Abstract
Objective:
To study the characteristics and prognostic implications of type 2 diabetes in different heart failure entities from a nationwide perspective.
Methods:
This observational study comprised 30,696 heart failure patients prospectively included in the Swedish Heart Failure Registry (SwedeHF) 2003–2011 from specialist care, with mortality information available until December 2014. Patients were categorized into three heart failure entities by their left ventricular ejection fraction (heart failure with preserved ejection fraction: ⩾50%, heart failure with mid-range ejection fraction: 40%–49% and heart failure with reduced ejection fraction: <40%). All-cause mortality stratified by type 2 diabetes and heart failure entity was studied by Cox regression.
Results:
Among the patients, 22% had heart failure with preserved ejection fraction, 21% had heart failure with mid-range ejection fraction and 57% had heart failure with reduced ejection fraction. The proportion of type 2 diabetes was similar, ≈25% in each heart failure entity. Patients with type 2 diabetes and heart failure with preserved ejection fraction were older, more often female and burdened with hypertension and renal impairment compared with heart failure with mid-range ejection fraction and heart failure with reduced ejection fraction patients among whom ischaemic heart disease was more common. Type 2 diabetes remained an independent mortality predictor across all heart failure entities after multivariable adjustment, somewhat stronger in heart failure with left ventricular ejection fraction below 50% (hazard ratio, 95% confidence interval; heart failure with preserved ejection fraction: 1.32 [1.22–1.43], heart failure with mid-range ejection fraction: 1.51 [1.39–1.65], heart failure with reduced ejection fraction: 1.46 [1.39–1.54]; p-value for interaction, p = 0.0049).
Conclusion:
Type 2 diabetes is an independent mortality predictor across all heart failure entities increasing mortality risk by 30%–50%. In type 2 diabetes, the heart failure with mid-range ejection fraction entity resembles heart failure with reduced ejection fraction in clinical characteristics, risk factor pattern and prognosis.
Introduction
Heart failure (HF) is a serious and increasingly prevalent condition causing major individual suffering and premature death worldwide. Diabetes not only increases the risk of developing HF 1 but also adversely affects prognosis when HF is established.2–4 Based on echocardiographic measurements including left ventricular ejection fraction (LVEF), HF has clinically been judged as heart failure with preserved ejection fraction (HFpEF; EF ⩾ 50%) and heart failure with reduced ejection fraction (HFrEF; EF < 40%). 5 Although pathophysiological mechanisms and associated risk factor profiles differ, HFpEF and HFrEF have been considered equally common and with a comparable prognosis.6–8 Recently, a new classification was introduced adding heart failure with mid-range ejection fraction (HFmrEF; EF: 40%–49%) to the already existing HFpEF and HFrEF in order to better understand the aetiology and pathophysiology in this ‘grey area group’. 5
Diabetes worsens the prognosis in both HFpEF and HFrEF,9–12 but existing data are somewhat sparse and mostly based on retrospective sub-group analyses of selected populations. In addition, analyses of HFmrEF with a focus on diabetes are so far lacking. Moreover, whether diabetes impacts prognosis differently in HFrEF and HFpEF has not been fully explored since few studies, if any, compared HFrEF and HFpEF within the same cohort, with diabetes as a primary objective.13–15 Ageing populations, changing risk factor pattern and improved survival in coronary artery disease may have influenced the proportion of patients with diabetes in HF populations and even the proportion and prognosis of HFpEF and HFrEF. 16 Updated information is important for future planning of healthcare demands.
This study aimed to investigate demographic characteristics and prognostic implications of type 2 diabetes (T2DM) in patients with HFpEF, HFmrEF and HFrEF in a nationwide contemporary Swedish HF population.
Methods
Data source
A detailed description of the Swedish Heart Failure Registry (SwedeHF; www.swedehf.se) has been published elsewhere. 17 A clinician-judged HF diagnosis is required for inclusion, and patients are reported via an online-based case report form. For this study, we selected patients registered for HF (68%) at hospital discharge or visit to a specialized HF outpatient clinic (32%) between 1 January 2003 and 30 September 2011, with a reported LVEF and arrived at a final population of 30,696 patients as detailed in Figure 1. Information on all-cause mortality until 31 December 2014 was generated through merging SwedeHF with the Swedish population registry. All patients were alive at least one day following their index date.
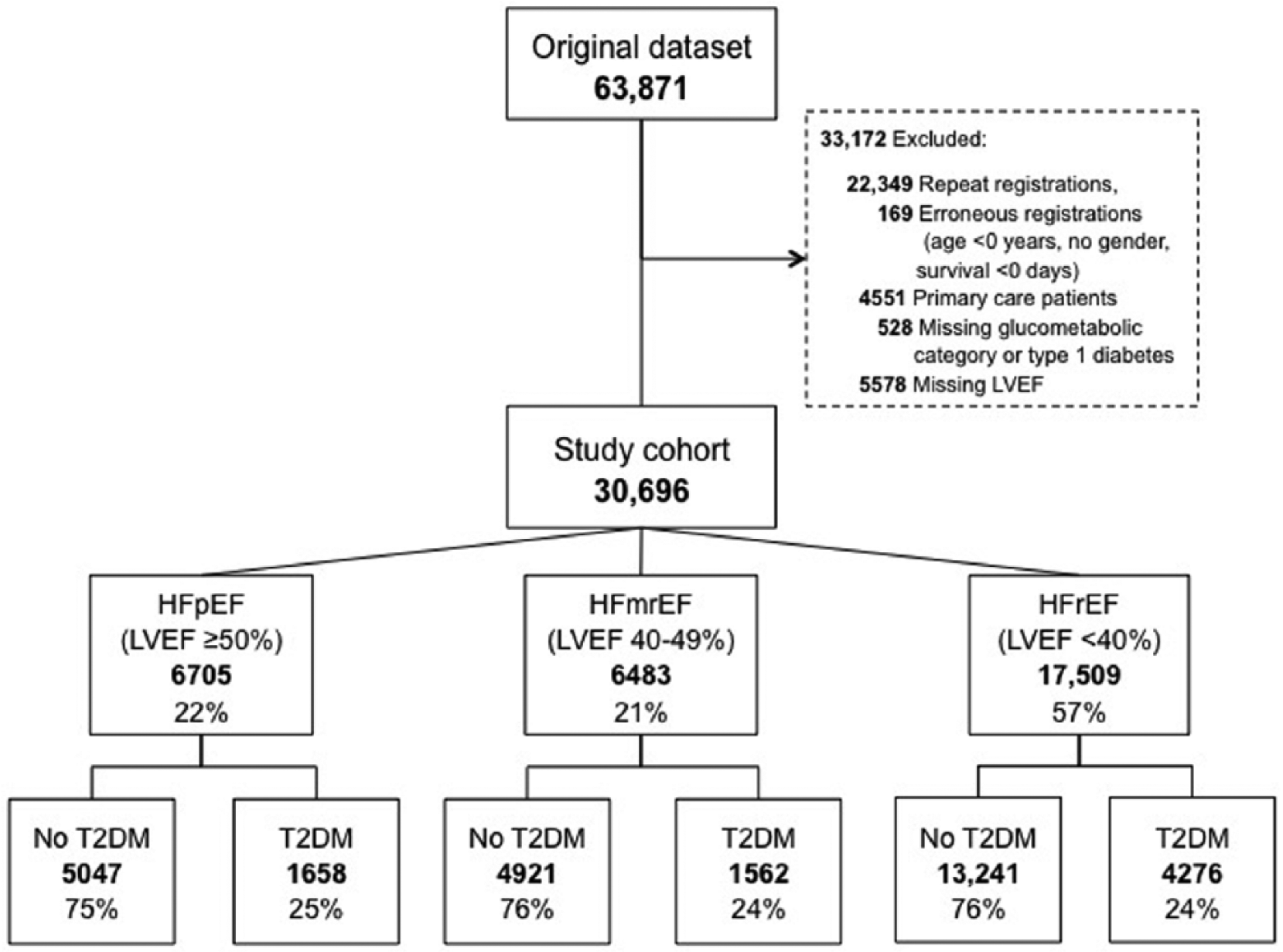
Flow-chart of the patient selection.
Definitions
Definitions were generally used as predefined in SwedeHF. HF was clinically diagnosed by the physician in charge according to the criteria used at the time of diagnosis, and the severity was based on the New York Heart Association (NYHA) functional classes I–IV. LVEF is reported to SwedeHF in four categories based on the most recent echocardiographically estimated LVEF: <30%, 30%–39%, 40%–49% and ⩾50%, continuous LVEF estimation was not available. For the present analysis HF entities were categorized according to the 2016 European Society of Cardiology (ESC) HF guidelines as preserved, mid-range and reduced LVEF; that is, HFpEF – LVEF ⩾50%, HFmrEF – LVEF 40%–49% and HFrEF – LVEF < 40%. 5 T2DM was defined as present or absent based on a confirmed history of this diagnosis and/or on-going glucose-lowering treatment. Comorbid conditions including hypertension, ischaemic heart disease (IHD), atrial fibrillation (AF), pulmonary disease, valvular heart disease and idiopathic dilated cardiomyopathy were defined as present or absent based on history of these diagnoses in the patient records. Renal function was based on serum creatinine and reported as estimated glomerular filtration rate (eGFR) according to the Modification of Diet in Renal Disease (MDRD) formula. 18 A cut-off for chronic kidney disease (CKD) was set at eGFR <60 ml/min/1.73 m2.
Statistical procedures
Analyses of statistical differences were undertaken comparing patients by diabetes status in the three HF entities (HFpEF, HFmrEF and HFrEF). Student’s t-test was applied for comparisons of continuous variables or when more than two independent groups by analysis of variance (ANOVA) verified by Kruskal–Wallis test and presented as mean and standard deviation when normal distribution. The non-normally distributed natriuretic peptides were logarithmically transformed prior to the t-test and were presented as median, range and 95% confidence interval (CI). Categorical variables were tested through chi-square test and presented as counts and proportions.
Survival analyses were performed using Log-rank test and are presented as age-adjusted estimated survival curves stratified by HF entity and T2DM. Mortality risk was estimated by means of crude and multivariate Cox proportional hazard regression generating hazard ratios (HRs) of mortality risk presented in a Forest plot. Adjustments were performed by age solely and extended by adjustment for important covariates in one additional model. Covariates were selected if they, in the entire cohort, had an unadjusted significance of p < 0.05, had less than 5% missing data and/or judged clinically relevant. The multivariable model (Model 1) included adjustments for T2DM, age, sex, level of care, duration of HF, weight, systolic and diastolic blood pressure, Hb class, eGFR class, IHD, hypertension, AF, pulmonary disease, valvular heart disease, revascularization, angiotensin converting enzyme inhibition (ACEi), angiotensin receptor blockade (ARB), mineralocorticoid receptor antagonist (MRA), beta-blockade, diuretics, statins, nitrates, aspirin and anticoagulants. The same model was applied in all sub-group analyses. A formal test of interaction between the three HF entities and T2DM was performed with Cox regression applying the same multivariate model as described above. A sensitivity analysis was performed after excluding all patients with valvular heart disease as well as in groups stratified by HF duration of more or less than 6 months. Mortality was also analysed by HFpEF versus HFrEF according to traditionally used definitions without including the HFmrEF entity (supplemental material). Moreover, a sub-group analysis was performed in the cohort with T2DM-stratifying groups by HF entity and the presence or absence of IHD (supplemental material). Separate analyses were performed according to level of care revealing no in-between group differences as regards to T2DM. In general, patients included from hospitals had a significantly higher mortality rate (data not presented). A two-sided probability of p < 0.05 was considered significant and is reported with 95% CI. Analyses were performed using SAS statistical software (version 9.4).
Ethical considerations
Establishment of SwedeHF and this specific analysis were approved by the Regional ethical review board at Linköping University (Dnr: 2011/1922-31/3 and M196-09) and conform to the declaration of Helsinki. In SwedeHF, individual patient consent is not required but patients are informed of registration and are allowed to opt out.
Results
As can be seen in Figure 1, the studied patient population (n = 30,696) included 22% with HFpEF (n = 6705), 21% with HFmrEF (n = 6483) and 57% with HFrEF (n = 17,509). Clinical characteristics in these three groups are detailed in Table 1. The proportion of T2DM was similar across the HF entities (HFpEF = 25%; HFmrEF and HFrEF = 24%). In general, patients with HFpEF were older, more often females with a larger proportion with hypertension and CKD compared with the patients with HFmrEF and HFrEF. The HFmrEF and HFrEF populations resembled each other regarding baseline characteristics with a larger proportion with reported IHD and preserved kidney function than those with HFpEF. In patients with T2DM, the major differences in characteristics were seen between those belonging to HFpEF versus those in the HFmrEF and HFrEF groups. Despite sometimes minor differences in statistical significance, no differences of clinical importance were seen between T2DM patients with HFmrEF and HFrEF apart from previous hypertension that was more common in HFmrEF than HFrEF, 63 versus 55%, p < 0.0001, and male sex that was more prevalent in HFrEF.
Baseline characteristics and treatment stratified by left ventricular ejection fraction (LVEF) and type 2 diabetes (T2DM).
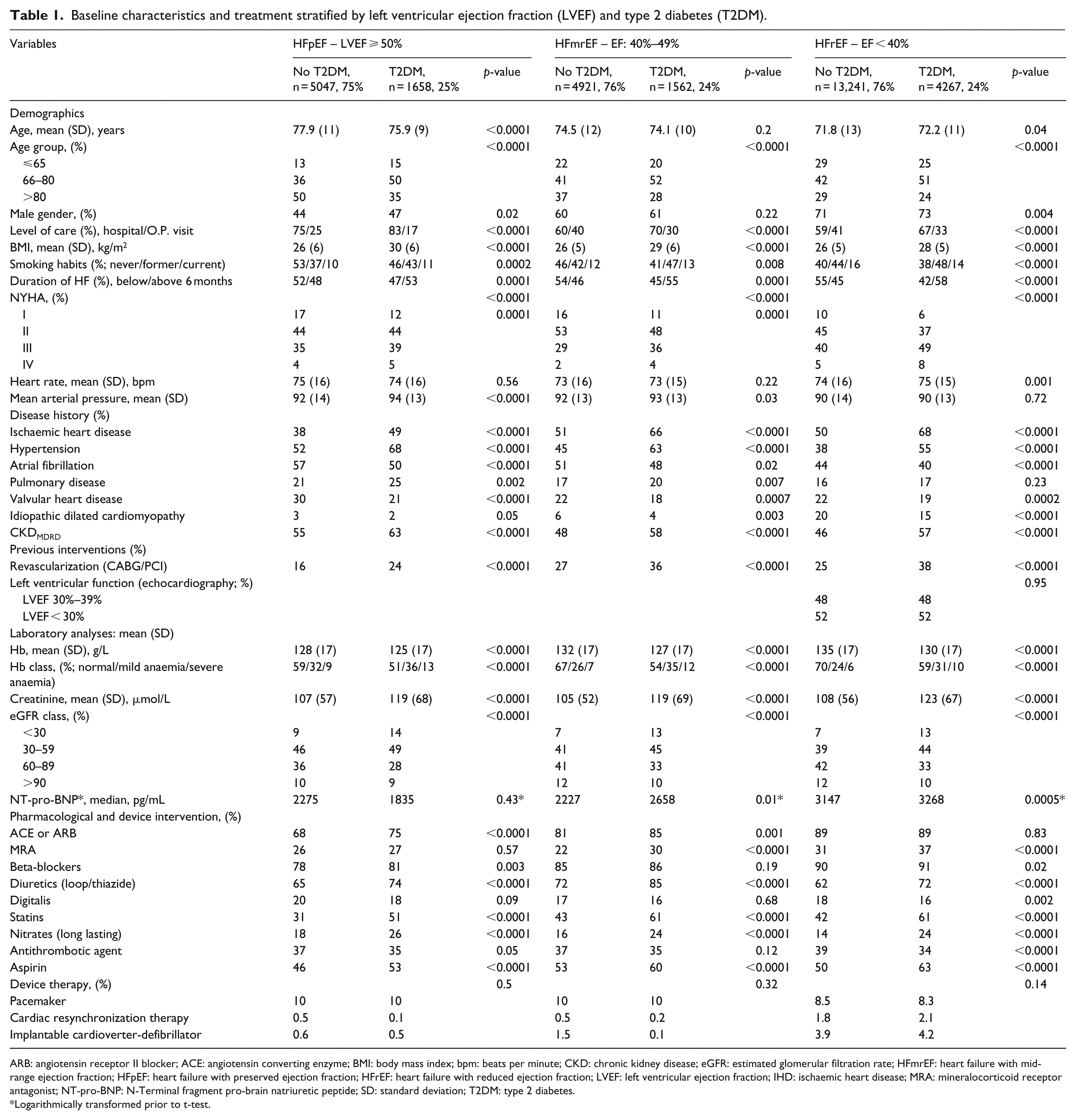
ARB: angiotensin receptor II blocker; ACE: angiotensin converting enzyme; BMI: body mass index; bpm: beats per minute; CKD: chronic kidney disease; eGFR: estimated glomerular filtration rate; HFmrEF: heart failure with mid-range ejection fraction; HFpEF: heart failure with preserved ejection fraction; HFrEF: heart failure with reduced ejection fraction; LVEF: left ventricular ejection fraction; IHD: ischaemic heart disease; MRA: mineralocorticoid receptor antagonist; NT-pro-BNP: N-Terminal fragment pro-brain natriuretic peptide; SD: standard deviation; T2DM: type 2 diabetes.
Logarithmically transformed prior to t-test.
Characteristics of HFpEF, comparison of T2DM versus no T2DM
Patients with T2DM were younger and less often females. They also more frequently had HF duration over 6 months, were more symptomatic (NYHA class III–IV), more often had a history of smoking and had a heavier risk factor burden with more overweight, IHD, hypertension, pulmonary disease and CKD. AF and valvular heart disease were less frequent. T2DM patients received more of pharmacological treatment and were more often revascularized.
Characteristics of HFmrEF, comparison of T2DM versus no T2DM
There was no difference in mean age. Patients with T2DM had a longer HF duration, more often NYHA class III–IV, slightly higher median N-Terminal fragment pro-brain natriuretic peptide (NT-pro-BNP; 2658 vs 2227 pg/ml, p = 0.01) and more comorbidity such as IHD, hypertension, CKD and pulmonary disease. AF and valvular heart disease were less common in T2DM. Patients with T2DM were more often revascularized and more frequently received treatment with renin angiotensin system (RAS) blockade, beta-blockers as well as statins and nitrates.
Characteristics of HFrEF, comparison of T2DM versus no T2DM
Among those with T2DM, male sex was slightly more common. In general, patients with T2DM had a longer HF duration, more often presented with NYHA class III–IV and slightly higher median NT-pro-BNP. The T2DM patients had a more frequent history of smoking, higher body weight and more frequently presented with IHD, hypertension and CKD, while AF was less common. Idiopathic dilated cardiomyopathy was more common in patients without T2DM (20% vs 15%). Pharmacological treatment did not differ between patients with and without T2DM regarding RAS blockade while patients with T2DM more often received beta-blockers, MRAs, statins and nitrates. Cardiac resynchronization therapy (CRT/ICD) was reported in 6% of the patients irrespective of T2DM.
Impact of T2DM on mortality in the three HF entities
The median follow-up time was 4 years (range: 0–12 years). Total mortality was higher and median survival time shorter in the presence of T2DM regardless of HF entity. Before adjustments the highest absolute mortality rate was seen in patients with HFpEF followed by those with HFmrEF and HFrEF (Log-rank p < 0.0001). However, after age adjustments, the most compromised survival was apparent in T2DM patients with HFrEF (Figure 2). After multivariate adjustment (Model 1; Figure 3) the HRs and 95% CIs of mortality associated with T2DM were in HFpEF – HR: 1.32 (1.22–1.43), HFmrEF – HR: 1.51 (1.39–1.65) and HFrEF – HR: 1.46 (1.39–1.54; p-value for interaction p = 0.0049).
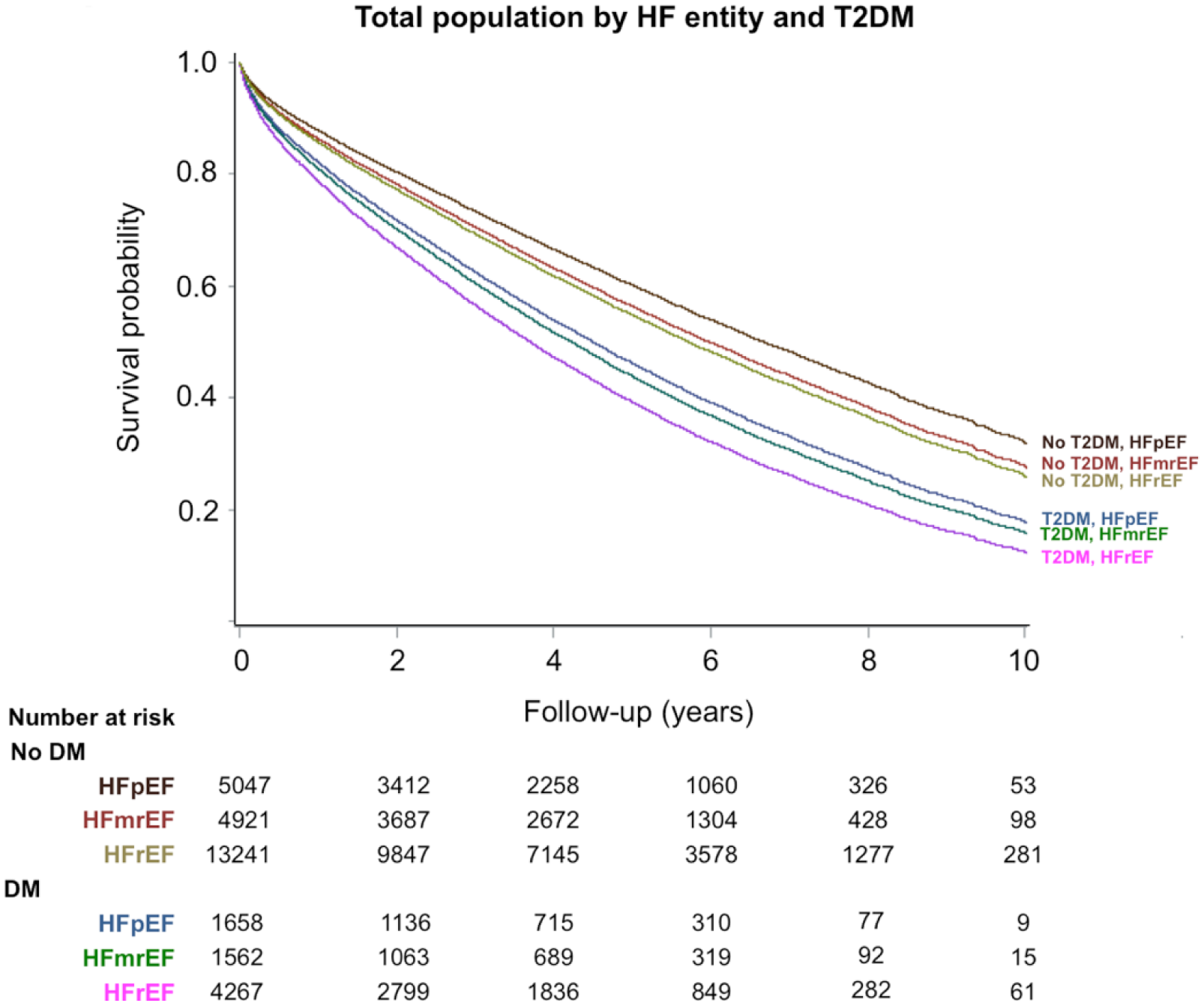
Age-adjusted long-term survival in the total population by heart failure entity and type 2 diabetes.
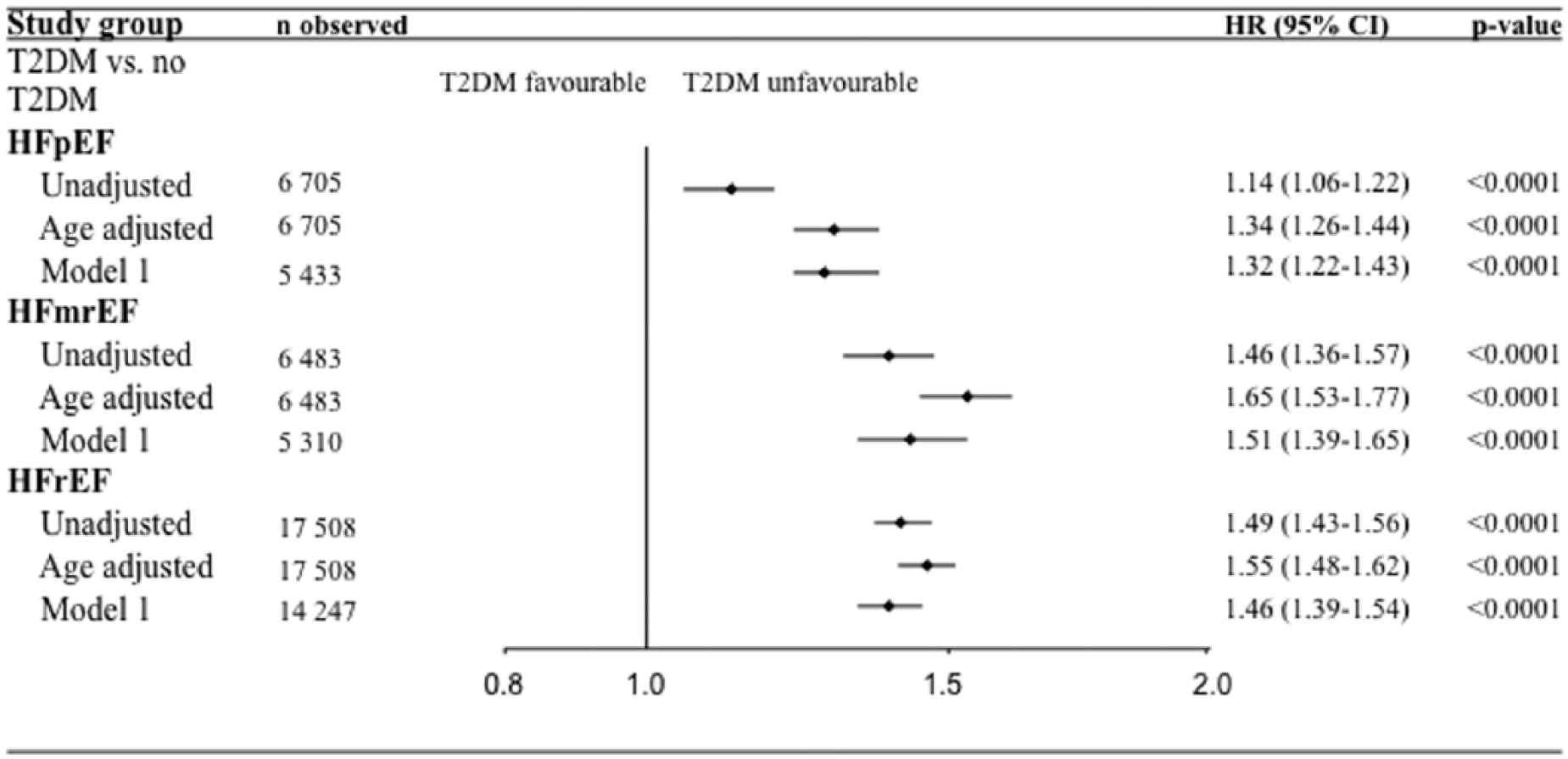
Forest plot of unadjusted and adjusted Cox proportional hazard ratios (HRs) of mortality by T2DM in HFpEF, HFmrEF and HFpEF, respectively.
When HFmrEF and HFpEF were analysed as one group, that is, including all patients with LVEF ⩾40%, the proportion of T2DM was 24% and the HR: 1.37 (1.22–1.55, adjustment model 1), while if HFmrEF was assembled with the HFrEF group (all with LVEF < 50%), the proportion of T2DM was unchanged, 24%, and the HR of mortality associated with T2DM was HR: 1.49 (1.36–1.64). Moreover, the prognosis deteriorated with further reduction of LVEF (Table 2). In T2DM, HFrEF also remained associated with worse survival than HFpEF when analysed according to the previous HF definition, dichotomized with LVEF cut-off ⩾/<40% or ⩾/<50% (i.e. HFmrEF entity omitted; Supplemental Figure).
Hazard ratios (HRs) for mortality in sub-groups of left ventricular function (LVEF) stratified by type 2 diabetes (T2DM).
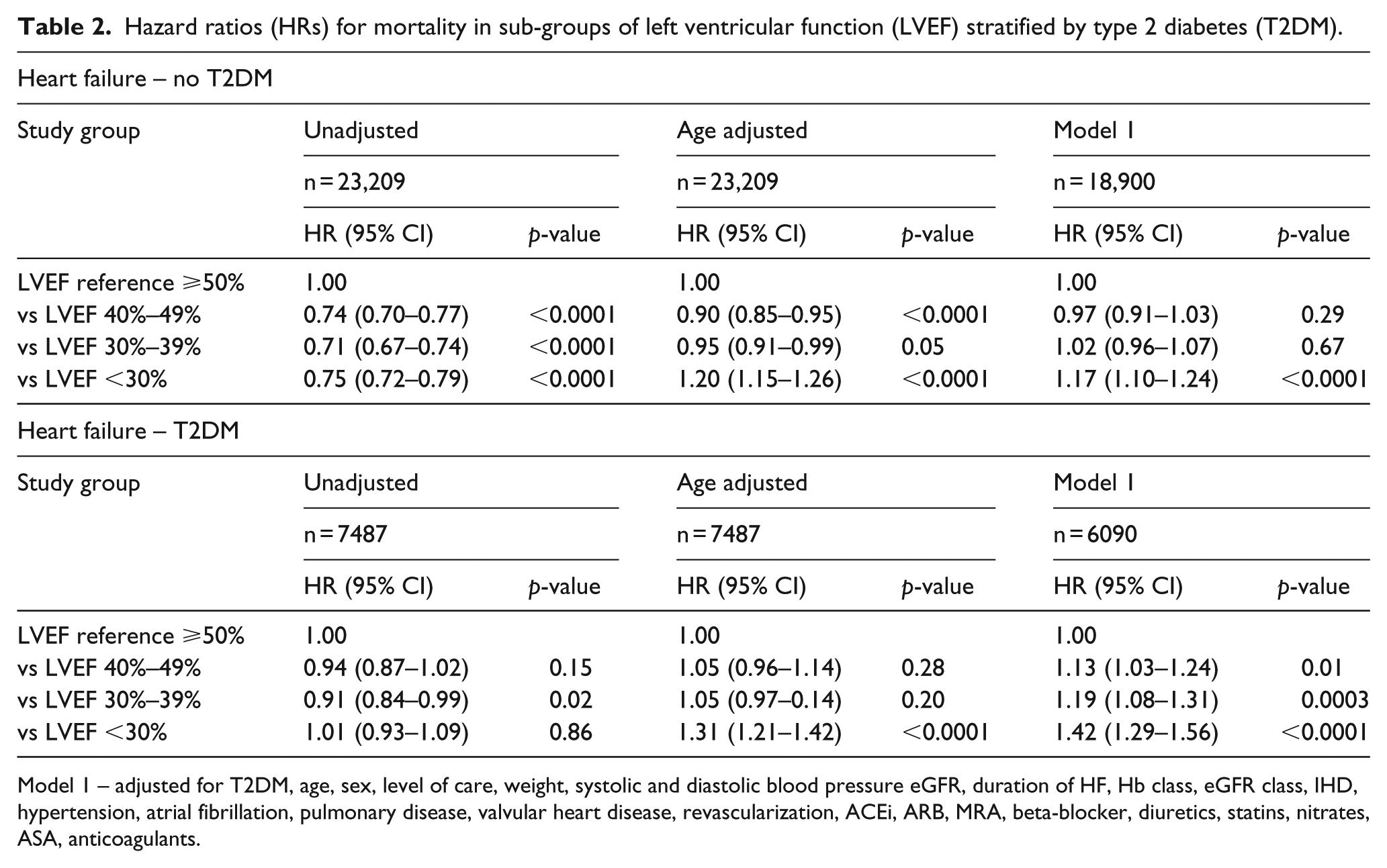
Model 1 – adjusted for T2DM, age, sex, level of care, weight, systolic and diastolic blood pressure eGFR, duration of HF, Hb class, eGFR class, IHD, hypertension, atrial fibrillation, pulmonary disease, valvular heart disease, revascularization, ACEi, ARB, MRA, beta-blocker, diuretics, statins, nitrates, ASA, anticoagulants.
Sensitivity analysis
The independent association of mortality in patients with T2DM remained robust after excluding history of valvular disease as well as when analysing in sub–groups with and without IHD. It was present also in those with short (<6 months) HF duration (Supplemental Tables S1 and S2).
Discussion
The main result of this analysis, applying a recently launched classification of HF in a contemporary HF population representing all-day practice, is that T2DM adversely impacts long-term prognosis across all HF entities. The negative impact was most pronounced in the HFmrEF and HFrEF groups where T2DM increased mortality risk by around 50% compared with 30% in HFpEF. These findings persisted even after adjustments for differences in age and the heavy comorbidity burden seen in patients with T2DM. Of interest is that baseline characteristics of the newly instituted HFmrEF largely resembled the HFrEF entity in patients with T2DM and with no substantial survival differences. A compromised systolic function, with an LVEF < 50% as cut-off, comprising three of four T2DM patients in the current population, severely impacted survival. The prognosis deteriorated with further reduction of the LVEF underlining the importance to prevent systolic dysfunction.
That T2DM was a somewhat stronger mortality predictor in HFrEF (and HFmrEF) than HFpEF contradicts the results from the Candesartan Heart failure-Assessment of Reduction in Mortality and morbidity (CHARM) trial in which the negative impact of T2DM on prognosis was significantly greater in HFpEF than HFrEF as reflected by a greater relative risk reduction in the former group. 13 The number of events was however higher in the HFrEF group irrespective of T2DM with similar absolute risk increase imposed by T2DM in HFpEF and HFrEF, possibly related to more important competing risks for HFrEF patients. A reason for the contradictory results may be different endpoints, cardiovascular (CV) death and worsening HF in CHARM versus total mortality in this study. Moreover, the CHARM population consisted of younger patients (mean age 67 vs 76 years in the HFpEF groups) with less advanced HF and fewer comorbidities compared with the present less-selected patient population. In addition, it is possible that the cut-off for HFpEF in CHARM, an LVEF ⩾40% versus the current ⩾50%, resulted in an HFpEF entity of T2DM patients with more severe prognosis. 13 Recent observations from the Prospective Comparison of ARNI With ACEi to Determine Impact on Global Mortality in Heart Failure (PARADIGM-HF) trial, including LVEF < 40%, are in line with this study showing, although only comparable in the HFrEF entity, a higher hazard of events related to diabetes with deteriorating LVEF. 19 This finding also resemble observations from a recent report from the Registro Nacional de Insuficiencia Cardiaca (RICA) registry, in which T2DM independently predicted 1-year survival with a trend towards higher mortality in HFrEF. 15
HFpEF have been reported to represent about half of an HF population.6,7 In this study, applying a cut-off of ⩾50%, about one-fifth of the T2DM population had HFpEF increasing to about two-fifths with a cut-off of ⩾40%. The general characteristics of the present HFpEF patients with T2DM are in line with those in previous reports, that is, older, with more females and a higher prevalence of hypertension and AF compared with their counterparts with HFrEF and in this cohort even HFmrEF.8,15
The phenotype and prognosis in patients with HFmrEF and T2DM was very similar to that of HFrEF apart from a somewhat larger proportion with IHD in the HFrEF group. In cohesion with the minor differences in baseline characteristics the long-term prognosis did not differ significantly between patients with HFmrEF and HFrEF in the T2DM-oriented analysis. Accordingly, the relevant question arises whether the introduction of the entity HFmrEF has any clinical value or if it is a matter of academic interest only, at least in the presence of T2DM. Future studies will provide further insights into this topic.
The reason behind the poor prognosis in T2DM patients with HF is most likely multifactorial and has yet to be fully elucidated in order to improve their treatment. Part of the adverse outlook does reasonably relate to their heavy burden of concomitant diseases especially IHD. Even in the absence of IHD, up to 90% of the T2DM patients have other comorbidities, among them are pulmonary disease, hypertension and AF. 20 T2DM did, however, remain an independent risk predictor of worse outcome after adjustment for comorbidities indicating that the dysglycaemic state in itself is driving HF development. Independent of IHD and hypertension diabetes has been associated with functional, biochemical and morphologic myocardial abnormalities. These perturbations have been referred to as diabetes cardiomyopathy.21–24 Myocardial dysmetabolism including impaired calcium handling, fibrosis and a reduced coronary flow reserve due to microvascular disease are among suggested reasons. Such abnormalities may explain the increased risk carried by T2DM patients both in HFpEF and HFrEF.23,24
We have previously reported that the disadvantageous prognosis for patients with T2DM could not be explained by differences in pharmacological treatment, 3 in fact, more extensive in the T2DM group, this study confirms that this pattern applies regardless of HF entity. There is, however, an underuse of device therapy (CRT/ICD) in HFrEF. 25 Further hope is provided by recent favourable effects of the sodium glucose transporter 2 (SGLT-2) inhibitor empagliflozin and canagliflozin26–28 with decreased risk of worsening HF or death in patients with T2DM. It seemed as if an important part of this effect was driven by a reduction in HF. Intensive research efforts are currently on-going for unravelling the underlying mechanisms. A plausible explanation, valid in both HFpEF and HFrEF, and accounting for the rapid onset of the effect, is a decrease in volume load related to osmotic diuresis and increased sodium excretion. On-going clinical trials such as the EMPEROR HF will provide further insight into the potential benefit of SGLT-2 inhibitors in HF patients without T2DM (www.clinicaltrials.gov).
In addition, interesting results were recently provided with the glucagon-like peptide 1 agonist liraglutide, which, in the LEADER trial, decreased CV mortality and morbidity in patients with T2DM and high CV risk. 29 A potential mechanism behind these benefits, which took some time to appear, is a retarded progression of vascular atherosclerosis. The impact on patients with compared to those without HF did not differ in this study.
Strengths and limitations
A major strength is the large population, representing a contemporary, everyday life HF population, with comprehensive data on important HF measures including LVEF, enabling a direct comparison between the three HF entities. The large size further enabled sub-group analyses of interest, showing consistency in our results. Important limitations are (1) the lack of information on serial measures and central reading of the echocardiograms as well as details of systolic and diastolic function such as E/e’ or global longitudinal strain, (2) no information on cause specific death, (3) NT-pro-BNP level could not be included in the multivariate analysis due to missing data and (4) lack of information related to T2DM including duration of the dysglycaemic condition, glucose control and glucose-lowering therapy. Finally, in the absence of a systematic screening with oral glucose tolerance testing, it is probable that the T2DM prevalence is underestimated.30,31
Conclusion
T2DM is an independent mortality predictor across all HF entities increasing mortality risk by 30%–50%. In T2DM, the HFmrEF entity resembles HFrEF in clinical characteristics, risk factor pattern and prognosis, indicating that the introduction of the mid-range entity, HFmrEF, may be unnecessary at least for clinical purposes, although this assumption needs further verification.
Supplemental Material
dvdres-mar-2018-00049-File008_S2 – Supplemental material for Type 2 diabetes and heart failure: Characteristics and prognosis in preserved, mid-range and reduced ventricular function
Supplemental material, dvdres-mar-2018-00049-File008_S2 for Type 2 diabetes and heart failure: Characteristics and prognosis in preserved, mid-range and reduced ventricular function by Isabelle Johansson, Ulf Dahlström, Magnus Edner, Per Näsman, Lars Rydén and Anna Norhammar in Diabetes & Vascular Disease Research
Supplemental Material
dvdres-mar-2018-00049-S1 – Supplemental material for Type 2 diabetes and heart failure: Characteristics and prognosis in preserved, mid-range and reduced ventricular function
Supplemental material, dvdres-mar-2018-00049-S1 for Type 2 diabetes and heart failure: Characteristics and prognosis in preserved, mid-range and reduced ventricular function by Isabelle Johansson, Ulf Dahlström, Magnus Edner, Per Näsman, Lars Rydén and Anna Norhammar in Diabetes & Vascular Disease Research
Footnotes
Author’s contributions
I.J. and A.N. developed the study design; U.D. and M.E. acquired the data; I.J. performed all statistical analyses under supervision by P.N. and A.N. All authors interpreted the results. I.J. and A.N. drafted the manuscript that was then reviewed and edited by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The study was enabled through unrestricted grants from the Swedish Heart-Lung Foundation and Stockholm County Council.
Supplemental Material
Supplementary files are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
