Abstract
Objective
To examine the role of rs10830963 and rs8192552 polymorphisms in melatonin receptor 1 B (MTNR1B) gene on the development of obesity and related comorbidities among adolescents in South-Eastern Turkey.
Methods
The present study included 200 unrelated adolescents (100 obese, 100 normal weight). The rs8192552 and rs10830963 polymorphisms in the MTNR1B gene were genotyped using a PCR SNaPshot assay.
Results
No statistically significant association was observed between MTNR1B gene rs8192552/rs10830963 polymorphisms and adolescent obesity. In adolescents with an rs8192552 E allele, homeostasis model assessment for insulin resistance (IR) level was lower and IR was less common. In morbidly obese adolescents with an rs8192552 E allele, total cholesterol level was lower. In obese adolescents with metabolic syndrome, plasma fasting glucose level was higher in rs10830963G allele carriers. In obese girls, body weight was lower in those with a rs10830963 C allele, whereas in obese boys, body weight and waist circumference were higher in those with a rs10830963 C allele.
Conclusions
The MTNR1B gene was not confirmed as an obesity susceptibility gene in adolescents. However, an association between the MTNR1B gene and IR/hypercholesterolemia/metabolic syndrome was observed in obese adolescents. A sex-specific effect on obesity was also identified.
Introduction
In recent years, obesity and related comorbidities such as insulin resistance (IR), type 2 diabetes mellitus (DM), dyslipidemia, hypertension and metabolic syndrome (MetS) have become among the most serious health concerns. Obesity is a complicated disease caused by genetic and environmental factors. 1 The imbalance between energy intake and expenditure is one of the main factors leading to obesity. Energy balance is provided by the combined effects of biological (e.g. circadian rhythm, sleep pattern) and environmental factors (e.g. dietary habits, physical activities). 2 There is a close relationship between circadian rhythm and metabolic regulation including 24-hour variations in glucose, insulin and leptin levels. Disturbances in sleep/circadian rhythms can therefore lead to changes in energy metabolism and may increase the risk of obesity, DM and MetS.3–6
The neurohormone melatonin is predominantly secreted from the pineal gland during the night and plays an important role in the regulation of circadian rhythm and sleep cycles.7–9 Melatonin also plays a role in the development of type 2 DM by modulating both insulin secretion and glucose homeostasis.3,7–9 Melatonin is secreted from the retina, pancreas (expressed in beta cells, upregulated in islet cells), adipose tissue and enteroendocrine cells of the gastrointestinal tract.4,7–9,10,11 Melatonin synthesis in the gastrointestinal tract is associated with nutrition-independent photoperiodic secretion from the pineal gland, while food intake results in increased tissue and plasma concentrations of melatonin. The release of melatonin from the gastrointestinal tract is regulated by a negative feedback effect of circulating melatonin levels. 12
The effect of melatonin is mainly modulated through the activation of two G-protein-coupled membrane receptors, melatonin receptor 1A (MTNR 1A) and melatonin receptor 1B (MTNR 1B).7,12 MTNR 1B inhibits insulin secretion via several intracellular messengers such as cAMP, cGMP and [Ca2+], and is located on chromosome 11q21-q22.7–9,10,12 A common variant of MTNR, the 1B rs10830963 polymorphism, is associated with elevated plasma fasting glucose (PFG) levels and increased risk of type 2 DM, independent of age and body mass index (BMI) status.13–15 Another polymorphism of MTNR 1B, rs8192552, has been found to be associated with increased prevalence of obesity and higher BMI/waist circumference values in Danish and French adults. 16
Supraphysiological doses of melatonin lead to the desensitization and internalization of melatonin membrane receptors, resulting in down-regulation of receptor production and loss of function. These effects subsequently lead to circadian rhythm impairment and sleep problems. 7 Sleep disturbances and chronic sleep deficit are independent risk factors for developing obesity. Although the underlying mechanism is not clear, this relationship may be attributable to changes in the sympathetic nervous system and in hormone concentrations.3,6,17,18 Neurohormonal changes in periods of reduced sleep affect appetite and energy storage.3,6,11,17,18
Therefore, we hypothesized that the rs10830963 and rs8192552 polymorphisms in the MTNR1B gene may play a role in the pathogenesis of obesity. Limited studies on the role of the MTNR1B gene in adolescent obesity have been reported to date, and the relationship of this polymorphism with obesity in Turkish populations has not yet been evaluated. Therefore, we aimed to examine the role of the rs10830963 and rs8192552 MTNR1B polymorphisms on the development of obesity and related comorbidities such as IR, MetS and dyslipidemia among adolescents in South-Eastern Turkey.
Materials and methods
Participants
A total of 100 unrelated obese adolescents and 100 age- and sex-matched normal weight control adolescents were selected from among those referred to the Well-Child Outpatient Clinic of Gaziantep University Hospital for routine health care visits. The sample size was estimated using a power calculation based on earlier studies. The minimum sample size was determined as 76 individuals for the rs8192552 polymorphism and 81 individuals for the rs10830963 polymorphism at a power level of 0.80 and an α error of 0.05.13,16 The study protocol was approved by the local ethics committee of Gaziantep University, and informed consent was obtained in accordance with the principles of the Declaration of Helsinki.
Anthropometric measurements (height, weight and waist circumference) were performed using a standardized protocol by the same researchers. A detailed clinical history, including laboratory findings, was obtained from the medical records of all adolescents. Exclusion criteria included any chronic disease, and receipt of medication, and dieting. The BMI was calculated as the weight divided by height squared (kg/m2). The standards of the International Obesity Task Force were used to diagnose obesity, 19 which was defined as BMI at or above the 95th percentile for adolescents of the same age and sex. Adolescents with a BMI at or above the 99th percentile were defined as morbidly obese. The evaluation of BMI was performed using the age- and sex-specific percentile standards of the Centers for Disease Control and Prevention. 20
Insulin resistance was determined as homeostasis model assessment for insulin resistance (HOMA-IR) >3.16. 21 Metabolic syndrome was defined according to the criteria of the International Diabetes Foundation. 22
Genotyping
A 2-mL EDTA-peripheral blood sample was obtained from each adolescent and genomic DNA was isolated from each sample using the salt fractionation procedure. 23 Amplification of the MTNR 1B rs8192552 and rs10830963 poymorphisms was performed using PCR. PCR products were verified using agarose (2%) gel electrophoresis and purified using NucleoFast 96 PCR clean-up plates (Macharey-Nagel GmbH & Co. KG, Düren, Germany). Genotyping of the SNPs was carried out using a SNaPshot multiplex system on an ABI 3130 capillary electrophoresis instrument (Applied Biosystems/Ambion Inc., Austin, TX, USA) according to the manufacturer’s protocol. The electropherograms were analyzed using GeneMapper 4.0 software (Applied Biosystems/Ambion Inc.).
Statistical analyses
For comparisons of genotype and allele frequencies between the obese adolescent and control groups, a chi-square test was used if expected cell frequencies were ≥5 and Fisher’s exact test was used if any expected counts were < 5 (Graphpad Instat version 3; Graphpad Software, San Diego, CA, USA). The Hardy–Weinberg equilibrium (HWE) was calculated using the DeFinetti computer program (http://ihg2.helmholtz-muenchen.de/cgi-bin/hw/hwal.pl). All data were analyzed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics for quantitative variables were given as means ± standard deviation, and allele frequencies and the distribution of genotypes were expressed as percentages. Clinical characteristics and MTNR1B gene polymorphisms were compared using the chi-square test. Multiple comparisons were calculated using the Mann–Whitney U test and Student’s t-test. Values of p<0.05 were accepted as statistically significant.
Results
The obese adolescents were aged 10 to 18 years (mean 13.6±2.4 years). Anthropometric measurements and Z scores (international reference standards of the World Health Organization) of obese adolescents are summarized in Table 1. Of the 100 obese adolescents, 50% (50/100) were female, and 59.0% (59/100) were morbidly obese. The clinical and laboratory characteristics of obese adolescents according to MTNR1B genotype are summarized in Table 2 and Table 3. The prevalence of IR was 47.0% (47/100) and that of MetS was 27.0% (27/100).
Anthropometric measurements and Z scores of obese adolescents.
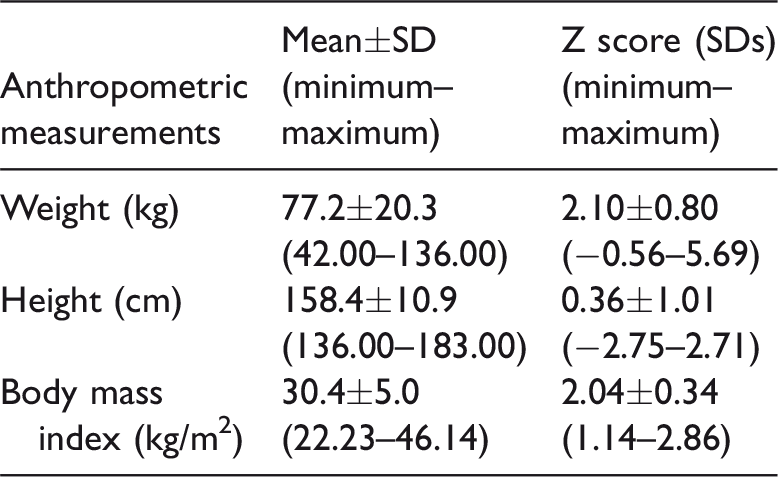
SD, standard deviation.
Clinical and laboratory characteristics of obese adolescents by MTNR1B rs8192552 polymorphism genotype.

SD, standard deviation.
Clinical and laboratory characteristics of obese adolescents by MTNR1B rs10830963 polymorphism genotype.

SD, standard deviation.
The comparison of genotype and allele frequencies of the MTNR 1B gene rs8192552 and rs10830963 polymorphisms between the obese adolescent and control groups are presented in Table 4. No statistically significant difference in genotype and allele frequency was observed between the obese adolescent and control groups (p>0.05) (Table 4).
Genotype/allele frequencies of rs8192552 and rs10830963 polymorphisms of MTNR1B among obese adolescents and control cases.

HWE, Hardy–Weinberg equilibrium; CI, confidence interval.
For individuals with an rs8192552 polymorphism, carriers of an E allele were less likely to have IR (non-carriers: 45/81, 55.6%; carriers: 2/12, 16.7%; p=0.012), and had lower HOMA-IR levels (non-carriers: 3.58±2.09 [0.56–15.50]; carriers: 2.53 ± 0.73 [0.98–3.99]; p=0.045) compared with non-carriers. In morbidly obese adolescents carrying an rs8192552 E allele, total cholesterol level was lower (carriers: 141.00±12.98 mg/dL [123–159]; non-carriers: 167.12±30.09 mg/dL (99–244); p=0.023).
For individuals with an rs10830963 polymorphism, total cholesterol level was lower among those carrying a G allele (carriers: 160.00±26.99 mg/dL [96–209]; non-carriers: 172.85±29.46 mg/dL [124–244]; p= 0.029). In obese girls, waist circumferance was unchanged but body weight was lower in cases with an rs10830963 C allele (carriers: 74.01±17.57 kg [44.20–126.00]; non-carriers: 86.86±6.57 kg [80.00–95.40]; p=0.045). Conversely, in obese boys, body weight and waist circumference were higher in cases with an rs10830963 C allele (body weight carriers: 80.82±22.95 kg [42.00–145.00]; non-carriers: 55.10±7.15 kg [46.85–59.60]; p= 0.024; waist circumference, carriers: 103.34±12.87 cm [75–135], non-carriers: 87.67±8.73 cm [78–95]; p=0.025). In obese adolescents with MetS, PFG levels were higher in rs10830963 G allele carriers (93.06±10.76 mg/dL [78–114]) compared with non-carriers (85.20±4.78 mg/dL [81–96], p=0.039).
Discussion
Obesity is a multifactorial disease characterized by a highly complex phenotype determined by genetic/environmental interactions (e.g. prenatal risk factors, socioeconomic factors, dietary and physical activity, sleep duration and screen time).1,2 Circadian rhythm impairment and sleep disturbances have previously been associated with metabolic dysfunction including obesity, DM and MetS.3,6,17,18 Melatonin plays an important role in the regulation of circadian rhythm and sleep.7–9,12 Alterations in the melatonin release mechanism may lead to changes in BMI in mammals, 11 although the underlying mechanism of by which melatonin regulates BMI remains unclear. One possible explanation implicates the activation of melatonin receptors; the activation of central receptors (e.g. in the pineal gland) may modulate the metabolic rate via sympathetic nerve activity or altered appetite. The activation of peripheral receptors (e.g. adipose tissue) may affect energy storage through adipocyte metabolism or by increasing the number of adipocytes. 11 Another possible mechanism may be related to decreased levels of leptin, which regulates appetite, food intake and energy expenditure as a result of short sleep duration following circadian rhythm impairment. 17
The MTNR1B rs8192552 polymorphism, located in exon 1, results in G24E mutation. 4 In recent years, several loci related to obesity have been identified, although rs8192552 has not previously been reported.9,10,24,25 Andersson et al. 16 demonstrated that the variant 24E allele is associated with increased prevalence of obesity and higher BMI/waist circumference values in adults. However, a relationship between the variant E allele and obesity among adolescents was not observed in the present study. To our knowledge, no study to date has investigated the relationship between obesity and the rs8192552 polymorphism of the MTNR1B gene in adolescents. Obesity-related differences observed between adults and adolescents may be largely attributable to age. Visceral adipose tissue and body weight increase with age whereas nocturnal plasma melatonin concentrations decrease, as a result of anatomical changes in circadian pacemaker neurons in the brain.11,18 It has also been shown that the minor 24E allele can lead to higher expression of MTNR1B in older individuals. 26 Additionally, gene–gene and gene–environment interactions can lead to modifications in DNA methylation patterns during the early years of life. 1 Our findings therefore suggest that adolescents with an MTNR1B gene E allele should be followed carefully by routine health care visits because of an increased likelihood of developing obesity in adulthood.
The MTNR1B rs8192552 variant E allele is associated with reduced PFG levels. 16 Our study demonstrated that a mutant 24 E allele may have a protective role against IR in obese adolescents. Although the underlying mechanisms of this protective effect are not fully understood, the mutant allele may alter glucose-stimulated insulin secretion and proinsulin conversion. 4 Another possible mechanism may relate to differential regulation of the melatonin receptor in specific target tissues with crucial functions in carbohydrate metabolism (e.g. adipose tissue, skeletal muscle, liver and hypothalamus).4,16 The protective effect also may be attributable to leptin, which is associated with IR independent of obesity by imparing insulin signaling. Leptin levels are increased in individuals with MetS, but in individuals with severe MetS, adipose tissue dysfunction may lead to decreased leptin levels. 17 It could be argued, therefore, that a mutant allele may lead to the development of relative hypoleptinemia via adipose tissue dysfunction. Elevated leptin has been shown to be an independent risk factor for hyperlipidemia. 17 Brydon et al. reported that MTNR1B activation in adipose tissue leads to increased lipid levels and adipocytes. 11 Surprisingly, we found that the mutant E allele was associated with reduced total cholesterol levels in morbidly obese adolescents. We speculate that a protective effect against hyperlipidemia may occur because of relative hypoleptinemia caused by adipose tissue dysfunction in morbidly obese adolescents. Our study illustrates that morbidly obese adolescents carrying an E allele may have reduced cardiometabolic risk of dyslipidemia, although further studies are required to confirm this effect.
The MTNR1B rs10830963 polymorphism, located in intron 1, results in a C/G mutation which is associated with increased MTNR 1B expression and mRNA levels in pancreatic ß cells. This mutation also leads to impaired insulin secretion and elevated PFG levels, independent of age and BMI status.4,13–15,26 Furthermore, increased MTNR 1B expression leads to disorders of the melatonin release mechanism that can alter BMI in mammals.7,11 However, previous studies among different ethnicities did not show any relationship between the MTNR1B rs10830963 minor allele (G) and obesity in children and adolescents.10,13,27,28 We also found no association between the MTNR 1B rs10830963 polymorphism and adolescent obesity. We observed a frequency of the MTNR1B rs10830963 minor allele (G) of 0.335 in our obese cases, which is higher than that in Caucasian (0.262), African American (0.091) and Hispanic (0.215) individuals 10 but similar to that in a German population (0.302) and lower than that observed in Chinese obese children and adolescents (0.4266).15,27 Allele frequency therefore appears to vary according to ethnicity, indicating that the effects of alleles on obesity and comorbidities require increased focus in specific ethnic groups. To the best of our knowledge, this is the first study to investigate the role of the MTNR1B gene in adolescent obesity and related comorbidities in Turkish populations. However, future studies in larger populations are required to verify the study findings.
The minor G allele of the MTNR1B rs10830963 polymorphism is significantly associated with increased PFG levels, independent of the presence of type 2 DM.13,15,26 Two different meta-analyses including 42 studies demonstrated that the rs10830963 polymorphism is associated with type 2 DM and impaired insulin secretion.8,9 Following stratification by ethnicity, a significant association was detected in a Caucasian population but not in an Asian population. 8 Liu et al. also confirmed the association between the G allele and PFG in a Shanghai but not in a Beijing subpopulation. 14 Obese adolescents carrying the G allele showed higher PFG levels among German, Caucasian and Hispanic populations, but not in African American individuals.10,26 We observed no evidence in rs10830963 polymorphism associated with genotype-dependent differences for PFG, insulin levels and IR among obese adolescents in South-Eastern Turkey. There are several possible explanations for the differences observed between studies. The frequencies of the risk alleles in the MTNR1B gene may differ between ethnic groups as described above. In our study, statistical power to detect a positive relationship may have been insufficient owing to the low frequency of the rs10830963 mutant G allele in our study population. In addition to socioeconomic and lifestyle factors, gene–gene or gene–environment interactions may also have been responsible for the differences observed between studies. Therefore, the findings of single locus studies are frequently contradictory. Furthermore, the prevalence of type 2 DM can vary according to ethnicity, and also, we examined only IR, which is associated with the prediabetic state. The prevalence of DM in our study population was unknown; this is a limitation of our study that may have contributed to the difference observed in our study compared with previous studies with respect to an association between rs10830963 polymorphism and PFG, insulin level and IR. Interestingly, we observed that in obese adolescents with MetS, PFG levels were higher in G allele carriers. Similar to our results, Lyssenko et al. 26 reported that the G allele was associated with elevated PFG levels in men with MetS. This relationship may be explained by the increase in leptin observed in individuals with MetS and its association with IR, independent of obesity. 17 Furthermore, early-onset obesity is known to be associated with increased risk for MetS, type 2 DM and cardiovascular disease in adulthood. 1 The present study indicates that obese adolescents with MetS carrying a G allele may have decreased insulin sensitivity arising from altered body fat distribution.
In previous studies, a relationship between the rs10830963 polymorphism and sex was not demonstrated.13,15,25 Lopez-Minguez et al. 28 did not find any relationship between the rs10830963 polymorphism and total dietary energy intake during 24 hours in overweight/obese women. However, Goni et al. 29 found that females carrying the CC genotype lose more weight on a hypocaloric diet compared with those carrying the CG/GG genotype. Interestingly, our results indicated that body weight was lower in obese girls carrying the C allele, whereas body weight and waist circumference were higher in obese boys carrying the C allele. The mechanisms underlying the effect of melatonin on body weight regulation appear to differ across animal studies. In hamsters, melatonin induces a decrease in fat mass in some species, while it induces an increase in fat mass in other species. 11 Based on this information, we hypothesized that the wild-type C allele may have a sex-specific effect and may have a protective role against morbid obesity in obese adolescent girls. However, this potential sex-specific effect of the MTNR1B gene requires further validation in larger studies.
The main limitations of this study were its small sample size and inclusion of adolescent individuals only. It is likely that the study may have been underpowered to identify an association between the investigated MTNR1B polymorphisms and obesity. Studies in children and adolescents can be difficult to conduct because of concerns about missing school and the requirement to fast for 12 hours prior to the evaluation of laboratory parameters, as well as a lack of information on the risk factors (e.g. prenatal/socioeconomic/environmental factors, dietary and physical activity) related to obesity. In addition, SNP studies have important limitations in the evaluation of associations, but are nevertheless widely used and can facilitate comparisons between studies. Despite its overall limitations, the main strength of our study is that it is the first to investigate the relationship between the MTNR1B gene and adolescent obesity in the Turkish population.
In conclusion, we could not confirm a relationship between the rs10830963 and rs8192552 polymorphisms in the MTNR1B gene and adolescent obesity. However, we observed that the mutant 24E allele of rs8192552 may have a protective role against IR and hypercholesterolemia in obese adolescents. We did not observe any relationship between rs10830963 polymorphism and IR, although we showed that obese adolescents with MetS carrying a G allele may have increased susceptibility to impaired PFG level. Finally, we observed that the wild-type C allele may have a sex-specific effect on obesity. Additional studies with a larger sample size in various populations are needed to confirm these results, supporting the usefulness of genetic testing for prevention of obesity and the identification of therapeutic targets.
Footnotes
Acknowledgements
We thank Assoc. Prof. Dr. Serdar Ceylaner from Intergen Genetics Center Ankara, Turkey for his collaboration.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
