Abstract
Objective
To evaluate the preventive effects of Moringa oleifera on metabolic syndrome (MS) in male Wistar rats.
Methods
MS was induced by feeding rats a high-fat diet and drinking water containing 10% fructose for 6 weeks. In the preventive group, M. oleifera was orally administered for 3 weeks prior to the induction of MS, while in the treatment group, M. oleifera was administered for 3 weeks after the onset of MS. The treatment groups were compared with a control group of untreated rats with induced MS. Fasting glucose, oral glucose tolerance, insulin tolerance, total cholesterol, triglycerides, abdominal circumference, and systolic and diastolic blood pressure were measured before and after MS induction and/or M. oleifera treatment.
Results
After the induction of MS, the control group had higher fasting glucose levels than the preventive group. No significant differences were observed in insulin tolerance, oral glucose tolerance, cholesterol, triglycerides, abdominal circumference, or systolic or diastolic blood pressure. Compared with untreated controls, rats in the treatment group had significantly improved glucose tolerance, triglycerides, and abdominal circumference.
Conclusions
M. oleifera treatment attenuates MS in Wistar rats.
Keywords
Introduction
Metabolic syndrome (MS) is a group of signs and symptoms related to glucose and lipid metabolism and blood pressure regulation, including insulin resistance and abdominal obesity. Insulin resistance, the main etiology related to MS, refers to the pathophysiological condition in which a normal concentration of secreted insulin is insufficient to produce adequate responses in peripheral target tissues, such as adipose tissue, muscle, and liver. 1 The prevalence of MS varies from 10% to 84% according to diagnostic criteria, ethnicity, race, age, and sex; a quarter of the world’s population is estimated to have MS. 2
Moringa oleifera (MO) is a tree native to India. Some populations consume it in their daily diet, whereas others use as a nutritional supplement and for medicinal purposes, mainly for diabetes.3–6 Treatment with MO ameliorates hyperglycemia and hypertriglyceridemia in diabetic rats7,8 and in patients with diabetes mellitus. 9 As MO is an accessible and inexpensive plant, it may be beneficial in complementary therapy for MS. The aim of this study was to determine whether the oral administration of ground leaves of MO prior to starting a high-fat diet prevents the development of MS, as well as the effect of MO consumption after MS is already established.
Materials and methods
Experimental animals
Thirty-three male Wistar rats (8 weeks old, mean weight 250 ± 50 g) were obtained from Envigo, Inc. (Huntingdon, UK) and housed under pathogen-free conditions with a standard 12-hour light/dark photoperiod at an environmental temperature of 22 ± 2°C. During the experimental period, food and water were supplied ad libitum except during food deprivation periods prior to blood sampling. The animals were maintained according to specific laboratory animal protocols at the Bioteria of the Centro Universitario de Investigaciones Biomédicas, Universidad de Colima, México. All experimental and animal management protocols were in accordance with the ethical standards and specifications of the Mexican Official Norm technical specifications for the production, care, and use of laboratory animals (NOM-062-ZOO-1999). We also followed the recommendations of the US National Institutes of Health for the care and use of laboratory animals. All protocols were approved by the ethics committee of the Universidad de Colima.
Induction of metabolic syndrome
To induce MS, rats were fed a high-fat diet (Research Diets, Inc., New Brunswick, NJ, USA) (Table 1) and drinking water containing 10% dissolved fructose for 6 weeks. This method has previously been used in various studies to induce MS in rats.10–12 After administration of the high-fat diet, the presence of MS was confirmed by the significant elevation of three or more of the following parameters with respect to initial values: area under the curve (AUC) of the oral glucose tolerance test, AUC of the insulin tolerance test, triglycerides (TG), total cholesterol (TC), abdominal circumference (AC), diastolic blood pressure (DBP), systolic blood pressure (SBP), and fasting glucose. 13 The control group had significantly (p < 0.05) higher averages for fasting glucose (78.01 ± 3.4 vs. 103 ± 3.8 mg/dL), AUC of the oral glucose tolerance test (13030.5 ± 436.4 vs. 15472.5 ± 308.4), and AC (15 ± 0.1 vs. 17.9 ± 0.4) after MS induction compared with initial values, and the MO group had significantly higher (p < 0.05) average values for fasting glucose (79.3 ± 4.5 vs. 102.4 ± 3.8), AUC of the oral glucose tolerance test (13095 ± 237.5 vs. 15518.3 ± 462.0), AUC of the insulin tolerance test (1814.1 ± 88.8 vs. 2126.6 ± 57.2), and AC (14.8 ± 0.2 vs. 17.9 ± 0.3) after MS induction compared with initial values.
Orally administered high-fat rodent diet with 60% kcal % fat*
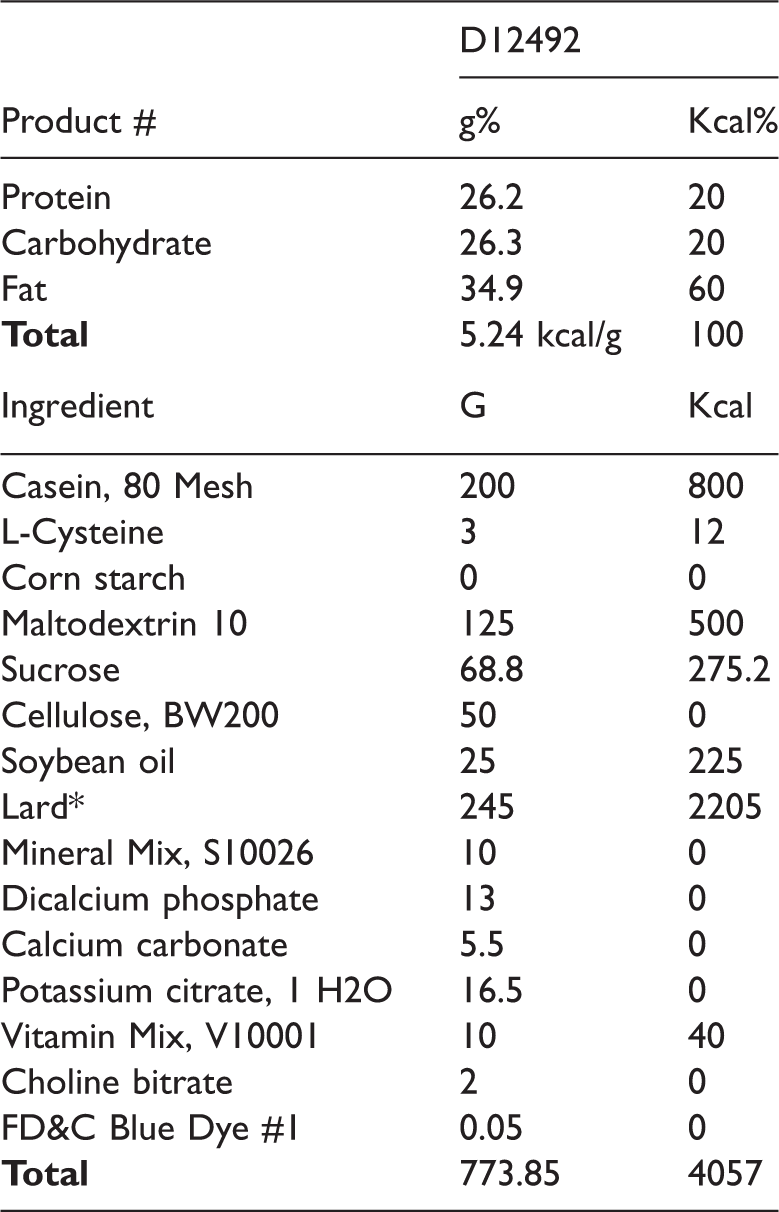
*Typical cholesterol in lard = 0.95 mg/gram. Cholesterol (mg)/4057 kcal = 232.8 cholesterol (mg)/kg = 300.8.
Experimental protocol
Three experimental groups were established. At the end of the study, the control group (n = 10), preventive group (n = 11), and treatment group (n = 9) comprised 30 animals in total. One rat from the control group and two rats from the treatment group died during the study period following respiratory failure. In the control group, MS was induced and no treatment was administered. The preventive group received MO leaf powder for 3 weeks before the MS induction diet was initiated. To assess the preventive effect of MO, final measurements from the preventive treatment group were compared with those of the control group at 9 weeks (at the end of MS induction) (Figure 1(a)). The treatment group received MO leaf powder (700 mg/kg body weight [bw]/day) for 3 weeks after MS induction. To determine the effect of MO in MS, the final measurements from the treatment group were compared with those taken at 12 weeks in the control group (3 weeks after MS induction) (Figure 1(b)).

Flow diagram of the study design. (a) Preventive experimental group: Moringa oleifera (MO) was administered for 3 weeks followed by high-fat diet (HFD) for 6 weeks. (b) Experimental group with metabolic syndrome: HFD was given for 6 weeks to induce metabolic syndrome followed by MO treatment for 3 weeks. (c) Control group: HFD for 6 weeks with no MO treatment at any time. Comparisons were made with the control group at the end of the HFD for the preventive group (9 weeks, a) or 3 weeks after inducing metabolic syndrome (12 weeks, b) for the treatment group (see dashed lines). Black bars represent periods of MO intervention, dashed bars indicate the experimental period with HFD, and white bars indicate periods without experimental intervention.
Plant material
Moringa oleifera Lam. leaves were purchased from local cultivators in the region of Rancho San Antonio, Sinaloa, Mexico (60 m.a.s.l., −107.663056 longitude, 25.041111 latitude), where the mean minimum annual temperature is 10.5°C, mean maximum temperature is 36°C, and mean annual rainfall is 790 mm.
Dose and administration
Powdered MO leaves were administered orally by gavage at a dose of 700 mg/kg bw/day at 9:00 am ± 30 minutes for 3 weeks. This dose was based on previous studies that demonstrated hypoglycemic effects in diabetic patients without toxicity and an allometric conversion for rats. Doses as high as 2000 mg/kg have been shown to not be toxic or lethal.14,15 For each animal, the powdered leaves were weighed daily using an analytical balance and the appropriate amount placed in 1.5-mL Eppendorf tubes. The powder was diluted in potable water and mixed by vortexing.
Measures of metabolic syndrome components
Fasting glucose (FG)
After 12 hours of fasting, a blood sample was drawn from the tip of the rat’s tail. Glucose was measured using an Accu-Check Active auto-analyzer (Roche, Mannheim, Germany).
Oral glucose tolerance test (OGTT)
Rats were administered 2 g glucose/kg bw by oral gavage. Blood samples were drawn after 0, 30, 60, and 120 minutes. Glucose concentrations were determined using an Accu-Check Active auto-analyzer. OGTT data were expressed as the AUC.
Insulin tolerance test (ITT)
Rats received a subdermal application of insulin (0.1 UI/kg bw; Eli Lilly and Company, Indianapolis, IN, USA). Blood samples were drawn after 0, 5, 15, and 30 minutes. Blood glucose concentrations were evaluated using an Accu-Check Active auto-analyzer. This method has a correlation of r = 0.984 (95% CI 0.974, 0.993) with respect to the hexokinase method. ITT data were expressed as AUC.
TC and TG
After 12 hours of fasting, blood samples were drawn from the tip of the rat’s tail. TC and TG levels were determined using an Accutrend Plus auto-analyzer (Roche Diagnostics, Indianapolis, IN, USA). Correlations of r ≥ 0.90 and p < 0.001 with regards to the reference method were found for TC and TG (Roche, Mannheim, Germany).
AC
AC was determined using a non-stretchable measuring tape and the technique described by Novelli et al. 16
Blood pressure
SBP and DBP measurements were taken under conditions intended to minimize stress. In a silent room, the rats were warmed at 36°C for 20 minutes to dilate their veins. The rats were then placed in an acrylic chamber with a heater at 30–32°C. Initial and final blood pressure were measured using a tail-cuff plethysmograph (Storage Pressure Meter model LE 5002; Panlab Harvard Apparatus, Barcelona, Spain). A clamp-type transducer and cuff were placed 2 cm from the proximal end of the rat’s tail. The potentiometer was then adjusted to stabilize the signal. When the rats were still, SBP and DBP were measured as the average of five consecutive measurements.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics, version 22 (IBM Corp., Armonk, NY, USA). The expressed variables were reported as mean and standard error. P < 0.05 was considered significant. To analyze the ITT and OGTT data, we calculated the AUC using the mathematical Tai model. 17 To assess differences in mean values between the beginning and the end of the intervention, we used a paired Student’s t-test for normally distributed data. To assess differences in the mean values recorded for MO intervention versus controls, we used the Student’s t-test for independent samples.
Results
Preventive effect of MO on rats with induced MS
Similar to the findings of studies on MO at equivalent doses or of the same duration in humans,8,14 we observed a hypoglycemic effect of MO in rats with induced MS. Following MS induction, the control group had higher fasting glucose than the preventive group (103 ± 3.8 vs. 80.09 ± 5.5 mg/dL; p < 0.05; Table 2). The two groups did not significantly differ with regard to the AUC for the ITT, OGTT, TC, TG, AC, SBP, or DBP (Table 2).
Preventive effects of Moringa oleifera (MO) powder on parameters of metabolic syndrome induced in rats
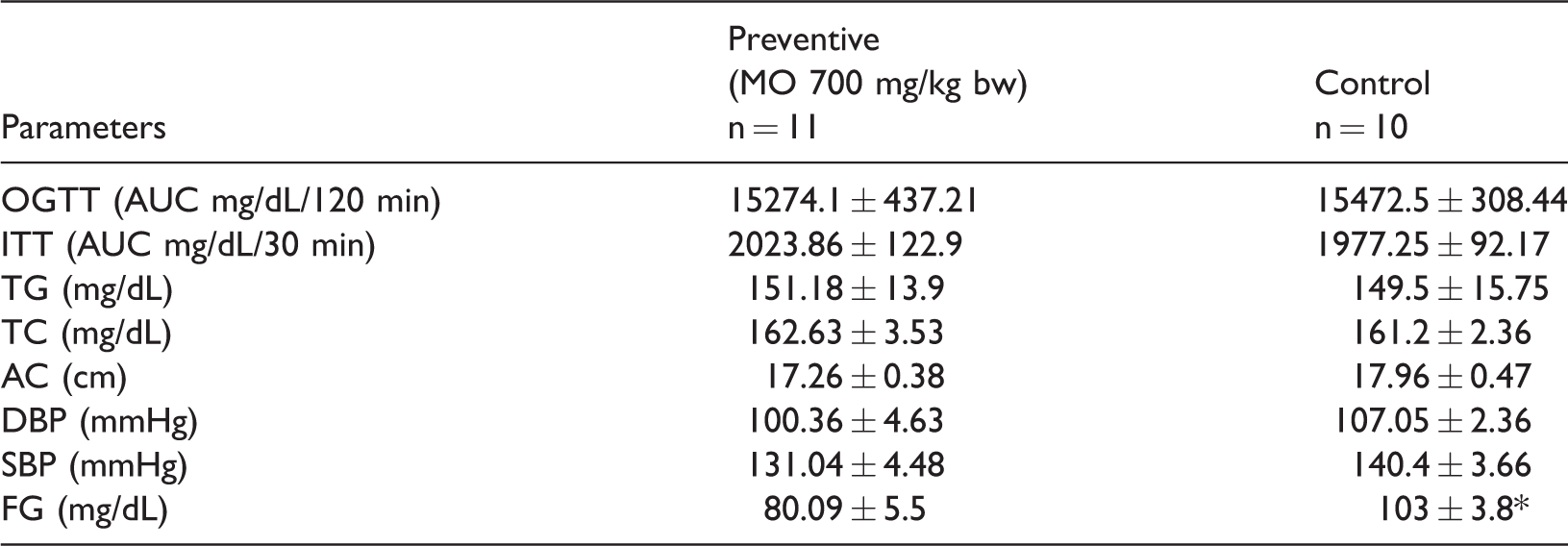
OGTT, oral glucose tolerance test; ITT, insulin tolerance test; AUC, area under the curve; TG, triglyceride; TC, total cholesterol; AC, abdominal circumference; DBP, diastolic blood pressure; SBP, systolic blood pressure; FG, fasting glucose.
*P < 0.05; Student’s t-test.
Values are presented as median ± SE.
Effect of MO on MS in rats
Following MO treatment, rats with induced MS exhibited a significantly reduced AUC for OGTT compared with the control group (12616 ± 316.8 vs. 13822.5 ± 213.93 mg/dL/120 min; p < 0.05; Figure 2c, d). At the end of the experiment, fasting glucose levels were lower in the treatment group than the control group, but these differences were not significant (Table 3). No significant differences in the ITT results were observed (Figure 2a, b). Compared with the control group, rats in the treatment group had significantly lower TG levels (140.56 ± 4.9 vs. 158.5 ± 1.4 mg/dL; p < 0.05) and AC (15.94 ± 0.3 vs. 17.2 ± 0.4 mg/dL; p < 0.05; Table 3).

Effect of Moringa oleifera (MO) powder in rats with induced metabolic syndrome. (a) Insulin tolerance test (ITT) results and (b) area under the curve. (c) Oral glucose tolerance test (OGTT) results and (d) area under the curve. In the group of rats treated with MO powder (700 mg/kg bw) after the induction of metabolic syndrome, blood glucose was measured prior to and 5, 15, and 30 minutes after insulin administration in the ITT, and prior to and 30, 60, and 120 minutes after glucose administration in the OGTT. Data are presented as the mean and standard error. *P < 0.05; Student’s t-test.
Parameters of metabolic syndrome after 3 weeks of treatment with Moringa oleifera (MO) powder

OGTT, oral glucose tolerance test; ITT, insulin tolerance test; TG, triglycerides; TC, total cholesterol; AC, abdominal circumference; DBP, diastolic blood pressure; SBP, systolic blood pressure.
Values are presented as median ± SE.
*P < 0.05; Student’s t-test.
Discussion
Oral administration of MO for 3 weeks in male rats with MS resulted in significantly reduced glucose tolerance, TG levels, and AC compared with untreated animals. However, 3 weeks of preventive MO administration decreased only fasting glucose levels compared with control rats. Previous studies have demonstrated that MO reduces hypoglycemia by minimizing gluconeogenesis and supporting the regeneration of damaged hepatocytes and pancreatic β cells in rats with diabetes induced by alloxan. 18 Chlorogenic acid is a compound found in MO that reportedly enhances insulin activity by triggering the AMP-activated protein kinase (AMPK) pathway. 19 Another compound found in MO leaves is kaempferol, 20 which improves glycolysis, glucose uptake, glycogen synthesis, AMPK activity, and GLUT-4 expression in skeletal muscle. 21 In diabetic mice, MO exerts an antioxidant effect and decreases the uptake of glucose by the intestines and skeletal muscle. 6
In our experiments in rats with established MS that were fed oral MO, no significant improvement in insulin sensitivity was observed by ITT, although the OGTT revealed lower glucose levels. This result may be explained by the presence of polyphenols in MO leaves that are related to the incretin effect at multiple levels, increasing GLP1 secretion and inhibiting dipeptidyl peptidase 4 (DPP4) with diverse effects on insulin sensitivity and secretion. 22 Paula et al. 23 identified insulin-like epitopes in protein fractions of MO that exhibited hypoglycemic effects when administered orally and intraperitoneally. Moreover, the effects of these insulin-like proteins are reportedly prolonged (similar to a long-acting insulin), likely because they are metabolized much more slowly than insulin itself as they are more resistant to hydrolysis and are likely not a substrate for insulin-degrading enzyme (IDE).
Rats that developed MS while on the high-fat/fructose diet and then received MO had test results similar to those that received preventive MO. Although the high-fat/fructose diet was not maintained in the groups with MS (control and treatment groups), TG levels and the AUC for OGTT were significantly lower in the group that received MO. These observed activities of MO may be attributable to chlorogenic acid, which reportedly improves lipoprotein lipase levels and reduces TG in diabetic rats. 24
MO also significantly reduced AC, possibly because MO powder contains about 12% (w/w) dietary fiber, which reduces gastric emptying by stimulating intestinal absorption and insulin secretion from β-cells.25,26 Similar results have been reported in rats treated with both metformin and MO, with a decrease in AC and food consumption observed compared with rats treated with metformin alone. 27
In experimental models of hypertension with a normal diet, however, MO treatment has produced contradictory effects. One study reported that MO had no effects on blood pressure, 28 but another study demonstrated an antihypertensive effect of MO, which was explained by the inhibition of IL-2 secretion and modulation of T-cell calcium signaling, an effect that was not observed in normotensive rats. 29 Thus, in the present study, although the rats had higher blood pressure after consuming the high-fat/fructose diet, they were not notably hypertensive, which may explain the lack of effectiveness of MO treatment on blood pressure.
In the present study, MO was administered for 3 weeks in male rats, but we did not examine the effects of a more prolonged preventive MO administration, different MO doses, or the effects of MO on female rats. Thus, we were unable to examine how MO may affect certain MS parameters over time.
Finally, we observed that MO-treated rats had significantly reduced glucose tolerance, TG levels, and AC compared with untreated animals. Moreover, 3 weeks of preventive MO treatment decreased fasting glucose levels. These effects were detectable 6 weeks after discontinuing MO administration.
The results of the present study may have been limited by several factors. First, the use of whole leaves makes it difficult to elucidate the mechanisms underlying the anti-MS effects of consuming MO. However, the main compounds of this plant, including chlorogenic acid, kaempferol, and fiber, have been identified in previous studies. The individual effects of these components on MS should be examined in future studies. Second, the term MS has recently been determined to refer to a single pathological entity in which treatment options for patients differ according to the manifestation of MS and the affected parameters in each individual. In addition, changes in lifestyle, including diet, are an established approach to the treatment of this disease, and a gold standard pharmacological treatment to control all parameters relating to MS is not available. 30 Third, a positive control group would require the administration of several different drugs, thus making it difficult to establish such a control. However, the main objective of this study was to determine whether the consumption of MO had a positive effect on MS in our experimental model.
In conclusion, the administration of MO for 3 weeks in male rats with induced MS resulted in significantly reduced glucose tolerance, TG levels, and AC compared with untreated animals. In addition, 3 weeks of preventive MO treatment decreased fasting glucose levels after MS induction.
Footnotes
Author contributions
MRS, MH, IDR, and ZU performed the statistical analyses. ML, YC, JAB, MHT, and KLQ collected the data. MH, MRS, and XT designed the experiments, contributed to the discussion, and wrote the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial aid from CONACyT-Mexico #268509 to MRS. YC, ML, and JABB received a CONACyT scholarship.
