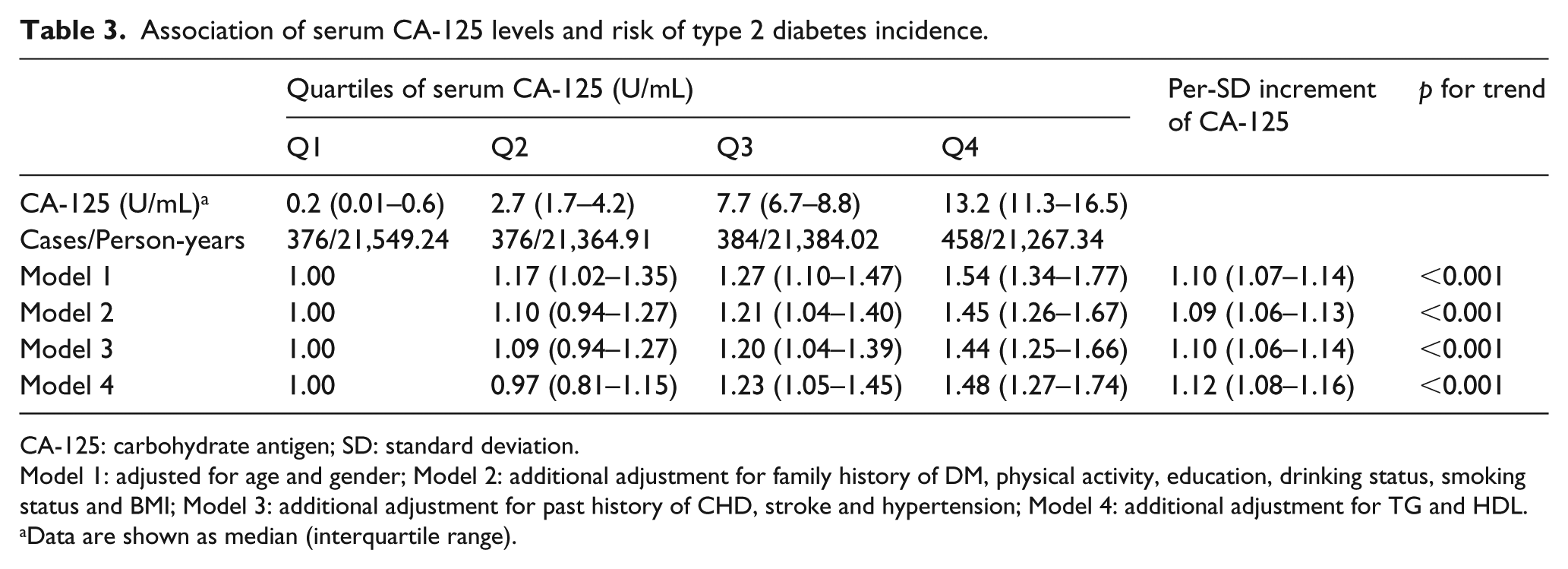
CA-125: carbohydrate antigen; SD: standard deviation.
Model 1: adjusted for age and gender; Model 2: additional adjustment for family history of DM, physical activity, education, drinking status, smoking status and BMI; Model 3: additional adjustment for past history of CHD, stroke and hypertension; Model 4: additional adjustment for TG and HDL.
Data are shown as median (interquartile range).
In addition, excluding individuals with prevalent CHD and stroke (n = 16,504) at baseline [Q2 vs Q1: HR = 1.00, 95% CI (0.83–1.21); Q3 vs Q1: HR = 1.23, 95% CI (1.03–1.48); Q4 vs Q1: odds ratio (OR) = 1.46, 95% CI (1.23–1.74); p for trend < 0.001; Supplemental Table 1] or excluding participants with incident T2DM during the first year (n = 18,896) of follow-up [Q2 vs Q1: HR = 0.97, 95% CI (0.81–1.16); Q3 vs Q1: HR = 1.23, 95% CI (1.04–1.45); Q4 vs Q1: OR = 1.52, 95% CI (1.29–1.78); p for trend < 0.001; Supplemental Table 2] did not materially alter the results. When T2DM was defined according to the World Health Organization criteria at follow-up, the association was slightly attenuated, but the relationship still remained statistically significant [Q2 vs Q1: HR = 0.89, 95% CI (0.74–1.07); Q3 vs Q1: HR = 1.17, 95% CI (0.99–1.39); Q4 vs Q1: OR = 1.33, 95% CI (1.12–1.57); p for trend < 0.001; Supplemental Table 3].
We subsequently conducted stratification analyses by gender, BMI, drinking status and smoking status. The associations between serum CA-125 levels and risk of T2DM seemed to be stronger among participants who are male, ever smokers and ever drinkers (all p for trend < 0.01; Supplemental Table 4); however, there were no significant interactions between serum CA-125 concentrations and gender, BMI, drinking status and smoking status (p for interaction = 0.81, 0.50, 0.37 and 0.63, respectively; Figure 1) on T2DM risk.
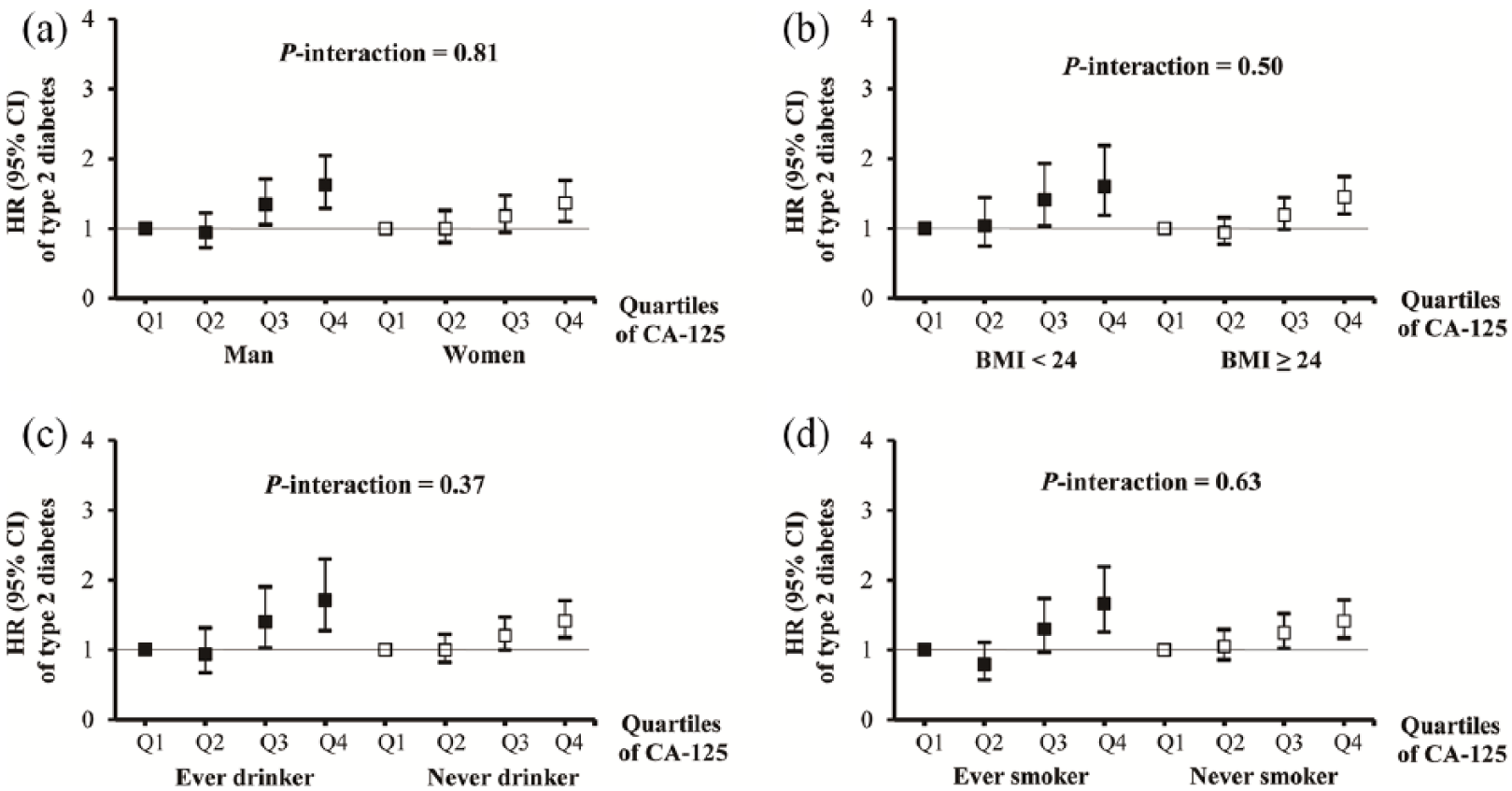
Associations between serum CA-125 and risk of T2DM incidence stratified by (a) gender, (b) BMI (in kg/m2), (c) drinking and (d) smoking among individuals of Dongfeng–Tongji cohort.
Discussion
To our knowledge, this study is the first prospective cohort study to investigate the relationship between serum CA-125 level and risk of incident T2DM, and the findings in this study indicated that serum CA-125 levels were positively correlated with incident T2DM risk.
In this prospective study, CA-125 concentrations tended to be higher in men, elders, smokers, drinkers or individuals without overweight/obesity or with lower levels of systolic blood pressure, diastolic blood pressure, and higher levels of TC and FPG. In line with previous studies,18,22 this study found that increased BMI was negatively associated with CA-125 concentration. The mechanism might be that individuals with a greater BMI have larger plasma volumes, decreasing the serum concentrations of soluble tumour markers, which was known as hemodilution.18,23
Consistent with previous findings,16,22,24 this study found that men, elders, smokers, drinkers, or participants without overweight/obesity or with lower blood pressure tended to have higher concentrations of serum CA-125. Serum CA-125 concentrations have been reported to be associated with glucose metabolism with controversial findings. Joo et al. 18 first reported that serum CA-125 concentrations were negatively correlated with fasting blood glucose levels, but no statistically significant association between serum CA-125 levels and IFG was observed in the observational study, conducted among 12,196 healthy Korean women. Afterward, a case-control study conducted among the Turkish population found that concentrations of serum CA-125 were higher in diabetic patients. 17 In contrast, another case-control study in Iran indicated that CA-125 levels were inversely correlated with diabetes status. 16 The case-control and cross-sectional designs hampered us to draw a causal conclusion between them. In addition, the Korean study 18 was conducted among women, which might not be representative of the general population. However, compared with previous study population (Korean, Turkish and Iran),16–18 Chinese population had relatively lower CA-125 levels. Therefore, the previous findings in the other studies might not be directly generalized to Chinese population.
The potential mechanisms of elevated serum CA-125 concentrations associated with increased risk of incident T2DM are still not clear. Donath and Shoelson 6 suggested that oxidative stress and inflammation may play a significant role in the development of T2DM. Previous studies suggested that CA-125 concentrations were elevated in response to several stimuli including mechanical stress and inflammatory stimuli.8,25 In vitro model also observed that CA-125 secretion was enhanced by several inflammatory stimuli. 26 In addition, serum CA-125 is not expressed in the normal pancreatic ducts, but CA-125 increased in pancreatic carcinoma and pancreatitis tissue,10,27 which indicated that CA-125 might play a potential role in the progression of these diseases. In addition, we observed that CA-125 level was positively associated with TLC in this study. TLC and NP have also been reported to be associated with the development of chronic inflammation. 28 Therefore, the positive relationship between CA-125 levels and incident T2DM might be attributed to inflammation. However, further studies are needed to elucidate the exact mechanisms.
The strengths of this study included the relatively large sample size, the prospective design and the ability to adjust some potential confounders. It would enable us to investigate the association between serum CA-125 levels and risk of incident T2DM with moderate statistical power and reduce the potential bias. Nonetheless, some limitations should be taken into consideration. First, this study was performed in middle-aged and elderly retired workers, thus the findings may not be representative of other ethnic or age groups. Second, the serum CA-125 concentration was measured only once at baseline; therefore, we could not account for within-individual variability in this study. Third, data on islet antibodies and pancreatic inflammation measures were not available, so we failed to classify the types of incident diabetes in this study and to further elucidate the mechanisms linking CA-125 and incident T2DM. However, participants in this study were middle-aged and elderly retired workers; therefore, the proportion of type 1 DM included in this study might be very limited.
Conclusion
In conclusion, in this prospective cohort study, we found a positive association between serum CA-125 concentrations and risk of T2DM. Serum CA-125 might be a potentially useful biomarker for risk assessment of T2DM. Further studies were needed to clarify the potential mechanisms behind the association.
Supplemental Material
Supplemental_Tables20190123 – Supplemental material for Serum carbohydrate antigen 125 levels and incident risk of type 2 diabetes mellitus in middle-aged and elderly Chinese population: The Dongfeng–Tongji cohort study
Supplemental material, Supplemental_Tables20190123 for Serum carbohydrate antigen 125 levels and incident risk of type 2 diabetes mellitus in middle-aged and elderly Chinese population: The Dongfeng–Tongji cohort study by Caizheng Yu, Qing Lei, Jing Wang, Xu Han, Fei Wang, Jing Yuan, Ping Yao, Sheng Wei, Youjie Wang, Yuan Liang, Xiaomin Zhang, Huan Guo, Handong Yang and Meian He in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank all study subjects for participating in this Dongfeng–Tongji (DFTJ) cohort study and all volunteers for assisting in collecting the samples and data.
Author contributions
CY, QL and MH designed the study, interpreted data and wrote the first draft of the paper; CY, JW, XH and FW took responsibility for the accuracy of the data analysis; JY, PY, SW, YW, YL, XZ, HG and HY performed data collection and designed the study’s analytic strategy. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the National Natural Science Foundation (NSFC-81522040 and 81473051), National Key R&D Program of China (2017YFC0907501) and the Program for HUST Academic Frontier Youth Team.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
