Abstract
Aims:
Type 1 diabetes entails increased cardiovascular morbidity and cardiac chamber sizes are associated with cardiovascular disease. The aim of this study was to compare cardiac chamber sizes in normoalbuminuric persons with type 1 diabetes to a background population without diabetes.
Methods:
In a cross-sectional study, we examined 71 normoalbuminuric persons with long-term type 1 diabetes without known cardiovascular disease using cardiac multi-detector computed tomography. Cardiac chamber sizes and left ventricular remodelling were compared to persons without diabetes from the Copenhagen General Population Study.
Results:
Participants were median (interquartile range) 54 (48–60) (type 1 diabetes) and 57 (50–64) (without diabetes) years old and 59% were men (both groups). Participants with type 1 diabetes had smaller left ventricular mass (−3.5 g/m2, 95% confidence interval −5.8 to −1.3) and left (−4.0 mL/m2, 95% confidence interval −6.9 to −1.0) and right (−11.7 mL/m2, 95% confidence interval −15.4 to −7.9) ventricular volumes in multivariable analyses (adjusted for age, sex, body composition, blood pressure and antihypertensive medication), but no differences in atrial volumes.
Conclusion:
Persons with long-term type 1 diabetes had smaller left ventricular mass and biventricular volumes, yet similar atrial sizes, compared to a background population without diabetes. These findings may reflect subclinical development of diabetic cardiomyopathy.
Keywords
Introduction
Persons with type 1 diabetes have increased risk of cardiovascular (CV) disease; however, the mechanisms behind the increased risk are not fully understood.1,2 Controversy exists as to whether the condition termed diabetic cardiomyopathy could partly be responsible for the reported poor prognosis in persons with type 1 diabetes. 3 In individuals without diabetes, alterations in left ventricular mass (LVM) and left ventricle (LV) remodelling, that is, changes in LV mass-to-volume ratio, carry prognostic information.4–6
There is a need for more studies evaluating the influence of type 1 diabetes on cardiac chamber sizes as previous studies are not in agreement: type 1 diabetes has been associated with decreased, 7 unaltered 8 or increased size of LVM, 9 and left ventricular diastolic volume (LVDV) has been reported to be decreased9–11 or unaltered 8 in type 1 diabetes. Interestingly, a very important confounder for the assessment of LV size has been shown to be albuminuria, which is associated with increase in LVM10,12–15 and with smaller7,11,14 or unaltered LVDV.12,13,15
To our knowledge, systematic examinations of differences in cardiac chamber sizes and LV remodelling in persons with type 1 diabetes and a background population without diabetes remain to be performed. Cardiac multi-detector computed tomography (MDCT) scan is a robust method for measurement of cardiac chamber anatomy.16,17 The aim of this study was to assess the influence of type 1 diabetes on cardiac chamber sizes and LV remodelling compared to a background population without diabetes.
Methods
This was a cross-sectional study that included normoalbuminuric, long-term type 1 diabetes persons examined with cardiac MDCT scans as part of previous studies.18,19 For comparison to the background population, participants without diabetes but of similar age and sex without known CV disease enrolled in the Copenhagen General Population Study (CGPS) were identified.
Study participants
Participants with type 1 diabetes were recruited from the Diabetes Unit, Rigshospitalet, Copenhagen University Hospital and Steno Diabetes Center, Gentofte.18,19 Inclusion criteria in the study were type 1 diabetes (according to the American Diabetes Association 20 criteria) for more than 10 years; age between 18 and 75 years; and HbA1c <86 mmol/mol (<10%). Exclusion criteria were albuminuria (elevated S-creatinine >120 µmol/L or urinary albumin-to-creatinine ratio >30 mg/g), known CV disease and clinical symptoms or electrocardiographic (ECG) signs of heart disease. Participants with type 1 diabetes underwent MDCT from November 2009 until July 2014.
Individuals without diabetes were identified from the CGPS 21 (a large, Danish cross-sectional population study), in which healthy individuals underwent MDCT from February 2010 and onwards. Inclusion criteria for MDCT scan were ⩾40 years of age and normal kidney function (S-creatinine <100 µmol/L). 16 In the current study, participants in the CGPS database were excluded if they had missing demographic data, non-evaluable MDCT scan (no contrast MDCT images or images in systolic phase defined as ⩽50% RR-interval), diabetes, previous history of CV disease or age <38 or >70 years (range of age for type 1 diabetes participants). Furthermore, women from the CGPS database were randomly selected (using SPSS 22 random sample function) until equal men–women ratio in the two cohorts. Persons from the CGPS underwent MDCT scan from February 2010 until February 2015 in the current study.
The studies followed the Declaration of Helsinki II and were approved by the Danish Scientific Ethical Committee (protocol numbers H-4-2009-091, H-2-2014-012 and H-KF-01-144/01) and the Danish Data Protection Agency (RH-2017-254). All participants gave oral and written informed consent.
Demographics
Questions regarding medical history and collection of blood samples were acquired as previously reported.16,18,19 Weight, height and blood pressure (BP) were also recorded. Body surface area (BSA) was calculated according to Du Bois’ formula (BSA = 0.007184 × height (cm)0.725 × weight (kg)0.425) and used for indexed values of the cardiac chamber measures. BP was measured at rest; the CGPS participants were measured at Herlev and Gentofte Hospital and at Rigshospitalet (BP was defined as mean value of the two measures), 16 and the participants with type 1 diabetes were measured at Rigshospitalet. 18
Cardiac MDCT
Participants were scanned with a 320-detector CT (Aquillon One, Toshiba Medical Systems, Tochigi, Japan) and analysed on dedicated software (Vitrea, Vital Images Inc., MN, USA). Non-contrast images were acquired for measurement of coronary artery calcification. Intravenous contrast media (Visipaque 70–110 mL) was infused adjusted for body weight and with a flow rate of 5 mL/s, followed by saline chaser (30–50 mL) for contrast examination, allowing measurement of LVM and cardiac chamber volumes. Image acquisition started automatically at a density threshold of 180 Hounsfield Units in the descending aorta. Detector collimation was set at 320 × 0.5 mm and 100 kV tube voltage. Images were acquired ECG-gated during diastolic best-phase (approximately 75% of RR-phase) and reconstructed with appropriate slice thickness and filtering.16,22
Chamber measurements
These measures were performed as previously described. 16 In brief, LVDV and right ventricular diastolic volumes (RVDV) were derived as manual correction of automated detection of the endocardial borders. Left and right atrial volumes (LAV and RAV), including appendage, were measured manually by tracing the endocardial borders on up to 20 tomographic slices. LVM was calculated as the LV myocardial volume, derived by manual correction of automated delineation of the epicardial and endocardial borders, multiplied with the gravity of myocardial tissue (assuming a tissue density of 1.055 g/mL). Interobserver variability using these methods was previously described.16,17,23 All chamber sizes were then converted to indexed values (I) by dividing the values with BSA (i.e. LVM/BSA = LVMI). Remodelling of LV was calculated as left ventricular mass per diastolic volume (LVM/DV).6,24
Coronary artery calcification
Coronary artery calcification was analysed according to the Agatston method using 3 mm tomography slices; calcium was identified as an area exceeding 130 Hounsfield units. 25 Participants in this study were dichotomised according to presence or absence of coronary artery calcification.
Statistical analysis
A two-tailed p-value <0.05 was considered significant. Dichotomous variables are listed as frequency (percentage) and group differences were calculated with Chi2 or Fisher exact test. Continuous, normally distributed variables are expressed as means ± standard deviation (SD) and analysed for group differences with Student’s t-test. Continuous, skewed data are expressed as median (interquartile range [IQR]) and analysed for group differences with Mann–Whitney U-test. Multivariable linear regression analyses were used for determination of the influence of type 1 diabetes; predefined determinants for cardiac chamber measurements were age, sex, diabetes status, body mass index (BMI), use of antihypertensive treatment and systolic BP.16,26 Missing data (i.e. one cardiac chamber was not fully visualised) were handled analysis-by-analysis. Statistical analyses were performed using SPSS 22 (IBM SPSS Statistics, New York, NY).
Sensitivity analyses were performed in order to examine if the differences found differences were present in a better matched, but smaller population: The patients with type 1 diabetes were matched on age, gender and use of antihypertensive medication in a 1:4 manner with the persons without diabetes. After this matching, two of the persons without diabetes with the least comparable BP to the patient with type 1 diabetes were excluded. This rendered 66 patients with type 1 diabetes, each matched with two controls without diabetes for the sensitivity analyses.
Results
Participants
A total of 71 persons with type 1 diabetes were included in this study. In the CGPS scan database, 3076 persons were eligible, had similar range of age and men–women ratio compared to the participants with type 1 diabetes.
Table 1 shows baseline characteristics of participants with type 1 diabetes and persons without diabetes. The persons without diabetes were slightly older, had higher BMI and BP and were less often treated with statins and antihypertensive medication. Participants with type 1 diabetes had diabetes duration of 34 ± 10 years and HbA1c of 63 ± 10 mmol/mol (7.9% ± 0.9%). Coronary artery calcification was detected in 53 (75%) of participants with type 1 diabetes compared to 1322 (43%) in persons without diabetes (p < 0.001).
Baseline characteristics of the background population without diabetes and participants with type 1 diabetes.
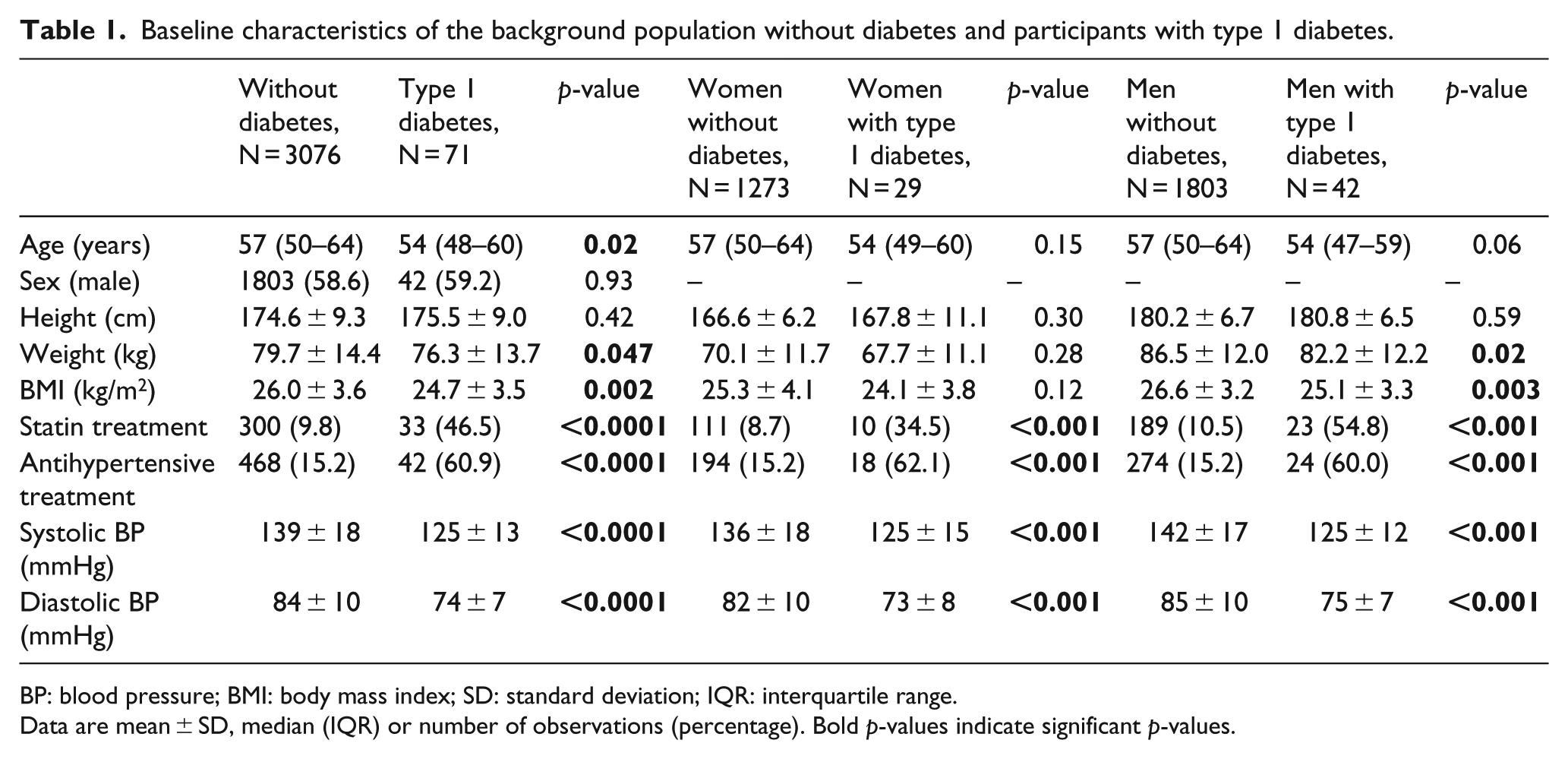
BP: blood pressure; BMI: body mass index; SD: standard deviation; IQR: interquartile range.
Data are mean ± SD, median (IQR) or number of observations (percentage). Bold p-values indicate significant p-values.
Comparison of cardiac chambers between persons with type 1 diabetes and persons without diabetes
Figure 1 illustrates values of indexed chamber volumes and indexed left ventricular mass (LVMI) stratified by gender. Men and women with type 1 diabetes had smaller LVMI and indexed right ventricular diastolic volume (RVDVI) than persons without diabetes. Only women had smaller indexed left ventricular diastolic volume (LVDVI). As a result of this, only men had a smaller LVM/DV, whereas women showed no difference in remodelling (Table 2). There were no differences in atrial sizes.
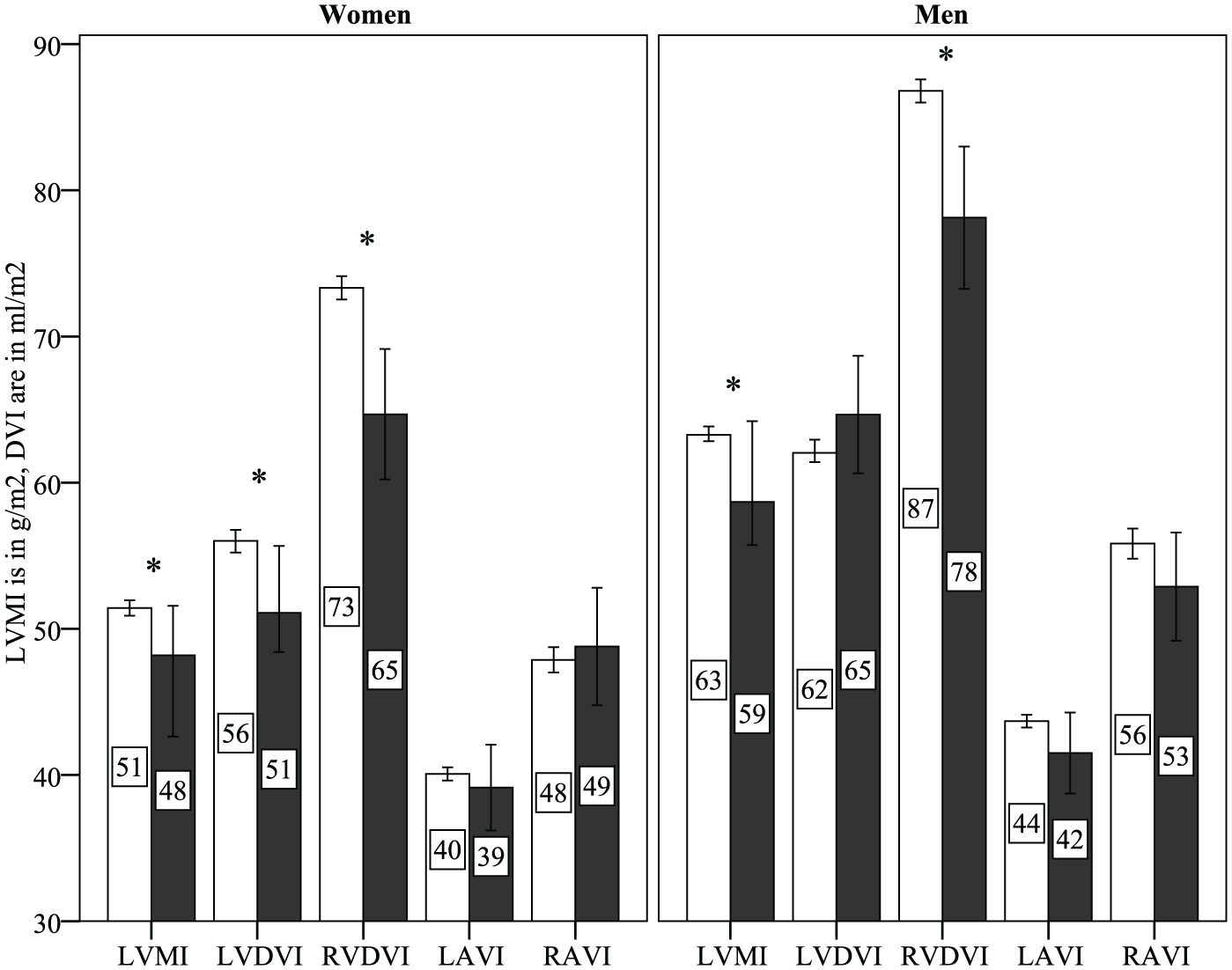
Cardiac chamber sizes in persons with type 1 diabetes and individuals without diabetes.
Comparison of cardiac chamber measurements between the background population without diabetes and persons with type 1 diabetes.
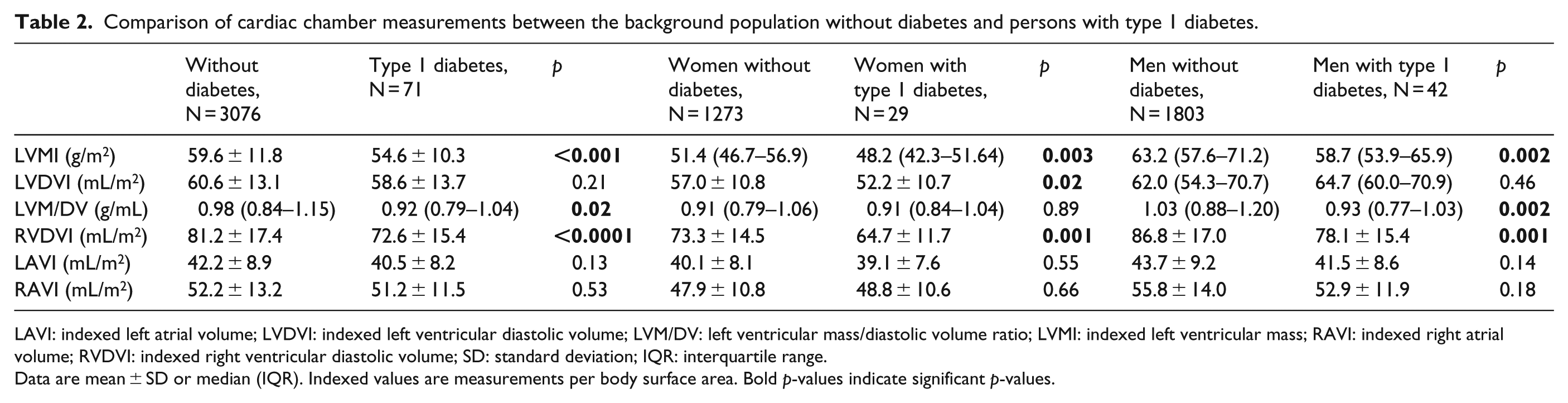
LAVI: indexed left atrial volume; LVDVI: indexed left ventricular diastolic volume; LVM/DV: left ventricular mass/diastolic volume ratio; LVMI: indexed left ventricular mass; RAVI: indexed right atrial volume; RVDVI: indexed right ventricular diastolic volume; SD: standard deviation; IQR: interquartile range.
Data are mean ± SD or median (IQR). Indexed values are measurements per body surface area. Bold p-values indicate significant p-values.
In multivariable linear regression analyses (Table 3), adjusted for age, sex, BMI, systolic BP and use of antihypertensive medication, type 1 diabetes was associated with smaller LVMI, LVDVI and RVDVI (all p < 0.01). Examination of interaction between diabetes status and sex for LVDVI (to examine the difference in Table 2) was not significant (p = 0.12). Adding the presence of coronary artery calcium to the model did not alter the results. There was a small difference of 1% (75% in persons with diabetes vs 74% in persons without diabetes, p = 0.02) in RR-phase between the two groups.
Influence of type 1 diabetes on cardiac chamber sizes compared to background individuals without diabetes.
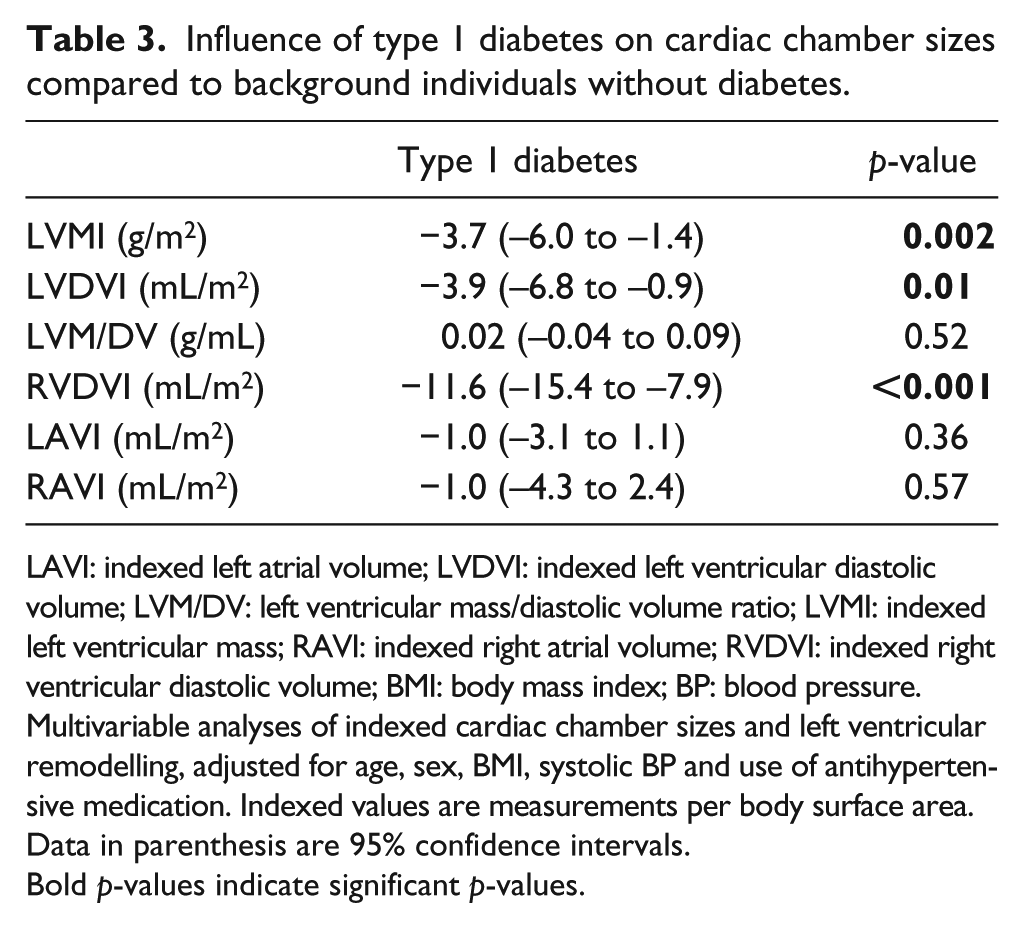
LAVI: indexed left atrial volume; LVDVI: indexed left ventricular diastolic volume; LVM/DV: left ventricular mass/diastolic volume ratio; LVMI: indexed left ventricular mass; RAVI: indexed right atrial volume; RVDVI: indexed right ventricular diastolic volume; BMI: body mass index; BP: blood pressure.
Multivariable analyses of indexed cardiac chamber sizes and left ventricular remodelling, adjusted for age, sex, BMI, systolic BP and use of antihypertensive medication. Indexed values are measurements per body surface area.
Data in parenthesis are 95% confidence intervals.
Bold p-values indicate significant p-values.
Results from the sensitivity analyses are shown in Supplemental Tables S1 and S2. There were no differences in the sensitivity analyses in age, gender or use of antihypertensive medication between the patients with type 1 diabetes and the matched controls and there were smaller differences in BMI, statin treatment and systolic BP. The differences shown in Figure 1 were also significant in the sensitivity analyses.
Discussion
In this work, we report for the first time that persons with type 1 diabetes have smaller ventricles yet with similar atrial cardiac chamber sizes when compared to a non-diabetic general population. We find 5 g/m2 smaller LVMI in participants with type 1 diabetes compared to persons without diabetes (p < 0.001) (Table 2). Men with type 1 diabetes had similar LVDVI compared to men without diabetes, whereas women had 5 mL/m2 smaller LVDVI, suggesting a gender-specific impact of diabetes on LV volume: this may be due to a wide range of confounders, for example, hormonal, and it would be interesting to see whether this finding is reproduced in the future. Persons with type 1 diabetes also had smaller RVDVI; this finding demonstrates that type 1 diabetes appears to structurally influence both cardiac ventricles. After adjustment for age, sex, BMI, BP and antihypertensive treatment, the type 1 diabetes condition remained associated with smaller cardiac LVM and biventricular volumes (Table 3). This finding correlates well with the previous report that higher HbA1c may be associated with smaller LVDVI in type 1 diabetes. 12
In a healthy, Danish population the sizes of ventricles decrease with age. 16 The persons with type 1 diabetes in this study have 5–10 mL/m2 smaller ventricles (both ventricles for women, but only right ventricle (RV) in men), which corresponds to an increase in ‘cardiac age’ of 10–20 years. 16 It has been speculated that the decrease in ventricular volumes with higher age in the general population could be due to myocardial stiffening accompanied by increasing diastolic dysfunction.16,27 These changes are hallmarks of the diabetic complication known as diabetic cardiomyopathy,3,28 which generally is difficult to quantify, but is in part caused by increased interstitial fibrosis. The fibrosis is occurring with increasing age, but is further enhanced in diabetes and consists of increased collagen deposition and cross-linking by interaction between collagen and glucose: the advanced glycation end-products.29,30 We do not have functional measures in these participants; hence, it is not possible to evaluate their systolic and diastolic functions, which would have been another clue of to what extent our findings may reflect diabetic cardiomyopathy. Nonetheless, the Thousand & 1 Study from Steno Diabetes Center (a cross-sectional study of persons with type 1 diabetes without known heart disease)7,31 revealed a high prevalence of diastolic ventricular dysfunction, which makes it plausible that our participants also have diastolic dysfunction.12,31 Interestingly, although LV was smaller in our cohort of type 1 diabetes participants, the average atrial sizes were not significantly different from the background population. This may reflect a relative atrial dilation, which is a frequent finding in individuals with restrictive LV filling and diastolic dysfunction. Thus, we speculate that our findings regarding ventricular mass and volumes, in these long-term, normoalbuminuric persons with type 1 diabetes, is a structural consequence of long-term diabetic cardiomyopathy.
Previous studies of persons with type 1 diabetes and LVMI have found normoalbuminuric participants to have both smaller 7 and similar LVMI 8 compared to controls; our finding supports evidence of smaller LVMI in long-term, normoalbuminuric persons with type 1 diabetes. Our result regarding smaller LVDVI in persons with type 1 diabetes is in accordance with previous results.7,11
RV heart failure is associated with increased cardiac mortality. 32 Type 1 diabetes has previously been associated with decrease in RV diastolic function; 33 but to our knowledge, this is the first description of a smaller RV in type 1 diabetes. Thus, this finding needs to be confirmed in other, larger studies in addition to the possible prognostic implications of this alteration in size. Our finding of a smaller RV may correlate well with reduced RV function, as we suspect our finding is due to increased myocardial fibrosis and thereby a stiffer myocardium.
It is necessary to investigate whether it is possible to normalise the sizes or reverse the possible restrictive cardiomyopathy in the individuals; future, randomised controlled studies should explore this. A possible study could include treatment with an angiotensin receptor blocker, as this has shown some effect on fibrosis and diastolic dysfunction a small study of patients with type 2 diabetes; 34 it would be interesting to investigate if this finding could be transferred to persons with type 1 diabetes for the prevention or even treatment of diabetic cardiomyopathy.
Cardiac chamber sizes are closely related to BP in addition to need of antihypertensive treatment and these factors should be considered when investigating cardiac morphology. In this study, the persons with type 1 diabetes had lower levels of BP and yet were more often treated with antihypertensive medication compared to the background population. This is likely due to the close BP monitoring, often yearly, of persons with type 1 diabetes. Furthermore, there is a lower threshold for initiating antihypertensive treatment in persons with type 1 diabetes than in the general population. 20 We suspect that these factors are the main explanations for the observed differences of BP (Table 1).
Limitations
We only have cardiac anatomical measures and not functional measures; hence, it is not possible to confirm our hypothesis of concomitant diastolic dysfunction. As this is a cross-sectional study, it is difficult to confirm whether the smaller sizes in participants with type 1 diabetes indeed are due to a faster decrease in sizes than in persons without diabetes – a longitudinal study is needed to confirm this hypothesis. Ideally, a study like this one would have included more participants with type 1 diabetes in order to account for more confounders in the multivariable analyses. A potential methodologic confounder could be image acquisition phase, the RR-phase, but a slight difference of 1% is not likely to have clinical impact. A potential confounder for the difference in cardiac chamber sizes found between study groups is difference in BP between the two populations. Nevertheless, we sought to adjust for this in the multivariable analyses and in the sensitivity analyses; the smaller ventricular sizes found in Figure 1 and Table 2 were still present after adjustments for BP in Table 3 and in Supplemental Table S2. Strengths in this study include a robust method for measurements of cardiac chambers, large N in the background population and exclusion of persons with albuminuria in order to avoid confounding from kidney damage.
Conclusion
In conclusion, we found smaller LVM and left and right ventricular volumes in long-term, normoalbuminuric persons with type 1 diabetes without known CV disease compared to a background population without diabetes – indicating that the consequence of type 1 diabetes is smaller ventricular volumes and mass. To our knowledge, this is the first study to describe this anatomical difference in right ventricular volume between type 1 diabetes and individuals without diabetes. We speculate that our findings are an expression of diabetic cardiomyopathy in these individuals.
Key messages
Type 1 diabetes entails increased risk of cardiovascular disease.
Cardiac chamber sizes and left ventricular remodelling carry prognostic information, but influence of long-term type 1 diabetes remains to be elucidated.
We find long-term, normoalbuminuric persons with type 1 diabetes to have smaller left ventricular mass and left and right ventricular volumes compared to the background population.
This is the first study to report smaller right ventricular volumes.
We argue that these findings may reflect diabetic cardiomyopathy.
Supplemental Material
Supplementary_tables_R2_CLEAN – Supplemental material for Cardiac ventricular sizes are reduced in patients with long-term, normoalbuminuric type 1 diabetes compared to the non-diabetic background population
Supplemental material, Supplementary_tables_R2_CLEAN for Cardiac ventricular sizes are reduced in patients with long-term, normoalbuminuric type 1 diabetes compared to the non-diabetic background population by Henrik Ø Hjortkjær, Tonny Jensen, Jannik Hilsted, Ulrik M Mogensen, Helle Corinth, Peter Rossing, Per E Sigvardsen, Andreas Fuchs, J Tobias Kühl, Børge G Nordestgaard, Lars Køber and Klaus F Kofoed in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors would like to thank the participants and the radiographic personnel at Rigshospitalet, Copenhagen University Hospital. This study has been supported by grants from the Arvid Nilssons Foundation, the AP Moeller Foundation and The Research Fund of Rigshospitalet. H.Ø.H., T.J., J.H., U.M.M., P.R., L.K. and K.F.K. made substantial contributions to conception and design. H.Ø.H., T.J., U.M.M., H.C., P.E.S., A.F., J.T.K. and B.G.N. made substantial contributions to acquisition or analysis of data. H.Ø.H., T.J., J.H. and K.F.K. made substantial contribution to interpretation of the data and drafting the article. U.M.M., P.R., H.C., P.E.S., A.F., J.T.K., B.G.N. and L.K. revised it critically for important intellectual content. All authors have approved the final version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: We report no conflicts of interest. Outside the submitted work, P.R. has received consultancy and/or speaking fees (to his institution) from AbbVie, Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Eli Lilly, Novo Nordisk and Sanofi Aventis and research grants from AstraZeneca and Novo Nordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by grants from the Arvid Nilssons Foundation, the AP Moeller Foundation and The Research Fund of Rigshospitalet.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
