Abstract
Background:
There is clinical and experimental evidence for altered adenosine signalling in the fetoplacental circulation in pregnancies complicated by diabetes, leading to adenosine accumulation in the placenta. However, the consequence for fetoplacental vasocontractility is unclear. This study examined contractility to adenosine of chorionic vessels from type 1 diabetes mellitus, gestational diabetes mellitus and normal pregnancies.
Methods:
Chorionic arteries and veins were isolated from human placenta from normal, gestational diabetes mellitus and type 1 diabetes mellitus pregnancies. Isometric tension recording measured responses to adenosine and the thromboxane A2 analogue U46619 (thromboxane A2 mediates fetoplacental vasoconstriction to adenosine). Adenosine and thromboxane prostanoid receptor protein expression was determined by immunoblotting.
Results:
Adenosine elicited contractions in chorionic arteries and veins which were impaired in both gestational diabetes mellitus and type 1 diabetes mellitus. Contractions to potassium chloride were unchanged. Adenosine A2A and A2B receptor protein levels were not different in gestational diabetes mellitus and normal pregnancies. Contractions to U46619 were unaltered in gestational diabetes mellitus arteries and increased in type 1 diabetes mellitus arteries. Overnight storage of vessels restored contractility to adenosine in gestational diabetes mellitus arteries and normalized contraction to U46619 in type 1 diabetes mellitus arteries.
Conclusion:
These data are consistent with the concept of aberrant adenosine signalling in diabetes; they show for the first time that this involves impaired adenosine contractility of the fetoplacental vasculature.
Background
Diabetes mellitus (DM) during pregnancies, maternal diabetes, is recognized as a hostile environment for the development of the embryo and fetoplacental circulation. It can result in foetal morbidity and, later, an increased risk of cardiovascular diseases.1,2 Maternal diabetes can be divided into pregestational and gestational diabetes mellitus (GDM). Pregestational DM refers to women previously diagnosed with either type 1 or type 2 diabetes mellitus (T1DM and T2DM, respectively) prior to the pregnancies. GDM, however, is defined as any degree of glucose intolerance with the onset of pregnancy. Optimal utero-placental and feto-placental blood flow, that is, efficient placental perfusion throughout gestation, is essential for oxygen and nutrient transport to the developing foetus. Human placenta at term from both pregestational type 1 and gestational diabetes have been shown to exhibit histopathological changes such as placental immaturity and abnormalities in both foetal and maternal placental vasculature.3–8 While placentae from pregnancies complicated by T1DM exhibited increased angiogenesis and leaky blood vessels with loss of key junctional adhesion molecules, GDM placentae were also found to show less severe, but still statistically significant downregulation of endothelial cell–cell junctional molecules.5,6
The human fetoplacental unit (umbilical cord and foetal placental vessels) is known to be devoid of nerves; therefore, only non-neuronal mechanisms can influence vascular tone and blood flow in this tissue. 9 Adenosine has been identified as one of the autacoids that is actively released in the foetal circulation and placental tissue of humans and other mammals,10–14 and there is evidence that its signalling is altered in pregnancies complicated by diabetes. A higher adenosine concentration in venous umbilical blood, but not in arterial umbilical blood, was detected in pregnancies with GDM compared to normal pregnancies, suggesting an accumulation of extracellular adenosine from the placenta. 15 The extracellular concentration of adenosine in human umbilical vein endothelial cell (HUVEC) cultures from GDM was also higher than in HUVECs from normal pregnancies, and evidence was presented that this involved reduced human equilibrative nucleoside transporter 1 (hENT1)-mediated adenosine uptake.16–19
Very little is known about the effect of diabetes on the fetoplacental vasomotor response to adenosine. The only report to date used human umbilical vein, the largest fetoplacental vessel, and showed that endothelium-dependent relaxation to adenosine involving A2A receptors was impaired in GDM. 19 Adenosine can also mediate contraction of the human placental vasculature (placental cotyledons and chorionic arteries and veins) via activation of A2B receptors following synthesis of thromboxane A2 or a related metabolite and subsequent activation of prostanoid thromboxane prostanoid (TP) receptors.20,21 In this study, responses to adenosine of chorionic vessels (arteries and veins) from type 1 and gestational diabetes were compared to those of normal pregnancies. The selectivity of any changes was assessed by investigation of vasocontractile responses to a thromboxane A2 analogue, U46619, and smooth muscle contractility of the placental vessels was assessed using a receptor-independent vasoconstrictor, potassium chloride (KCl). Adenosine and TP receptor protein expression levels were measured using immunoblotting. In light of the evidence for increased placental adenosine levels in maternal diabetes, 15 we also sought to investigate whether any changes in contractility to adenosine of the chorionic vessels could be recovered after an extended period in the absence of exposure to extracellular adenosine, namely, by overnight storage in physiological solution.
Methods
Human placenta samples
This study was based on pregnancies monitored at Queen’s Medical Centre (QMC), Nottingham, UK, and had the approval of the QMC and University Hospital NHS Trust Ethics Committee (Ref: 0G010101; PI-LL). The study was carried out in accordance with the declaration of Helsinki. Pregnant women with singleton pregnancy scheduled for elective Caesarean section were approached for written informed consent. The study groups included pregnant women who had pre-existing type 1 diabetes (T1DM), those diagnosed as having gestational diabetes (GDM) in the second trimester by either a fasting plasma glucose level of ⩾5.6 mmol/L or a 2-h plasma glucose level of ⩾7.8 mmol/L 22 and those who had normal, uncomplicated term pregnancies. Placentae were taken from elective sections only in order to avoid any parturition signals that may alter vascular reactivity. Pregnant women with asthma, smokers and pregnancies with other maternal or foetal conditions complicating the pregnancies (hypertension, preeclampsia and placenta praevia) were excluded.
Placentae taken were from full-term (>37 weeks) pregnancies. No significant difference was found in placental weight or birth weight between the three study groups. The GDM group was treated with metformin (3) or diet (4), while all T1DM were insulin treated. HbA1 were <6.8% in the diabetic groups.
Placentae (with cord clamped) were brought to the laboratory within 30 min of the delivery and transferred to a dissecting bath with physiological salt solution (PSS), which had been gassed with 5% CO2 in air and adjusted to pH 7.4. The composition of the PSS was (mM): 127.76 NaCl, 25 NaHCO3, 4.69 KCl, 2.4 MgSO4, 1.18 KH2PO4, 6.05 glucose, 0.034 ethylenediaminetetraacetic acid (EDTA) and 1.6 CaCl2. One superficial chorionic artery and one chorionic vein were carefully dissected from each placenta. Segments were derived between first- and third-order branches of chorionic vessels (diameter 1–2 mm). Two vessel ring segments (length ~0.5 cm) were obtained from each artery and vein from each placenta. The ring segments were used both as fresh tissues and after overnight-storage. The purpose of the overnight storage was to determine whether an extended washout could affect adenosine-mediated contractility. A total of 20 human term placenta from normal (n = 8), GDM (n = 7) and T1DM (n = 5) pregnancies were prepared for the contractility studies. The fresh ring segments were quickly set up in organ baths for contractility studies, while overnight-stored ring segments were kept at 4°C in PSS containing 2% Ficoll (type 7000, Sigma, Dorset). 23
Contractility studies
Ring segments were suspended in a jacketed organ bath filled with PSS, gassed with 5% CO2 in air and maintained at 37°C. The segments were suspended between two wires passed through the vessel lumen. The lower wire was fixed to a glass supporting rod, while the upper wire was connected via thread to a force transducer (Lectro-Med, Letchworth, UK) for isometric tension recording, linked to a PowerLab data acquisition unit (AD Instruments Ltd, Hastings, UK) via an amplifier. Segment rings were put under tension of 2.0 g for 45 min equilibration, with PSS washout every 15 min, and allowed to relax to a resting tension of around 1.0–1.5 g.21,24
Segment rings were initially contracted with two to three 60-mM KCl applications, with washout, at ~20-min intervals. The last KCl response was used for analysis. After the final KCl application was washed out, the tissue was rested for 15–30 min until it reached its resting tension of 1.0–1.5 g.21,24 Preliminary studies showed that contractile responses to adenosine of the chorionic vessels rapidly desensitize, thus cumulative concentration-response curves could not be carried out. Thus, adenosine was initially applied to the vessel segments at a concentration of 100 µM as this consistently produced a robust and reproducible response which could be used to initially assess tissue responsiveness to adenosine. The adenosine was rapidly washed out as soon as a peak contraction was reached (about 3–5 min) and then left for 20 min before the next adenosine application. This was followed by the non-cumulative addition of adenosine (1 µM–1 mM) to elicit concentration-dependent contractions of chorionic vessels 21 (the order of adenosine applications is shown in Figure 1). Lower concentrations of adenosine have been reported in blood from human umbilical artery and vein (~0.2–0.7 µM). 15 Contractions were evoked within 3–5 min of adenosine applications, and once the maximal contraction was reached, the tissues were rapidly washed. Applications were at 20-min intervals to minimize desensitization of the adenosine-induced contraction. At the end of the experiment, another 30 min was allowed before the cumulative addition of incremental doses (1 nM–30 µM) of U46619 at 6- to 10-min intervals.
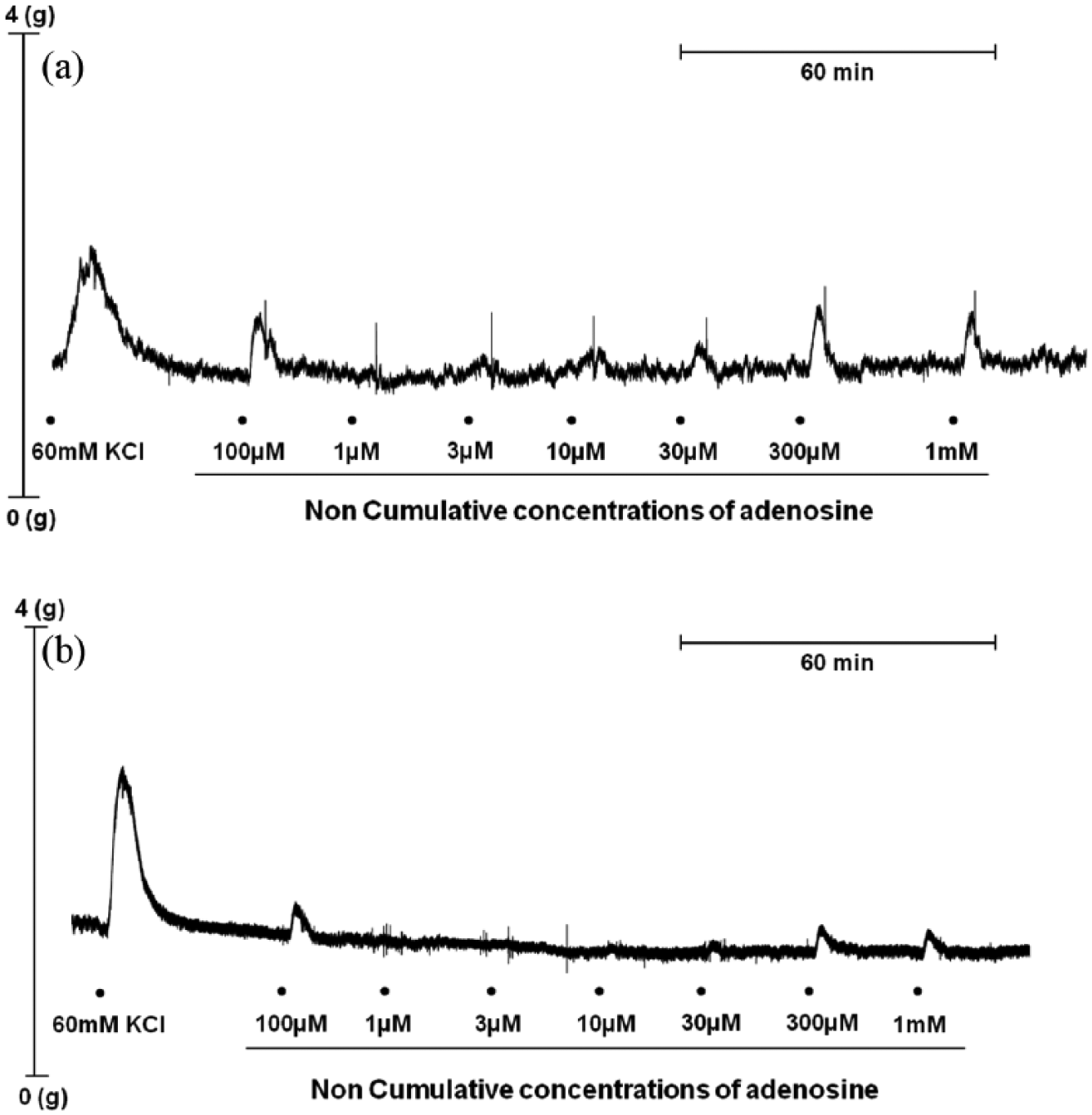
Representative traces showing contractions to adenosine of isolated chorionic arteries from placenta of (a) normal and (b) gestational diabetes (GDM) pregnancies. Adenosine was added as non-cumulative applications at concentrations of 1 µM–1 mM. Adenosine was washed out as soon as the contractile response had plateaued (4–5 min), and this was followed by an ~20-min recovery period before the next concentration was added.
Receptor quantification
Frozen chorionic vessel segments from GDM and normal groups were quantified for adenosine receptor subtypes (A2A, A2B and A3), and TP receptors by immunoblotting using standard methods. 25 The antibodies used for the receptor quantifications were as follows: anti-A2A antibody (rabbit polyclonal AA 373-391, Antibodies-online.com, 1:1000) tested positive against rat striatum, anti-A2B antibody (goat polyclonal sc-7505, Santa Cruz, Dallas, Texas, 1:500) tested positive against pig bladder, anti-A3 antibody (rabbit polyclonal A4229, Sigma, Dorset, 1:1000) tested positive against A3-transfected Chinese hamster ovary (CHO) cells and rabbit polyclonal anti-thromboxane (TBX)A2 (rabbit monoclonal ab134959, Abcam, Cambridge, 1:500) antibody tested positive against pig heart. Full immunoblots and positive controls are shown in Supplementary Figures 1–3. A2B receptor secondary antibody used was IRDye 800CW donkey anti-goat polyclonal (925-32214, LI-COR). A2A and A3 receptor secondary antibody was goat anti-rabbit 800CW (925-32211, LI-COR). Three different antibodies against the A1 receptor, from Sigma, Abcam and Alomone, were used but due to their lack of specificity, the samples were not quantified for A1 receptor expression. Anti-GAPDH was used (mouse monoclonal G8795, Sigma, Dorset); secondary antibody used was IRDye 680CW goat anti-mouse (926-68070, LI-COR, Nebraska, USA).
Data quantification and statistical analysis
Contractile responses were recorded in grammes of tension. Contractile responses to adenosine and U46619 were expressed as a percentage of the final KCl response and plotted as concentration-response curves. All data are presented as mean ± standard error of mean (s.e.mean) with number of placentae (n). Prism software version 6.04 (GraphPad Software, USA) was used to plot graphs and for statistical analysis. From concentration-response curves of each vessel segment, the agonist concentration producing 50% of the maximal response (EC50) and maximal response (Rmax) were determined. Concentration-response curves between study groups were tested using two-way analysis of variance (ANOVA) followed by Fisher’s least significant difference (LSD) test. EC50 and Rmax were tested using one-way ANOVA followed by Fisher’s LSD test. In a few vessel segments, response curves to adenosine and U46619 did not plateau so Rmax and EC50 values could not be derived, and this accounts for the different n numbers in the figures and their corresponding tables. Immunoblotting data were analysed using Student’s unpaired t-test. Significance was established by a p-value < 0.05.
Results
KCl contractions of chorionic vessels are similar in GDM and T1DM and after overnight storage
Contractile responses to KCl (60 mM) of arteries and veins were not significantly different between the control vessels, GDM and T1DM (p > 0.05, ANOVA) (Table 1). Given the evidence for an accumulation of adenosine from the placenta, 15 some vessels were stored in Krebs solution overnight, to determine whether this extended washout period affected adenosine-mediated contractility. Overnight storage had no significant effect on contractile responses to KCl of the arteries and veins of normal, GDM and T1DM vessels compared to their corresponding fresh vessels (p > 0.05, ANOVA). Thus, the contractile machinery of the vascular smooth muscle of chorionic arteries and veins is fundamentally unimpaired by maternal diabetes.
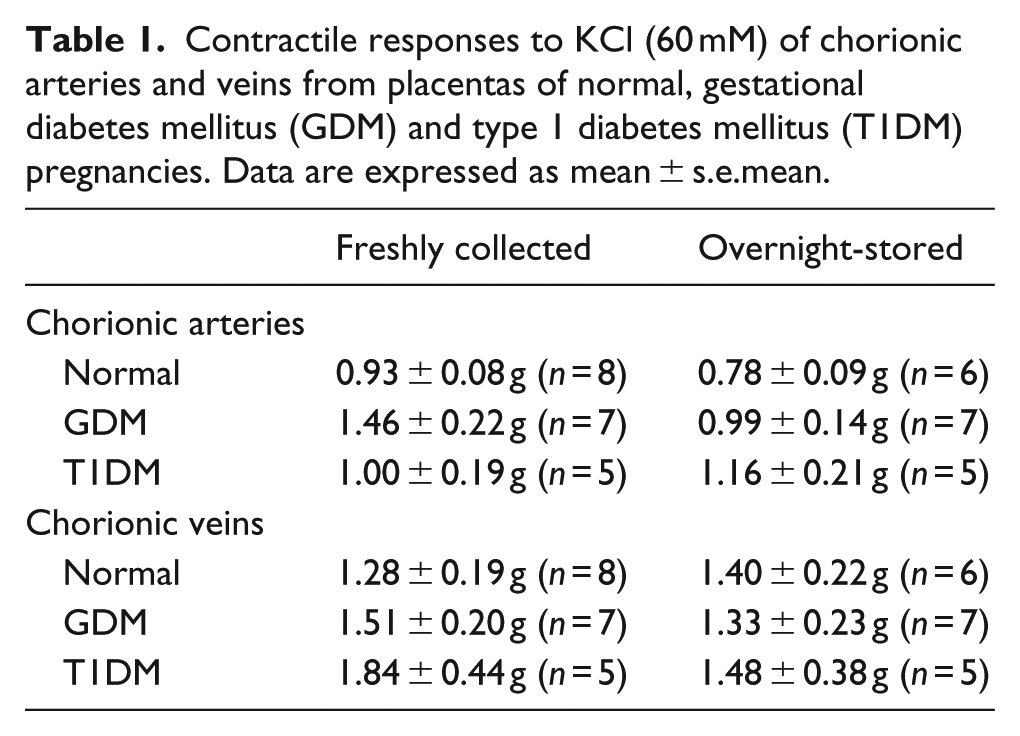
Contractile responses to KCl (60 mM) of chorionic arteries and veins from placentas of normal, gestational diabetes mellitus (GDM) and type 1 diabetes mellitus (T1DM) pregnancies. Data are expressed as mean ± s.e.mean.
Adenosine contractions are impaired in GDM arteries and veins
Adenosine (1 µM–1 mM) elicited concentration-dependent contractions in the chorionic arteries and veins [Figures 1, 2(a) and 3(a)] as reported by others. 21 Contractions to adenosine of fresh chorionic arteries from GDM were smaller than those of the normal arteries (p < 0.0001, ANOVA) [Figure 2(a)]. Contractile responses of chorionic veins were also smaller in freshly prepared GDM compared to normal veins (p < 0.001; ANOVA) [Figure 3(a)]. After overnight storage, there was an increase in responses to adenosine in GDM arteries, and the difference in contractions to adenosine between the GDM and normal arteries was abolished [Figure 2(b)]. After overnight storage contractions to adenosine remained smaller for chorionic veins from GDM compared to those of the normal vessels (p < 0.01; ANOVA) [Figure 3(b)]. Thus, adenosine contractility is impaired in chorionic arteries and veins in GDM, and overnight storage reverses the impaired contractility in chorionic arteries, but not veins.
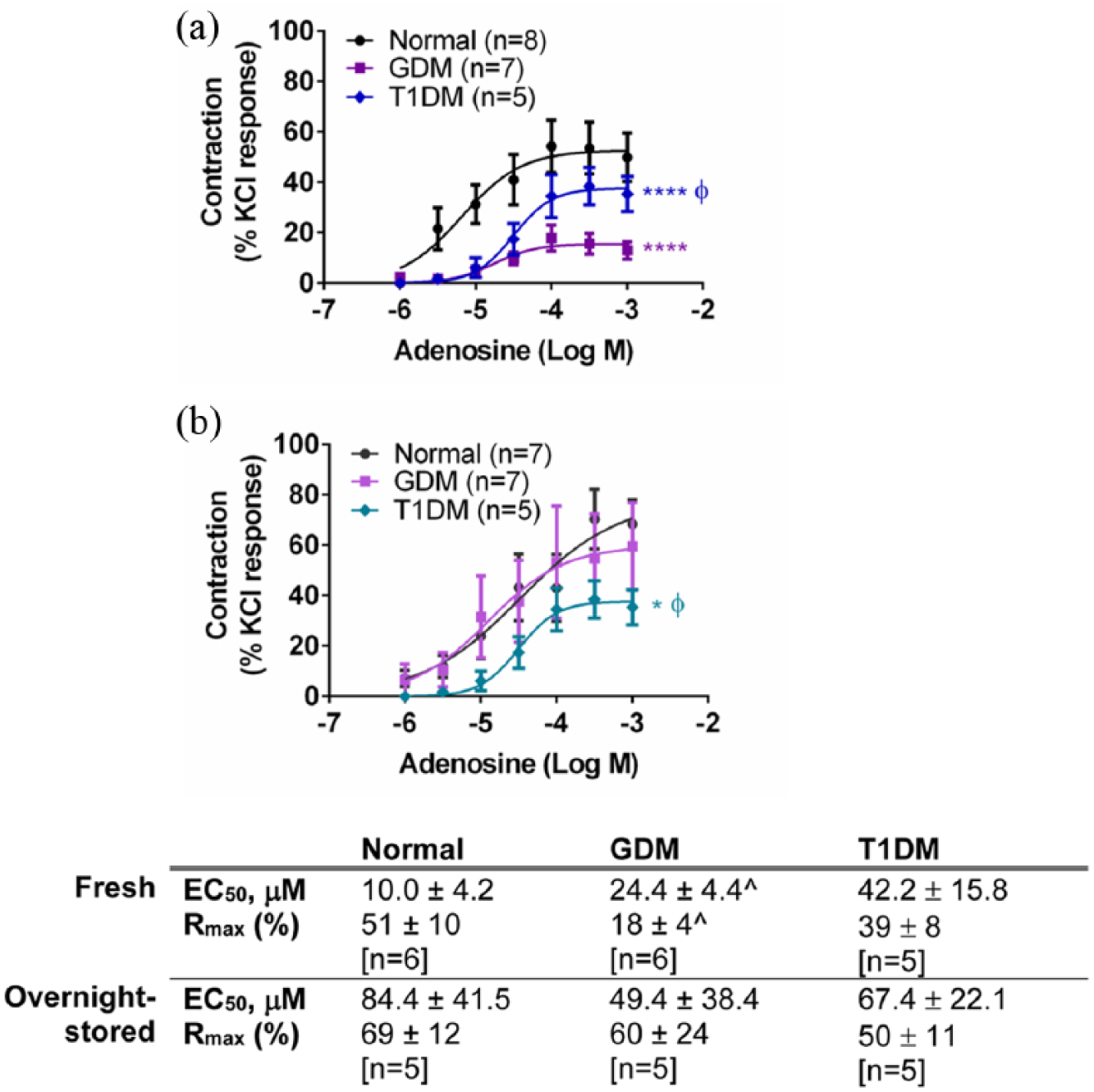
Response to adenosine in (a) fresh and (b) overnight-stored chorionic arteries from gestational diabetes mellitus (GDM), type 1 diabetes mellitus (T1DM) and normal pregnancies. Table indicates potencies (EC50) and maximum response to adenosine (Rmax). Values are mean ± s.e.mean with the number of placentae (n) shown in parentheses. Contractile response is expressed as a % of the contraction to potassium chloride (KCl, 60 mM).
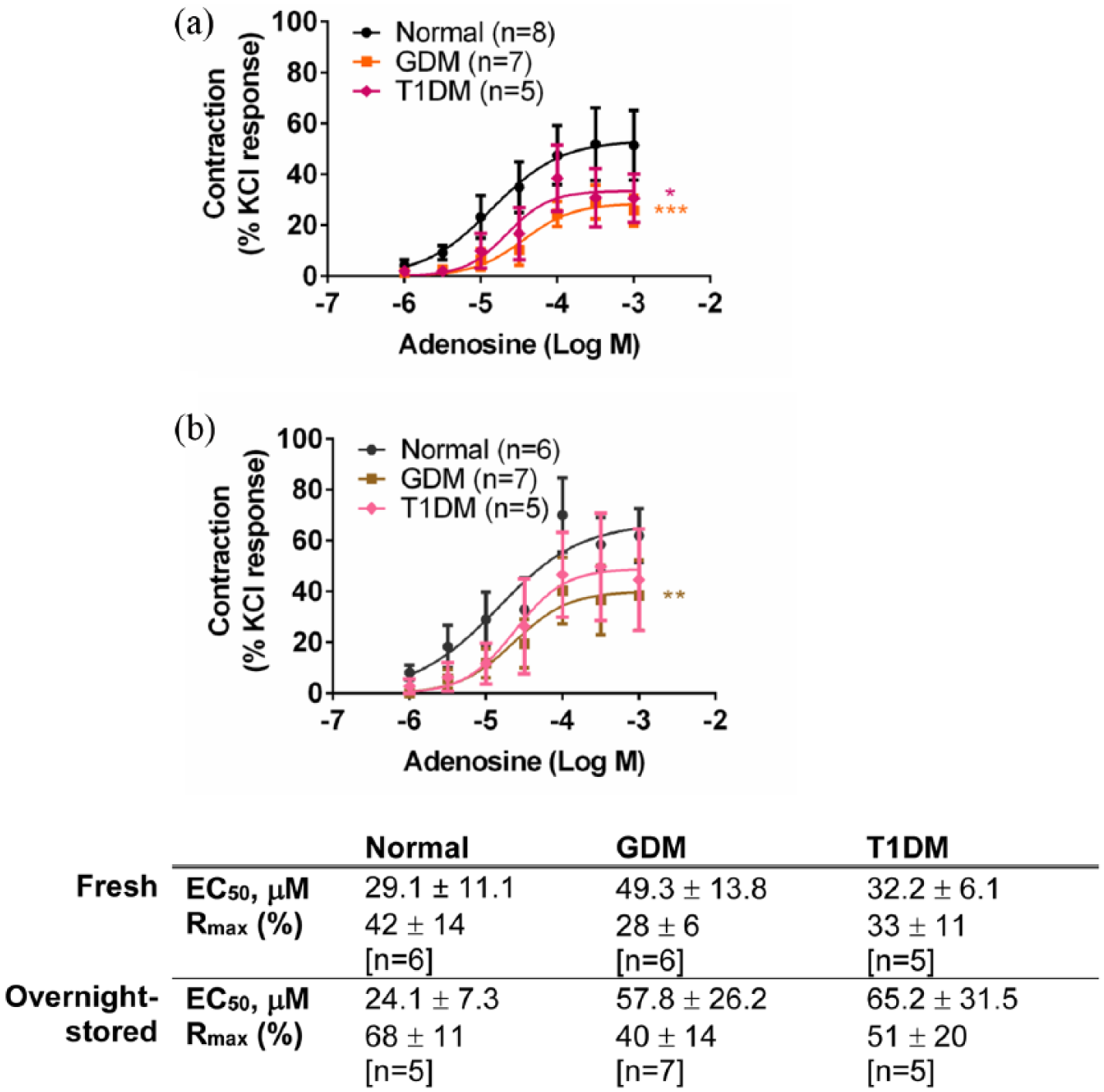
Response to adenosine in (a) fresh and (b) overnight-stored chorionic veins from gestational diabetes mellitus (GDM), type 1 diabetes mellitus (T1DM) and normal pregnancies. Table indicates potencies (EC50) and maximum response to adenosine (Rmax). Values are mean ± s.e.mean for number of placentae (n) shown in parentheses. Contractile response is expressed as a % of the contraction to potassium chloride (KCl, 60 mM).
Adenosine contractions are impaired in T1DM arteries and veins
Contractions to adenosine in the chorionic arteries from T1DM were smaller than those of the arteries of normal pregnancies (p < 0.0001, ANOVA) [Figure 2(a)]. Contractile responses to adenosine of the chorionic veins were also smaller between the T1DM and normal groups (p < 0.05, ANOVA) [Figure 3(a)]. After overnight storage, the significant difference in contractions to adenosine between the T1DM and normal arteries was still apparent (p < 0.05, ANOVA) [Figure 2(b)]. After overnight storage, the responses to adenosine remained smaller in T1DM compared to normal veins, although not significantly so (p > 0.05, ANOVA) [Figure 3(b)]. Thus, contractile responses to adenosine are impaired in both chorionic arteries and veins in T1DM, and this is not fundamentally altered by overnight storage.
Effect of GDM on vasocontractile responses to U46619
U46619 (1–300 nM) elicited concentration-dependent contractions in the chorionic arteries and veins (Figures 4 and 5). Contractile responses to U46619 were similar between GDM and normal groups in the arteries [Figure 4(a)] (p > 0.05, ANOVA) and were reduced in the GDM veins (p < 0.0001, ANOVA) [Figure 5(a)]. After overnight storage, responses to U46619 in the chorionic arteries (normal and GDM) were increased overall [see Figure 4(a) and (b)]. However, the responses to U46619 in the arteries between GDM and normal groups remained similar [Figure 4(b)] (p > 0.05, ANOVA). After overnight storage, contractile responses to U46619 in chorionic veins also remained smaller compared to the normal veins (p < 0.01, ANOVA) [Figure 5(b)].
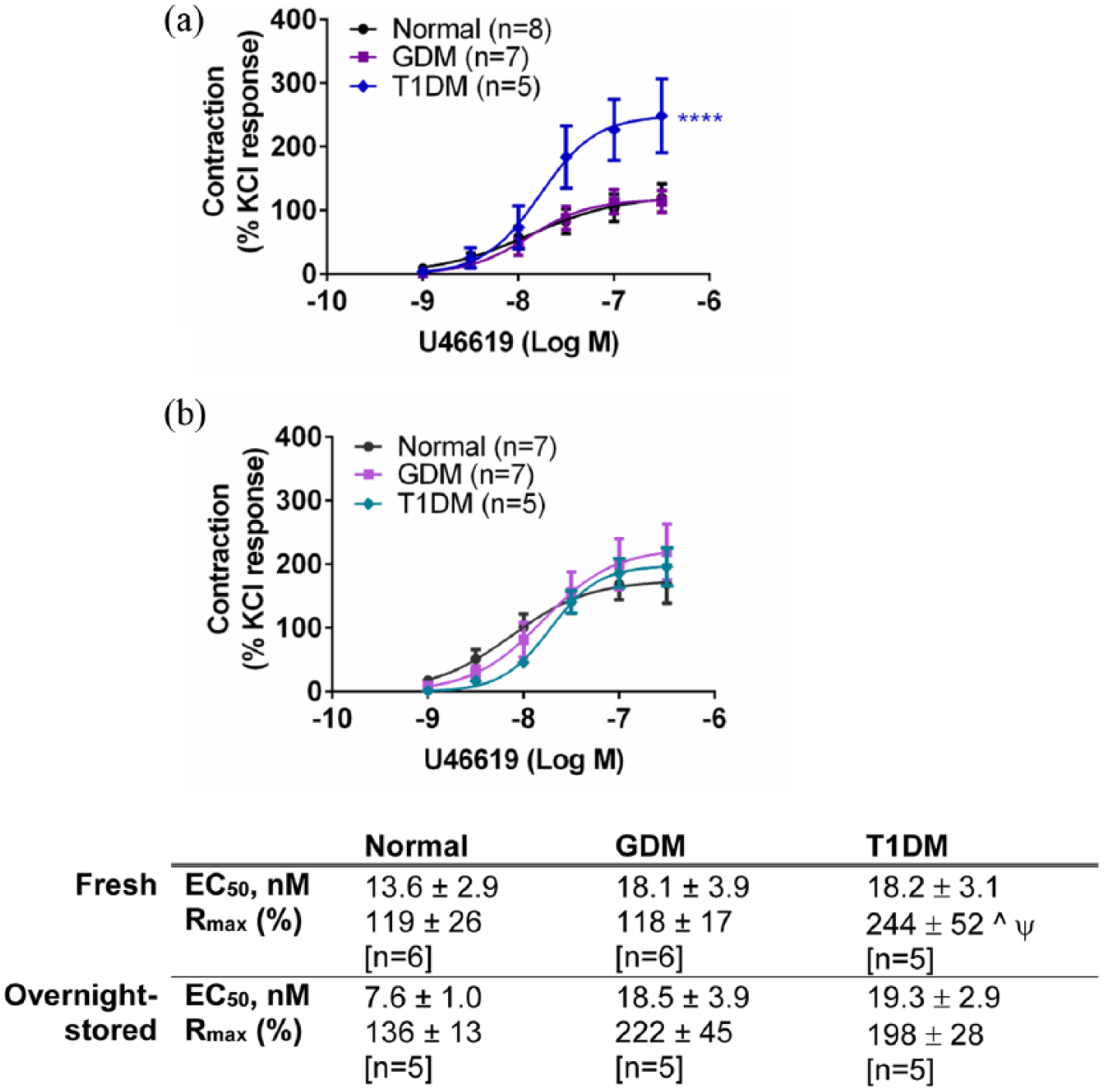
Response to U46619 in (a) fresh and (b) overnight-stored chorionic arteries from gestational diabetes mellitus (GDM), type 1 diabetes mellitus (T1DM) and normal pregnancies. Table indicates potencies (EC50) and maximum response to U46619 (Rmax). Values are mean ± s.e.mean for the number of placentae (n) shown in parentheses. Contractile response is expressed as a % of the contraction to potassium chloride (KCl, 60 mM).
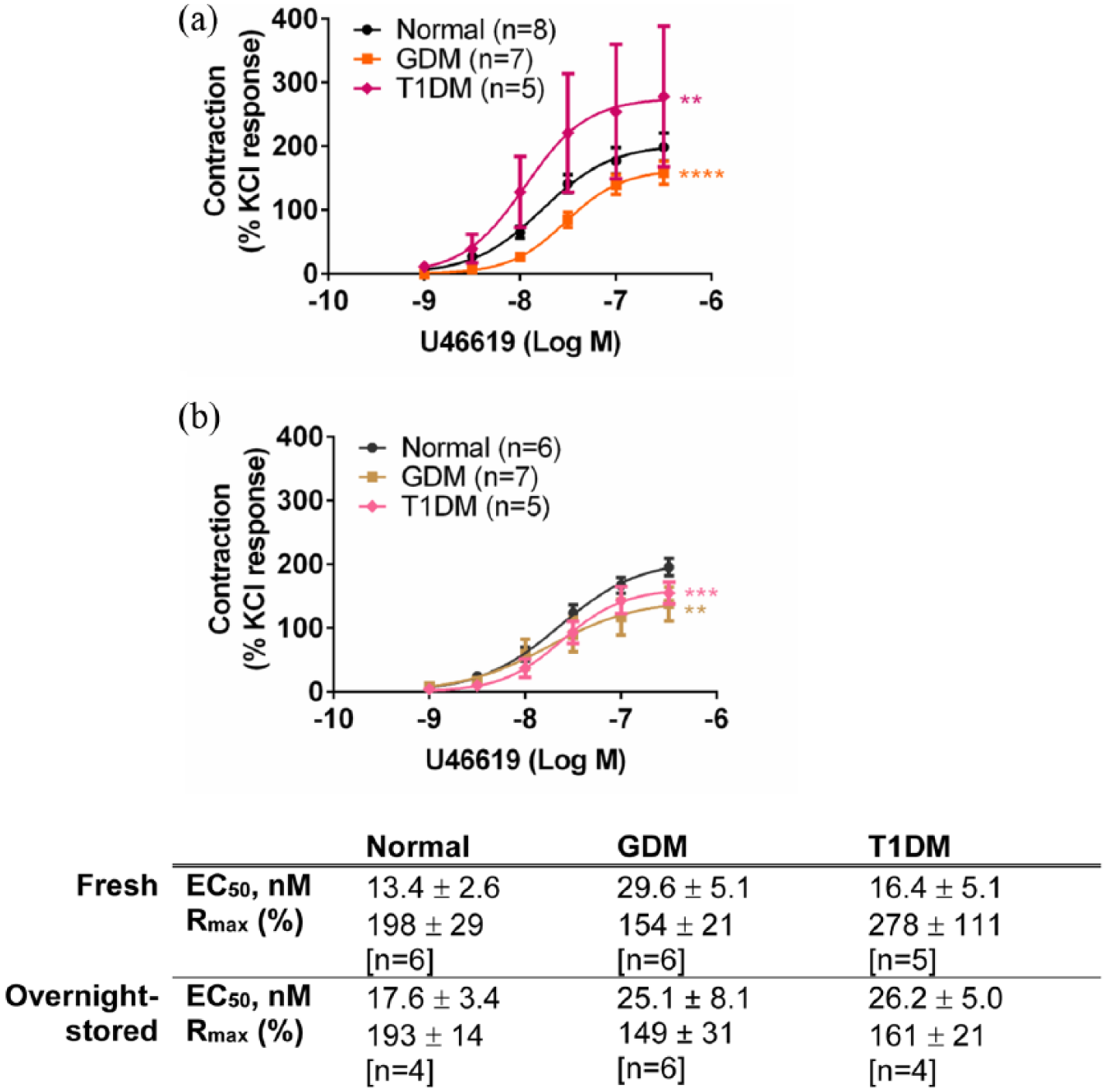
Response to U46619 in (a) fresh and (b) overnight-stored chorionic veins from gestational diabetes mellitus (GDM), type 1 diabetes mellitus (T1DM) and normal pregnancies. Table indicates potencies (EC50) and maximum response to U46619 (Rmax). Values are mean ± s.e.mean for the number of placentae (n) shown in parentheses. Contractile response is expressed as a % of the contraction to potassium chloride (KCl, 60 mM).
U46619 contractions are augmented in T1DM arteries and veins
Contractile responses to U46619 of the chorionic arteries were larger in the T1DM group compared to normal and GDM (p < 0.0001, ANOVA) with a significantly greater Rmax in the T1DM group compared to the normal and GDM groups (p < 0.05, ANOVA) [Figure 4(a)]. Contractile responses to U46619 in chorionic veins were also greater in T1DM compared to the normal group (p < 0.01, ANOVA) [Figure 5(a)]. Overnight storage normalized the differences in the responses between the normal and T1DM chorionic arteries, such that responses to U46619 were no longer significantly different between the two groups (p > 0.05, ANOVA) [Figure 4(b)]. After overnight storage, the responses to U46619 were slightly reduced in the veins in T1DM compared to the normal group (p < 0.001, ANOVA) [Figure 5(b)].
Immunoblotting for receptor quantification in normal and GDM vessels
Expression levels of A2A, A2B and A3 receptor protein were investigated using immunoblotting in normal and GDM arteries and veins. A1 receptor protein was not investigated as we could not demonstrate specificity of the antibodies. Bands were seen at ~45 kDa for the A2A receptor protein and ~50 kDa for the A2B receptor protein, which is similar to their predicted molecular weights of 45 kDa (Figure 6). The band size of the A3 receptor protein was ~50 kDa in both chorionic arteries and veins, which is greater than its predicted molecular weight of 36 kDa. There was no significant difference between normal and GDM arteries and veins with regard to expression of A2A and A2B receptor protein (Figure 6). A3 receptor protein expression was similar in normal and GDM arteries, but was lower in veins of GDM compared to normal pregnancies (p < 0.05, Student’s t-test) (Figure 6). Immunostaining for the TP receptor protein was observed at ~50 kDa, which is similar to its predicted molecular weight of 44 kDa. TP receptor protein expression was lower in GDM arteries compared to normal arteries (p < 0.05, Student’s t-test) [Figure 7(a)], but expression levels were similar in normal and GDM veins [Figure 7(b)].
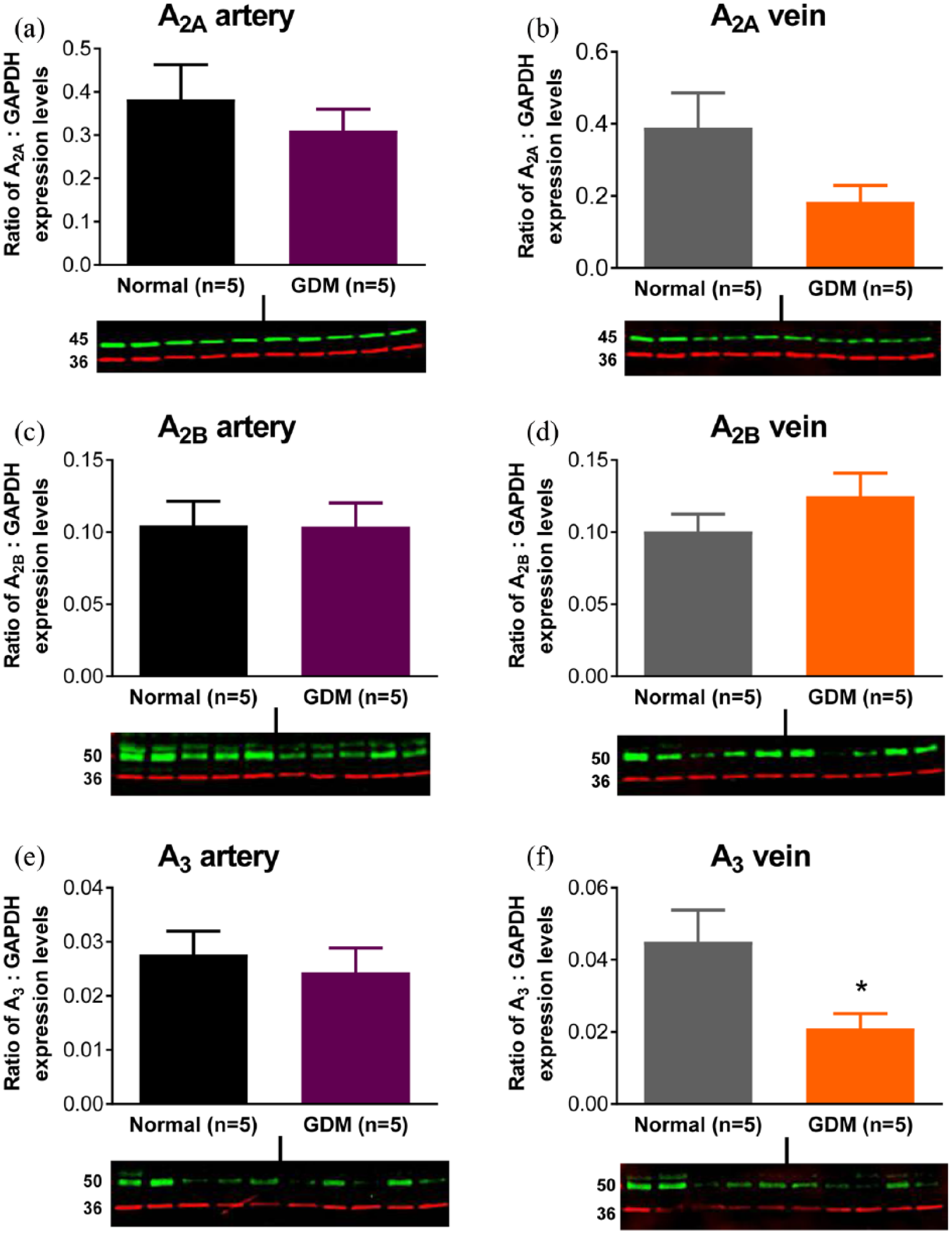
Expression levels of A2A (~45 kDa) (a, b), A2B (~50 kDa) (c, d) and A3 (~50 kDa) (e, f) adenosine receptor proteins in chorionic arteries (a, c, e) and chorionic veins (b, d, f) from term placenta of normal and gestational diabetic mellitus (GDM) pregnancies. Blots show bands for adenosine receptor proteins (green) and GAPDH (~36 kDa) (red). Data are expressed as the ratio of A2A to GAPDH band intensities and are the mean ± s.e.mean of five normal and five GDM samples.
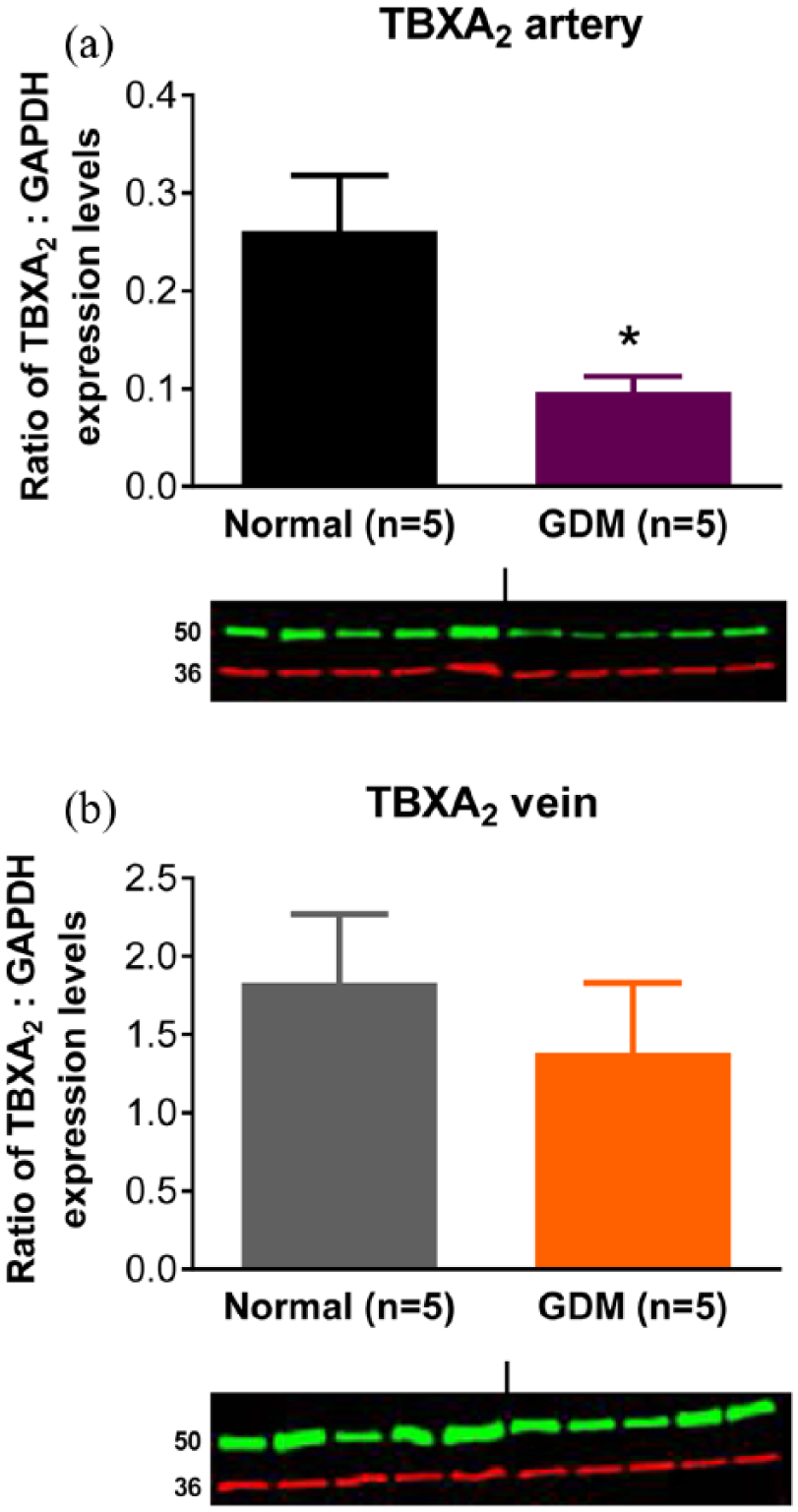
TP receptor protein (TBXA2 ~50 kDa) and GAPDH (~36 kDa) expression levels in (a) chorionic arteries and (b) chorionic veins. Ratio of expression levels of TBXA2 to GAPDH in chorionic arteries (a) and chorionic veins (b) from term placenta of normal and gestational diabetic mellitus (GDM) pregnancies based on the intensities of the bands. Data are expressed as the ratio of TBXA2 protein to GAPDH band intensities and are the mean ± s.e.mean of five samples.
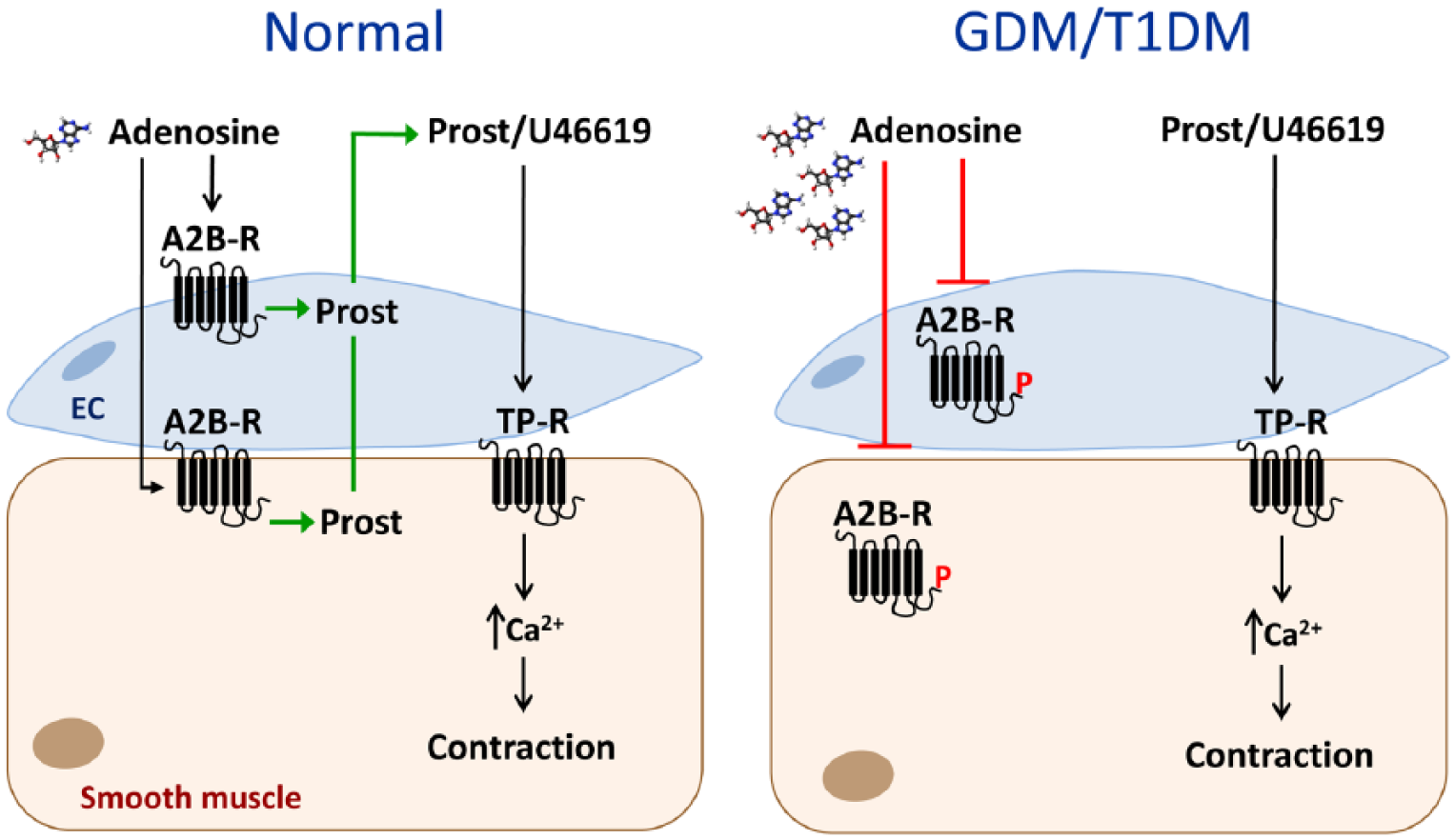
Summary diagram showing a putative pathway whereby adenosine contractility of the chorionic vasculature is impaired in maternal diabetes. Adenosine causes contraction of normal chorionic vessels through activation of A2B receptors (A2B-R) on the endothelium and smooth muscle, release of prostanoids (Prost) and activation of thromboxane receptors (TP-R). Thromboxane A2 is the principal endogenous agonist of TP-R and U46619 is a thromboxane A2 analogue. In gestational diabetes mellitus (GDM) and type 1 diabetes mellitus (T1DM), an accumulation of adenosine causes loss of the response to adenosine, possibly through receptor desensitization/downregulation, likely involving receptor phosphorylation (P). The contractile pathway involving prostanoid/U46619 activation of TP-R remains intact.
Discussion
This is the first study to investigate responses to adenosine in human chorionic vessels from pregnancies complicated by two types of maternal diabetes, GDM and T1DM. The main finding in this study is that there is an impaired contractile response to adenosine in chorionic arteries and veins isolated from pregnancies with GDM and T1DM compared to vessels from normal pregnancies. Contraction to adenosine of human chorionic arteries and veins is mediated by A2B receptors and a release of thromboxane A2 or a related metabolite. 21 Here, we show that in chorionic arteries of both GDM and T1DM, the impaired adenosine contraction does not involve impaired signalling via TP receptors.
This study provides further evidence that adenosine induces vasoconstriction in human-isolated chorionic vessels and cotyledons.20,21,26 Both the potency and maximal responses to adenosine in chorionic arteries and veins from normal pregnancies (artery EC50 10 µM, Rmax 51%; vein EC50 29 µM, Rmax 42%) are similar to those obtained by Donoso and colleagues (artery EC50 38 µM, Rmax 43%; vein EC50 38 µM, Rmax 41%). 21 Others have reported relaxation of the human fetoplacental vasculature to adenosine, but either at substantially higher concentrations (EC50 ~1 mM adenosine) in isolated chorionic vessels 27 or as a slow response (developed over 20–30 min) in perfused cotyledons. 28 Biphasic responses of human isolated chorionic vessels to adenosine, with initial contraction (within 4 min) followed by relaxation (within 10–15 min), were also reported, 26 and similarly in the ovine placental vasculature. 29 The human placental vasculature expresses all four adenosine receptors, A1, A2A, A2B and A3.21,30,31 The contractile adenosine receptor in human chorionic arteries and veins has been characterized as the A2B subtype, located largely on the endothelium, which activates the cyclooxygenase pathway to mediate synthesis of thromboxane A2 or a related metabolite, whereas the relaxation appears to involve smooth muscle A2/A3 receptors.21,28 In vivo, the vascular responses are likely to depend on the adenosine levels, and endothelial integrity, such that an acute increase in adenosine will cause predominantly vasoconstriction, whereas a sustained increase in adenosine will elicit additionally/predominantly relaxation, and this will help with the regional distribution of blood; an overall increase in the placental concentration of adenosine would, therefore, be expected to dampen adenosine-mediated blood flow regulation if accompanied by significant adenosine receptor desensitization.
Here, we report, for the first time, impaired contractions to adenosine of chorionic vessels (arteries and veins) from both GDM and T1DM, suggesting a systemic alteration of adenosine signalling in the fetoplacental circulation in maternal diabetes. This did not involve a generalized impairment of contractility of the vascular smooth muscle since responses to KCl, a receptor-independent vasoconstrictor, were similar between the normal and diabetic groups. Moreover, the impaired responsiveness to adenosine in chorionic arteries was selective with respect to responses to U46619, which were unaltered in GDM and increased in T1DM chorionic arteries. Selective impairment of the response to adenosine was also observed in chorionic veins from T1DM, since U46619 mediated responses were augmented. In GDM, veins impaired responsiveness to U46619 accompanied the impaired contractile response to adenosine. Adenosine contractions involve the production and actions of thromboxane A2, 21 but these data suggest that impaired adenosine contractions in artery and vein in T1DM, and in artery in GDM, do not involve defective signalling via TP receptors.
We used immunoblotting to investigate whether altered adenosine receptor expression could underlie the impaired contractile responses of the chorionic arteries and veins from GDM. There is a precedent for changes in placental adenosine receptor expression in complicated pregnancies since, in preeclampsia, the protein and messenger RNA (mRNA) of all four adenosine receptors were increased in placental biopsies and villous explants. 31 The band sizes of ~45 kDa for the A2A receptor protein and ~50 kDa for the A2B receptor protein are similar to their predicted molecular weights of 45 kDa, and to the bands observed in human placental biopsies (45 kDa, A2A; 52 kDa, A2B). 31 The band size of the A3 receptor protein at ~50 kDa in both chorionic arteries and veins is greater than its predicted molecular weight of 36 kDa. Immunostaining for the A3 receptor protein in human placental biopsies 31 showed a band of ~36 kDa and an additional higher molecular weight band which could correspond to our band of ~50 kDa. We found no difference in A2B (and A2A) adenosine receptor protein expression levels between GDM and normal chorionic arteries and veins. It is possible that the impaired contractility to adenosine in GDM (and possibly T1DM) involves A2B receptor desensitization/internalization, 32 perhaps as a consequence increased extracellular levels of adenosine in the GDM placenta. 15 Rapid fade of contractile responses to P2Y1 receptor activation in human chorionic arteries was attributed to its exit from membrane lipid rafts and internalization. 33 In contrast, contractile responses of the human chorionic vasculature to U46619 and PGF2α are sustained.34–36 A limitation of our study is that we do not have data for receptor protein expression in chorionic vessels from T1DM, but the data with GDM show that changes in agonist-induced contractile function can occur without a change in the level of receptor protein.
Whether the impaired contractility to adenosine in chorionic vessels from GDM and T1DM involves changes in plasma glucose concentrations is unclear. Maternal hyperglycaemia induces foetal hyperglycaemia and subsequent changes in the foetal circulation such as lower oxygen content and saturation. 37 However, hypoxia has been shown to increase mRNA expression of the trophoblast A2B receptor (which was shown to mediate proliferation, invasion and activation of cAMP response element binding protein). 38 A high glucose concentration impairs vascular endothelial function in vitro, 39 and since endothelium removal attenuates adenosine-mediated contractions in chorionic vessels, 21 this could contribute to the aberrant adenosine-mediated contractions in maternal diabetes. However, the blood glucose levels of the volunteers in our study are likely to have been well controlled, and others have reported no change in glucose in umbilical vein blood from normal and gestational diabetic pregnancies. 17 There is evidence that insulin receptors regulate placental vascular adenosine transport and nitric oxide generation,15,19,40 and investigation of the impact of this in the study groups is an important avenue for future investigation.
Overnight storage in physiological solution reversed some of the changes, restoring contractility to adenosine in the GDM chorionic arteries. This recovery of function may involve resensitization/upregulation of contractile A2B receptors due to the removal of locally released or circulating adenosine. Extracellular levels of adenosine in the placental circulation may be elevated in maternal diabetes, an effect involving downregulation of placental hENT1,15–17 and this may contribute to the reduced adenosine responses. An important limitation of this study is that adenosine levels were not measured in the tissue/blood so we do not have evidence that the differences that we have observed are a consequence of changes in adenosine levels. Changes in adenosine and adenosine triphosphate (ATP) release and metabolism could also be important in local elevations in placental vascular adenosine concentration. 41 KCl contractions were unaffected by overnight storage, suggesting that the recovery of function involved specific changes relevant to adenosine signalling. Reduced responses to KCl after overnight storage have reported by others, 42 but we did not see this. Overnight storage did not, however, reverse impaired contractility to adenosine in GDM chorionic veins, or in T1DM chorionic arteries and veins, suggesting more complex changes in contractility.
Contraction to U46619 in GDM arteries was unchanged, and in veins it was modestly reduced. This did not correlate with changes in TP receptor protein levels which were reduced in arteries and unchanged in veins. Interestingly, contractile responses to U46619 were much larger in normal veins than in arteries (Rmax 198% veins; Rmax 119% arteries), which correlates with the greater (approximately 10-fold) TP receptor expression in the veins. Contraction to U46619 was enhanced in chorionic vessels from T1DM, in both arteries and veins, and this was reversed by overnight storage. In contrast, impaired vasoconstrictor responses to U46619 of perfused cotyledons from women with pregestational diabetes has been reported, attributed to a reduction in the affinity but not the number of placental TP receptors; placental thromboxane production was unchanged. 43 The reason for the increase in U46619 responses observed in this study in T1DM arteries and veins, but not in GDM vessels, is unclear. However, it is possible that the opposite changes in responses to adenosine and U46619 in T1DM vessels reflect a compensatory mechanism as placental vasoactive autacoids are known to work in concert to ensure optimum placental haemodynamic function in vivo.
Despite the evidence for a vasocontractile role of adenosine in the human placental vasculature,20,21 there is an emphasis in the literature on adenosine-mediated placental vasorelaxation, possibly because adenosine is a vasodilator in most other vascular beds. 44 This study reinforces that overall control of fetoplacental haemodynamics by adenosine will involve the coordinated actions of vasocontractile as well as vasodilator adenosine receptors, likely differently expressed throughout the placental vascular tree as shown for the purine/pyrimidine nucleotide receptors P2Y1 and P2Y2. 45 The impaired adenosine-mediated contractility of chorionic vessels in maternal diabetes reported in this study might lead to a shift in balance towards adenosine-mediated placental vasorelaxation, although evidence for impaired adenosine relaxation in the umbilical vein in GDM, 19 suggests that adenosine-mediated blood flow regulation overall may be dampened.
Conclusion
This study reaffirms the presence of adenosine-induced vasocontraction in human chorionic vessels. The study has provided the first evidence of an altered contractile response to adenosine in chorionic vessels from pregnancies complicated by both pregestational and gestational diabetes. Impaired responses to adenosine in both chorionic arteries and veins suggest a systemic alteration of adenosine signalling in the fetoplacental circulation which may be contributed by both foetus and placenta in response to the maternal diabetes, and which may involve desensitization/downregulation of adenosine receptors (Figure 8). Our study contributes to a better understanding of differential function of the vasculature in diabetes.
Supplemental Material
dvdres-feb-2018-00026-File010 – Supplemental material for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes
Supplemental material, dvdres-feb-2018-00026-File010 for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes by Azlina A Razak, Lopa Leach and Vera Ralevic in Diabetes & Vascular Disease Research
Supplemental Material
dvdres-feb-2018-00026-File011 – Supplemental material for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes
Supplemental material, dvdres-feb-2018-00026-File011 for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes by Azlina A Razak, Lopa Leach and Vera Ralevic in Diabetes & Vascular Disease Research
Supplemental Material
dvdres-feb-2018-00026-File012 – Supplemental material for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes
Supplemental material, dvdres-feb-2018-00026-File012 for Impaired vasocontractile responses to adenosine in chorionic vessels of human term placenta from pregnant women with pre-existing and gestational diabetes by Azlina A Razak, Lopa Leach and Vera Ralevic in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors are grateful to L Stoddart for the donation of anti-A3 receptor antibody. The authors are very grateful to L Latif for technical assistance with the immunoblotting. Human tissue was obtained through the clinical connections and under the project licence of L.L. V.R. and A.A.R. designed the research study. A.A.R. performed the research and analysed the data. A.A.R. and V.R. wrote the initial draft. L.L. critically evaluated and commented on the results. All the authors read, modified and approved the final version of the manuscript.
Availability of data and material
All data generated or analysed during this study are included in this published article (and in its supplementary information file).
Consent for publication
Not applicable; no individual’s data are reported.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics approval and consent to participate
This study had the approval of the QMC and University Hospital NHS Trust Ethics Committee (Ref: 0G010101; PI-LL) and was carried out in accordance with the declaration of Helsinki. Pregnant women gave written informed consent.
Funding
A.A.R. was supported by the Islamic Development Bank under the Merit Scholarship Programme.
Supplementary Data
Supplementary files are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
