Abstract
Purpose:
Copeptin and insulin-like growth factor binding protein-1 analysed at admission for a myocardial infarction in patients with type 2 diabetes mellitus predicts cardiovascular events. The present aim was to study the association between copeptin and insulin-like growth factor binding protein-1, the development of the levels over time, and if the predictive value remained when measured at hospital discharge and 3 months thereafter.
Methods:
Copeptin and insulin-like growth factor binding protein-1 were analysed in patients (median age = 70, male = 68%) with type 2 diabetes mellitus + myocardial infarction at admission (n = 393), discharge (n = 309) and 3 months later (n = 288). The primary endpoint was cardiovascular event (cardiovascular death/non-fatal myocardial infarction/stroke) with the three time points as separate baselines.
Results:
The median copeptin levels were 21.8 pmol/L at admission, 8.5 pmol/L at discharge and 8.4 pmol/L after 3 months, while insulin-like growth factor binding protein-1 levels continued to increase. There were significant correlations between the biomarkers at all occasions. During an average follow-up of 2.5 years, copeptin, but not insulin-like growth factor binding protein-1, predicted cardiovascular event at all occasions in unadjusted analyses. Copeptin remained as a predictor at discharge and after 3 months in the final multiple model (including: heart failure/age/creatinine clearance).
Conclusion:
The relationship between copeptin and insulin-like growth factor binding protein-1 during the initial phase of a myocardial infarction persisted in a less-stressful situation, and copeptin remained as a prognostic indicator at discharge and 3 months later.
Introduction
People with type 2 diabetes mellitus (T2DM) are predisposed to develop cardiovascular disease, such as myocardial infarction (MI), along with a more severe prognosis when such events occur. 1 The reasons for this are not fully known, making the search for novel risk factors and risk markers important and among them different hormonal axes have attracted attention. The present report focuses on the vasopressin and the insulin growth factor-1 (IGF-1) axes.
Copeptin, a more stable part of the pre-pro-vasopressin molecule secreted in equal amount to vasopressin, 2 is a negative prognostic marker in MI, both for individuals with and without T2DM.3,4 Furthermore, activation of the vasopressin system may play a role in the development of glucose abnormalities.5,6 In a previous report from the Diabetes Mellitus Insulin-Glucose Infusion in Acute Myocardial Infarction 2 trial (DIGAMI 2), 4 copeptin was a predictor of prognosis when measured at admission for acute myocardial infarction (AMI). Moreover, copeptin correlated with insulin-like growth factor binding protein-1 (IGFBP-1) and was the strongest prognostic predictor of these two biomarkers. This suggests a pathophysiological relation between the two hormonal systems, a relation initially proposed by Catrina et al., 7 demonstrating increasing levels of IGFBP-1 in patients with diabetes insipidus receiving desmopressin (a vasopressin analogue) infusions. Since both vasopressin and IGFBP-1 levels are affected by physiological stress,8,9 it is of interest to study whether the findings in the acute phase of an MI remain in the less-stressful state during the post-MI period.
The primary aim of the present epidemiological substudy of the DIGAMI 2 was to investigate if the association between copeptin and IGFBP-1 in patients with T2DM and AMI remains at the time for hospital discharge and 3 months thereafter. The secondary aims were to explore the copeptin and IGFBP-1 levels over time and to study if the prognostic impact of these biomarkers remained during the post-MI period.
Methods
The DIGAMI 2 was a multicenter, prospective randomized, open trial with blinded evaluation comparing three different glucose-lowering strategies in patients with T2DM and a suspected AMI. 10 Patients were suspected of AMI if they had symptoms (chest pain lasting more than 15 min during the last 24 h) and/or ST-segment changes in two or more leads. In total, 1253 patients were randomized to receive (1) acute insulin–glucose infusion followed by an insulin-based long-term glucose control (n = 474), (2) insulin–glucose infusion followed by standard glucose control (n = 473) and (3) routine glucose management according to local practice (n = 306). During a median follow-up of 2.5 (1.1–3.0) years, no significant difference in mortality and morbidity among the three groups was observed. In a biochemistry substudy, 575 of the DIGAMI 2 patients had blood samples stored and analysed at hospital admission, at the time for hospital discharge as well as 3 and 12 months thereafter, regardless of treatment allocation. In the present report, copeptin and IGFBP-1 were studied at three time points: hospital admission (n = 393), hospital discharge (n = 309) and 3 months later (n = 288). The admission data have been reported previously. 4
Laboratory analyses
Copeptin concentration was measured with a sandwich immunofluorescence assay (LUMI test CT-proAVP, BRAHMS AG, Henningsdorf/Berlin, Germany). The lower detection limit was 0.4 pmol/L, and the functional assay sensitivity (<20% interassay coefficient of variation) <1 pmol/L. 11 The serum IGFBP-1 concentrations were determined by radioimmunoassay according to the method of Póvoa et al., 12 with a sensitivity of 3 μg/L and intra-assay and interassay coefficients of variation of 3% and 10%, respectively. Creatinine clearance (mL/min) was calculated using the Cockcroft–Gault formula.
Outcomes
Copeptin and IGFBP-1 were studied in relation to three of the adjudicated endpoints in DIGAMI 2. 10 The primary endpoint was a composite of cardiovascular events (cardiovascular death and non-fatal MI or stroke) while the fatal and non-fatal events separately served as secondary endpoints.
Statistical methods
Continuous variables are presented as median and interquartile ranges (IQRs), and categorical data are presented as number and percentages unless otherwise stated. Associations between copeptin and IGFBP-1 were assessed using Spearman’s rank correlation test. Wilcoxon signed-rank test was used to evaluate the difference in biomarker levels over time.
The relationship between copeptin, IGFBP-1 and the primary and secondary endpoints was assessed using Cox’s proportional hazard regression and is presented as hazard ratio (HR) and 95% confidence intervals (CI). Because of skewed distributions, the copeptin and IGFBP-1 values were log-transferred prior to analysis. Each of the three defined time points – hospital admission, discharge and 3 months thereafter – was used as the baseline for the subsequent prognostic evaluation. Two multiple models were used where the first model involved both biomarkers. Heart failure, age and creatinine clearance were chosen from known predictors of outcome in the DIGAMI 2 trial to be included in the final multiple model, together with copeptin, based on stepwise adjustments of the admission analyses.4,10 The same set of variables was used for all endpoints at all time points. A two-tailed p < 0.05 was considered significant. All analyses were done using SAS, version 9.3 (SAS Institute).
Ethical considerations
The local ethics committee of the Karolinska Institutet, Stockholm, Sweden, approved the study protocol that complies with the Declaration of Helsinki. All recruited participants provided their written informed consent for the initial study and the follow-up.
Results
Baseline characteristics
The clinical and biochemical characteristics for the patients at admission have been presented elsewhere. 4 In brief, 70% were men, the median age was 68 years, body mass index (BMI) was 28 kg/m2, and the median duration of T2DM was 6–7 years. Of the entire study population, 38% of the patients had a history of previous MI, 20% had previous congestive heart failure and 22% were current smokers. In total, 34% were treated with insulin at baseline, 25% with metformin and 39% sulfonylureas.
Copeptin and IGFBP-1 levels
Figure 1(a) illustrates copeptin levels at the three time points of measurement, of which the admission levels have been published. 4 The median level at admission (21.8 pmol/L) was significantly higher than the corresponding level at the subsequent time points (discharge: 8.5 pmol/L; 3 months: 8.4 pmol/L).
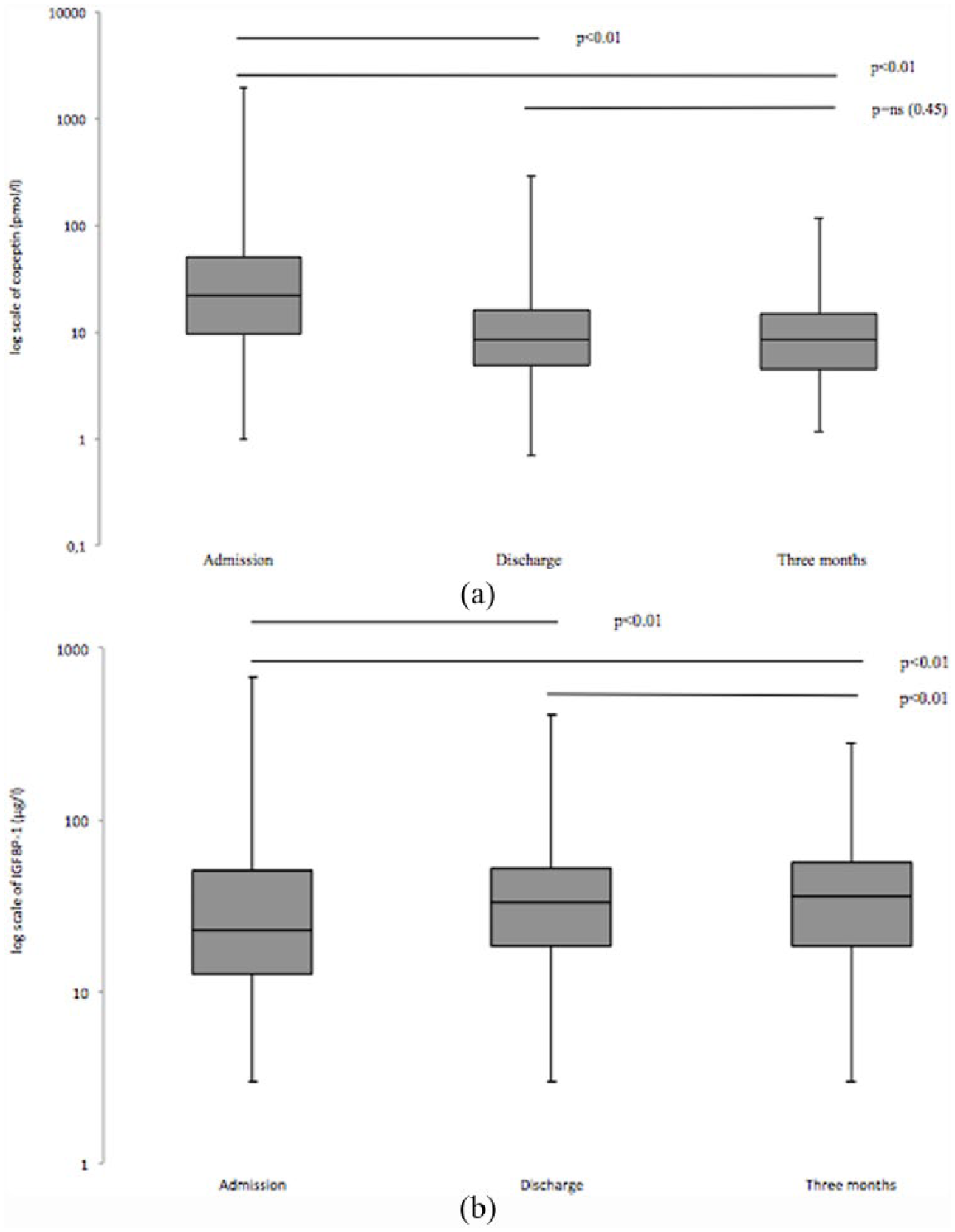
(a) The median copeptin levels at admission for MI (n = 393; 21.8 pmol/L), at discharge (n = 309; 8.5 pmol/L) and after 3 months (n = 288; 8.4 pmol/L). Boxes span the 25%–75% percentile, and whiskers outline minimum and maximum levels. Wilcoxon signed-rank test was used to evaluate difference over time. (b) The median IGFBP-1 levels at admission for MI (n = 393; 23.0 µg/L), at discharge (n = 309; 33.0 µg/L) and at 3 months (n = 288; 36.0 µg/L). Boxes span the 25%–75% percentile, and whiskers outline minimum and maximum levels. Wilcoxon signed-rank test was used to evaluate difference over time.
There was a correlation between copeptin at admission and at the time for hospital discharge (r = 0.47, p < 0.01) and between admission and after 3 months (r = 0.45, p < 0.01) as well as between hospital discharge and 3 months (r = 0.68, p < 0.01).
Figure 1(b) illustrates IGFBP-1 levels at the three different time points, of which the admission values have been published. 4 The median IGFBP-1 level at admission was 23.0 µg/L, at discharge 33.0 µg/L and after 3 months 36.0 µg/L.
The IGFBP-1 levels at hospital admission correlated with those at the time for hospital discharge (r = 0.43, p < 0.01) and after 3 months (r = 0.45, p < 0.01) and there was a correlation between IGFBP-1 measured at hospital discharge and 3 months later (r = 0.62, p < 0.01).
There was a positive correlation between copeptin and IGFBP-1 at hospital admission (r = 0.53, p < 0.01) and discharge (r = 0.31, p < 0.01) as well as after 3 months (r = 0.14, p = 0.02).
Prognostic influence of copeptin and IGFBP-1
The median follow-up time was 2.5 (1.1–3.0) years from admission during which 95 (24.2%) patients died, whereof 77 (19.6%) from cardiovascular causes. A non-fatal re-infarction occurred in 59 (15.0%) and a non-fatal stroke in 25 (6.4%) of the patients.
In unadjusted analyses, copeptin predicted the primary composite endpoint, cardiovascular events as well as its individual components cardiovascular death and non-fatal MI or stroke at all time points (Table 1). Increasing levels of IGFBP-1 predicted cardiovascular events and cardiovascular death at admission and discharge. IGFBP-1 only predicted non-fatal re-infarction or stroke at discharge.
Unadjusted and adjusted predictive ability of copeptin and IGFBP-1 assessed by Cox proportional hazard regression at admission, discharge and after 3 months.
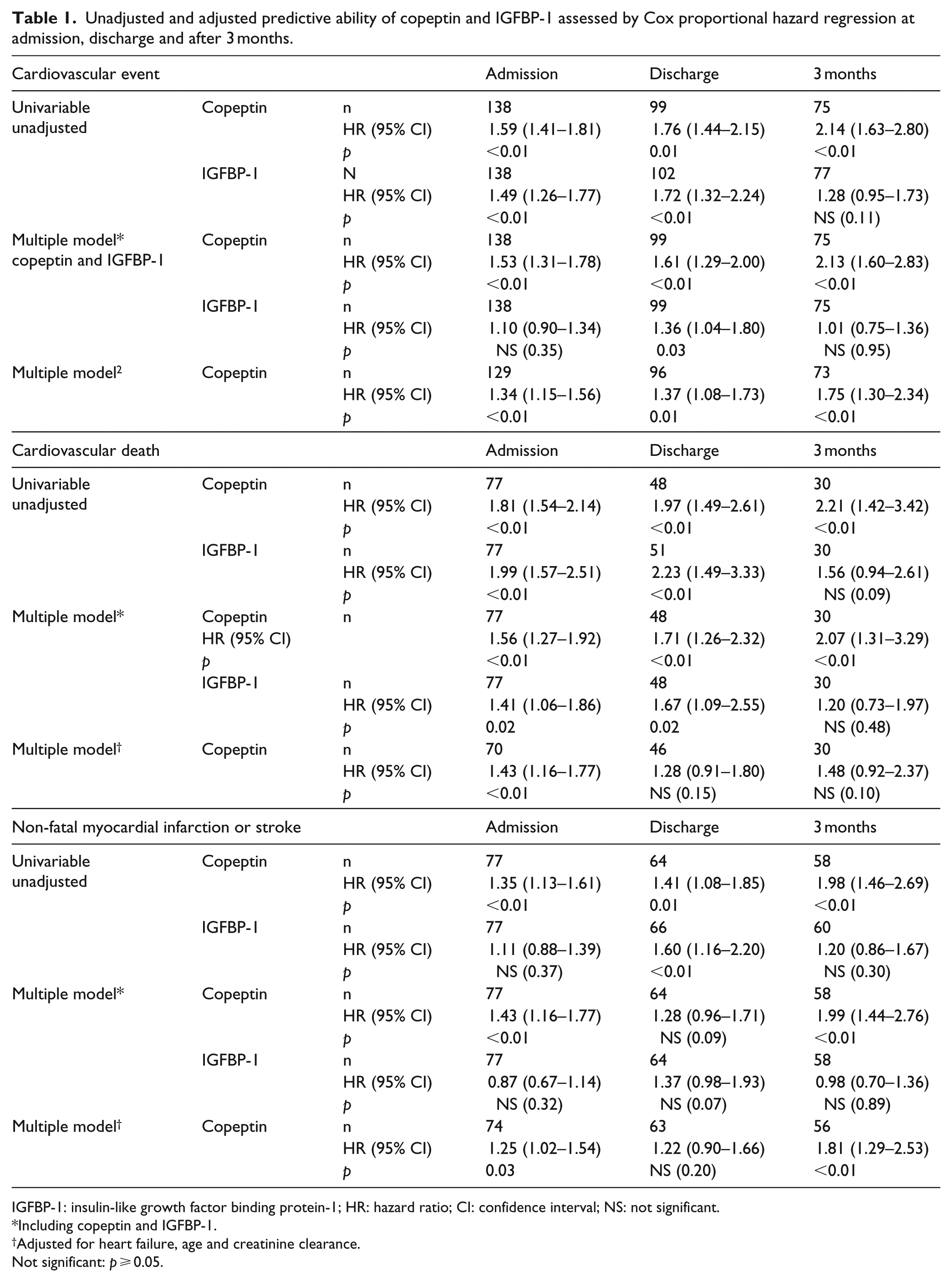
IGFBP-1: insulin-like growth factor binding protein-1; HR: hazard ratio; CI: confidence interval; NS: not significant.
Including copeptin and IGFBP-1.
Adjusted for heart failure, age and creatinine clearance.
Not significant: p ⩾ 0.05.
In a multiple model including both biomarkers, copeptin was a predictor of all events at all time points apart from non-fatal re-infarction and stroke at discharge, while IGFBP-1 remained a predictor for cardiovascular event at discharge and for cardiovascular death at admission and discharge (Table 1).
In the final multiple model including heart failure, age and creatinine clearance, copeptin remained an independent predictor for cardiovascular event at discharge and after 3 months. Copeptin was a predictor for cardiovascular death at admission and predicted non-fatal re-infarction or stroke at admission and after 3 months.
Discussion
This report expands previous knowledge on copeptin and IGFBP-1 in patients with T2DM and MI by adding information on the levels of these biomarkers as well as their predictive power in the stable post-MI period. The main findings were that the previously reported relation between copeptin and IGFBP-1 levels at admission persisted at the time for hospital discharge and 3 months later. Furthermore, copeptin, but not IGFBP-1, remained as a predictor for cardiovascular events when measured at discharge and 3 months after the MI.
The present finding that the correlation between copeptin and IGFBP-1 during the acute phase of an MI 4 was maintained during the post-MI period supports the hypothesis that there is an interaction between the vasopressin and IGF-1 hormonal axes. 7 IGFBP-1 production is up-regulated in response to an increase in pro-inflammatory cytokines, oxidative stress and N-terminal prohormone of brain natriuretic peptide (NT-proBNP), which all are triggered by physiological stress 9 such as at the onset of an AMI. It is mainly down-regulated by the inhibitory effects of insulin. 13 The finding of an association between copeptin and IGFBP-1 not only during the stressful state of an AMI but also thereafter indicates that other factors, such as vasopressin, might influence the IGF-1 axis. This assumption gains further support by the findings that copeptin was a stronger predictor than IGFBP-1 at all studied time points.
Both the IGF-1 and the vasopressin axes seem to be activated in the presence of dysglycemia. For example, IGFBP-1 may improve glucose homeostasis by enhancing insulin sensitivity, 14 where as vasopressin activation, measured as elevated copeptin levels, has shown to be a risk marker for T2DM. 15 Furthermore, copeptin is higher among patients with T2DM than in those without.6,16 Taken together with the present findings, it seems reasonable to conclude that activation of the vasopressin system relates to T2DM. Moreover, that a possible pathway is via the IGF-1 axis via increased secretion of IGFBP-1.
Vasopressin is a stress hormone, 8 which is the likely explanation that the highest levels of copeptin were seen at admission for an AMI in accordance with a report from Khan et al. 3 These authors reported that the highest copeptin levels were obtained at admission for an AMI in 980 patients (p < 0.01: day 1 vs day 5). 3 In the present investigation, the copeptin levels stabilized and reached similar levels at the time of hospital discharge as when measured 3 months later in the post-MI period. Interestingly, the copeptin levels at that time still exceeded levels reported in healthy populations,17,18 suggesting that the increase in copeptin in not only secondary to stress. A possible explanation is that all patients in the present cohort had T2DM. The present findings are hypothesis generating and warrant further studies in both epidemiological and mechanistic settings.
A limitation with this report is that not all patients had copeptin and/or IGFBP-1 levels available at all time points by logistic reasons (deaths, unwillingness to participate and improper sample storage). Nevertheless, copeptin remained as a significant predictor for cardiovascular events at discharge and 3 months later even after adjusting for known confounders.
Conclusion
The relationship between copeptin and IGFBP-1 observed in the acute phase of an MI is sustained over time. These results support the hypothesis of a connection between vasopressin, expressed as copeptin, and the IGF-1 system measured as IGFBP-1 at least in patients with diabetes. Moreover, it confirms the capacity of copeptin as a predictor of future cardiovascular events in T2DM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Authors’ contribution
M.I.S. and L.G.M researched and prepared data, wrote and edited the manuscript. K.B., S.-B.C., A.N. and V.G. contributed to the discussion and reviewed the manuscript. M.I.S. is the guarantor of this work and, as such, has full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by unconditional grants from the Swedish Diabetes Foundation, The Swedish Society of Medicine, the Swedish Heart-Lung Foundation, the Family Erling-Persson Foundation and the Signe and Olof Wallenius Foundation.
