Abstract
The purpose of this study was to understand the influence of sustained intravitreal vascular endothelial growth factor neutralisation on the retinal and choroidal vasculature in diabetic eyes. Ins2Akita diabetic mice received five intravitreal injections of anti-mouse vascular endothelial growth factor antibody or goat immunoglobulin G (0.2 µg/µL/eye) over a 4-month period. Retinal and choroidal vascular changes were analysed by confocal microscopy of tissue flat-mounts. Retinal gene expression of vascular endothelial growth factor family members (vascular endothelial growth factors A, B, C and D), vascular endothelial growth factor receptors (sVEGFR-1 and VEGFR-2) and tight junctions (claudin 1, 2, 5; occludin and zonula occludens-1) were analysed by quantitative reverse transcription polymerase chain reaction. Vascular endothelial growth factor A and claudin 5 were significantly increased in diabetic retinae. Gene expression was unaffected by anti-vascular endothelial growth factor treatment. The number of acellular vessels was increased in diabetic retinae and reduced following anti-vascular endothelial growth factor treatment. Retinal and choroidal vascular density and area were unaffected by sustained vascular endothelial growth factor neutralisation. Our results suggest that five consecutive intravitreal anti-vascular endothelial growth factor injections do not cause significant vascular changes in the retina and choroid in diabetic and non-diabetic mice.
Introduction
Diabetic retinopathy (DR) is the most common microvascular complication of diabetes mellitus and remains a major cause of vision impairment and blindness in the working age population.1,2 DR is marked by progressive dysfunction of the retinal neurovascular unit associated with prolonged hyperglycaemia resulting in neuroglial damage, vasodegeneration and impaired tissue perfusion. Increased activation of inflammatory pathways and/or retinal hypoxia drive the advanced, sight-threatening stages of diabetic macular oedema (DMO) and proliferative DR (PDR).3–5 Chronic retinal microvascular damage results in elevation of intraocular levels of vascular endothelial growth factor A (VEGF), which plays a major role in the pathogenesis of DR by promoting blood-retinal barrier (BRB) disruption and neovascularisation. 6 Anti-VEGF agents were approved for the treatment of DMO in 2012, and more recently for PDR via repeated intravitreal injections. Prior to this, laser photocoagulation was the historic standard of care. 7 Anti-VEGF treatment has been shown to substantially improve visual and anatomic outcomes without the ocular side effects associated with laser treatment8,9 and has become the preferred treatment option for DMO and PDR patients.
Despite its pathophysiologic role, VEGF is a key regulator of many physiological processes and exerts cytoprotective effects on a large number of cell types including endothelial, epithelial and neuronal cells. 10 Therapeutic approaches to blocking VEGF signalling long term in retinal diseases can therefore be associated with detrimental side effects which may in part be due to the loss of VEGF-induced cytoprotection. 11 While anti-VEGF therapy is generally considered safe, systemic side effects including hypertension, proteinuria, and thromboembolism12,13 and local side effects such as intraocular pressure elevation, vitreous haemorrhage, retinal detachment and vascular occlusion13,14 have been reported. The overall risk of ocular vascular events following anti-VEGF injection was shown to be higher in the diabetic population. 14 This may be due to the fragile nature of the retina to sustained high blood sugar levels, thus highlighting a safety concern of long-term anti-VEGF therapy especially for DR patients.
Neurodegeneration has been suggested as an adverse effect of anti-VEGF treatment. We and others have shown that prolonged intravitreal VEGF neutralisation accelerates diabetes-induced retinal neurodegeneration.11,15 Furthermore, a recent study showed that anti-VEGF administration for 2 years in DMO patients was associated with reduced retinal nerve fibre layer thickness suggestive of retinal ganglion cell apoptosis. 16 In addition to neurons, endothelial cells also rely on VEGF for normal physiological function. Retinal pigment epithelial (RPE)–secreted VEGF has been shown to be involved in maintaining the choriocapillaris 17 and in vitro studies have shown that anti-VEGF treatment increased endothelial cell apoptosis 18 while VEGF addition protected endothelial cells from high glucose exposure induced apoptosis. 19 In order to further understand the influence of sustained VEGF neutralisation on the retinal and choroidal vasculature in vivo, we investigated the long-term adverse effects of multiple intravitreal anti-VEGF injections in the Ins2Akita mouse model of type 1 diabetes.
Materials and methods
Animals
Male heterozygous diabetic Ins2Akita mice of C57BL/6J background (Cat 003548 – originally purchased from Jackson Laboratory, Bar Harbor, USA) and age- and gender-matched wild type non-diabetic littermates were used in this study. The heterozygous Ins2Akita mice developed robust hyperglycaemia by 4.5 weeks of age. 20 Ins2Akita heterozygous males were bred with female C57BL/6J mice and diabetes was confirmed by blood glucose test (>250 mg/dL) at 6 weeks of age. All mice were housed in pathogen-free conditions on a 12/12 h light/dark cycle, with free access to food and water. All procedures were conducted under the regulation of the UK Home Office Animals (Scientific Procedures) Act 1986 and were approved by the Ethical Review Committee of Queen’s University Belfast. This study was conducted in compliance with the Association for Research in Vision & Ophthalmology Statement for the Use of Animals in Ophthalmology and Vision Research.
Intravitreal injection
Intravitreal injections were performed as previously described.11,21 Briefly, 1 μL of goat anti-mouse VEGF164 (200 ng/μL/eye; endotoxin-free, AF-493-NA; R&D Systems, Abingdon, UK) or endotoxin-free goat immunoglobulin G (IgG) (200 ng/μL; AB-108C; R&D Systems) in phosphate buffered saline (PBS) was injected into the vitreous of both eyes of 3-month-old diabetic Ins2Akita or non-diabetic C57BL/6J littermates using a repeating dispenser (PB-600-1; Hamilton Bonaduz AG, Bonaduz, Switzerland). The AF-493-NA antibody has been shown to efficiently neutralise the bio-activity of mouse VEGF164 in vivo and the ND50 is 0.05–0.15 µg/mL. 22 The dose of AF-493-NA was chosen based on our previous study. 11 Five intravitreal injections per eye (2.5–3 weeks between injections) were performed over a 4-month period. Animals were culled 4 weeks after the last injection at 7 months of age.
Two sets of experiments were performed. In the first experiment, animals were culled by CO2 inhalation and eyes were enucleated. Left eyes were processed for immunohistochemistry while right eyes were processed for RNA extraction. Age-matched mice that did not receive any intravitreal injections served as full controls (non-diabetic: n = 4 mice per group; diabetic: not injected n = 4, IgG injected n = 2 and anti-VEGF injected n = 3). In the second experiment, animals were administered a terminal dose of sodium pentobarbital (120 mg/kg) via intraperitoneal injection and blood vessels were directly labelled by cardiac perfusion using a solution containing 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (DiI) (non-diabetic: n = 3 mice per group; diabetic: IgG n = 3 and anti-VEGF n = 5). 23
Quantitative reverse transcription polymerase chain reaction (PCR)
Retinae were dissected immediately after CO2 inhalation and snap frozen in Buffer RLT (Qiagen, UK) containing β-Mercaptoethanol (10 µL/mL buffer RLT) as recommended by the manufacturer. Total RNA was extracted using Qiagen’s RNeasy Mini kit (Qiagen, UK) and reverse transcribed (300 ng of RNA per sample) into cDNA using a Superscript II Reverse Transcriptase kit (Invitrogen, UK) as per manufacturer’s instructions. cDNA samples were stored at −20°C until further use. Quantitative PCR was carried out in a total reaction volume of 10 µL containing 2 µL of cDNA (prior diluted 1:10 in water), 5 µl of 2× SYBR green (Fermentas, Canada), 0.5 µl of 10 mM forward and reverse primer and 2.5 µL RNase/DNase free water. Reactions were performed in triplicate using the LightCycler 480 Real-Time PCR System (Roche Diagnostics Ltd, West Sussex, UK). Expression of target genes relative to reference genes was calculated using the advanced relative quantification analysis module of the LightCycler 480 software 1.5 (Roche Diagnostics Ltd) based on the delta-delta CT method. Four housekeeping genes were tested (β-actin, 18S, RPLP0 and SDHA). Relative expression levels (relative to RPLP0 as this was the most stable housekeeping gene across samples) are presented in graphs. Primer sequences are shown in Table 1.
List of primer sequences used in this study.
Immunohistochemistry
Eyes were enucleated and fixed in 2% paraformaldehyde (Sigma-Aldrich, UK) for 2 h. After washing in PBS, retinae were carefully dissected from the eye cups, washed in PBS and immersed in permeabilisation buffer (10% foetal calf serum and 3% TritonX-100 in PBS) for 3 days at 4°C with mild agitation. Retinae were extensively washed in PBS and incubated in primary antibody staining solution [Biotinylated Griffonia Simplicifolia Lectin I isolectin B4 (1:100 dilution, Vector Laboratories, UK) and rabbit anti-collagen IV (1:200 dilution, Bio-Rad AbD Serotec, UK) in 10% foetal calf serum and 0.5% TritonX-100 in PBS] for 5 days at 4°C with mild agitation. Retinae were washed in PBS and incubated in secondary antibody [Fluorescein Streptavidin (1:100, Vector Laboratories, UK); Donkey Anti-Rabbit IgG (H + L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 594 (1:200, Invitrogen, UK) in 10% foetal calf serum and 0.5% TritonX-100 in PBS] for 24 h at 4°C with mild agitation. Retinal tissue was washed in PBS and flat-mounted in VECTASHIELD Antifade Mounting Medium (Vector Laboratories, UK) on glass slides. Z-stack confocal images of the retinal flat-mounts were taken by confocal microscopy (C1-Nikon Confocal Microscope, Eclipse TE200-U, Nikon UK Ltd, Surrey, UK) at 40× magnification. Four images per retina were acquired (one image in each quadrant). Acellular vessels (identified as Collagen IV positive but isolectin B4 negative) were counted manually in each z-stack image using Image J software (National Institutes of Health, Bethesda, MD) and normalised to the total image area. The average number of acellular vessels per retina was calculated and the average number per treatment group was presented in the graphs.
Cardiac perfusion
Cardiac perfusion was performed using a published protocol 23 with modifications. DiI stock solution was prepared by dissolving 100 mg of DiI crystal (Sigma-Aldrich, UK) in 16.7 mL of 100% ethanol on a rocker overnight and stored in the dark at room temperature. The chest cavity was opened under deep terminal anaesthesia (120 mg/kg sodium pentobarbital) and mice were perfused through the left ventricle with 10 mL of PBS containing 10 units/mL heparin followed by 10 mL of 1% glucose solution containing 200 μL of DiI stock solution using a peristaltic pump (10 mL/min). Following perfusion, eyes were enucleated immediately and fixed in 2% paraformaldehyde (Sigma-Aldrich, UK) for 2 h. Fixed ocular tissue was washed in PBS, retinae were dissected and flat-mounted in VECTASHIELD Antifade Mounting Medium (Vector Laboratories) on glass slides. Flat-mounted retinal tissue was examined by confocal microscopy (C1-Nikon Confocal Microscope), with 12 images collected per retina (three images in each quadrant in the centre, middle and peripheral retina).
RPE was removed from the choroid by incubation in 0.05% Trypsin-ethylenediaminetetraacetic acid (EDTA) (1×; Thermo Fisher Scientific, UK) at 37°C, for 20 min, followed by gentle brushing with fine paint brushes. Choroids were flat-mounted in VECTASHIELD Antifade Mounting Medium (Vector Laboratories) on glass slides, and examined by confocal microscopy (C1-Nikon Confocal Microscope). In all, 15–30 images per choroid were captured (5–10 images at each location: centre, middle and peripheral choroid).
Image analysis
All images were analysed using Image J software (Fiji image processing package). Two-dimensional (2D) images of each retinal vascular layer were reconstructed from z-stacks. For vessel density analysis, a 3 × 3 grid was applied to each image and the number of times the vertical lines were crossed by a vessel were counted. For vessel area analysis, the area covered by blood vessels was calculated as a percentage of total image area. Average number of crossings/percentage of vessel area was calculated per retina/choroid and the average number per treatment group was presented in the graphs. Full details of the Image J macro used for the retinal vasculature analysis can be found in https://github.com/pedriniedoardo/retinal_vascular_image_analysis
Statistical analysis
Statistical analysis was performed using GraphPad Prism software. Data were checked for normality and differences between non-diabetic and diabetic groups were analysed using Mann–Whitney U test. Differences between treatment groups (not injected vs IgG injected vs anti-VEGF injected) were analysed using Kruskal–Wallis test followed by post hoc Dunn’s multiple comparison test. Thus, p < 0.05 was considered statistically significant.
Results
The effect of intravitreal VEGF neutralisation on the expression of VEGF family genes and tight junction genes
The expression of VEGF-A was significantly higher in the retinae of diabetic Ins2Akita mice (with 6 months of diabetes duration) when compared to age- and sex-matched non-diabetic littermates. Soluble VEGF receptor 1 (sVEGFR-1) gene expression was also significantly increased under diabetic conditions. The expression of VEGF-B, -C, -D or VEGFR-2 was not significantly altered in diabetic retinae [Figure 1(a) and (b)]. VEGF-B was the most highly expressed VEGF variant in the retina, whereas VEGF-C and -D were the lowest expressed variants.
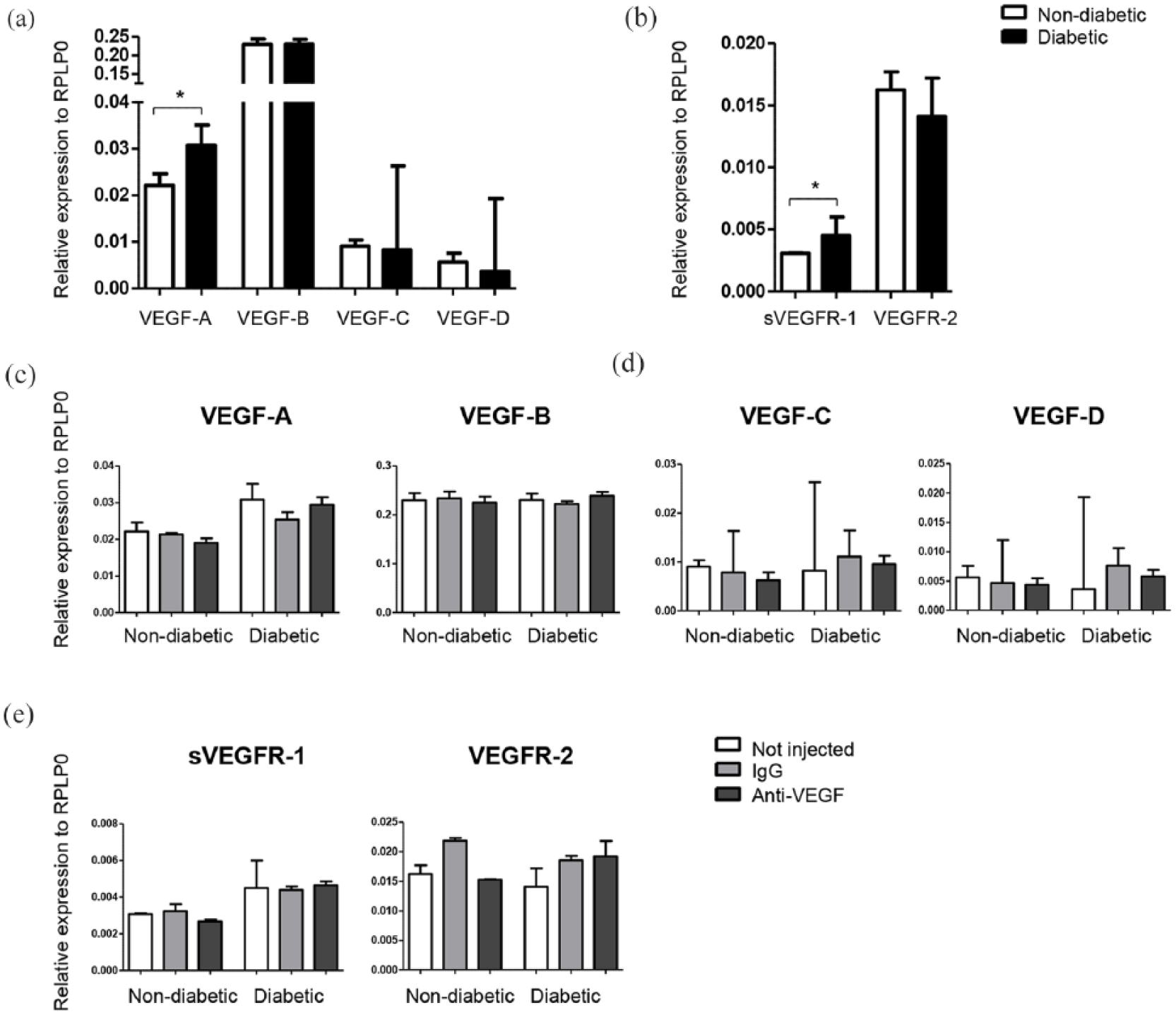
VEGF variants and VEGF receptor gene expression in the retina of control and Ins2Akita mice with/without intravitreal anti-VEGF injections. Expression of (a) VEGF variants and (b) receptors in the retina of Ins2Akita diabetic mice and non-diabetic littermates. Gene expression of (c) VEGF variants and (d) VEGF receptors after five intravitreal injections with IgG control or anti-VEGF antibody. Median + interquartile range are shown. Non-diabetic: n = 4 mice per group; diabetic: not injected n = 4, IgG n = 2 and anti-VEGF n = 3. Mann–Whitney U test was performed to compare non-diabetic versus diabetic groups [(a) and (b)]. Kruskal–Wallis test was performed to compare treatment groups [(c) and (d)]. *p < 0.05.
Intravitreal injections of the anti-VEGF antibody AF-493-NA did not significantly affect the expression of any of the VEGF isoforms [Figure 1(c)] or sVEGFR-1 and VEGFR-2 gene expression [Figure 1(d)]. However, there was a trend of increased VEGFR-2 expression following anti-VEGF treatment in diabetic retinae [Figure 1(d)].
Occludin was most highly expressed in the mouse retina, followed by claudin-5, zonula occludens-1 (ZO1) and claudin-1/2 [Figure 2(a)]. The expression of claudin-5 was significantly increased in the retina of diabetic Ins2Akita mice compared to non-diabetic littermate controls [Figure 2(a)]. The expression of other tight junction genes remained unchanged in the diabetic retina. Intravitreal anti-VEGF treatment did not significantly affect the expression of any of the tight junction genes tested [Figure 2(b)].
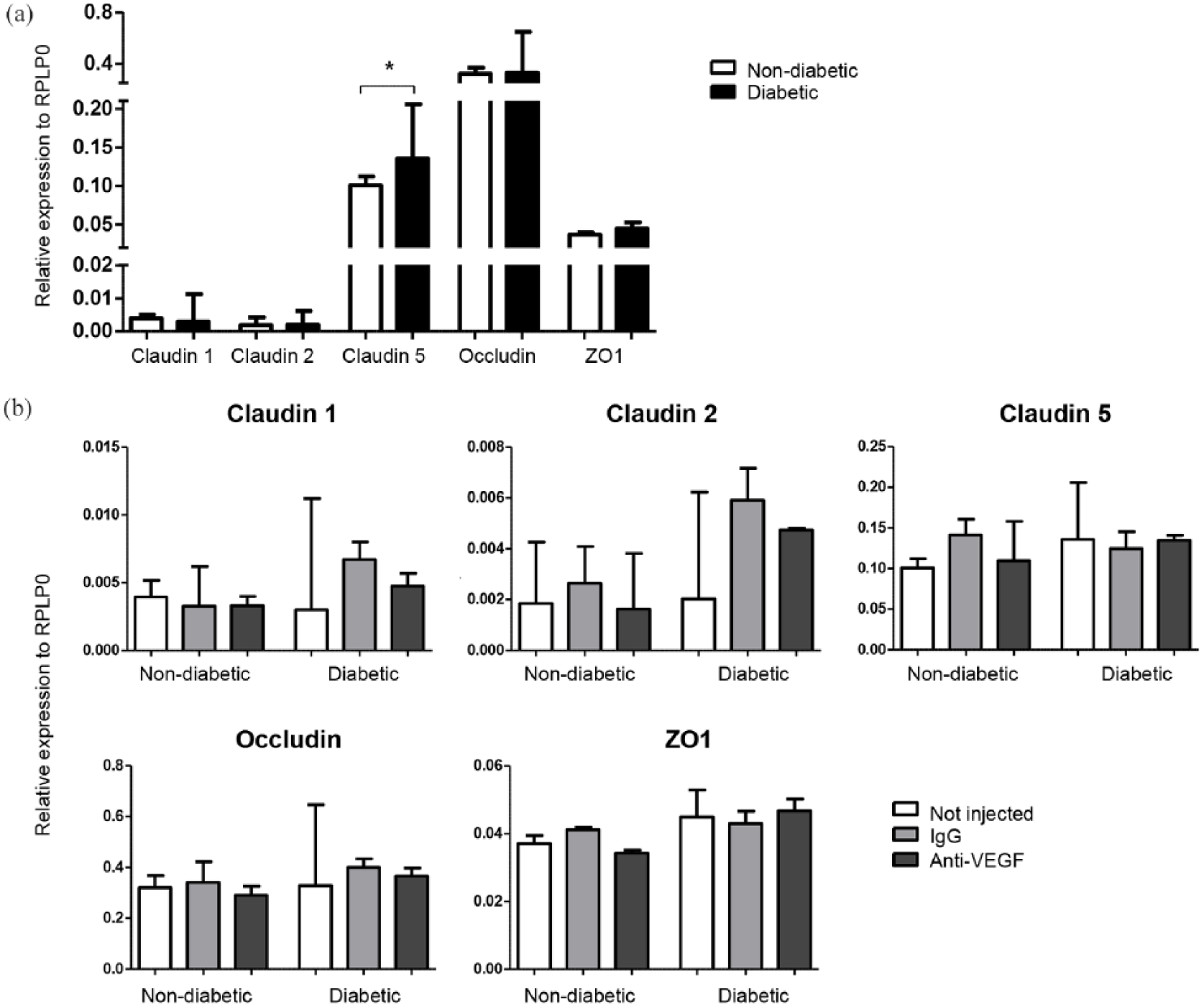
Tight junction gene expression in the retina of control and Ins2Akita mice with/without intravitreal anti-VEGF injections. (a) Expression of tight junction genes in the retina of Ins2Akita diabetic mice compared to non-diabetic littermates. (b) Gene expression of tight junction genes after five intravitreal injections with IgG control or anti-VEGF antibody. Median + interquartile range are shown. Non-diabetic: n = 4 mice per group; diabetic: not injected n = 4, IgG n = 2 and anti-VEGF n = 3. Mann–Whitney U test was performed to compare non-diabetic versus diabetic groups (a). Kruskal–Wallis test was performed to compare treatment groups (b). *p < 0.05.
The effect of intravitreal VEGF neutralisation on the retinal vasculature
Retinal blood vessels were stained for collagen IV and isolectin B4 [Figure 3(a)]. The number of collagen IV positive and isolectin B4 negative acellular vessels was statistically significantly increased in the retina of Ins2Akita diabetic mice compared to non-diabetic littermates [Figure 3(b)]. Acellular vessels were reduced in diabetic mice following intravitreal injection of VEGF over a 4-month period; however, the difference was not statistically significant. The number of acellular vessels in non-diabetic littermates was not affected by the treatment [Figure 3(c)].
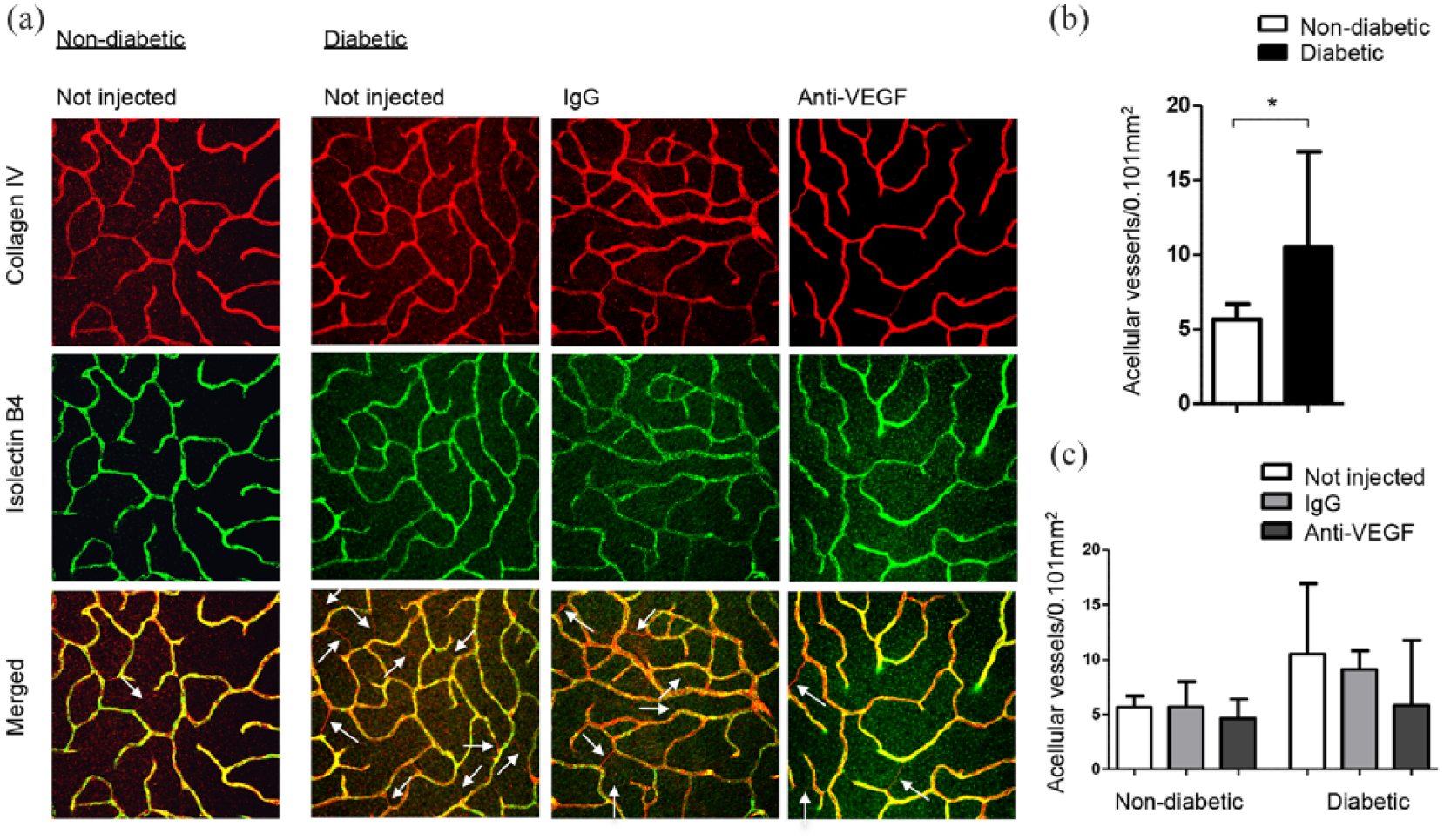
Retinal acellular vessels in the retina of control and Ins2Akita mice with/without intravitreal anti-VEGF injections. (a) Representative images show collagen IV positive and isolectin B4 negative acellular vessels. (b) Acellular vessels quantified throughout the z-stack image in the retina of Ins2Akita diabetic and non-diabetic littermates. (c) Acellular vessels quantified in the same way after five intravitreal injections with IgG control or anti-VEGF antibody. Median + interquartile range are shown. Non-diabetic: n = 4 mice per group; diabetic: not injected n = 4, IgG n = 2 and anti-VEGF n = 3. Mann–Whitney U test was performed to compare non-diabetic versus diabetic groups (b). Kruskal–Wallis test was performed to compare treatment groups (c). *p < 0.05.
No significant differences in vessel density or vessel area were detected in DiI stained retinal blood vessels in Ins2Akita diabetic mice when compared to non-diabetic littermate controls [Figure 4(a)]. Intravitreal injection of anti-VEGF Ab did not affect the vessel density or vessel area in diabetic Ins2Akita mice or non-diabetic littermates [Figure 4(b)].
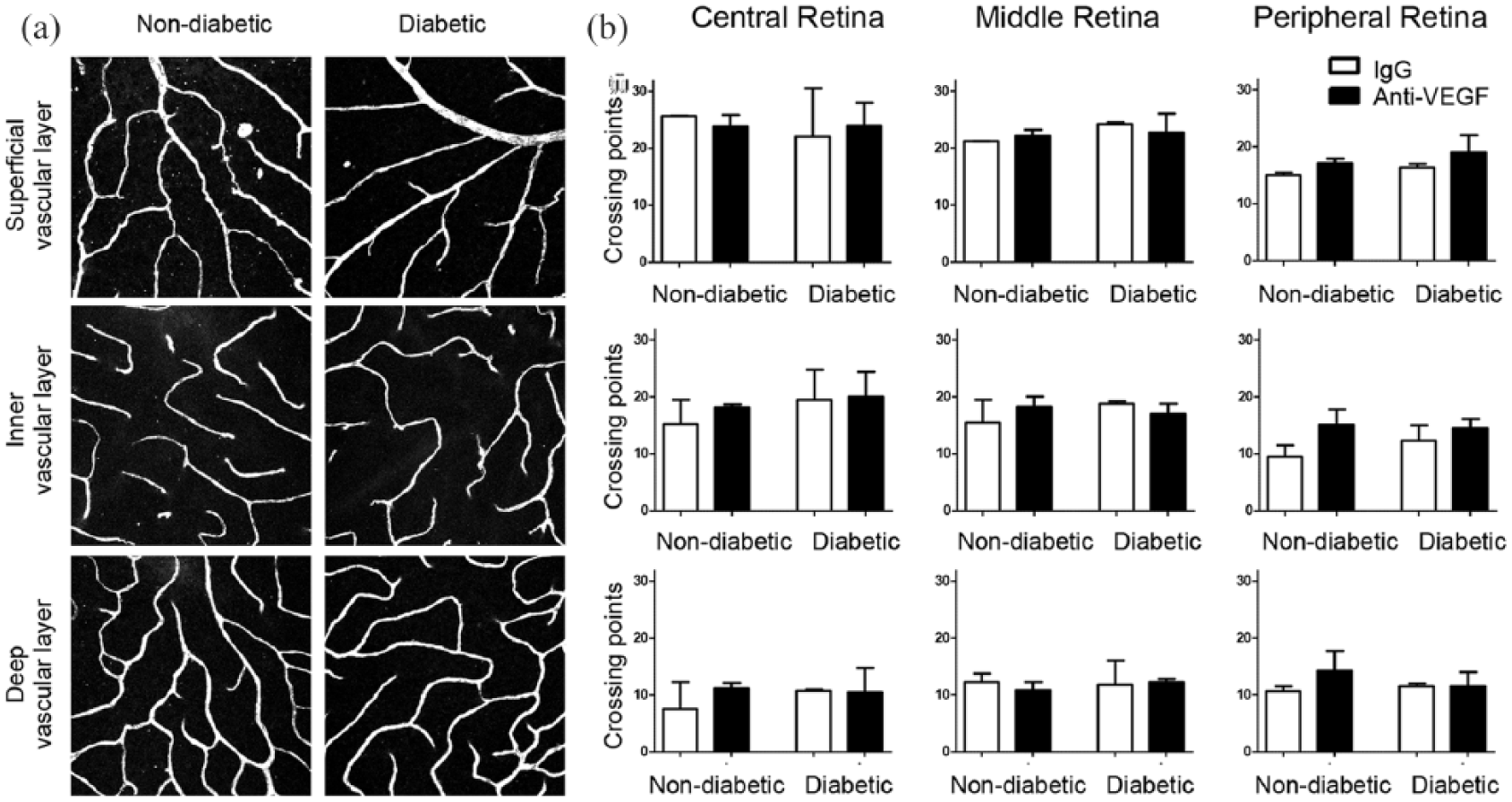
Retinal vasculature in the control and Ins2Akita mice following five intravitreal injections of goat IgG or anti-VEGF antibody. (a) Representative images of DiI stained vessels from a z-stack taken in the peripheral retina and separated into the three vascular layers. (b) Vessel density analysis of the superficial, inner and deep vascular layers in the central, middle and peripheral retina after five intravitreal injections with IgG control or anti-VEGF antibody. Median + interquartile range are shown. Non-diabetic: n = 3 mice per group; diabetic: IgG n = 3 and anti-VEGF n = 5. Kruskal–Wallis test was performed.
Analysis of vessel density and area in images of collagen IV stained retinal vessels (i.e. Figure 3) confirmed our results from the DiI perfusion study (data not shown). We also did not observe any differences between non-injected groups and IgG injected groups (data not shown).
The effect of intravitreal VEGF neutralisation on the choroidal vasculature
Choroidal blood vessels were examined in choroidal flat-mounts following cardiac perfusion with DiI, and vessel area was quantified in the central, middle and peripheral choroid [Figure 5(a)]. Vessel density in the central and middle choroid was ~98%, while only ~90% vessel coverage was observed in the peripheral choroid. No differences in vessel area were found when comparing the choroidal vasculature of Ins2Akita diabetic mice to non-diabetic littermate controls [Figure 5(b)]. Choroidal blood vessel area was unaffected by anti-VEGF injections in both non-diabetic and diabetic mice [Figure 5(b)].
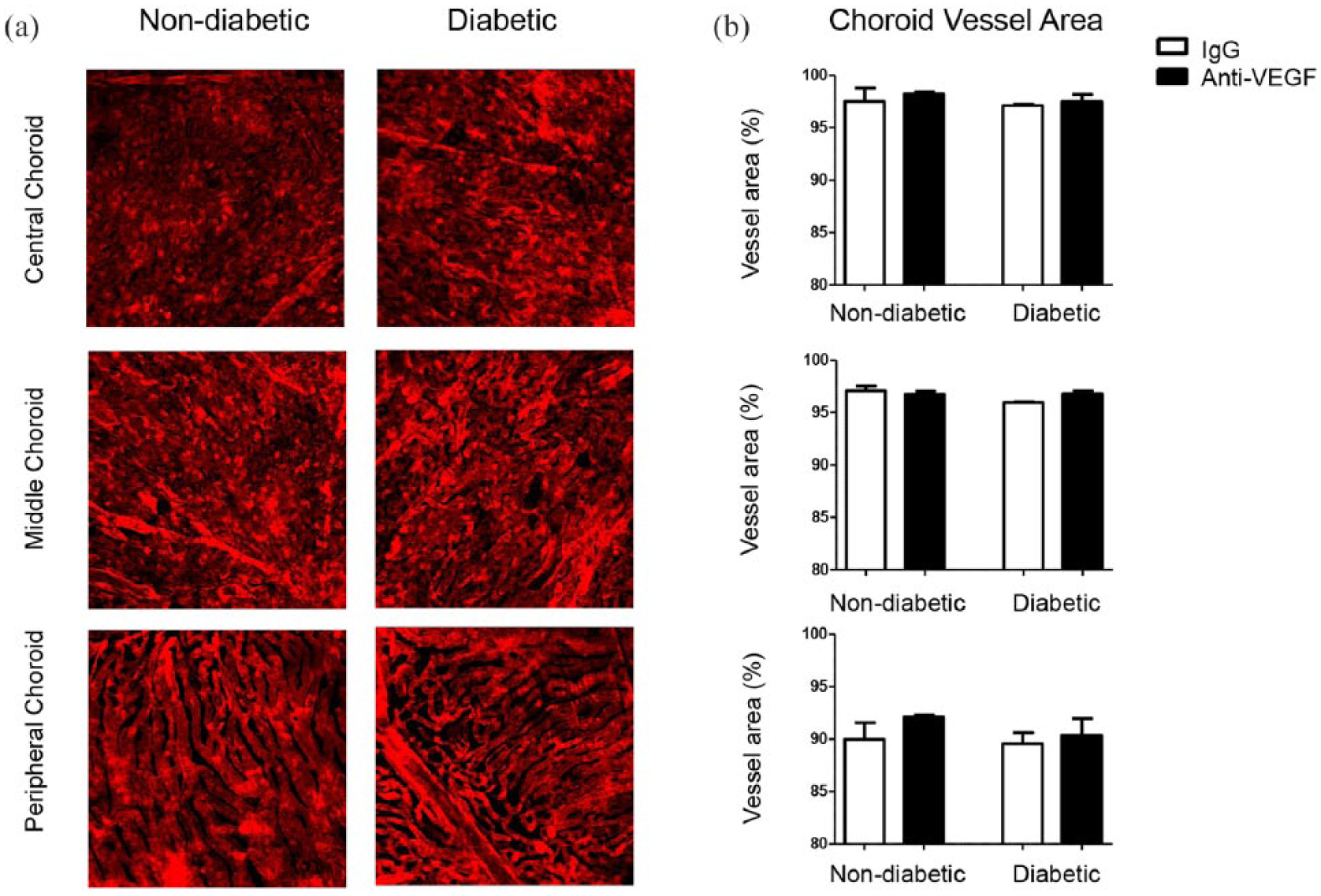
Choroidal vasculature in the control and Ins2Akita mice following five intravitreal injections of goat IgG or anti-VEGF antibody. (a) Representative images of DiI stained vessels in the central, middle and peripheral choroid. (b) Vessel area analysis in the choroid after five intravitreal injections with IgG control or anti-VEGF antibody. Median + interquartile range are shown. Non-diabetic: n = 3 mice per group; diabetic: IgG n = 3 and anti-VEGF n = 5. Kruskal–Wallis test was performed.
Discussion
The Ins2Akita mouse exhibits vascular, neural, and glial abnormalities and has been widely used as a model of early DR.24–26 In this study, we confirm that Ins2Akita diabetic mice have significantly elevated mRNA levels of VEGF-A in the retina when compared to non-diabetic controls [Figure 1(a)]. 26 VEGF-A is the main factor for physiological and pathological angiogenesis, and exerts most of its actions via VEGF receptor 2 (VEGFR-2; also known as KDR and FLK1). 27 In this study, we observed elevated mRNA levels of sVEGFR-1 in the retina of Ins2Akita diabetic mice when compared to non-diabetic controls [Figure 1(b)]. Generally, sVEGFR-1 sequesters the free form of VEGF-A, and acts as a negative regulator of angiogenesis. 28 However, sVEGFR-1 was increased in the aqueous humour of macular oedema patients 29 and may influence vascular permeability through inflammatory pathways.30,31 In addition to VEGF-A, -B, -C and -D are also members of the VEGF family. 27 VEGF-B is a potent survival factor for different cells including vascular and neuronal cells.32,33 VEGF-B levels were increased in the vitreous of diabetic patients with ocular disease suggesting it may play a role in the pathogenesis of DR. 34 In this study, VEGF-B was the most highly expressed VEGF family member in the retina; however, the expression levels were unaffected after 6 months of hyperglycaemia duration in the Ins2Akita mice. VEGF-C and -D play a minor role in angiogenesis but are critical regulators for lymphangiogenesis. 13 VEGF-C has been identified as a trophic factor for neuronal cells in the brain 35 and both VEGF-C and VEGF-D were shown to promote diabetic wound healing in the db/db diabetic mouse model. 36 VEGF-C and -D were the lowest expressed VEGF family members in the retina, and their expression levels were not significantly affected by diabetes in this study.
Tight junction transmembrane proteins (e.g. occludins and claudins) between vascular endothelial cells represent a major component of the inner BRB that regulates molecular exchange between the retina and circulation. 37 Claudin-1, -2 and -5 have been identified as the most prominent claudins of the inner BRB. 38 The expression of tight junctions has been shown to be modulated by high glucose in in vitro and in vivo studies.39,40 Occludin and ZO1 were reported to be decreased in streptozotocin-induced diabetic mice 41 and rats. 42 Another study in diabetic rats reported the downregulation of occludin and claudin-5 but observed increased levels of ZO1 following hyperglycaemia. 39 In the mouse model of oxygen-induced retinopathy, increased mRNA and protein levels of claudin-2 and -5 were associated with their abnormal localisation and distribution in retinal blood vessels. 38 In this study, we observed significantly increased levels of claudin-5 in the retina of diabetic animals compared to controls [Figure 2(a)]. This may be suggestive of disorganised tight junctions or a compensatory mechanism to withstand the compromised BRB.
While we observed significantly increased numbers of acellular vessels, indicative of endothelial cell drop out in diabetic mice [Figure 3(b)], we did not observe any significant structural changes (e.g. vessel density and area) in the retinal or choroidal vasculature when comparing diabetic with non-diabetic mice. Our results suggest that Ins2Akita diabetic mice display early vascular changes following 6 months of hyperglycaemia (elevated levels of VEGF-A and claudin-5 and increased number of acellular vessels), and we used this model to investigate the effects of sustained VEGF neutralisation in retinal and choroidal vasculature.
Anti-VEGF therapy is the current standard of care for DMO, resulting in a significant improvement in visual acuity. 8 However, since VEGF is an important maintenance and neuroprotective factor, 10 long-term VEGF inhibition is associated with safety concerns and potential side effects as described in preclinical11,15 and clinical studies.13,16 In this study, anti-VEGF treatment did not have any effects on VEGF-A, -B, -C or -D gene expression in diabetic or non-diabetic animals. There was a trend of increased VEGFR-2 gene expression in the diabetic retina following intravitreal anti-VEGF treatment; however, the difference was not statistically significant [Figure 1(d)]. Similarly, in a previous study, repeated anti-VEGF treatment of human umbilical vein endothelial cells in vitro under hypoxic conditions resulted in unchanged VEGF-A levels but increased VEGFR-2 levels. 43 The increased VEGFR-2 expression may be a compensatory mechanism in response to VEGF depletion.
VEGF is known to regulate the expression of tight junction genes44,45 and anti-VEGF treatment restored the VEGF-induced decrease of claudin-1 expression in immortalised bovine retinal endothelial cells. 45 In our study, the expression of tight junction genes in the retina were unaffected by sustained intravitreal VEGF neutralisation in both non-diabetic and diabetic mice [Figure 2(b)].
VEGF is also important for choroidal homeostasis. The absence of soluble VEGF isoforms in mice resulted in age-dependent choroidal vascular degeneration. 17 In DMO patients, sustained intravitreal anti-VEGF therapy over a 6-month period was associated with significant thinning of the choroid. 46 We have demonstrated that VEGF neutralisation over 4 months did not cause significant vascular changes in the retina or choroid of diabetic and non-diabetic mice (Figures 4 and 5). As VEGF-B is highly expressed in the mouse retina, and the antibody used in this study (AF-493-NA) only recognises VEGF-A, retinal and choroidal blood vessels may be protected by VEGF-B when VEGF-A is neutralised.
We observed less acellular vessels in diabetic animals following anti-VEGF therapy although the difference was not statistically significant [Figure 3(c)]. While VEGF has important cytoprotective properties, VEGF overproduction may alter the pro-survival signalling pathway of VEGF in retinal endothelial cells, 47 resulting in loss of endothelial cells and pericytes as seen in DR, 1 thus highlighting the need to tightly control endogenous VEGF levels to maintain endothelial homeostasis. 48 Normalised VEGF levels through sustained VEGF neutralisation may account for the trend in the reduction of acellular vessels observed in this study. Similarly, anti-VEGF treatment in diabetic rats also caused a reduction in acellular vessels. 49 In patients, anti-VEGF therapy has been associated with reduced rates of capillary closure and non-perfusion which may be explained by re-endothelialisation of persistent basement membranes and reduced VEGF-induced leukostasis.50–52
Our results are limited by the relatively short life span of mice and the duration of diabetes studied. In the clinic, diabetic patients may receive intravitreal anti-VEGF therapy over extensive time-frames. The effect of VEGF neutralisation on the retinal and choroidal vasculature over extended time periods (e.g. several years of treatment) in human patients warrants further investigation. However, mice have a shorter life span compared with humans, and therefore, after the age of adulthood onset in mice (after 12 weeks of age), one human year is equivalent to 2.60 mice days. 53 Anti-VEGF neutralisation was performed over a 4-month period in this study which corresponds to 46 human adult years. Nevertheless, caution must be taken when translating our results into clinical practice due to the fact that diabetic mice only develop mild vascular degeneration. In summary, our data suggest that in a preclinical model of diabetes, sustained VEGF neutralisation for over 4 months does not have negative adverse effects on the structure of retinal and choroidal blood vessels.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Diabetes UK (13/0004729).
