Abstract
Background:
To evaluate the renoprotective effects of canagliflozin, we assessed the albuminuria-lowering effect in Japanese type 2 diabetes patients with chronic kidney disease (CKD).
Methods:
In this prospective, open-label, parallel-group study, type 2 diabetes patients with CKD were randomized to receive either oral canagliflozin (100 mg/day) or usual care (control group) for 52 weeks. Endpoints included changes in urinary albumin-to-creatinine ratio (UACR), other urinary biomarkers, laboratory parameters, and adverse events.
Results:
Both groups included 20 patients in the analysis. Mean changes in UACR was −83 (−266 to −31) mg/gCr and 27 (−11 to 131) mg/gCr, in the canagliflozin and control groups, respectively (p = 0.004). Urinary liver-type free acid binding protein, N-acetyl-β-
Conclusion:
Canagliflozin was associated with slower progression of kidney disease and reduction in albuminuria and tubulointerstitial markers in diabetes patients with CKD.
Introduction
Sodium glucose cotransporter 2 (SGLT2) inhibitors are expected to have renoprotective effects based on the possible mechanism that SGLT2 inhibitors reduce hyperfiltration by blocking proximal tubule sodium reabsorption and activating tubuloglomerular feedback (TGF). SGLT2 inhibitors may decrease inflammatory, fibrotic and hyperplastic response of proximal tubular cells by blocking glucose reabsorption into proximal tubular cells. 1 Furthermore, a 30% to 40% reduction in albuminuria was found.2,3 However, the effects of SGLT2 inhibition are unclear with regard to other kidney-related biomarkers, such as those associated with tubulointerstitial damage. In this study, we aimed to examine precisely the renoprotective effects of the SGLT2 inhibitor canagliflozin by measuring albuminuria, tubulointerstitial biomarkers, estimated glomerular filtration rate (eGFR) and other metabolic parameters in type 2 diabetes patients with chronic kidney disease (CKD) who are treated with renin-angiotensin system (RAS) inhibitors.
Materials and methods
The study protocol was approved by the Ethics Committee of Nihon University Itabashi Hospital, and all patients provided written informed consent (Clinical Trial Registration number: UMIN000031454). The study protocol was designed in accordance with the Declaration of Helsinki.
Enrollment criteria were as follows: (1) type 2 diabetes with nephropathy under stable glycaemic control, defined as a haemoglobin A1c (HbA1c) level <10.0% after administration of oral hypoglycaemic agents and/or insulin injection therapy for 12 consecutive weeks; (2) albuminuria: urinary albumin-to-creatinine (Cr) ratio (UACR) 30–2000 mg/gCr (average of two consecutive measurements recorded during the 8-week pretreatment period); (3) eGFR of 45–89 mL/min/1.73 m2; and (4) hypertension treated with a fixed dose of RAS inhibitors for at least 12 weeks before the study treatment began. Exclusion criteria were (1) age <20 years or >80 years, (2) HbA1c ⩾10.0% and (3) severe heart failure, angina, myocardial infarction or stroke occurring within 6 months before enrollment in the trial.
This prospective, open-label, parallel-group, single-centre study was conducted between June 2016 and December 2017, and eligible patients were randomized to receive oral canagliflozin (100 mg/day) or usual care for 52 weeks. Before randomization, patients received fixed doses of conventional antidiabetic drugs (oral hypoglycaemic agents and/or insulin) for 12 weeks, and these drugs were continued during the 52-week treatment period. The primary efficacy endpoint was change in albuminuria as assessed by UACR. Secondary endpoints included changes in vital signs and laboratory tests during the study and safety. Clinical parameters were measured by routine clinical chemistry procedures using commercially available assay kits. eGFR was calculated according to the following formula for Japanese patients: 4 eGFR (mL/min/1.73 m2) = 194 × serum Cr−1.094 × age−0.287(× 0.739 for women).
Urinary albumin levels were measured by the immunoturbidimetric assay. Urinary liver-type fatty acid binding protein (L-FABP) was measured by a specific chemiluminescent enzyme immunoassay (CLEIA; SRL, Inc., Tokyo, Japan) in the same urine sample, and values were expressed relative to the urinary Cr concentration. Urinary N-acetyl-β-
Statistical analyses
Data are expressed as the mean ± standard deviation or median (interquartile range) as appropriate. Continuous variables were compared using the Student’s t test or the Mann–Whitney U test, and categorical variables were compared using the χ2 test or Fisher’s exact test. Statistical significance was set at p < 0.05. All analyses were performed using JMP software version 12 (SAS Institute Ltd., Cary, NC, USA).
Results
Subjects
A total of 42 patients were randomly assigned to the canagliflozin group (n = 21) or the control group (n = 21). One patient in the canagliflozin group was admitted to hospital due to complete atrioventricular (AV) block, and one patient in the control group was admitted to hospital due to pneumonia. Both of these patients were excluded from the final analysis, so 20 patients were analysed in each group. There were no significant differences between the two groups in baseline demographic, hemodynamic or anthropometric variables; cardiovascular comorbidities; or medications (Table 1). The canagliflozin dose was not changed in any patient and none of the patients in the canagliflozin group started another antidiabetic drug.
Patient backgrounds and results.
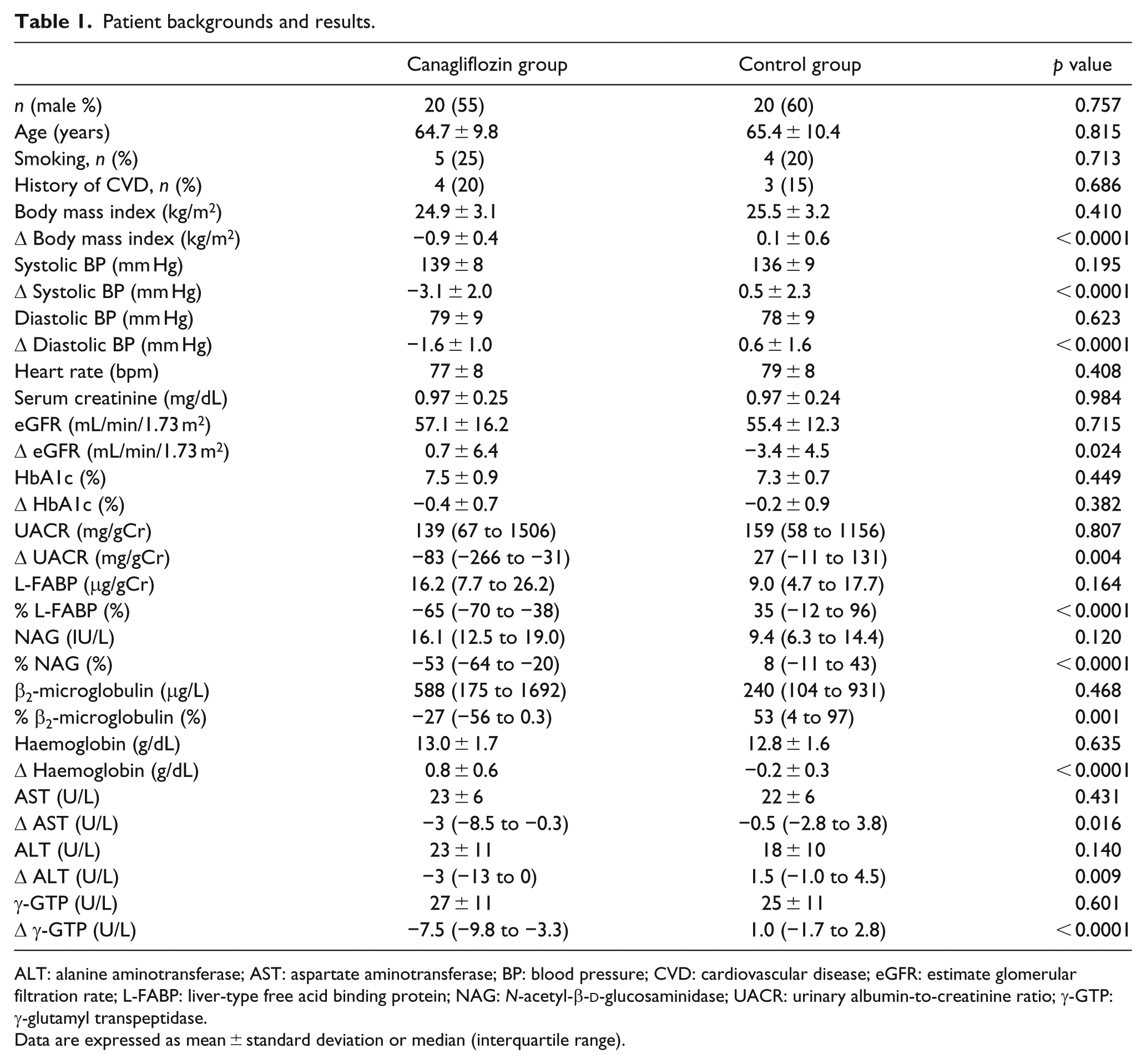
ALT: alanine aminotransferase; AST: aspartate aminotransferase; BP: blood pressure; CVD: cardiovascular disease; eGFR: estimate glomerular filtration rate; L-FABP: liver-type free acid binding protein; NAG: N-acetyl-β-
Data are expressed as mean ± standard deviation or median (interquartile range).
Effects on kidney parameters
UACR decreased significantly from 139 (67–1506) mg/gCr at baseline to 38 (20–675) mg/gCr at week 52 in the canagliflozin group (p < 0.0001) but remained unchanged in the control group, with values of 159 (58–1156) mg/gCr and 194 (63–1050) mg/gCr at baseline and week 54, respectively. The mean changes in UACR were −83 (−266 to −31) mg/gCr and 27 (−11 to 131) mg/gCr, in the canagliflozin and control groups, respectively (p = 0.004). The least squares (LS) mean change in UACR was −220.8 mg/gCr and 81.4 mg/gCr, in the canagliflozin and control groups, respectively.
There was a significant reduction in urinary L-FABP, NAG and β2MG levels in the canagliflozin group. Conversely, urinary β2MG and L-FABP levels were significantly increased in the control group. The percent change in urinary L-FABP, NAG and β2MG all decreased significantly in the canagliflozin group (−65%, −53% and −27%, respectively), but not in the control group (35%, 8% and 53%, respectively).
Decreases in eGFR from baseline were observed more in the canagliflozin group compared with control group at week 4. The reductions in eGFR with canagliflozin were largest at week 4 and then trended back towards baseline over the 26-week treatment period. LS mean changes in eGFR at the end of the study were 0.7 and −3.4 mL/min/1.73 m2 in the canagliflozin and control groups, respectively (p = 0.024).
Glycaemic control and other clinical parameters
HbA1c (%) decreased significantly from 7.5 ± 0.9 at baseline to 7.1 ± 0.9 at week 52 in the canagliflozin group (p < 0.05) but remained unchanged in the control group. Of note, a significant decrease in body mass index (BMI), systolic and diastolic BP was observed in the canagliflozin group. In addition, canagliflozin was associated with a significant increase in haemoglobin concentrations. Aspartate aminotransferase (AST), alanine aminotransferase (ALT) and γ-glutamyl transpeptidase (γ-GTP) levels significantly decreased in the canagliflozin group but not in the control group.
Safety
None of the patients exhibited significant adverse events (AEs) such as symptomatic hypoglycaemia, urinary tract infection, genital infection or limb amputation. Canagliflozin was well tolerated and was not discontinued in any of the patients.
Discussion
In this study, canagliflozin reduced UACR by 42% after 52 weeks. Furthermore, urinary L-FABP, NAG and β2MG levels were also reduced by canagliflozin treatment. These findings are the first, to our knowledge, to show that canagliflozin improves kidney parameters, including albuminuria and tubulointerstitial markers.
SGLT2 inhibitors may confer renoprotection by directly lowering blood glucose levels, activating TGF and/or potentially inducing weight loss through volume normalization, and improved BP control. 5 Heerspink et al. 6 suggested that the decrease in UACR after SGLT2 inhibition is a result of reduced intraglomerular pressure. In patients with diabetic nephropathy, the development of progressive diabetic kidney disease has been associated with changes in proximal tubules, renal function and prognosis and showed a higher correlation with structural lesions in the tubulointerstitium than with glomerular changes. 7 Various proximal tubule pathophysiological stresses, such as proteinuria, induce upregulation of human L-FABP gene expression, thereby resulting in increased proximal tubular L-FABP excretion and increased urinary L-FABP excretion. 8 Severe proteinuria or ischemia can also exacerbate tubulointerstitial damage by causing an overload of free fatty acids in the proximal tubules. 8 Urinary L-FABP is an important marker of tubulointerstitial changes in diabetic nephropathy, while oxidative stress is a pathogenic factor underlying diabetic complications such as nephropathy. 9 Because this study showed that urinary L-FABP was reduced by canagliflozin treatment, canagliflozin could prevent the progression of tubulointerstitial damage. Nakamura et al. 10 reported that insulin and reactive oxygen species increase SGLT2 expression and glucose uptake in proximal tubular cells. Our study showed increased haemoglobin level even in patients with impaired kidney function. Although we did not confirm an increase in serum erythropoietin concentration – which has been shown in patients treated with dapagliflozin 11 – the observed increase in hematocrit in this study would likely deliver more oxygen to tissues. 12 Therefore, SGLT2 may play a role in various tubular injuries. However, further studies would be needed to clarify that increased haemoglobin contributed to the maintenance of GFR and prevention cardiovascular events, which were shown in previous large-scale studies.
This study has several limitations. First, the number of patients included in this single centre design was limited. However, this study demonstrated that a new approach using several urinary biomarkers could clarify the distinct characteristics of glomerular and tubulointerstitial damage. Future multicenter studies with large cohorts should be conducted to reassess our findings. Second, the dose of canagliflozin in this study was set at only 100 mg daily because this is the maximum dose approved in Japan. Therefore, we could not compare the efficacy of canagliflozin 100 and 300 mg.
In conclusion, canagliflozin treatment leads to a reduction in albuminuria, urinary L-FABP, NAG and β2MG levels in diabetes patients with CKD, regardless of simultaneous RAS inhibition. These findings suggest that canagliflozin may be an appropriate treatment option for patients with diabetes and CKD.
Footnotes
Acknowledgements
Publication of this report was not financially supported by any grant. Study concept and design: M.A., H.T.; data acquisition: M.A., H.T., Y.Y., C.N., T.F., R.T., T.M., N.M.; data analysis/interpretation: M.A., H.T.; statistical analysis: M.A. All authors contributed important intellectual content during manuscript drafting or revision, and accept accountability for the overall work, ensuring that questions pertaining to the accuracy or integrity of the work were appropriately investigated and resolved. M.A. takes responsibility that this study is reported honestly, accurately and transparently; that no important aspects of the study have been omitted; and that any discrepancies in the study have been explained.
Declaration of conflicting interests
M.A. has received honoraria from Mitsubishi Tanabe Pharma Corp. The other authors have no conflicts of interest to declare. Clinical Trial Registration number: UMIN000031454.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
