Abstract
Objective
To evaluate whether diabetic retinopathy can be reversed after aflibercept, based on improvements in diabetic macular edema, hard exudates (HEs) of the posterior pole, and retinal microaneurysms (MAs).
Methods
This was a single-center retrospective study of 30 patients (34 eyes) with severe non-proliferative diabetic retinopathy (NPDR) who were treated between August and October 2018. Best-corrected visual acuity (BCVA), central foveal thickness (CFT), area of HEs, and number of MAs were compared before and after treatment.
Results
The mean patient age was 61.4 ± 7.1 years; 14 patients (46.7%) were men. The mean number of injections per patient was 3.5 ± 0.5. The time between the last injection and the last follow-up was 82 days (range, 78–110 days). Six months after the first intravitreal injection, significant improvement was observed in BCVA (from 0.70 ± 0.18 to 0.42 ± 0.19 logMAR), CFT (from 377.17 ± 60.41 to 261.21 ± 31.50 µm), and number of MAs (from 182.2 ± 77.4 to 101.5 ± 59.6). Observations over 6 months after the first intravitreal injection showed a statistically significant reduction in the area of HEs (P = 0.007). No adverse events occurred during the treatment period.
Conclusion
Diabetic retinopathy might be partially reversed by aflibercept treatment, as indicated by BCVA, CFT, number of MAs, and area of HEs.
Keywords
Introduction
Type 2 diabetes mellitus (DM) is a common endocrine disorder characterized by variable degrees of insulin resistance and deficiency, which results in hyperglycemia. 1 Potential complications of type 2 DM include cardiovascular diseases, neuropathy, nephropathy, retinopathy, and elevated mortality. 2 The worldwide prevalences of type 2 DM are 9% in men and 7.9% in women. 3 Type 2 DM is intimately associated with obesity, hyperlipidemia, hypertension, and detrimental lifestyle habits that are characteristic of Western diets.2,4–6 With improvements in life expectancy and living standards in China, the prevalence of diabetes mellitus (DM) has gradually increased, reaching an estimated 110 million individuals (approximately 9% to 12%); complications related to DM are also becoming more prevalent. 7
Diabetic retinopathy (DR) is an eye disease caused by retinal microangiopathy, which occurs at 5 to 10 years after disease onset in most patients with DM.8–10 DR is one of the most common fundus vascular diseases in Chinese individuals >50 years of age and has gradually become one of the major eye diseases leading to blindness.11,12 DR can be classified according to disease severity: 1) no apparent retinopathy; 2) mild non-proliferative DR (NPDR); 3) moderate NPDR; 4) severe NPDR; and 5) proliferative DR.10,13,14
Vascular endothelial growth factor (VEGF) contributes to DR development in patients with DM. 15 Hence, VEGF inhibitors have constituted breakthrough treatments for DR. Indeed, the intravitreal injection of anti-VEGF agents (e.g., ranibizumab) is sufficient for management of DR.16–18 Intravitreal injection of ranibizumab causes reperfusion in ischemic regions of the retina, suggesting that DR may be reversed after treatment with ranibizumab; 19 however, this hypothesis has not been fully tested for other anti-VEGF agents. The CLARITY non-inferiority trial showed that aflibercept is an effective alternative for patients with proliferative DR. 20 Aflibercept is a human recombinant fusion protein 21 that acts as a high-affinity soluble decoy receptor for VEGF and placental growth factor. 22 In the context of DR, VEGF activation enhances endothelial cell permeability, proliferation, survival, and migration.23,24 Therefore, VEGF scavenging with a decoy receptor prevents the activation of VEGF receptors on endothelial cells. 22
To explore whether retinal changes caused by ranibizumab could be elicited by other anti-VEGF agents, this retrospective study evaluated whether DR could be reversed after aflibercept treatment based on improvements in diabetic macular edema, hard exudates (HEs) of the posterior pole, and retinal MAs.
Materials and methods
Patients
This retrospective study included patients with severe NPDR who were treated at Qilu Hospital of Shandong University between August 2018 and October 2018. The Ethics Committee of Qilu Hospital of Shandong University approved this study. The requirement for informed patient consent was waived due to the retrospective study design.
The inclusion criteria were: 1) presence of type 2 DM with hemoglobin A1c <10% and blood pressure <160/90 mmHg; 2) diagnosis of severe NPDR (at least 20 bleeding points in a single quadrant of the retina, visible vein beads in at least two quadrants, or retinal capillary non-perfusion area exceeding five disc areas8,10) on the basis of fundus examination with a VISCAM200 Fundus camera (Carl Zeiss GmbH, Oberkochen, Germany), fundus fluorescein angiography with a Heidelberg SPECTRALIS (INNOVA Medical Ophthalmics, North York, Canada), and optical coherence tomography with a CIRRUS HD-OCT (Carl Zeiss GmbH); and 3) no previous fundus treatment such as retinal photocoagulation or intravitreal injection of anti-VEGF drugs or hormones. The exclusion criteria were poor image quality due to media turbidity and/or concurrent non-diabetic retinal vascular disease.
Treatment
All patients received an intravitreal injection of aflibercept (2 mg, 3 + pro re nata). Aflibercept was injected once per month for 3 months and when necessary thereafter, based on reductions in best-corrected visual acuity (BCVA; measured using an RT-3100 Global Phoropter [Nidek Co., Ltd., Tokyo, Japan]) and changes in central foveal thickness (CFT).
Data collection
Age, sex, side, number of injections, BCVA before and after injection, CFT, area of HEs, and number of MAs were collected from medical records. Color photos of the fundus were measured with an area measurement tool contained in the image analysis software that accompanied the fundus camera. The same experienced doctor manually demarcated the boundary line of all HEs within the 30° range of retina centered on the macular fovea to clearly capture the area of each HE, then added the areas of all HEs in this range to obtain the total area of HEs. All images were reviewed in a blinded manner after they had been shuffled to prevent recognition and sequential analysis of the same patient. Comparisons of BCVA, CFT, HEs, and MAs before and after injection were performed. To unify the comparison of results before and after examination within and between patients, observations of peripheral retinopathy were excluded. The area of HEs and number of MAs were only counted within the 30° range of the retina, with the central macular fovea as the center of the region of analysis. All results were reviewed by a senior ophthalmologist. Because MAs and bleeding were sometimes difficult to distinguish, the observations were confirmed by comparison of color fundus photographs and fundus fluorescein angiography findings. Adverse events during the treatment period were also recorded.
Statistical analysis
Statistical analysis was carried out using SPSS Statistics for Windows, version 21.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as means ± standard deviations and analyzed using the paired t-test. Categorical variables were expressed as n (%) and analyzed using the chi-squared test. P < 0.05 was considered statistically significant.
Results
Thirty patients (34 eyes; mean age, 61.4 ± 7.1 years) were included in this study. Their detailed characteristics are shown in Table 1. Six months after the first injection, BCVA, CFT, and number of MAs all showed significant improvement (all P < 0.001; Table 2, Figure 1). Observations over the 6-month period after the first intravitreal injection showed a significant reduction in the area of HEs (P = 0.007). Notably, HEs were completely absorbed in eight patients (Table 3, Figure 2). Finally, no adverse events occurred during the treatment period.
Characteristics of patients with diabetic retinopathy who received aflibercept in this study

Data are shown as mean ± standard deviation, unless otherwise indicated.
MAs, microaneurysms.
Changes in BCVA, CFT, and number of MAs during treatment

BCVA, best-corrected visual acuity; CFT, central foveal thickness; MA, microaneurysm.

Changes in the number of retinal microaneurysms (MAs) before and after treatment. (a) Early-stage fundus fluorescein angiography image, taken before treatment. (b) Early-stage fundus fluorescein angiography image, taken after 6 months of treatment. The number of MAs is significantly reduced. MAs have completely disappeared in some areas (yellow arrows) and non-perfusion areas show reperfusion (blue arrows)
Changes in the area of HEs during treatment

HEs, hard exudates.
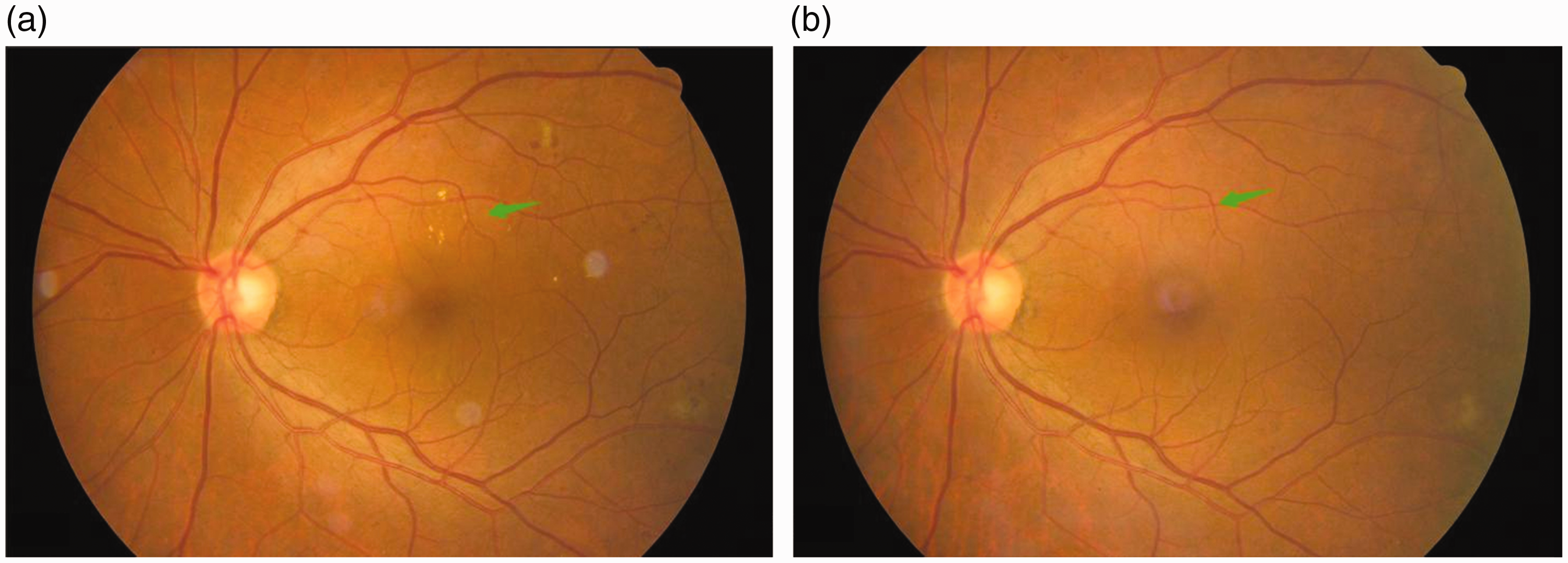
Changes in the area of hard exudates (HEs) before and after treatment. (a) Fundus photograph taken before treatment. (b) Fundus photograph taken after 6 months of treatment. The area of HEs was considerably reduced or absent (green arrows)
Discussion
There is evidence to suggest that DR is reversible after treatment with anti-VEGF agents, 19 but this hypothesis was not previously confirmed for aflibercept. Therefore, this study aimed to evaluate whether DR can be reversed after aflibercept treatment based on changes in diabetic macular edema, HEs of the posterior pole, and MAs. The results suggested that NPDR could be partially reversed by aflibercept treatment, as demonstrated by BVCA, CFT, number of MAs, and area of HEs.
In this retrospective study, we found that treatment of DR with intravitreal injections of aflibercept could reverse both morphological and functional DM-related changes in the retina. These findings are consistent with those of a previous study concerning intravitreal ranibizumab injections. 19 Indeed, anti-VEGF therapy is now a recognized treatment for diabetic macular edema; it can significantly inhibit the growth of new blood vessels, reduce edema, and eventually improve vision.16–18 Our results demonstrated improvements in BCVA and CFT after aflibercept treatment, in agreement with the findings of the CLARITY trial that compared aflibercept with pan-retinal photocoagulation;20,25,26 our results are also consistent with the findings of a phase IV study in which a treat-and-extend regimen of aflibercept was found to improve visual acuity and retinal edema in eyes with Type 3 neovascularization, with good tolerability over 52 weeks. 27 Previous studies showed that when an anti-VEGF treatment was administered to patients with retinal venous obstruction, the anti-VEGF group displayed delayed progression of non-perfusion areas and showed improved reperfusion, compared with the control group. 28 Importantly, our retrospective study was unable to determine causality, but its results imply that aflibercept can considerably modify DR; the findings require confirmation in future prospective studies.
The reperfusion of retinal non-perfusion areas has also been observed in prospective studies of patients with DR following anti-VEGF treatment; 29 however, there have been no reports concerning improvements in HEs and MAs, which are common in patients with DR, after such treatment. Our results showed that patients with NPDR experienced reductions in the area of HEs and in the number of MAs after 6 months of treatment with aflibercept. Previous studies have shown that the mechanism of MAs associated with proliferative DR, including local structural fragility of the vascular wall accompanied by vasodilation and the proliferation of local vascular endothelial cells, involves the thickening of substrate membrane in blood vessels and the loss of peripheral cells. 30 In primates, elevated concentrations of endothelial cell growth factors in the vitreous body can lead to the formation of MAs. 31 Therefore, the reduction in number of MAs was presumed to be related to the intravitreal injection of aflibercept; the mechanism might involve apoptotic degradation after the loss of cytokines in proliferative endothelial cells following anti-VEGF treatment. Nevertheless, due to the limitation of the retrospective study design, we could not explore the molecular mechanism underlying this reduction in MAs; our present findings provide an important basis for subsequent prospective studies.
HEs constitute a fundus manifestation of DR, primarily consisting of dilated capillaries, as well as lipid and protein components of MA leakage.8–10 HEs are generally deposited under the outer and inner retina. Increased numbers of HEs can enhance the risk of visual impairment, while severe HEs in the central retina can enhance the risk of subretinal fibrosis. 32 We presume that the cause of the reduction in HE area after anti-VEGF treatment might have been related to the lower number of MAs and alleviation of leakage due to reperfusion in the non-perfusion area, but we cannot rule out the possibility that anti-VEGF treatment directly acted on HEs and promoted their absorption.
This study had some limitations. First, the sample size was small and no control group was included, which limits the interpretation and generalizability of the results. Second, the retrospective design prevented assessment of causal relationships, although we suspect that injections might promote morphological improvements by enhancing the health of retinal blood vessels; this hypothesis should be tested in future studies. Third, we could not investigate whether the degeneration of abnormal vascular endothelial cells contributed to the reversal of DR after anti-VEGF treatment. Larger, prospective studies with longer follow-up are needed to examine this point. Fourth, changes in the peripheral retina are affected by the examination angle and patient cooperation with physicians. Therefore, to unify the standard of comparison, only a 30° area with macular fovea as its center was selected for comparison.
In conclusion, our results suggest that DR can be reversed by aflibercept treatment, as demonstrated by BVCA, CFT, number of MAs, and area of HEs.
