Abstract
Objective:
We examined the risk of myocardial infarction associated with glucose-lowering therapy among diabetes patients with and without obstructive coronary artery disease.
Methods:
A cohort of patients with type 1 or type 2 diabetes (n = 12,030), who underwent coronary angiography from 2004 to 2012, were stratified by presence of obstructive (any stenosis ⩾50%) coronary artery disease and by type of diabetes treatment: diet, non-insulin treatment and insulin (±oral anti-diabetics). The primary endpoint was myocardial infarction. Adjusted hazard ratios were calculated using diet-treated patients without coronary artery disease as reference.
Results:
In patients without coronary artery disease, risk of myocardial infarction was similar in patients treated with non-insulin medication (adjusted hazard ratio 0.70, 95% confidence interval 0.27–1.81) and insulin (adjusted hazard ratio 0.76, 95% confidence interval 0.27–2.08) as compared to diet only. In patients with coronary artery disease, the risk of myocardial infarction was higher than in the reference group and an incremental risk was observed being lowest in patients treated with diet (adjusted hazard ratio 3.79, 95% confidence interval 1.61–8.88), followed by non-insulin medication (adjusted hazard ratio 5.42, 95% confidence interval 2.40–12.22), and highest in insulin-treated patients (adjusted hazard ratio 7.91, 95% confidence interval 3.51–17.82).
Conclusion:
The presence of obstructive coronary artery disease defines the risk of myocardial infarction in diabetes patients. Glucose-lowering therapy, in particular insulin, was associated with risk of myocardial infarction only in the presence of coronary artery disease.
Introduction
Current guidelines recommend primary prophylactic treatment (statins, antihypertensive treatment and antiplatelet therapy) together with lifestyle interventions in diabetes patients at increased cardiovascular risk.1–3 While guidelines differ on the definition of high-risk diabetes patients, the American Diabetes Association 1 , European Society of Cardiology 2 and International Diabetes Federation 3 do agree on the following risk factors: older age, hypertension, dyslipidemia and albuminuria. Due to uncertainties concerning the benefits of aspirin, their recommendations are ambiguous concerning use of antiplatelet therapy, with aspirin as a primary preventive, for high-risk diabetes patients. 4
The landmark study by Haffner et al. 5 reported the risk of myocardial infarction (MI) in diabetes patients to be equal to that of patients without diabetes with a previous MI. A subsequent Danish nationwide study confirmed these results and suggested that diabetes patients receiving glucose-lowering therapy should be considered for an intensive primary prevention intervention. 6 However, the study did not take into account whether patients had coronary artery disease (CAD).
A Danish study, based on the Western Denmark Heart Registry (WDHR), demonstrated that cardiovascular risk in diabetes patients depends on the presence or absence of obstructive CAD, but did not stratify these results by type of glucose-lowering therapy. 7 The current study combined assessment of glucose-lowering therapies with information on CAD.
Research design and methods
Data sources
We used the WDHR to identify all diabetes patients who underwent coronary angiography (CAG) between 1 July 2004 and 31 December 2012. 8 The WDHR is a clinical database of information on all adult (⩾15 years) patients in Western Denmark (population of 3.3 million persons) referred for diagnostic, invasive and surgical procedures, including CAG, percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG), since 1999. This Registry was recently described in detail. 9 The WDHR is an Internet-based online system, in which data are entered by physicians using their patients’ unique 10-digit identifier. 10 This personal identifier, assigned to each Danish citizen at birth and to residents upon immigration, is included in every Danish medical database. The following databases were used in the current study: the Danish Civil Registration System, which contains data on all Danish citizens’ vital status (dead, alive or emigrated); the Danish National Patient Registry, which records all inpatient and outpatient hospital diagnoses; and the Danish National Health Service Prescription Database, which provides data on all reimbursed prescriptions redeemed at Danish pharmacies.8,11,12
Diabetes
Diabetes patients were identified through the WDHR and the Danish National Health Service Prescription Database. We were unable to distinguish between type 1 and type 2 diabetes. Diabetes patients were divided into three groups according to treatment. Insulin treatment was defined as receipt of insulin (±supplementary oral anti-diabetes medication), as recorded in the WDHR, or redemption of ⩾1 prescription for insulin ± non-insulin diabetes medication [Anatomical Therapeutic Chemical (ATC) codes in Supplemental Material, Table S1] within 6 months before or 1 month after CAG, as recorded in the Danish National Health Service Prescription Database. Non-insulin treatment was defined as oral diabetes treatment recorded in the WDHR or redemption of ⩾1 prescription for non-insulin diabetes medication [including oral agents and glucagon-like peptide 1 (GLP-1) receptor agonists], as recorded in the Danish National Health Service Prescription Database, and no insulin prescription within 6 months before or 1 month after CAG. Dietary treatment was defined as non-pharmacological dietary treatment recorded in the WDHR and no redeemed prescription for insulin or non-insulin diabetes medication within 6 months before or 1 month after CAG.
CAD
Obstructive CAD was defined as ⩾1 coronary artery with ⩾50% angiographic lumen narrowing ascertained from the WDHR. Absence of CAD was defined as no stenosis in all coronary arteries or mild angiographic lumen narrowing (<50%) in a single coronary vessel.
Patient selection
Patients with a CAG procedure registered in the WDHR between 1 July 2004 and 31 December 2012 were included in this study (Figure 1). If a patient underwent multiple CAG examinations during the study period, the first CAG was considered the index procedure. Patients without diabetes (n = 82,980) and patients <18 years old (n = 2) were excluded. Patients with diffuse CAD, defined as diffuse atherosclerosis in >1 coronary vessel with <50% stenosis, also were excluded (n = 1360). In addition, we excluded 424 patients due to high likelihood of misclassification (i.e. patients classified as having ‘no CAD’ despite a recorded history of previous MI, PCI and/or CABG). We performed two sensitivity analyses in which the latter 424 patients were classified as either having or not having obstructive CAD (data not shown). Patients who either died or emigrated <30 days after CAG (n = 525) also were excluded from the study.
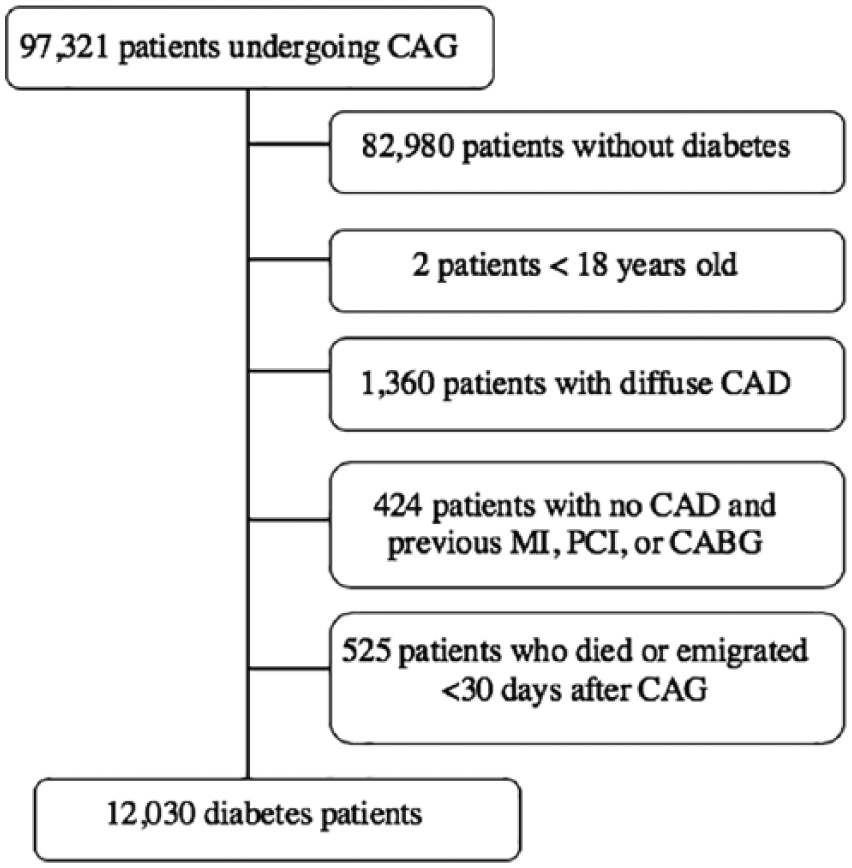
Flowchart of selection of diabetes patients who underwent CAG in Western Denmark between 1 July 2004 and 31 December 2012.
Comorbidity
Comorbidity was assessed using the Charlson Comorbidity Index score, based on ICD-10 codes (Supplemental Material, Table S2) registered in the Danish National Patient Registry 5 years before the study inclusion date. 13 A modified Charlson’s Comorbidity Index score (excluding ‘diabetes’ and ‘diabetes with end-organ damage’) was estimated for each patient on the CAG date.
Medication
Records of treatment with a statin, aspirin, adenosine diphosphate receptor (ADP) inhibitor, angiotensin-converting enzyme (ACE) inhibitor/angiotensin II receptor blocker (ARB) and β-blocker were obtained from the Danish National Health Service Prescription Database (ATC codes in Supplemental Material, Table S1). 12 Medical treatment was defined as reimbursement of a redeemed prescription between 6 months before and 1 month after the index CAG.
MI
MI events were identified in the Danish National Patient Registry using the ICD-10 code for MI (Supplemental Material, Table S2). Follow-up began 30 days after CAG since a previous study showed less validity of the MI diagnosis within 30 days after CAG due to interhospital patient transfers and initial double registration of MI. 14 The sensitivity (94%) and specificity (99%) of the MI diagnosis 30 days after CAG are high. 14
All-cause death
Patients’ vital status (dead, alive or emigrated) was ascertained through the Danish Civil Registration System. 15
Statistical analyses
Follow-up began 30 days after the CAG procedure due to the above-mentioned uncertainties about the MI diagnosis before this. For MI, follow-up continued until MI, death, emigration or end of follow-up (31 December 2012), whichever occurred first. For death, follow-up continued until death, emigration or end of follow-up. Event rates for MI and death were estimated per 100 person-years. We used Cox proportional hazard regression to estimate unadjusted and adjusted hazard ratio (HR and adjHR) of MI, using patients without obstructive CAD, who were receiving dietary treatment, as reference. The proportional hazard assumption was met in the case of MI, but not for death. We therefore used the modified Poisson regression, with the event as the outcome and the natural log of person-years as the offset, to estimate unadjusted and adjusted incidence rate ratio (IRR and adjIRR) of death. 16 We adjusted for age, sex, the modified Charlson Comorbidity Index score, antihypertensive treatment, smoking status, statin treatment, ADP inhibitor treatment and aspirin treatment. We conducted a subgroup analysis using patients with obstructive CAD, who were treated with diet, as reference. We also performed a test for trend in each endpoint stratified for CAD status. A subgroup analysis for patients with diffuse CAD is provided in Supplemental Material, Table S3. We used Stata/IC software version 13.1 (StataCorp, College Station, TX, USA) for all statistical analyses.
Results
A total of 12,030 diabetes patients were included in the analyses. Of these, 8672 (72.1%) had obstructive CAD and 3358 (27.9%) had no obstructive CAD at angiography. Among patients with obstructive CAD, 3229 (37.2%) were treated with insulin (±non-insulin diabetes medication), 4669 (53.8%) received non-insulin diabetes medication and 774 (8.9%) received non-pharmacological dietary treatment. Among patients without obstructive CAD, 1104 (32.8%) were treated with insulin (±non-insulin diabetes medication), 1847 (55.0%) received non-insulin diabetes medication and 407 (12.1%) received non-pharmacological dietary treatment. Median follow-up-time was 3.0 years (interquartile range 1.0–5.0 years), with maximum follow-up of 8.4 years.
Baseline characteristics
Baseline characteristics at time of CAG are summarized in Table 1. Patients with obstructive CAD were more often male, older and active smokers than patients without obstructive CAD. Moreover, patients with obstructive CAD were more often treated with aspirin, statins, β-blockers and ADP inhibitors.
Baseline characteristics of diabetes patients with and without obstructive CAD as ascertained by CAG, by type of diabetes treatment, Denmark, 2004–2012 (%, unless otherwise indicated).
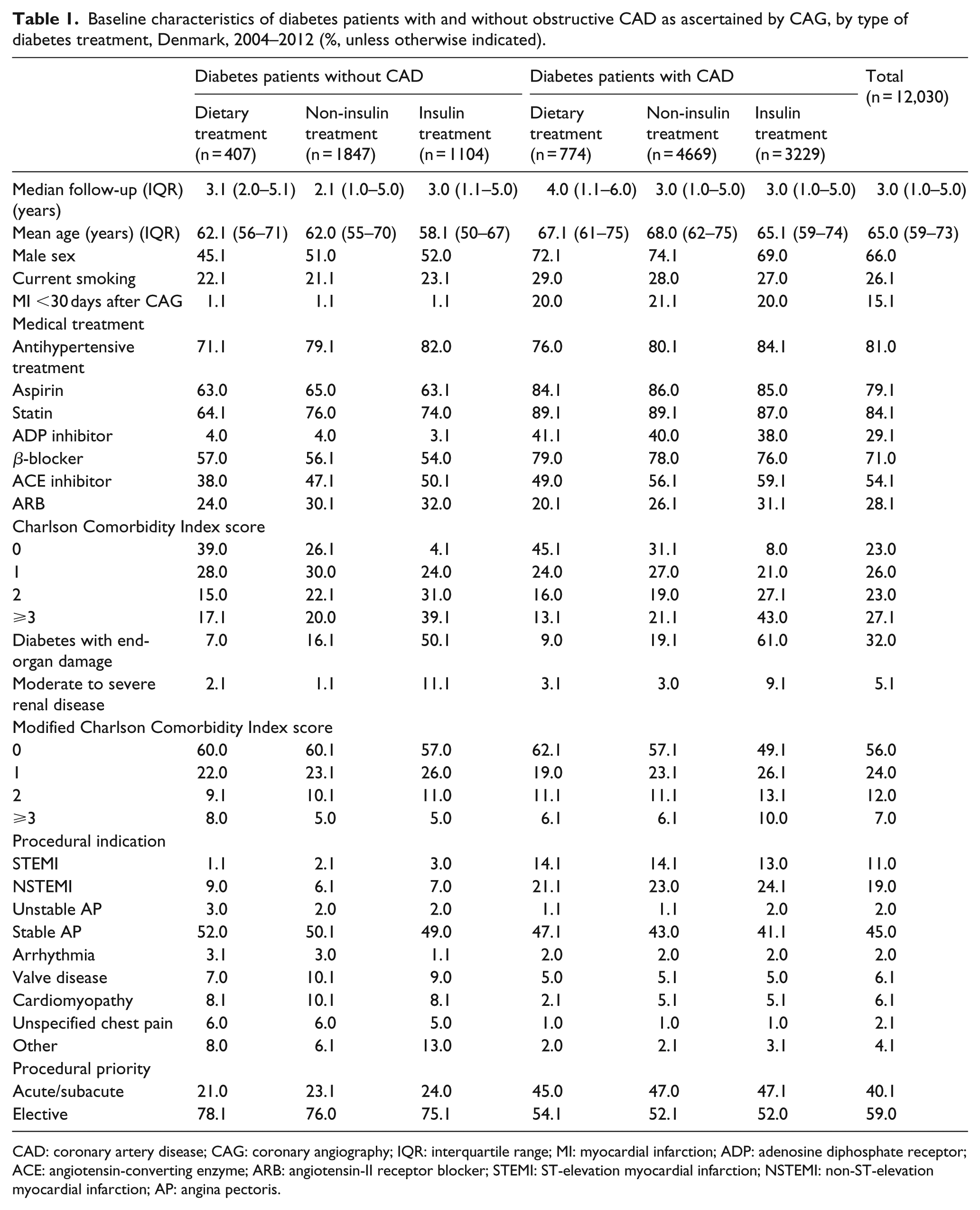
CAD: coronary artery disease; CAG: coronary angiography; IQR: interquartile range; MI: myocardial infarction; ADP: adenosine diphosphate receptor; ACE: angiotensin-converting enzyme; ARB: angiotensin-II receptor blocker; STEMI: ST-elevation myocardial infarction; NSTEMI: non-ST-elevation myocardial infarction; AP: angina pectoris.
Clinical endpoints
Numbers of events during the follow-up period stratified by the presence or absence of obstructive CAD and diabetes treatment strategy are summarized in Table 2. Cumulative 5-year incidence curves of MI and death, representing up to 75 percentiles, are shown in Figure 2.
Number of events and risk of myocardial infarction and death in patients with diabetes, by the presence or absence of obstructive CAD and type of treatment.
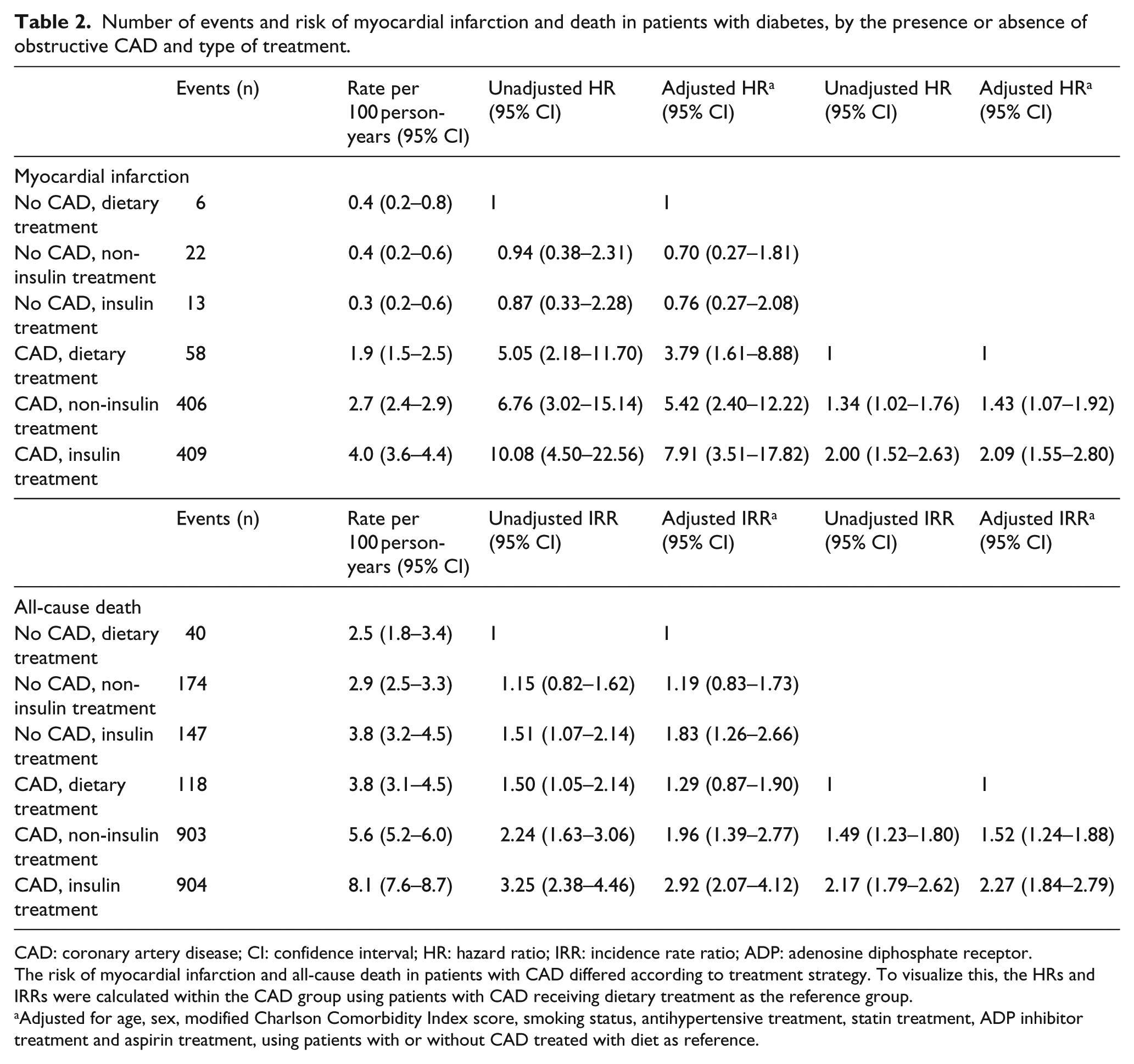
CAD: coronary artery disease; CI: confidence interval; HR: hazard ratio; IRR: incidence rate ratio; ADP: adenosine diphosphate receptor.
The risk of myocardial infarction and all-cause death in patients with CAD differed according to treatment strategy. To visualize this, the HRs and IRRs were calculated within the CAD group using patients with CAD receiving dietary treatment as the reference group.
Adjusted for age, sex, modified Charlson Comorbidity Index score, smoking status, antihypertensive treatment, statin treatment, ADP inhibitor treatment and aspirin treatment, using patients with or without CAD treated with diet as reference.
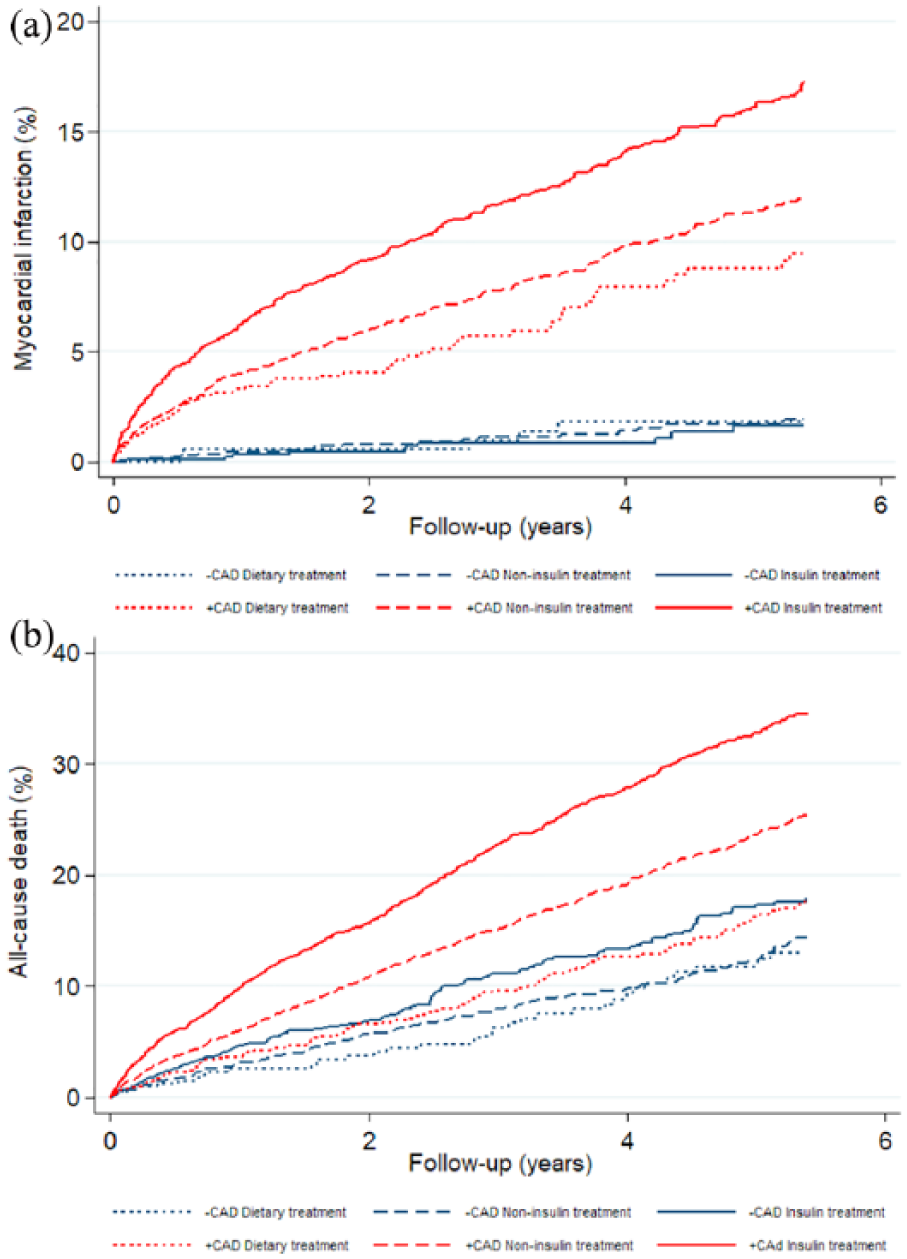
Accumulated rates of myocardial infarction (a) and all-cause death (b) in diabetes patients with (+CAD) and without (–CAD) obstructive coronary artery disease (CAD) treated with insulin, non-insulin diabetes medication or diet-only. The follow-up period represents the 75th percentile.
Patients without obstructive CAD treated with diet (reference group) had the lowest risk of MI (0.4 per 100 person-years). In patients without obstructive CAD, risk of MI was comparable to the reference group among patients treated with non-insulin medication [0.4 per 100 person-years; adjHR 0.70, 95% confidence interval (CI) 0.27–1.81] and insulin (0.3 per 100 person-years; adjHR 0.76, 95% CI 0.27–2.08). In patients with obstructive CAD, the risk of MI was higher than in the reference group and an incremental risk was observed being lowest in patients treated with diet (1.9 per 100 person-years; adjHR 3.79, 95% CI 1.61–8.88), followed by patients treated with non-insulin medication (2.7 per 100 person-years; adjHR 5.42, 95% CI 2.40–12.22) and highest in insulin-treated patients (4.0 per 100 person-years; adjHR 7.91, 95% CI 3.51–17.82).
Patients without obstructive CAD treated with diet (reference group) also had the lowest risk of death (2.5 per 100 person-years). In patients without obstructive CAD, risk of death was comparable to the reference group among patients treated with non-insulin medication (2.9 per 100 person-years; adjIRR 1.19, 95% CI 0.83–1.73), but higher for insulin-treated patients (3.8 per 100 person-years; adjIRR 1.83, 95% CI 1.26–2.66). As for MI, patients with obstructive CAD had incremental risks of death with diet (3.8 per 100 person-years; adjIRR 1.29, 95% CI 0.87–1.90), non-insulin medication (5.6 per 100 person-years; adjIRR 1.96, 95% CI 1.39–2.77) and insulin treatment (8.1 per 100 person-years; adjIRR 2.92, 95% CI 2.07–4.12).
For patients with obstructive CAD and receiving diet, non-insulin and insulin treatment, respectively, the incremental risk of MI and death among diabetes patients was highly significant (ptrend < 0.001 for MI and ptrend < 0.001 for death). For patients without obstructive CAD and receiving diet, non-insulin and insulin treatment, respectively, there was no incremental risk of MI (ptrend = 0.81) but still an incremental risk of death (ptrend < 0.005).
Sensitivity analyses
The results of the two sensitivity analyses with different handling of possibly misclassified patients showed no major impact on risk of MI or death (data not shown).
Discussion
The results of the present study confirmed that the presence or absence of obstructive CAD defines the risk of MI in diabetes patients. More importantly, we also demonstrated that choice of diabetes treatment – insulin, non-insulin diabetes medication and diet – was associated with an incremental risk of MI in diabetes patients with obstructive CAD, but not in diabetes patients without obstructive CAD. Thus, assessment of CAD can be used to stratify diabetes patients into those at high risk and those at low risk of MI, while diabetes treatment differentiates MI risk only in patients with obstructive CAD.
Haffner et al.’s 5 landmark study suggested that diabetes patients without prior MI had risks of MI and death similar to non-diabetes patients with previous MI. Since then, prophylactic treatment of cardiovascular risk factors such as hypertension and dyslipidemia in diabetes patients has been intensified,1,2 perhaps contributing to the lower diabetes-related risk of MI found in subsequent studies.17–19 Schramm et al. 6 used Danish national registries to compare patients requiring glucose-lowering therapy with non-diabetes patients with a previous MI. They concluded that diabetes patients receiving glucose-lowering therapy exhibited a cardiovascular risk comparable to non-diabetes patients with a prior MI and consequently suggested that a requirement for glucose-lowering therapy should prompt intensive prophylactic treatment for cardiovascular diseases. However, a closer look at the data showed that risk of MI in the diabetes group was nearly half as great as the risk for non-diabetes patients with a prior MI, while risk of stroke was similar. 6 Unlike the current study, Schramm et al.’s study did not take the presence or absence of obstructive CAD into account. Thus, the approach suggested by Schramm et al. implies that patients without obstructive CAD, and therefore at low risk of MI, are considered as high-risk patients.
Based on our WDHR cohort, we recently reported that diabetes and non-diabetes patients had the same low risk of MI in the absence of obstructive CAD as ascertained by CAG, while diabetes patients with obstructive CAD had a particularly high risk of MI. Saely et al. 20 studied a cohort of 750 patients undergoing CAG (mean follow-up period of 2.2 years) and consistently found that the presence of ⩾50% stenosis at CAG was a strong independent predictor for vascular events, while the diabetic state itself was not a significant independent predictor for vascular events. Moreover, if coronary atherosclerosis was already present, diabetes added a further risk to that of obstructive CAD. Likewise, using coronary computer tomography angiography (CCTA) to determine CAD status, Van Werkhoven et al. 21 examined 616 patients (mean follow-up period of 20 months) and found a cardiac event rate of 0% in both diabetes patients and non-diabetes patients without obstructive CAD; the cardiac event rate increased to 36% in non-diabetes patients with obstructive CAD and was highest (47%) in diabetes patients with obstructive CAD. These results underscore the importance of accounting for the presence or absence of obstructive CAD when estimating cardiovascular risk in diabetes patients.
The current study confirms previous findings showing that glucose-lowering therapy is associated with MI risk, but adds that this association only appears in patients with obstructive CAD.6,7 Thus, by assessing the presence or absence of obstructive CAD, it seems possible to identify a large number of diabetes patients receiving glucose-lowering treatment as being at low risk of MI. Going forward, diabetes patients may be risk-stratified according to the presence of obstructive CAD and hence receive modified prophylactic treatment. Intensified prevention may prove beneficial in diabetes patients at high risk of MI, that is, those with obstructive CAD.
Most diabetes patients should receive lifestyle interventions, glucose-lowering therapy, statins and antihypertensive treatment as primary prophylaxis according to guidelines.1–3 Patients with established cardiovascular disease should further receive a sodium-glucose cotransporter-2 (SGLT-2) inhibitor or GLP-1 analogue. 22 Until recently, glucose-lowering therapies failed to reduce the risk of macrovascular disease. However, randomized clinical trials, such as the EMPA-REG [(Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients)], LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results) and SUSTAIN-6 (Trial to Evaluate Cardiovascular and Other Long-term Outcomes with Semaglutide in Subjects with Type 2 Diabetes) demonstrated a lower risk of cardiovascular morbidity and mortality among patients with type 2 diabetes at high risk for cardiovascular events when treated with these study drugs on top of standard care.23–25 Noticeable, SGLT-2 inhibitors only have moderate benefits on atherosclerotic cardiovascular events seemingly confined to patients with established CAD, whereas they have stronger benefits on reducing hospitalization for heart failure regardless of existing CAD or a history of heart failure. 26 These SGLT-2 inhibitors and GLP-1 analogues may also be appropriate as secondary prophylaxis in diabetes patients with obstructive CAD. In contrast, benefit of primary prophylactic treatment in low-risk diabetes patients without obstructive CAD will be difficult to document based on the low event rates found in this study. In our cohort, 64.5% of patients without obstructive CAD continued aspirin treatment after CAG. The disadvantages of aspirin, such as increased risk of gastrointestinal bleeds, might outweigh the benefits in diabetes patients without obstructive CAD, as recently documented by the large-scale ASCEND (A Study of Cardiovascular Events in Diabetes) study. 1
Routine screening for CAD in asymptomatic diabetes patients is controversial.27,28 The American Diabetes Association currently advises against screening for CAD in asymptomatic patients based on the lack of evidence. 1 Only a single randomized clinical trial has examined screening for CAD in asymptomatic using CCTA. 29 This trial found that screening for CAD was not beneficial in preventing cardiac events and death. However, patients were randomized prior to CCTA. This approach means that a large proportion of low-risk diabetes patients with normal or non-obstructive CAD were included. The consequences were that the event rates were lower than expected, and that the study was underpowered. There are currently no studies that have used CCTA to identify high-risk asymptomatic patients followed by randomization of these to novel secondary prophylactic strategies, such as SGLT-2 inhibitors and GLP-1 analogues, with documented effect in symptomatic patients with established CAD. The current study included symptomatic diabetes patients and it primarily shows that presence of obstructive CAD, as assessed by imaging, can be used for risk stratification beyond the currently available tools. However, secondary prophylactic intervention in asymptomatic diabetes patients with confirmed CAD, as assessed by CCTA, needs to be confirmed in prospective randomized clinical trials with adequate power.
In the current study, CAD status was determined by CAG. While this invasive approach yields information about obstructive CAD, detailed information about vessel wall characteristics and non-obstructive CAD is insufficient compared to CCTA. A previous study has shown that anatomic characteristics of CAD according to CCTA confer valid prognostic information, ensuring accurate risk stratification among diabetes patients. 30 Nevertheless, this strategy requires further investigation.
In the present study, we observed increased mortality in patients without CAD treated with insulin compared to patients without CAD treated with non-insulin medication or diet. However, the MI rate was similar in these three groups. Patients with type 2 diabetes treated with insulin typically have more co-morbidities as also found in this study where diabetes-related end-organ damage, including chronic renal disease, was more frequent among insulin-treated patients. Therefore, the increased mortality in insulin-treated patients without CAD is likely associated with these co-morbidities rather than caused by ischemic heart disease.
A limitation of this study is that we could not differentiate between type 1 and type 2 diabetes. However, only patients with type 2 diabetes were treated through diet or received non-insulin diabetes medication, while a combination of patients with type 1 and type 2 diabetes patients received insulin ± non-insulin diabetes medication. Furthermore, we were unable to determine the duration of diabetes. The Framingham Heart Study demonstrated that duration of diabetes increased coronary mortality independently of other coexisting risk factors. 31 Several other studies showed consistent results.32,33 Moreover, in the current study, we could not differentiate between various types of non-insulin anti-diabetes treatments. The significant association between glucose-lowering therapy, particularly insulin, and cardiovascular risk dependent on CAD status is most likely due to the duration and/or severity of diabetes rather than a direct effect of the specific glucose-lowering therapy. Patients with diffuse non-significant CAD were excluded from the main analysis as we were uncertain whether to characterize these patients as having CAD or not. A separate analysis was provided in the Supplement, indicating that diffuse non-significant CAD may be considered a comparable risk factor to patients with significant obstructive CAD. Another concern is that the current study was limited to a maximum follow-up of 8.4 years and the interpretation of the data cannot yet be extended beyond this period. A previous study observed many additional complications during extra follow-up as patients were older and had a longer duration of diabetes. 34 Finally, the treating physician evaluated classification of CAD by CAG, which probably is associated with a minor degree of inter-observer variation.
In conclusion, the major finding of this study is that the presence or absence of obstructive CAD defined the risk of MI in diabetes patients, while glucose-lowering therapy, particularly insulin, was associated with risk of MI only in the presence of obstructive CAD. Among diabetes patients, the presence of obstructive CAD was thus a stronger predictor of MI than the requirement for glucose-lowering therapy.
Supplemental Material
Supplementary_Material – Supplemental material for Association between anti-diabetes treatments and cardiovascular risk in diabetes patients with and without coronary artery disease
Supplemental material, Supplementary_Material for Association between anti-diabetes treatments and cardiovascular risk in diabetes patients with and without coronary artery disease by Christine Gyldenkerne, Kevin Kris Warnakula Olesen, Morten Madsen, Troels Thim, Lisette Okkels Jensen, Bent Raungaard, Henrik Toft Sørensen, Hans Erik Bøtker and Michael Maeng in Diabetes & Vascular Disease Research
Footnotes
Declaration of conflicting interests
MM has received fees for advisory board meetings and lectures from Novo Nordisk, Bristol-Myers Squibb, AstraZeneca, Boehringer Ingelheim and Bayer. KKWO has received speaking fees from Bayer.
Ethical approval
This study complies with the Declaration of Helsinki and was approved by the Danish Data Protection Agency (record number 2015-57-0002, identification number AU420).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This is an academic study funded by the Aarhus University Hospital, Department of Cardiology, Research Unit.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
