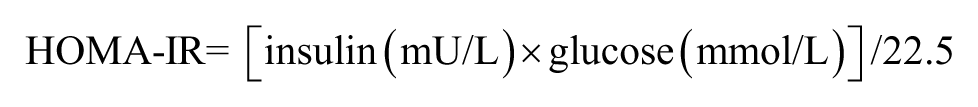Abstract
Background:
Reaven originally described the clustering of insulin resistance/hyperinsulinaemia, obesity (particularly visceral), altered cytokine levels, glucose intolerance, hypertriglyceridaemia and low high-density lipoprotein cholesterol. Subsequently, a potentially highly atherogenic small, dense low-density lipoprotein was also reported. We have studied the effect of bariatric surgery on this and other risk factors for atherosclerosis.
Methods:
Forty patients (20 with type 2 diabetes mellitus) undergoing bariatric surgery were studied before and 1 year after bariatric surgery.
Results:
Twelve months after bariatric surgery, median body mass index had decreased from 49.5 to 36.5 kg/m2, fasting insulin from 21.3 to 7.8 mU/L and insulin resistance (homeostatic model assessment of insulin resistance) from 5.9 to 1.8 (all p < 0.001). Thirteen out of 20 patients had remission from type 2 diabetes mellitus. Highly sensitive C-reactive protein, interleukin-6, fasting triglycerides (p < 0.001) and small, dense low-density lipoprotein (p < 0.001) decreased, while high-density lipoprotein cholesterol increased (p < 0.001) significantly, irrespective of having type 2 diabetes mellitus and/or being treated with statin therapy before surgery.
Conclusion:
The association between marked weight loss and change in insulin resistance and hyperinsulinaemia with the change in small, dense low-density lipoprotein and interleukin-6 warrants further investigation. Bariatric surgery provides a model for investigating the mechanisms linking insulin resistance/hyperinsulinaemia to atherosclerosis.
Keywords
Introduction
The advent of reliable plasma insulin assays in the 1960s provided the opportunity for Gerald Reaven et al. 1 to discover the association between the insulin response to carbohydrate feeding and serum triglyceride levels. Higher insulin responses were associated with higher triglyceride levels. 1 Reaven and others went on to report that a constellation of other abnormalities was also associated with an exaggerated insulin response in addition to hypertriglyceridaemia, including type 2 diabetes mellitus (T2DM) or a predisposition to it, high-density lipoprotein cholesterol (HDL-C), non-alcoholic steatohepatitis, hypertension, hyperuricaemia and raised plasminogen activator inhibitor-1 (PAI-1), fibrinogen and highly sensitive C-reactive protein (hs-CRP).2–5 This, he termed ‘Syndrome X’, 2 although it is now more widely known as the metabolic syndrome, particularly when associated with obesity. Initially, Reaven’s hypothesis stated that increased insulin levels were the cause of the hypertriglyceridaemia, because insulin was at that time believed to stimulate hepatic very low-density lipoprotein (VLDL) secretion. 1 However, later experiments with adult hepatocytes maintained in tissue culture showed that the primary effect of insulin on hepatic VLDL secretion was inhibitory.6–8 Thus, the hypertriglyceridaemia was due to insulin resistance, rather than hyperinsulinaemia, as previously proposed by Himsworth 9 many years previously. Reaven thus modified his hypothesis and extended it to state that both muscle and the liver must be resistant to the action of insulin, at least in relation to their diminished capacity for the uptake of glucose, and therefore, the increased insulin levels were a response to overcome the insulin resistance. That hepatic insulin resistance could also explain the hypertriglyceridaemia of metabolic syndrome and T2DM, was later confirmed in human studies of VLDL kinetics.10–12 Throughout his life Reaven continued to argue, however, that it was impossible using available techniques and models to separate the effects of insulin resistance from the effects of too much insulin in humans. 13 It is known that insulin resistance due to inherited insulin receptor defects results in reduced glucose uptake and hyperinsulinaemia, but does not lead to hypertriglyceridaemia or hepatic steatosis. 14 However, after its uptake by functioning receptors, insulin regulates multiple intracellular pathways through several signalling mechanisms. 15 It therefore remains entirely possible that some of these are resistant to insulin, whereas, as Reaven originally postulated, others are over-stimulated by the hyperinsulinaemia which develops to maintain euglycaemia. When the increased delivery of insulin is inadequate to overcome insulin resistance to glucose uptake, T2DM develops, but the insulin levels are much higher than in healthy, non-obese, people without diabetes. 16
The provision of evidence that hyperinsulinaemia/insulin resistance is causal for hypertriglyceridaemia and the other components of the metabolic syndrome was, until the advent of bariatric surgery, hampered by the lack of a means of dramatically reversing it. Drugs which decrease insulin resistance tend to have multiple other actions and the effect of weight reduction through dietary restriction can only be studied when a large proportion of failures are excluded. Bariatric surgery provides a means of substantially and consistently reversing hyperinsulinaemia/insulin resistance. While the substantial decrease in adiposity may explain this reversal of hyperinsulinaemia/insulin resistance, additional mechanisms such as changes in gut hormone profiles due to intestinal bypass may contribute. We, and others, have previously reported a decrease in elevated levels of inflammatory cytokines and an increase in adiponectin after bariatric surgery.17–20 These cytokines emanate from adipose tissue and those from visceral adipose tissue, particularly interleukin-6 (IL-6), arrive at the liver via the portal circulation, 21 and are believed to be responsible for hepatic resistance to insulin-mediated glucose uptake. 22
Quite why the metabolic syndrome is associated with an increased risk of atherosclerotic cardiovascular disease (CVD) has never been fully explained. Increases in low-density lipoprotein cholesterol (LDL-C) and in its major protein moiety, apolipoprotein B (ApoB), which are definitely causal, are not a feature of metabolic syndrome.2,23 Moderate hypertriglyceridaemia, typical of the metabolic syndrome has proved controversial as a cause of CVD, 24 and the role of high-density lipoprotein (HDL) in atherogenesis is currently being re-evaluated. 25 We have previously made a preliminary report of a decrease in small, dense low-density lipoprotein (SD-LDL) following bariatric surgery. 18 SD-LDL is increased in hypertriglyceridaemia 26–28 and is particularly susceptible to atherogenic modifications, such as oxidation and glycation.29,30 Inflammatory cytokines associated with atherothrombosis may also make a major contribution to CVD in metabolic syndrome. 31 In the present study, we have undertaken a comprehensive assessment of the effect of bariatric surgery on insulin secretion and insulin resistance in relation to the change in SD-LDL and IL-6, an upstream regulator of C-reactive protein (CRP), 4 as there is emerging evidence from Mendelian randomisation studies that it has a longer term association with CVD than hs-CRP.32,33 We have sought to establish the basis for the change in insulin secretion and insulin resistance after bariatric surgery, to further understand the basis of the Reaven hypothesis.
Methods
Study design and patient recruitment
This study was a prospective observational cohort study. Forty patients (8 men and 32 women) with obesity were recruited from the pre-bariatric surgery clinic at Salford Royal Hospital, a Tier 4 specialist weight management service in the North West of England. They all underwent Roux-en-Y laparoscopic gastric bypass surgery.
Prior ethical approval was sought and granted by the Central Manchester Research and Ethics Committee. All patients were given detailed information about the study and each person provided informed consent before taking part in the study.
Patient assessments
Patients were asked to attend a morning appointment (between 09:00 and 10:30 h) having fasted from 22:00 h at baseline and then 6 and 12 months after bariatric surgery. At each visit, a detailed medical history including medication used was assessed. Each participant underwent measurement of their weight and height and body mass index (BMI) was calculated. Blood pressure was measured after resting in a seated position for 15 min, using an Omron HEM 705-CP semiautomatic oscillometric recorder. Fasting venous blood was collected at each visit. Metabolic syndrome was defined using the current revision from the National Cholesterol Education Programme Adult Treatment Panel III. 23
Complete remission from type 2 diabetes was determined 12 months post-operatively with glycated haemoglobin (HbA1c) below 6.0% (42 mmol/mol) and no active pharmacological therapy, as per the American Diabetes Association consensus statement. 34
Laboratory procedures and analyses
HbA1c and fasting glucose were assessed in the biochemistry laboratory at Manchester University Hospitals NHS Foundation Trust using routine methods. The remaining samples were processed in the Cardiovascular Research Lab at the University of Manchester. Laboratory procedures and measurements were carried out according to our previously described protocol. 18 Serum and ethylenediaminetetraacetic acid (EDTA)-plasma were isolated by centrifugation at 2000 × g for 15 min at 4°C within 2 h of blood collection. Aliquots for biochemical analysis were frozen at −80°C.
Total cholesterol was measured using the cholesterol oxidase phenol 4-aminoantipyrine peroxidase method and triglycerides by the glycerol phosphate oxidase phenol 4-aminoantipyrine peroxidase method. HDL-C was measured using a second-generation homogeneous direct method (Roche Diagnostics, Burgess Hill, UK). LDL-C was estimated using the Friedewald formula. 35 ApoB was measured using immunoturbidimetric assays (ABX Diagnostics, Shefford, UK). All these tests were performed on a Cobas Mira analyser (Horiba ABX Diagnostics, Nottingham, UK).
Small, dense low-density lipoprotein apolipoprotein B (SD-LDL ApoB; LDL particles of density > 1.044 g/mL) was isolated from plasma and ultracentrifuged at 100,000 r/min (435,680 × g) for 5 h at 4°C using a Beckman Optima TLX bench top ultracentrifuge fitted with TLA 120.2 fixed angle rotor (Beckman Coulter UK). 36 ApoB in SD-LDL was then determined using an immunoturbidimetric assay (ABX Diagnostics). SD-LDL is thus expressed in terms of the plasma concentration of its ApoB component.
An in-house, antibody sandwich enzyme-linked immunosorbent assay (ELISA) technique using anti-human CRP antibody, calibrators and controls from Abcam (Cambridge, UK) was used to measure hs-CRP. IL-6 was measured by ELISA using kits from R&D Systems (Abingdon, UK). The upper limit (95th percentile) for IL-6 in plasma was 3.1 pg/mL. 37 Plasma insulin was measured with Mercodia ELISA kits from Diagenics Ltd. (Milton Keynes, UK). Homeostatic model assessment of insulin resistance (HOMA-IR) was used to assess insulin resistance, 38 using the formula
The laboratories participated in the UK National External Quality Assessment Service (UKNEQAS, Birmingham, UK) for quality control of general blood chemistry.
Statistical analysis
SPSS for Mac (Version 23.0; IBM SPSS Statistics, IBM Corp., Armonk, NY) and GraphPad Prism (Version 7.00; GraphPad Software, La Jolla, CA, USA) were used for analysis of data. Tests for normality were done using the Shapiro–Wilk test, visualisation of histograms and Q-Q plots. When more than two time points were being compared, one-way ANOVA was used for parametric data and Friedman’s two-way analysis of variance by Ranks was used for non-parametric data. Specific post hoc pairwise comparisons were done using the Bonferroni correction in SPSS. The McNemar test was used to compare paired categorical variables. The percentage change in variables was determined as the absolute difference between measurements 12 months after surgery and baseline divided by the baseline value (and multiplied by 100). Correlation analysis was done using Pearson’s test for parametric and Spearman’s test for non-parametric data. A p-value of <0.05 was considered to be statistically significant.
Results
Clinical characteristics
The baseline and post-operative clinical measures are given in Table 1. The mean age of participants was 48 years. BMI, waist circumference and systolic blood pressure were reduced significantly (p < 0.05), with no significant change in diastolic blood pressure (p = 0.15). Of the 40 patients, 20 had T2DM pre-operatively and remitted completely in 13 out of 20 (65%) patients 12 months after surgery (p < 0.001). There was a significant reduction in the number of participants meeting the diagnostic criteria for the metabolic syndrome (p < 0.001) and there was a trend towards reduction in the use of lipid-lowering drugs (p = 0.06) after bariatric surgery.
Clinical parameters before and after bariatric surgery.
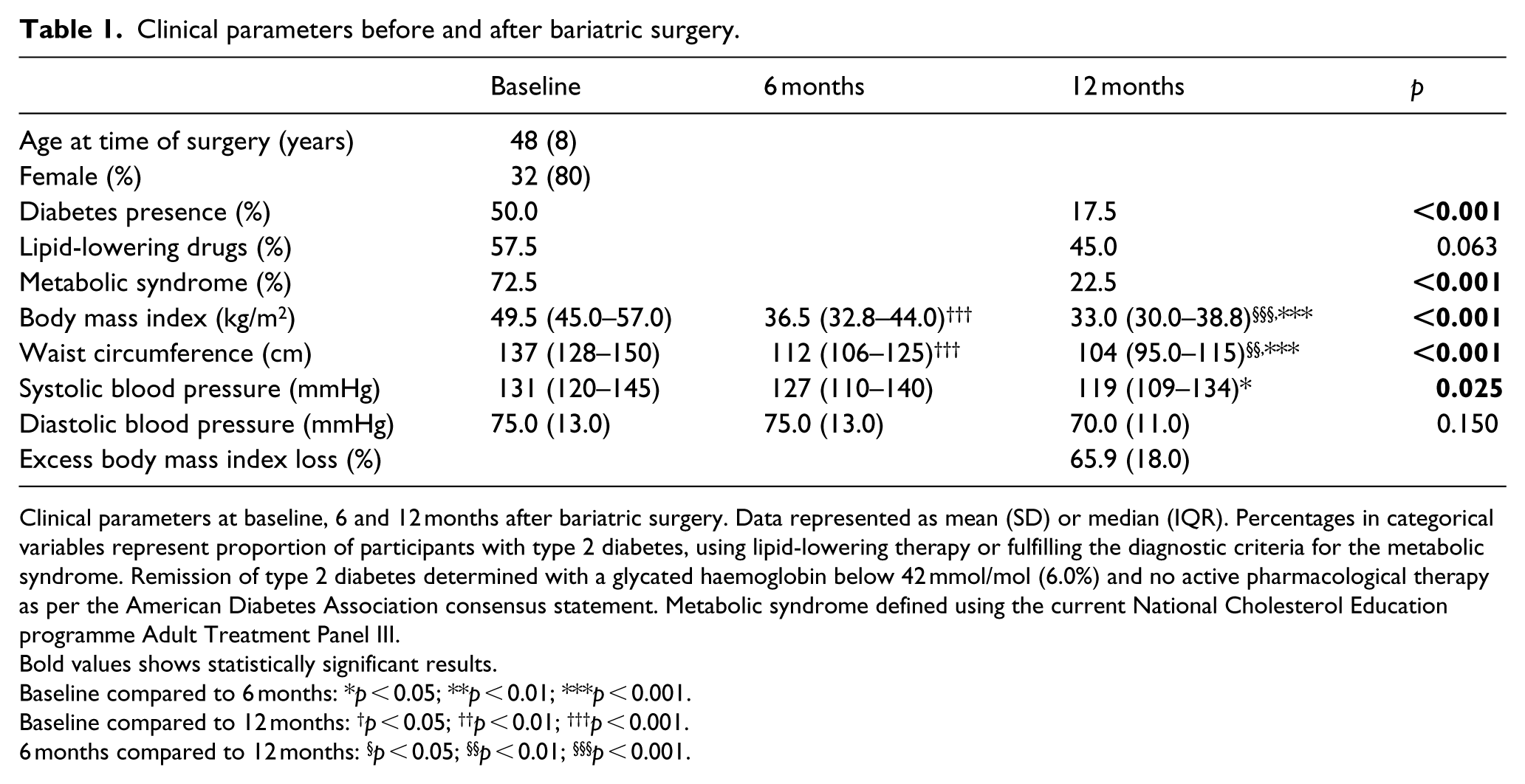
Clinical parameters at baseline, 6 and 12 months after bariatric surgery. Data represented as mean (SD) or median (IQR). Percentages in categorical variables represent proportion of participants with type 2 diabetes, using lipid-lowering therapy or fulfilling the diagnostic criteria for the metabolic syndrome. Remission of type 2 diabetes determined with a glycated haemoglobin below 42 mmol/mol (6.0%) and no active pharmacological therapy as per the American Diabetes Association consensus statement. Metabolic syndrome defined using the current National Cholesterol Education programme Adult Treatment Panel III.
Bold values shows statistically significant results.
Baseline compared to 6 months: *p < 0.05; **p < 0.01; ***p < 0.001.
Baseline compared to 12 months: †p < 0.05; ††p < 0.01; †††p < 0.001.
6 months compared to 12 months: §p < 0.05; §§p < 0.01; §§§p < 0.001.
Laboratory measurements
Results of the laboratory measurements are shown in Table 2 and Figure 1. In the entire cohort, there were significant (p < 0.05) reductions in the triglycerides, HDL-C, SD-LDL ApoB, hs-CRP, IL-6, HbA1c, glucose, insulin and HOMA-IR, 12 months post-operatively with intermediate values at 6 months. Total ApoB levels were significantly reduced only at 12 months and total cholesterol and LDL-C did not change significantly.
Laboratory values before and after bariatric surgery.
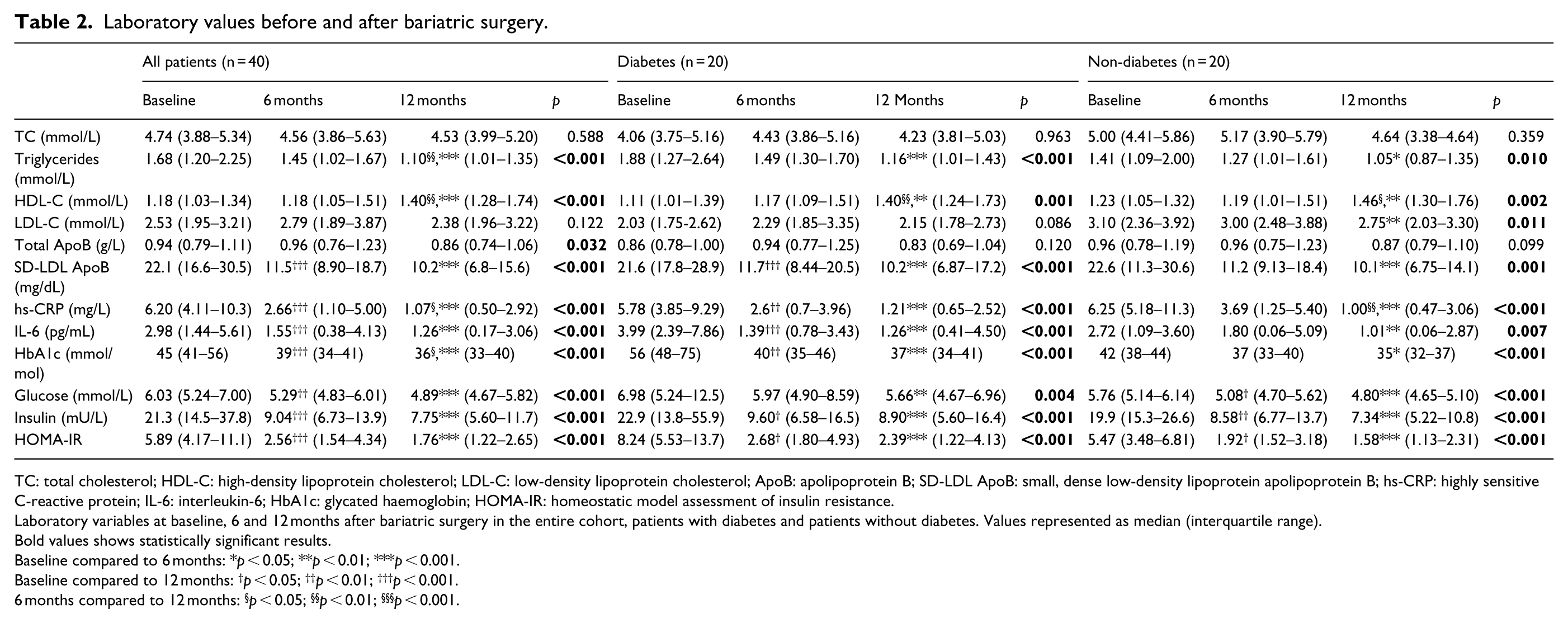
TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; ApoB: apolipoprotein B; SD-LDL ApoB: small, dense low-density lipoprotein apolipoprotein B; hs-CRP: highly sensitive C-reactive protein; IL-6: interleukin-6; HbA1c: glycated haemoglobin; HOMA-IR: homeostatic model assessment of insulin resistance.
Laboratory variables at baseline, 6 and 12 months after bariatric surgery in the entire cohort, patients with diabetes and patients without diabetes. Values represented as median (interquartile range).
Bold values shows statistically significant results.
Baseline compared to 6 months: *p < 0.05; **p < 0.01; ***p < 0.001.
Baseline compared to 12 months: †p < 0.05; ††p < 0.01; †††p < 0.001.
6 months compared to 12 months: §p < 0.05; §§ p < 0.01; §§§p < 0.001.
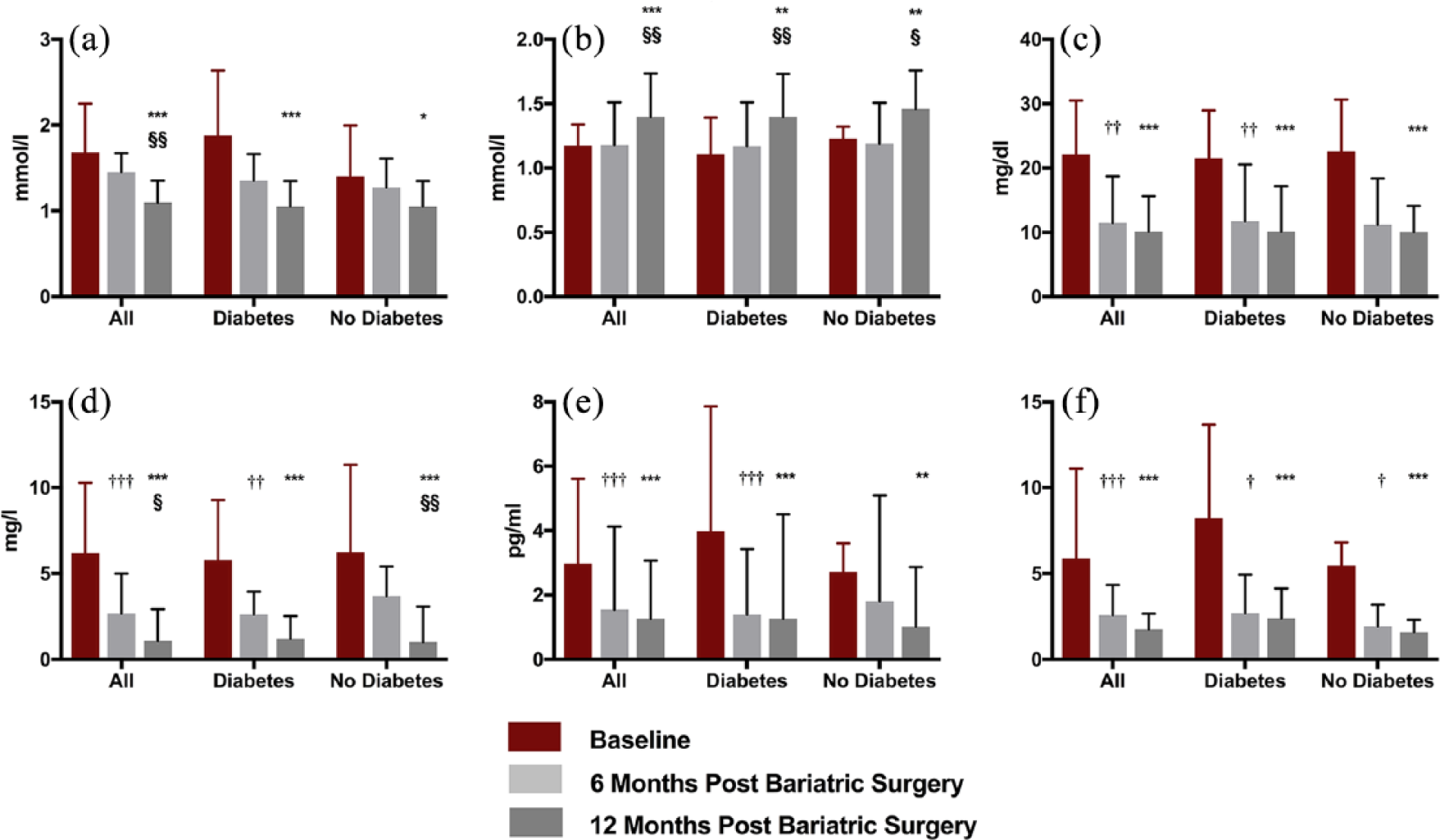
The responses to bariatric surgery in the whole 40 patients and in those with and without type 2 diabetes separately before and at 6 and 12 months post-operatively in (a) fasting triglycerides, (b) high-density lipoprotein cholesterol (HDL-C), (c) small, dense low-density lipoprotein apolipoprotein B (SD-LDL ApoB), (d) highly sensitive C-reactive protein (hs-CRP), (e) interleukin-6 (IL-6) and (f) insulin resistance (HOMA-IR). Bars are median + 75th percentile.
Subgroup analysis of the patients with and without diabetes showed similar results, except patients without diabetes had a significant reduction (p = 0.01) in LDL-C compared to those with diabetes. The significant improvements in triglycerides, HDL-C, SD-LDL ApoB, hs-CRP, IL-6, HbA1c, glucose, insulin and HOMA-IR were seen both in patients using statins (n = 23) and those not on statins (n = 17). Those using statins had higher serum total ApoB, reflecting a more severe dyslipidaemia phenotype. Patients not on statin therapy showed a greater reduction in total ApoB, but this was not statistically significant (p = 0.12) (Supplementary Table 1).
Relationships between fasting insulin, HOMA-IR and other metabolic variables
Correlations of values at baseline and 12 months post-operatively
The relationships at baseline between fasting insulin and BMI are illustrated in Figure 2(a) and with HOMA-IR and BMI in Figure 2(c). The association between post-operative fasting insulin and BMI is shown in Figure 2(b) and for HOMA-IR and BMI in Figure 2(d).
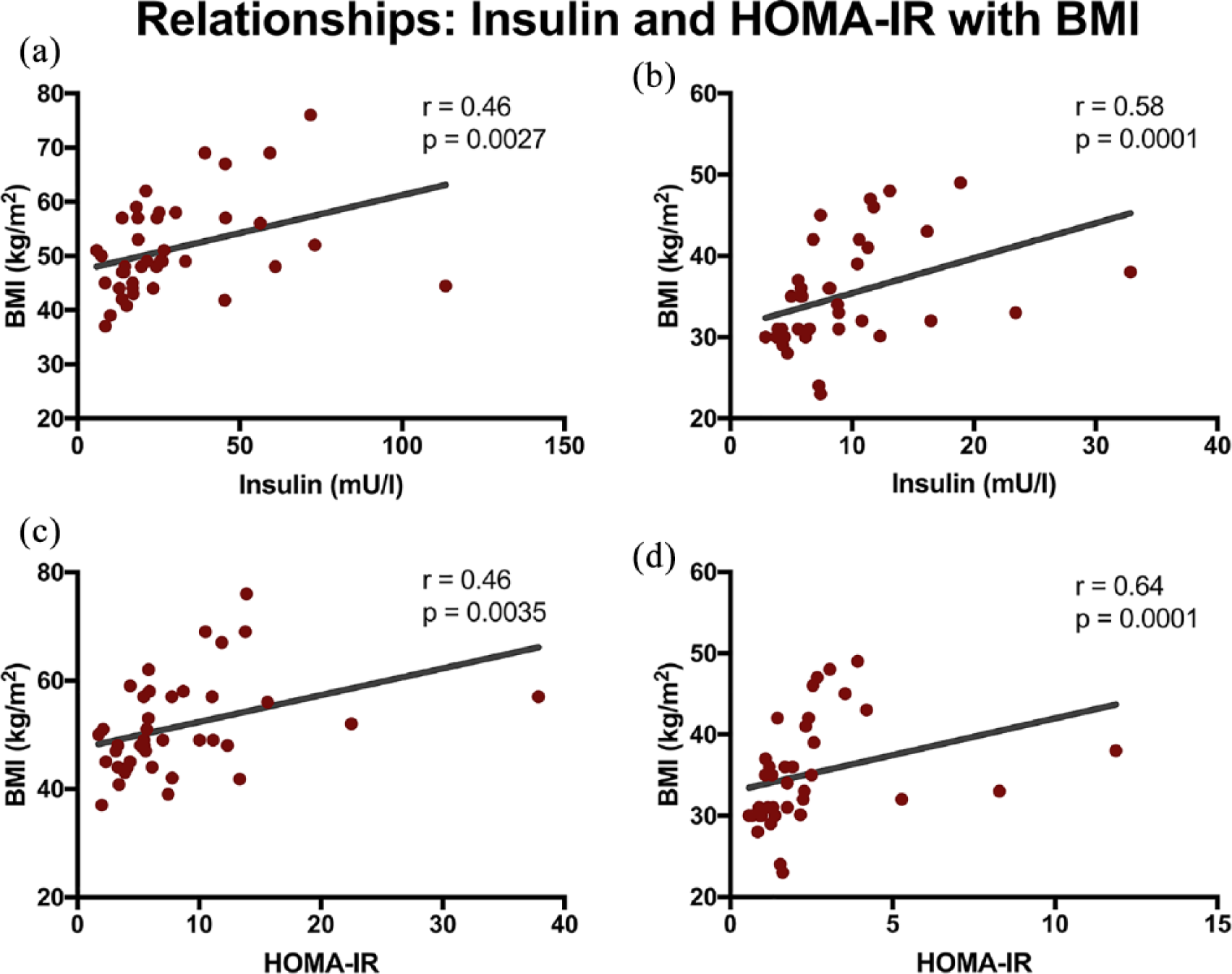
Fasting insulin and homeostatic model assessment of insulin resistance (HOMA-IR) as a function of body mass index (BMI) at baseline [panels (a) and (c)] and 12 months after bariatric surgery [panels (b) and (d)].
Both pre-operatively and 1 year post-operatively, fasting insulin levels correlated significantly with HDL-C levels (r = –0.37; p = 0.02 and r = –0.40; p = 0.01, respectively). HOMA-IR measurements showed a significant association with triglycerides (r = 0.34; p = 0.03) and HDL-C (r = –0.39; p = 0.01) at baseline and at 12 months (triglycerides r = 0.36; p = 0.02, HDL-C r = –0.42; p = 0.007). Triglycerides were not significantly correlated with insulin (r = 0.22; p = 0.18) at baseline, but were weakly correlated at 12 months post-operatively (r = 0.31; p = 0.05). Pre-operatively, IL-6 correlated with BMI (r = 0.43; p = 0.009), but not with insulin levels or HOMA-IR. The relationship with BMI was weaker post-operatively (r = 0.31; p = 0.07) possibly because of the loss of visceral fat. IL-6 correlated with hs-CRP post-operatively (r = 0.42; p = 0.007).
Association of change in hyperinsulinaemia and insulin resistance
The percentage change (Δ) in fasting insulin levels between the pre- and post-operative state was related to Δtriglycerides (r = 0.36; p = 0.03), Δhs-CRP (r = 0.42; p = 0.01), ΔIL-6 (r = 0.41; p = 0.02) and ΔBMI (r = 0.43; p = 0.007). There were also significant correlations between ΔHOMA-IR with Δtriglycerides (r = 0.33; p = 0.04), Δhs-CRP (r = 0.37; p = 0.02) and ΔIL-6 (r = 0.37; p = 0.03).
Discussion
This study shows that a marked reduction in hyperinsulinaemia/insulin resistance in obese people after bariatric surgery ameliorates not only raised triglycerides, hs-CRP and low HDL-C, features of the Reaven syndrome, 2 but also SD-LDL and IL-6, irrespective of the presence of T2DM or statin therapy.
SD-LDL concentration increases with triglyceride levels. 26 It is a cholesterol-depleted low-density lipoprotein (LDL), the presence of which is not evident from measurement of LDL-C. It contributes to total serum ApoB and is the cause of hyperapobetalipoproteinaemia, 39 but the majority of ApoB-containing lipoproteins are of lower density so that even when total ApoB is measured its presence may not be obvious. It is likely that it is the cause of the association between hypertriglyceridaemia and CVD.24,26,27 SD-LDL is more susceptible both to oxidative and glycative modification than less dense LDL species.29,30 Both oxidatively modified and glycated LDL, unlike more buoyant, unmodified LDL, are rapidly taken up via scavenger receptors on macrophages in tissue culture to become foam cells similar to those in atheromatous lesions. 40 In our series of patients, the SD-LDL concentration of 22.1 mg/dL at baseline declined to 10.2 mg/dL 12 months after bariatric surgery, restoring its levels close to the median value in a healthy population (14 mg/dL for men and 9 mg/dL for women). 41
A raised level of hs-CRP is recognised as a feature of metabolic syndrome 2 and is closely associated to the risk of developing CVD. 4 However, evidence that hs-CRP is causally related to CVD has not been forthcoming. 4 On the other hand IL-6, a major regulator of hepatic CRP secretion, unlike CRP, has been found in Mendelian randomisation studies to be linked to atherosclerosis.4,32,33 Many adipokines are released both from peripheral and visceral adipose tissue, which have the potential to contribute to hepatic insulin resistance and secretion of CRP, but those released from visceral fat may have a special place in the genesis of the metabolic syndrome, because they arrive at the liver through the portal vein and may do so at higher concentration than those arriving by the hepatic artery after dilution in the systemic circulation. IL-6 was found in much higher concentration in portal blood than in systemic arterial blood by Fontana et al. 21 in patients undergoing gastric bypass surgery for obesity. Tumour necrosis factor-α, resistin, macrophage chemoattractant protein-1 and adiponectin concentrations were similar in the portal vein and radial artery. Portal vein IL-6 concentration also correlated directly with systemic CRP. The decrease in IL-6 after bariatric surgery, suggests that it could be associated with the decrease in hs-CRP and other features of hepatic insulin resistance. Recently, reduction in IL-6 levels similar to that reported here, but achieved by administration of a monoclonal antibody to interleukin-1β, was reported to be associated with decreased atherosclerotic CVD incidence. 31
The findings of this and earlier reports in which the effects of the decrease in insulin resistance accompanying weight loss on components of the metabolic syndrome have been investigated,17–20 provide powerful support for Reaven’s hypothesis. Bariatric surgery which induces the most dramatic decreases in hyperinsulinaemia/insulin resistance is a model which could yield an even greater understanding of, for example, the mechanism by which atheroma risk is increased. Our finding of a decrease in SD-LDL should lead to exploration of the effects of weight loss due to bariatric surgery on modified, potentially highly atherogenic LDL subspecies, such as oxidised and glycated LDL. Furthermore, it could lead to some resolution of the conflict which exists between which components of the metabolic (Reaven) syndrome are due to resistance to insulin (too little insulin action) and which are due to hyperinsulinaemia itself (too much insulin action). In recent years, it has often been forgotten that, because insulin regulates several intracellular signalling pathways controlling a variety of processes, while its effects will be deficient in pathways resistant to it, in others the high levels of insulin produced to attempt to maintain euglycaemia may hyperstimulate non-resistant pathways. An example might be the regulation of sex hormone–binding globulin (SHBG), which is decreased in insulin resistance, leading to increased free androgen levels in both men and women.42,43 This at least partly explains the androgenisation of insulin resistant women and thus their male pattern (visceral; central) obesity and hirsutism. Despite the association of insulin resistance with decreased SHBG, however, tissue culture experiments with human hepatocytes reveal insulin to have an inhibitory action on SHBG production.44,45 Thus, unlike the VLDL production pathway where insulin resistance decreases the inhibitory effect of insulin despite the increase in its concentration in response to resistance to glucose uptake, the pathway for the production of SHBG must escape resistance to the action of insulin and be inhibited by hyperinsulinaemia. The model of bariatric surgery may provide opportunities for futher study of this mechanism as it may for other phenomena associated with the hyperinsulinaemia of insulin resistance, such as the pathways linking metabolic syndrome to hyperuricaemia. 46
It has been difficult to distinguish components of the metabolic syndrome due to resistance to insulin action and those hyperstimulated by the ensuing increase in insulin secretion to maintain euglycaemia as first discussed in Kim and Reaven. 13 We found that the strength of the associations between change in components of the metabolic syndrome, such as triglycerides and HDL-C, and change in fasting insulin and HOMA-IR were similar with perhaps the suggestion that triglyceride concentration was more closely related to insulin resistance and HDL-C to fasting insulin levels. It has been suggested that the insulin clamp technique would be a better means of assessing insulin resistance rather than HOMA-IR, which relies on the ratio between fasting insulin and glucose. 38 However, this argument is less persuasive when it is considered that in insulin clamping, the insulin is administered into the systemic rather than the portal circulation into which it is secreted physiologically. 47 This means that physiologically the liver is subject to much higher levels than peripheral tissues and the insulin clamp is thus measuring insulin resistance to glucose uptake in tissues, such as skeletal muscle, while hepatic uptake is relatively unaffected by insulin arriving by the hepatic artery. Presumably, HOMA-IR, however, represents the contribution of both hepatic and peripheral glucose disposal. At the time of bariatric surgery, direct measurement of hormones and metabolites in portal venous blood can be undertaken, 21 but repetition of this after weight loss is not possible, at least in human models. Nonetheless, discovering which processes are resistant to insulin and which are over-stimulated by the accompanying hyperinsulinaemia could be important therapeutically.
We conclude that bariatric surgery provides an excellent model to dissect Reaven’s hypothesis, to discover the mechanisms by which its components, such as raised SD-LDL and IL-6, cause atherosclerosis. It also raises questions about the respective roles of insulin resistance and of hyperinsulinaemia on different components of the metabolic syndrome.
Supplemental Material
Supplementary_Table_1_DVDR_(2) – Supplemental material for Bariatric surgery as a model to explore the basis and consequences of the Reaven hypothesis: Small, dense low-density lipoprotein and interleukin-6
Supplemental material, Supplementary_Table_1_DVDR_(2) for Bariatric surgery as a model to explore the basis and consequences of the Reaven hypothesis: Small, dense low-density lipoprotein and interleukin-6 by Safwaan Adam, Yifen Liu, Tarza Siahmansur, Jan H Ho, Shaishav S Dhage, Rahul Yadav, John P New, Rachelle Donn, Basil J Ammori, Akheel A Syed, Rayaz A Malik, Handrean Soran and Paul N Durrington in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors acknowledge support from The Manchester Comprehensive Local Research Network and The National Institute for Health Research/Wellcome Trust Clinical Research Facility. The authors are also grateful to the biomedical scientists in the Clinical Biochemistry Department at Manchester University NHS Foundation Trust for their assistance with laboratory measurements.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.S. and P.N.D. have served as consultants to pharmaceutical companies marketing lipid-lowering drugs and have received travel expenses, payment for speaking at meetings and funding for research from some of these companies. None of the other authors have any relevant conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research donation from AMGEN and the Lipid Diseases Fund.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.