Abstract
Objective:
To investigate the long-term prognostic value of insulin-like growth factor-binding protein 1 in patients with acute myocardial infarction.
Methods:
Patients (n = 180) with admission glucose < 11 mmol/L without previously known diabetes admitted for an acute myocardial infarction in 1998–2000 were followed for mortality and cardiovascular events (first of cardiovascular mortality/acute myocardial infarction/stroke/severe heart failure) until the end of 2011 (median 11.6 years). Fasting levels of insulin-like growth factor-binding protein 1 at day 2 were related to outcome in Cox proportional hazard regression analyses.
Results:
Median age was 64 years, 69% were male and median insulin-like growth factor-binding protein 1 was 20 µg/L. Total mortality was 34% (n = 61) and 44% (n = 80) experienced a cardiovascular event during a median follow-up time of 11.6 years. After age adjustment, insulin-like growth factor-binding protein 1 was associated with all-cause (1.40; 1.02–1.93, p = 0.039) and cancer mortality (2.09; 1.15–3.79, p = 0.015) but not with cardiovascular death (p = 0.29) or cardiovascular events (p = 0.57). After adjustments also for previous myocardial infarction, previous heart failure and body mass index, insulin-like growth factor-binding protein 1 was still associated with all-cause mortality (1.38; 1.01–1.89, p = 0.046).
Conclusion:
In patients with acute myocardial infarction without previously known diabetes, high insulin-like growth factor-binding protein 1 was associated with long-term all-cause and cancer mortality but not with cardiovascular events.
Keywords
Introduction
Cardiovascular (CV) disease is the most common cause of death in the world accounting for one third of all mortality causes. 1 Individuals with newly discovered glucose abnormalities and known diabetes have a high risk of CV disease and also an increased rate of adverse events after an acute coronary syndrome.2,3 To identify high-risk patients and further explore the underlying causes for long-term risk, the importance of the insulin-like growth factor 1 (IGF-1) system and insulin-like growth factor-binding protein 1 (IGFBP-1) has been explored. Circulating IGFBP-1 is mainly derived from the liver where insulin regulates the production by inhibiting its transcription.4,5 IGFBP-1 is therefore closely related not only to hyperinsulinaemia, insulin resistance and glucose abnormalities, but also to future CV events.6–8 Still, the relationship of IGFBP-1 and CV disease remains conflicting. Low levels of IGFBP-1 have been associated with future CV risk in patients with diabetes, 9 while high levels of IGFBP-1 seemed to be related to mortality in the elderly, 10 critically ill patients 11 and after an acute myocardial infarction (AMI) in patients with known diabetes.12,13 Whether this also applies to individuals with AMI without known diabetes remains less well explored.
The major aim of this report was to evaluate the relation between IGFBP-1 and the long-term prognosis after an AMI in patients without previously known diabetes, testing the hypothesis that IGFBP-1 is positively associated to mortality in a cohort with a large proportion of newly detected abnormal glucose tolerance (AGT).
Research design and methods
This analysis is based on the Glucose Tolerance in Patients with Acute Myocardial Infarction (GAMI) trial described previously in detail. 14 A total of 181 patients with AMI, admission capillary blood glucose < 11.1 mmol/L and without known diabetes were prospectively enrolled when admitted to Karolinska and Västerås Hospital coronary care units in 1998–2000. The exclusion criteria were age > 80 years and serum creatinine > 200 µmol/L. 14 All patients were treated for their AMI according to established international guidelines at that time. 14 Fasting levels of biochemical parameters, including IGFBP-1, were analysed on the first morning after admission (day 2) and at discharge (day 4–5). A standardised oral glucose tolerance test (OGTT) was performed in 167 patients on the day of hospital discharge and patients were classified as normal glucose tolerance (NGT), impaired glucose tolerance (IGT) or newly discovered diabetes (DM) according to the World Health Organization (WHO) definition 1999. Patients with IGT and DM were merged as having AGT.
Laboratory analyses
Glucose concentration was measured in whole capillary blood immediately after sampling by means of a HemoCue® portable photometer (HemoCue AB, Ängelholm, Sweden).
HbA1c was analysed by high-performance liquid chromatography on whole blood applied on a filter paper (Boehringer-Mannheim Scandinavian AB, Bromma, Sweden). The upper normal limit was 5.3% (MonoS) and the coefficients of variation were <3%. HbA1c will be reported as MonoS and IFCC (mmol/mol) standards.
Plasma IGF-1 was analysed by an in-house radioimmunoassay according to Bang et al. 15 Since IGF-I declines with age, thus age dependent, the IGF-I values are expressed as deviation from the mean of age-matched controls similar to T-score. The standardised IGF-1 score (IGFSD) was calculated as IGFSD = [log(IGF-1) + 0.00625 × age – 2.555]/0.104. 16
Plasma IGFBP-1 was analysed according to the method of Póvoa et al. 17 by radioimmunoassay where the sensitivity was 3 µg/L and the intra- and inter-assay coefficients of variation were 3% and 10%, respectively.
Plasma lipids were measured in the fasting state by standard methods at the local laboratory.
Plasma insulin and intact proinsulin were quantified with commercially available immunoassays (DAKO Ltd, Cambridgeshire, UK). Insulin resistance was estimated at a fasting steady state by homeostasis model assessment [HOMA; homeostasis model assessment of insulin resistance (HOMA-IR)]. Insulinogenic index (IGI) was calculated as the difference between 30-min and 0-min plasma insulin divided by the difference between 30-min and 0-min plasma glucose at OGTT.
Outcome measures and definitions
Patients were followed until 31 December 2011 with one patient lost to follow-up during this time leaving 180 patients as the final cohort. Information on CV events was derived from hospital and outpatient clinical records supplemented by a telephone interview with the patient or a close relative. Information on the cause of death was derived from death certificates obtained from the Swedish National Death Registry and additionally checked against the available hospital records.
Mortality reasons were classified as CV (caused by AMI/stroke/aortic dissection or sudden death without any obvious reason), cancer or other.
AMI was diagnosed according to national guidelines. A re-infarction was classified as an AMI > 72 h after the index infarction.
Stroke was defined as a neurological deficit observed by a physician and persisting > 24 h without any other disease explaining the symptoms.
Severe congestive heart failure (CHF) was defined as necessitating hospital admission for heart failure with intensified treatment.
Major CV event was defined as the first occurrence of re-infarction/AMI, stroke, severe CHF or CV death.
AGT was defined as either IGT or diabetes at the OGTT.
Statistics
Baseline characteristics are presented as median (lower and upper quartiles) for continuous variables and numbers and percentages for categorical variables. The Chi-square or, when appropriate, Fisher’s exact test and Wilcoxon rank-sum test were used to compare baseline characteristics between groups. Associations between IGFBP-1 at day 2 and other variables was analysed by Spearman’s rank-order correlation. The independent prediction of IGFBP-1 for a future event was analysed before and after age adjustments in Cox proportional hazard regression models. IGFBP-1 was log-transformed before analyses to avoid the influence of extreme values. If still statistically significant after age adjustments, we also adjusted for important clinical variables as previous MI (prior to the index event), previous heart failure, body mass index (BMI) and AGT. It was estimated that with few mortality events due to cancer (n = 18) further analyses beyond age adjustments would not be appropriate. Competing risks for cause-specific mortality and major CV event were analysed by estimates of the cumulative incidence function (CIF). In addition to be analysed as a log-transformed continuous variable, IGFBP-1 was also dichotomised and examined by gender-specific median levels. To further identify the prognostic capacity and a predictive level of IGFBP-1 at day 2, quintiles of IGFBP-1 were analysed in terms of mortality. Kaplan–Meier curves were computed to assess time to mortality and major CV event by the gender-specific median levels of IGFBP-1. A two-sided p value < 0.05 was accepted as statistically significant. Analyses were conducted using the SAS statistical program (SAS version 9.4; SAS Institute, Cary, NC, USA).
Ethical consideration
The local ethics committee of Karolinska Institutet approved the study protocol that complies with the Declaration of Helsinki. All recruited participants provided their written informed consent for the initial study and the follow-up.
Results
Baseline characteristics
The median age was 64 years (interquartile range (IQR): 57–71) and 69% were male. In total, 20% had a previous history of MI, 8% of CHF and 3% of stroke. Among patients performing OGTT, 32% (n = 54) had NGT and 68% (n = 113) had AGT (IGT − 58; diabetes − 55). Median level of IGFBP-1 at day 2 (n = 177) was 19 µg/L (IQR: 10–39 µg/L) for men and 23 µg/L (IQR: 16–36 µg/L) for women. The corresponding values for IGFBP-1 at discharge (n = 160) were 19 µg/L (IQR: 10–29 µg/L) for men and 22 µg/L (IQR: 15–32 µg/L) for women. Clinical and laboratory characteristics for patients by median level of IGFBP-1 at day 2 are presented in Table 1. Patients with elevated levels of IGFBP-1 (above median) were older (65 vs 62 years, p = 0.032), with lower low-density lipoprotein (LDL; 3.6 vs 3.9 mmol/L, p = 0.028) and had previous hypertension less often (22% vs 41%, p = 0.007). There was no difference in the proportion of AGT (61% vs 64%, p = 0.875) although those with IGFBP-1 below the median had a higher proportion of newly detected diabetes (40% vs 26%, p = 0.043). Baseline characteristics by quintiles of IGFBP-1 at day 2 are presented in Supplemental Table 1. IGFBP-1 at day 2 correlated positively to adiponectin, age, previous heart failure and cortisol and inversely to insulin and IGF-1 (data not shown).
Clinical and laboratory characteristics in patients divided into those above and below the median (men − 19 µg/L, IQR: 10–39 µg/L; women − 23 µg/L, IQR: 16–36 µg/L) for IGFBP-1 at day 2.
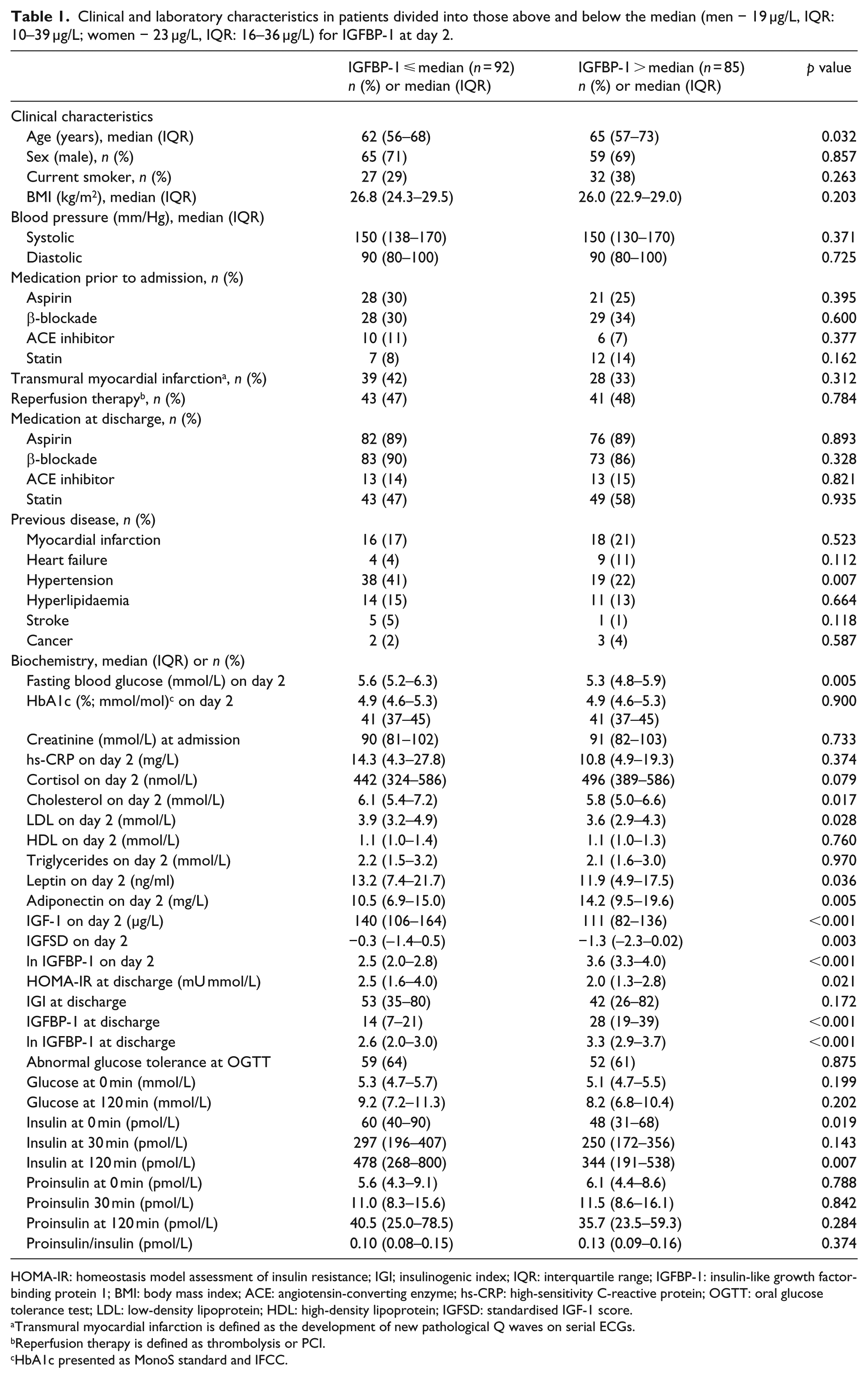
HOMA-IR: homeostasis model assessment of insulin resistance; IGI; insulinogenic index; IQR: interquartile range; IGFBP-1: insulin-like growth factor-binding protein 1; BMI: body mass index; ACE: angiotensin-converting enzyme; hs-CRP: high-sensitivity C-reactive protein; OGTT: oral glucose tolerance test; LDL: low-density lipoprotein; HDL: high-density lipoprotein; IGFSD: standardised IGF-1 score.
Transmural myocardial infarction is defined as the development of new pathological Q waves on serial ECGs.
Reperfusion therapy is defined as thrombolysis or PCI.
HbA1c presented as MonoS standard and IFCC.
Outcome
Patients were followed during a median time of 11.6 years (IQR: 9.6–12.1). Events are presented in Table 2. In the total cohort, all-cause mortality was 34% (n = 61) and 44% (n = 80) experienced a major CV event. All-cause mortality and death due to cancer were more common in patients with elevated levels of IGFBP-1 compared to those with low IGFBP-1, while CV events were equally distributed.
Events in patients by level of IGFBP-1. Each event is recorded only once. Mortality causes are presented as the proportion of the total mortality. Three patients had missing IGFPB-1 values.
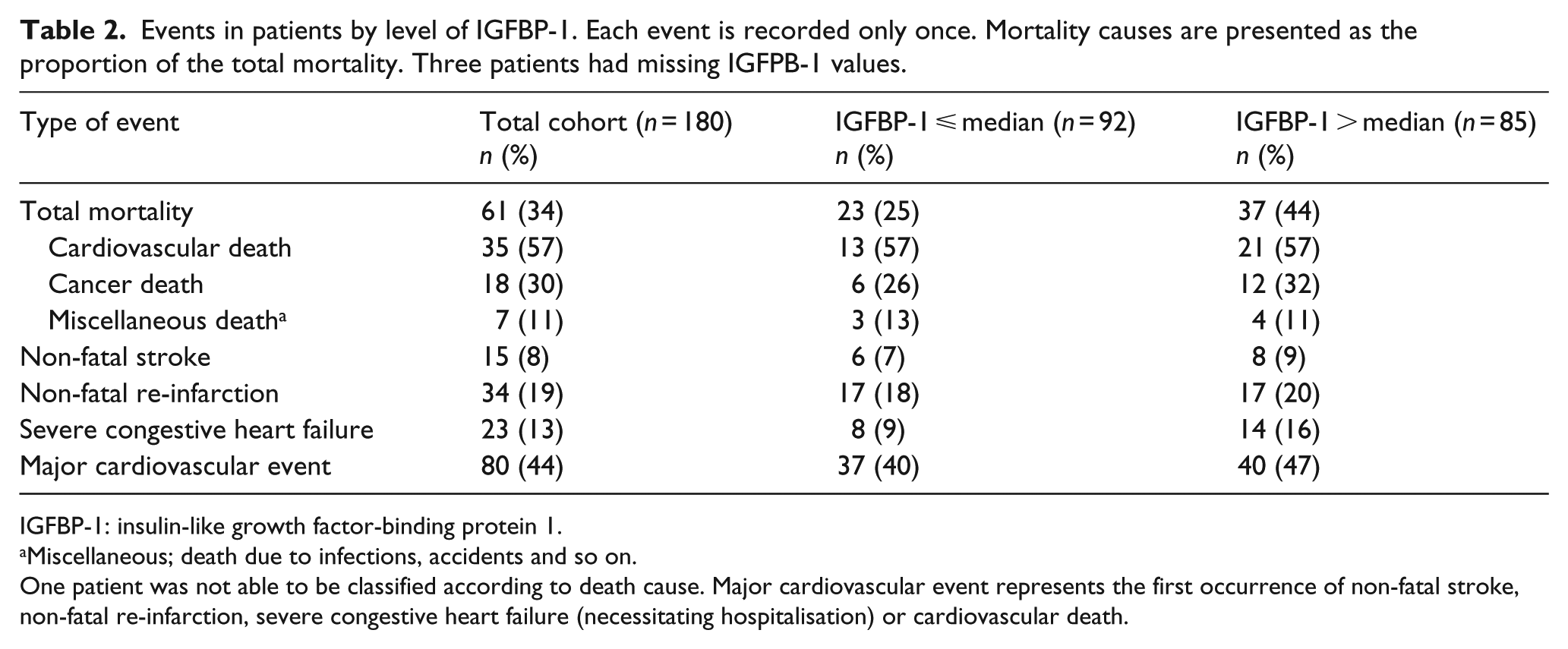
IGFBP-1: insulin-like growth factor-binding protein 1.
Miscellaneous; death due to infections, accidents and so on.
One patient was not able to be classified according to death cause. Major cardiovascular event represents the first occurrence of non-fatal stroke, non-fatal re-infarction, severe congestive heart failure (necessitating hospitalisation) or cardiovascular death.
Univariate and age-adjusted Cox proportional hazard regression analyses are presented in Table 3. After age adjustment, IGFBP-1 at day 2 predicted all-cause mortality [hazard ratio (HR): 1.40; 95% confidence interval (CI): 1.02–1.93, p = 0.039] and cancer mortality (2.09; 1.15–3.79, p = 0.015) but not CV events (p = 0.57) or CV mortality (p = 0.29). When further adjusting for previous MI, previous heart failure and BMI, IGFBP-1 was still associated with all-cause mortality (1.38; 1.01–1.89, p = 0.046), but not when also including AGT (1.34; 0.97–1.87, p = 0.080). When competing risk analyses were performed, IGFBP-1 at day 2 was not associated with cancer death (age adjusted − 1.93; 0.96–3.88, p = 0.065). IGFBP-1 at day 2 corresponding to the highest quintile (n = 37; 43–231 µg/L) was associated with increased mortality rate (2.65; 1.17–6.00, p = 0.019; Supplemental Figure 1). In age-adjusted analyses, IGFBP-1 at discharge did not remain a significant predictor for any outcome (Table 3). IGFSD at day 2 was associated with death due to cancer (0.70; 0.53–0.93, p = 0.015; age adjusted − 0.72; 0.54–0.95, p = 0.019) but was not associated with all-cause mortality (p = 0.67), CV mortality (p = 0.09) or CV events (p = 0.94).
Predictors of future events in patients, unadjusted and age-adjusted Cox proportional hazard regression models (HR; 95% CI).
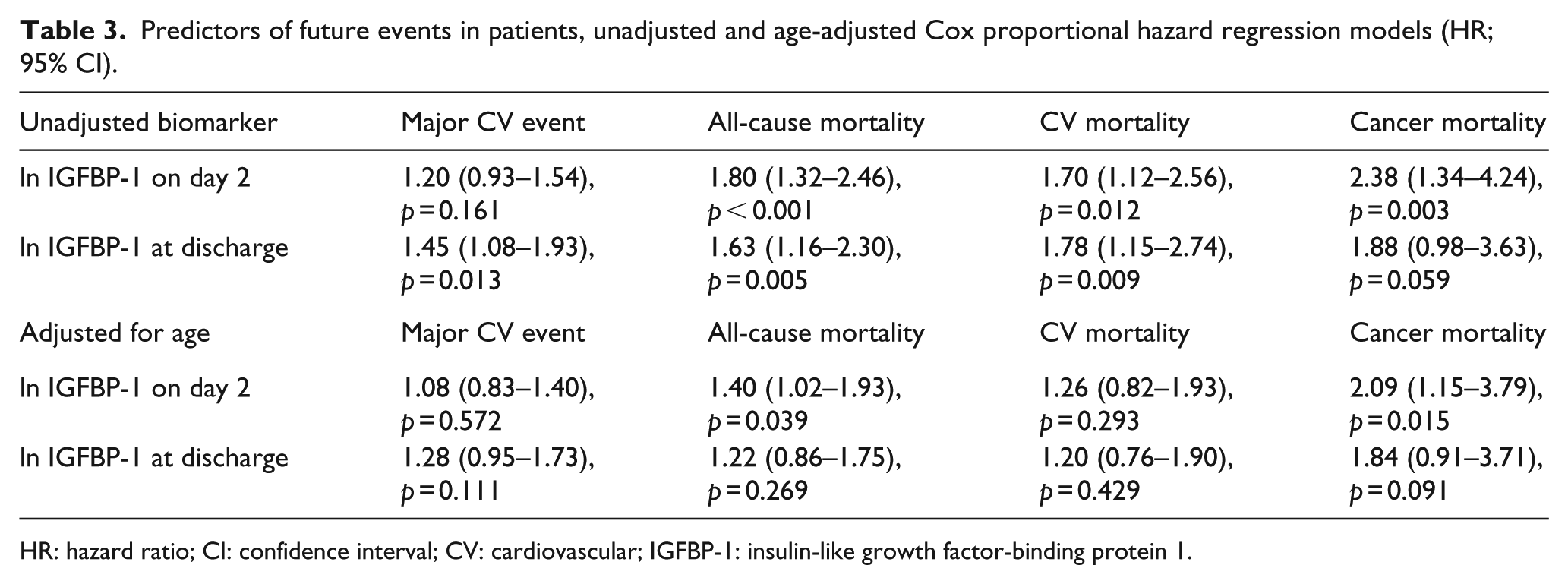
HR: hazard ratio; CI: confidence interval; CV: cardiovascular; IGFBP-1: insulin-like growth factor-binding protein 1.
Kaplan–Meier curves for long-term outcome
Kaplan–Meier estimates for time to all-cause mortality, death due to cancer and CV events by sex-specific medians of IGFBP-1 at day 2 are depicted in Figure 1(a) to (c). Mortality rate was higher in patients with elevated levels of IGFBP-1 at day 2 (log-rank test p = 0.012; Figure 1(a)), but there was no significant difference in CV event-free survival (log-rank p = 0.388; Figure 1(c)). Kaplan–Meier estimates for time to all-cause mortality, death due to cancer and CV events by sex-specific medians of IGFBP-1 at discharge are presented in Supplemental Figure 2(a) to (c) with higher mortality rate in patients with elevated levels of IGFBP-1 at discharge (log-rank test p = 0.007; Figure 2(a)).
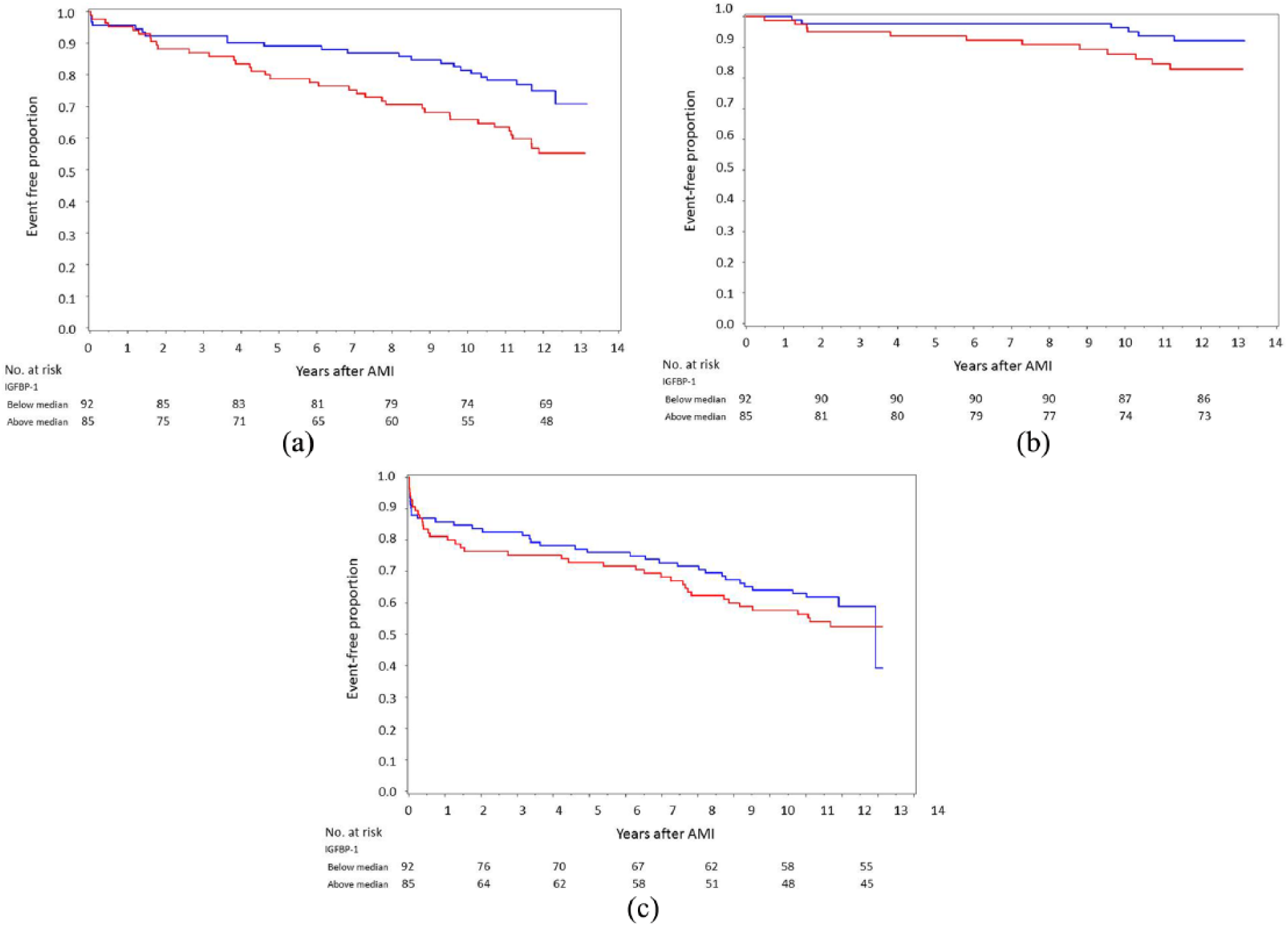
Kaplan–Meier curves for IGFBP-1 at day 2 (n = 177) divided into patients above (red) and below (blue) the median (men − 19 µg/L, women − 23 µg/L) related to (a) all-cause mortality (log-rank test p = 0.012), (b) cancer death (log-rank test p = 0.068) and (c) CV events (log-rank test p = 0.388).
Discussion
The major finding was that IGFBP-1 levels during the initial phase of an AMI were associated with higher long-term all-cause and cancer-induced mortality in the present cohort of patients with AMI and a substantial proportion of newly detected AGT. In contrast, IGFBP-1 was not associated with future CV events. Accordingly, IGFBP-1 levels in the higher range in the acute setting seem to identify a more compromised and severely diseased patient rather than being a marker for increased insulin resistance and risk for future CV complications.
The median level of IGFBP-1 in the present cohort was low (19 µg/L in men and 23 µg/L in women) when compared to age-matched healthy controls (24 µg/L in men and 33 µg/L in women) suggesting that the majority of these subjects had the metabolic syndrome with insulin resistance and hyperinsulinaemia. 6 Low levels of IGFBP-1 are associated with the metabolic syndrome 18 and increased risk of developing CV disease in patients without diabetes, 7 but the prognostic value of IGFBP-1 in patients with an acute CV event without known diabetes is to the best of our knowledge not explored. Elevated levels of IGFBP-1 above the mean in healthy controls represent oxidative stress, inflammation, catabolism, malnutrition,10,11 low insulin production and possibly renal impairment, 19 all factors that may hamper the prognosis in patients with an acute coronary event.
In the DIGAMI 2 trial, studying patients with known diabetes and AMI, there was a positive association between elevated levels of IGFBP-1 and CV death. 12 DIGAMI 2 recruited a large study population (n = 1253), of which 102 patients died from CV disease in comparison with only 35 patients in this study. A possible explanation to the present lack of relation may therefore be the lack of power. Furthermore, DIGAMI 2 included patients with long-standing diabetes and with glucose-lowering therapies including intense insulin treatment at the coronary care unit, a treatment known to increase IGFBP-1. In DIGAMI 2, the median level of IGFBP-1at day 2 was 23 µg/L and at discharge it was 33 µg/L to compare with the present median levels of 20 µg/L at day 2 and at discharge. Accordingly, in DIGAMI 2 there was a more potent rise of IGFBP-1 than in our study, maybe related to the active glucose lowering treatment in DIGAMI 2.
IGFBP-1 is proposed to be linked to inflammation. Experimental studies demonstrated that IGFBP-1 is regulated by cytokines and reactive oxygen species (ROS). 20 Under physical stress, epinephrine and noradrenaline stimulate the production of IGFBP-1 in humans. 21 During a stressful condition like an acute CV event, as in this study, patients with pathologically high IGFBP-1 (⩾43 µg/L, quintile 5) were those with the highest mortality. Moreover, patients with elevated levels of IGFBP-1 were older with a lower HOMA index and a lower insulinogenic index compared to patients with lower IGFBP-1. Accordingly, it seems that IGFBP-1 captures high-risk patients. There was a positive correlation between IGFBP-1 and age, previous heart failure, cortisol and adiponectin. Faxén et al. 22 demonstrated that IGFBP-1 correlated with the N-terminal pro b-type natriuretic peptide (NT-proBNP) in a heart failure population; however, there is no information on NT-proBNP in this cohort. We have recently demonstrated that adiponectin is associated with increased long-term mortality after an AMI. 23 Like adiponectin, IGFBP-1 may be more related to severe comorbidities and serve as a marker of patients at increased risk rather than signalling CV events.
IGFBP-1 at day 2 was associated with total mortality also when adjusting for other clinically relevant factors, such as age, BMI, previous MI and previous heart failure but not when AGT was entered into the model. IGFBP-1 at discharge was associated with all-cause mortality but did not remain predictive when adjusting for age. The most likely explanation for this would be lack of statistical power. The Kaplan–Meier estimates for time to all-cause mortality by IGFBP-1 at day 2 and at discharge have similar appearance only with different number of individuals and events. Furthermore, there were no differences in age in patients with available IGFBP-1 at day 2 (n = 177) compared to those with IGFBP-1 at discharge (n = 160) and also in associations between other variables and IGFBP-1 at day 2 or at discharge (data not shown). IGFBP-1 may therefore be a constant predictor of dismal prognosis, not only taken in the initial phase of an acute coronary syndrome but also at discharge or even in the stable phase after an AMI although the present material is not enough to confirm such an association.
IGFBP-1 correlated inversely with IGFSD. IGFSD was negatively and IGFBP-1 was positively associated with cancer mortality in Cox proportional hazard regression analyses. Low IGF-1 seems to protect against cancer according to previous reports.24,25 Analysing the predictive role of both IGF-1 and IGFBP-1, Kaplan et al. 26 demonstrated that decreasing IGF-1 level over time did not predict death due to cancer, whereas increasing IGFBP-1 levels predicted an increased risk of cancer mortality even after extensive adjustments for age, sex, race, diabetes, BMI, creatinine, albumin and C-reactive protein (CRP). The small sample size, and accordingly few cancer-related deaths, limits the ability to perform further adjustments than for age, making it difficult to further assess the significance of the strong correlation between IGFBP-1 and IGF-1 in this study. Signorello et al. 27 demonstrated a positive association between IGFBP-1 and prostate cancer and Gianuzzi et al. 28 reported on high IGFBP-1 levels in patients with ovarian cancer. The levels of IGFBP-1 may vary depending on the type of tumours. 29 Unfortunately, there was no such information available in this study as only overall cancer has been analysed. Although the mechanisms behind an increased risk of cancer are not known, one may speculate in the regulating role of IGFBP-1 on the growth hormone (GH) axis and cell growth. Increased IGFBP-1 levels cause an increase in GH secretion which may locally increase IGF-1 production and stimulate cell growth.
Strengths and limitations
Strengths of the GAMI study are the well-characterised cohort including the careful ascertainment of glucose tolerance and evaluation of future events. In addition, as men have lower levels of IGFBP-1 than women, IGFBP-1 was dichotomised in gender-specific medians to compare high and low IGFBP-1. Additional strength is that the patients did not receive any glucose-lowering treatment before sampling that could have affected the IGFBP-1 levels. The major limitation is the small sample size and limited numbers of events despite a decade of follow-up. We may therefore, as underlined, unintentionally have overlooked an association with CV events. The small sample size limits the ability to evaluate prognostic cut-off levels and also resulted in limited possibilities for adjustments in Cox proportional hazard regression models. When competing risk analyses were performed, age-adjusted IGFBP-1 at day 2 was not statistically significantly associated with cancer death. The most likely explanation for this would be the lack of statistical power with few cancer-related deaths. The results should therefore be confirmed in larger cohorts. Another limitation is that the treatment during follow-up is not known or whether this was different in the two groups. Those with IGFBP-1 levels below median had more signs of metabolic syndrome and insulin resistance and may have therefore been treated differently and thus protected from severe events. Finally, the cohort study design hampers the possibility to attribute causality to the association between IGFBP-1 and mortality, even when adjusting for known established CV risk factors in the multivariate model.
Conclusion
In patients with AMI and a high proportion of newly detected AGT, IGFBP-1 in the initial phase is associated with long-term outcome with an increased total mortality risk and was also related to death due to cancer. Accordingly, IGFBP-1 levels in the higher range in the acute setting seems to mirror individuals at a high risk for a dismal prognosis rather than serving as a marker of increased insulin resistance or risk for future CV events.
Supplemental Material
dvdres-dec-2017-00190-Supplementary_File – Supplemental material for Elevated levels of insulin-like growth factor-binding protein 1 predict outcome after acute myocardial infarction: A long-term follow-up of the glucose tolerance in patients with acute myocardial infarction (GAMI) cohort
Supplemental material, dvdres-dec-2017-00190-Supplementary_File for Elevated levels of insulin-like growth factor-binding protein 1 predict outcome after acute myocardial infarction: A long-term follow-up of the glucose tolerance in patients with acute myocardial infarction (GAMI) cohort by Viveca Ritsinger, Kerstin Brismar, Linda Mellbin, Per Näsman, Lars Rydén, Stefan Söderberg and Anna Norhammar in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
We would like to thank Inga-Lena Wivall, RN, and Elvi Sandberg, RN, at Department of Molecular Medicine and Surgery and Department of Endocrinology, Metabolism and Diabetes for analyses of IGFBP-1 and IGF-I; Christina Edman Jönsson, RN, at the Cardiology Unit at Karolinska University Hospital Solna; and Eleni Tanoglidi MD at the Department of Medicine and Centre for Clinical Research, Central Hospital Västerås, for assistance in collecting events. All authors have made substantial contributions to this paper. All authors took part in the interpretation of the results. L.R. and A.N. participated in the study design and collection of patients. IGFBP-1 and IGF-I levels were analysed by K.B. V.R. participated in the collection of events and analysed the death certificates together with A.N. V.R. managed the database and performed the statistical analyses. P.N. provided supervision and support within the frameworks of statistical analyses. K.B. contributed with expert knowledge on IGFBP-1. V.R. and A.N. finalised the manuscript after adjustments by all authors.
Declaration of conflicting interests
L.R. has received research support from the Swedish Heart-Lung Foundation, Swedish Diabetes Foundation, Karolinska Institute, Stockholm County Council, Swedish Medical Assembly, Private donators, Amgen, Bayer AG, Boehringer-Ingelheim, Merck and Novo Nordisk, and honoraria for lectures and expert group meetings from Bayer AG, Boehringer-Ingelheim, MSD and Sanofi. A.N. has received honoraria for expert group participation from Astra Zeneca, MSD, Novo Nordisk and Boehringer Ingelheim. V.R. has received honoraria from Astra Zeneca, Novo Nordisk and Boehringer Ingelheim for expert group participation. K.B., L.M., P.N. and S.S. report no conflicts.
Funding
This work was supported by the Swedish Heart-Lung Foundation, the Department of Research and Development Region Kronoberg, Västerås Hospital, Family Erling-Persson foundation, Medical Research Council and the Family Kamprad Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
