Abstract
Aim:
Glomerular hyperfiltration usually occurs early in development of kidney complications in diabetes. To understand hyperfiltration as a marker of renal disease progression in type 2 diabetes mellitus, we aimed to examine association between glomerular hyperfiltration (estimated glomerular filtration rate ⩾ 120 mL/min/1.73 m2) and rapid renal decline (annual estimated glomerular filtration rate loss ⩾ 3 mL/min/1.73 m2).
Methods:
This was a prospective cohort comprising 1014 patients with type 2 diabetes mellitus attending a Diabetes Centre of a regional hospital in 2002–2014. A separate prospective cohort, comprising 491 patients who attended Diabetes Centre or primary-care polyclinics, was used for validation. We performed binary mediation analysis to examine role of hyperfiltration on relationship between baseline haemoglobin A1c and rapid renal decline.
Results:
Among patients in discovery cohort, 5.2% had baseline hyperfiltration. Over mean follow-up of 6 years, 22.9% had rapid glomerular filtration rate decline. Baseline hyperfiltration was significantly associated with greater odds of rapid renal decline after adjusting for demographics, diabetes duration and clinical covariates (odds ratio: 2.57; 95% confidence interval: 1.21–5.46;
Conclusion:
Glomerular hyperfiltration is an independent risk factor of rapid renal decline. It mediates the association between increasing haemoglobin A1c and rapid renal decline.
Introduction
Diabetic kidney disease (DKD) remains as one of the major complications in type 2 diabetes mellitus (T2DM). About 25%−40% of patients with T2DM develop kidney complications. 1 The problem is more pronounced in Asians who are more susceptible to DKD than other ethnicities such as Caucasians. 2 As such, there is a strong impetus to understand the natural history of DKD, which is of particular importance in preventing the onset and progression to end-stage renal disease (ESRD).
Notably, glomerular hyperfiltration is an early manifestation in the clinical course of DKD.3,4 There is no established estimated glomerular filtration rate (eGFR) cut-off for hyperfiltration, but it can vary from 120 to 150 mL/min/1.73 m2. 5 Likewise, the prevalence of glomerular hyperfiltration in T2DM ranged widely from 0% to 73%.6–8 Such marked heterogeneity in prevalence is likely attributed to differing definition of hyperfiltration and distribution of factors like ageing, control of haemoglobin A1c (HbA1c), diabetes duration and glomerular filtration rate (GFR) methodology. 9
It was suggested that glomerular hyperfiltration might predispose to development of DKD.4,10 However, understanding the putative role of glomerular hyperfiltration has been hampered by small sample sizes and insufficient duration of follow-up
9
in addition to the inability to control confounding. There is also limited information on the link between hyperfiltration and GFR trajectory as previous studies have mainly focused on albuminuria as the renal outcome.
9
A few studies have reported an association between hyperfiltration and subsequent GFR evolution in T2DM with discordant results.7,11,12 For example, the study by Silveiro et al.
12
observed that patients with non-insulin-dependent diabetes and hyperfiltration were more likely to experience decline in GFR, whereas the study by Chaiken et al.
11
reported that hyperfiltration in patients with T2DM did not confer higher risk for development of diabetic nephropathy (DN). The inconsistency may be attributed to issues like limited sample size (as low as
The clinical implications in proving hyperfiltration as an early marker of DKD constitute another compelling reason for investigating hyperfiltration as a key haemodynamic driver of accelerated GFR decline. It is timely to revisit the pathogenic role of hyperfiltration given the recent emergence of sodium-glucose co-transporter 2 (SGLT2) inhibitors for treatment in diabetes mellitus (DM). There is mounting evidence that these agents confer specific reno-protective benefit against GFR decline by several mechanisms, one of which being attenuation of glomerular hyperfiltration and therefore in-tandem, ameliorating intra-glomerular hypertension. 7
We aim to examine the relationship between glomerular hyperfiltration and rapid GFR decline. As hyperglycaemia has been proposed as one of the key pathogenic factors of hyperfiltration and that insulin treatment could reverse early hyperfiltration at the initial stage of type 1 diabetes mellitus (T1DM),7,13 we also aim to study the role of hyperfiltration as a hypothesised mechanism accounting for the association between glycaemic burden and rapid renal decline.
Methods
Study population
The DN Cohort Study (https://www.ktph.com.sg/cru/section_page/357/97) was a prospective observational cohort study which enrolled adult subjects with T2DM attending the Diabetes Centre in the Alexandra Health Private Limited (AHPL) regional hospital in Singapore from 2002 to 2014 (‘discovery cohort’). The inclusion criteria were as follows: ⩾3 eGFR results, ⩾2 years of follow-up duration and urinary albumin-to-creatinine (uACR) ratio of ⩽300 mg/g at baseline. A total of 1014 subjects met the inclusion criteria.
Data collection
Demographic, clinical and medication [including renin–angiotensin system (RAS) antagonist, insulin, statins and fibrates] information were obtained from a standard questionnaire or extracted from patient’s medical records. Trained nurses measured blood pressure (BP) using a standard sphygmomanometer using an appropriate cuff size in the sitting position. The body mass index (BMI) was derived by dividing weight in kilograms by the square of height in metres.
Blood and spot urine samples were collected and sent to the hospital laboratory accredited by the College of the American Pathologists (CAP). Serum creatinine, low-density lipoprotein cholesterol (LDL-cholesterol) and triglycerides (TG) were quantitated with enzymatic colorimetric test (Roche Cobas® C501); HbA1c with Tina-quant Haemoglobin A1c Gen.3 (Roche Cobas® C501); and urinary albumin with immunoturibidmimetric assay (Roche Cobas® C501). Measurements were taken at recruitment and at multiple time points during follow-up. GFR was estimated using the CKD-EPI (CKD Epidemiology Collaboration) equation. 14 Hyperfiltration was defined as the eGFR ⩾ 120 mL/min/1.73 m2. This cut-off has been used to study the association of renal hyperfiltration and eGFR decline.5,8 The slope of the linear regression of all eGFR readings over time in years for each patient was assessed and expressed as mL/min/1.73 m2 per year. 15 Rapid GFR decline was defined as eGFR loss of ⩾3 mL/min/1.73 m2 based on the previous literature. 16
A separate cohort (SMART2D: https://www.ktph.com.sg/cru/section_page/494) was also used for determining the association of hyperfiltration with rapid GFR decline (‘validation cohort’). This comprised adult subjects attending the Diabetes Centre in the Alexandra Health Private Limited (AHPL) regional hospital or a primary-care polyclinic in the Northern region of Singapore between August 2011 and November 2013. The patients were included in the analysis if they had ⩾3 eGFR results and uACR was ⩽300 mg/g at baseline. A total of 491 subjects met the inclusion criteria. Clinical and laboratory measurements were taken at multiple time points till April 2016. Blood and urine samples were quantitated at the same hospital laboratory using assays similar to those for the DN cohort.
The study was approved by the National Healthcare Group Domain Specific Review Board in Singapore. All participants provided written informed consent.
Statistical analysis
Analyses were performed using STATA Version 14.0 (STATA Corporation, College Station, TX). Categorical variables were expressed as number (percentage), and continuous variables as mean values ± standard deviation (SD) or median (interquartile range) as appropriate. Differences in patient characteristics were compared using chi-square test for categorical variables and student
Multivariable logistic regression was used to study the relationship between hyperfiltration and rapid eGFR decline, adjusting for systolic blood pressure (SBP), HbA1c, LDL-cholesterol, uACR, age, gender, race, duration of diabetes, use of RAS antagonist and insulin. These variables were included in the model based on biological plausibility or
Mediation analysis was performed to examine the role of hyperfiltration on the relationship between glycaemic control and rapid GFR decline in the validation cohort. This involved Baron and Kenny
17
three-step framework to examine (1) if baseline HbA1c was associated with rapid GFR decline, (2) if baseline HbA1c was associated with hyperfiltration and (3) if the association between baseline HBA1c and rapid GFR decline was attenuated upon inclusion of hyperfiltration in the model. The commands ‘binary_mediation’ and ‘bootstrap’
Results
The baseline characteristics of the patients in the discovery cohort were as follows: mean age: 54.1 ± 11.1 years; 60.9% males; 69.8% Chinese, 12.0% Malay and 18.2% Indian; mean DM duration: 10.1 ± 7.7 years; mean BMI: 26.6 ± 5.0 kg/m2; mean HbA1c: 8.3% ± 1.8%; mean SBP: 132.5 ± 17.2 mm Hg; mean LDL-cholesterol: 2.8 ± 0.8 mmol/L; median TG: 1.4 mmol/L (1.0–2.0); median eGFR: 93.4 mL/min/1.73 m2 (79.9–104.4); and median uACR: 19 mg/g (8–48). Patients who had hyperfiltration at baseline tended to be younger, females, have shorter DM duration, higher HbA1c and LDL-cholesterol but lower SBP (
Baseline characteristics in the discovery cohort.
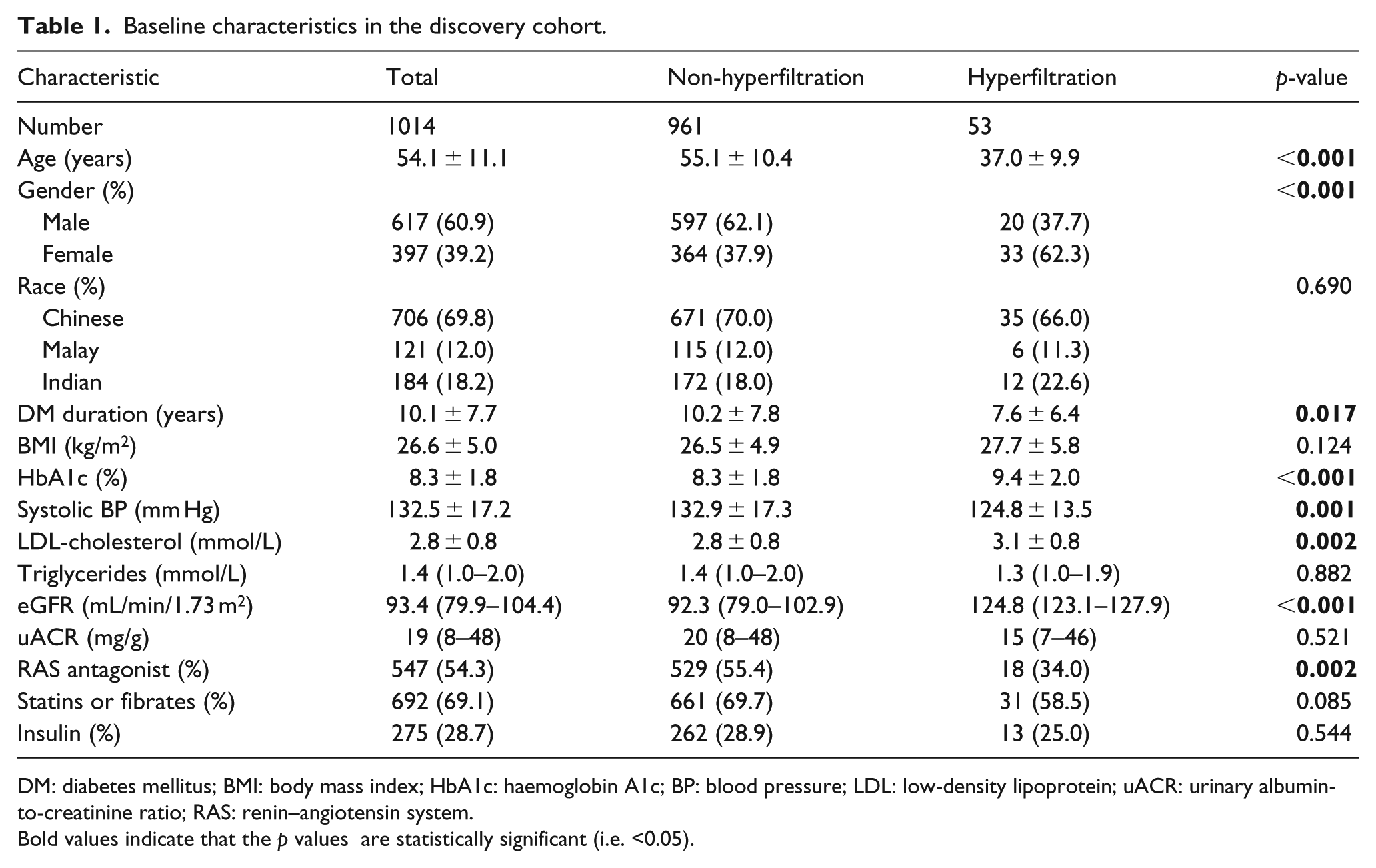
DM: diabetes mellitus; BMI: body mass index; HbA1c: haemoglobin A1c; BP: blood pressure; LDL: low-density lipoprotein; uACR: urinary albumin-to-creatinine ratio; RAS: renin–angiotensin system.
Bold values indicate that the
Over a mean follow-up period of 6.0 ± 2.2 years in the discovery cohort, 5.2% had baseline hyperfiltration and 22.9% rapid GFR decline. Patients with rapid GFR decline tended to be older, of Malay ethnicity and more adverse risk profile in terms of DM duration, HbA1c, SBP, LDL-cholesterol and uACR (
Characteristics stratified by rapid eGFR decline in the discovery cohort.
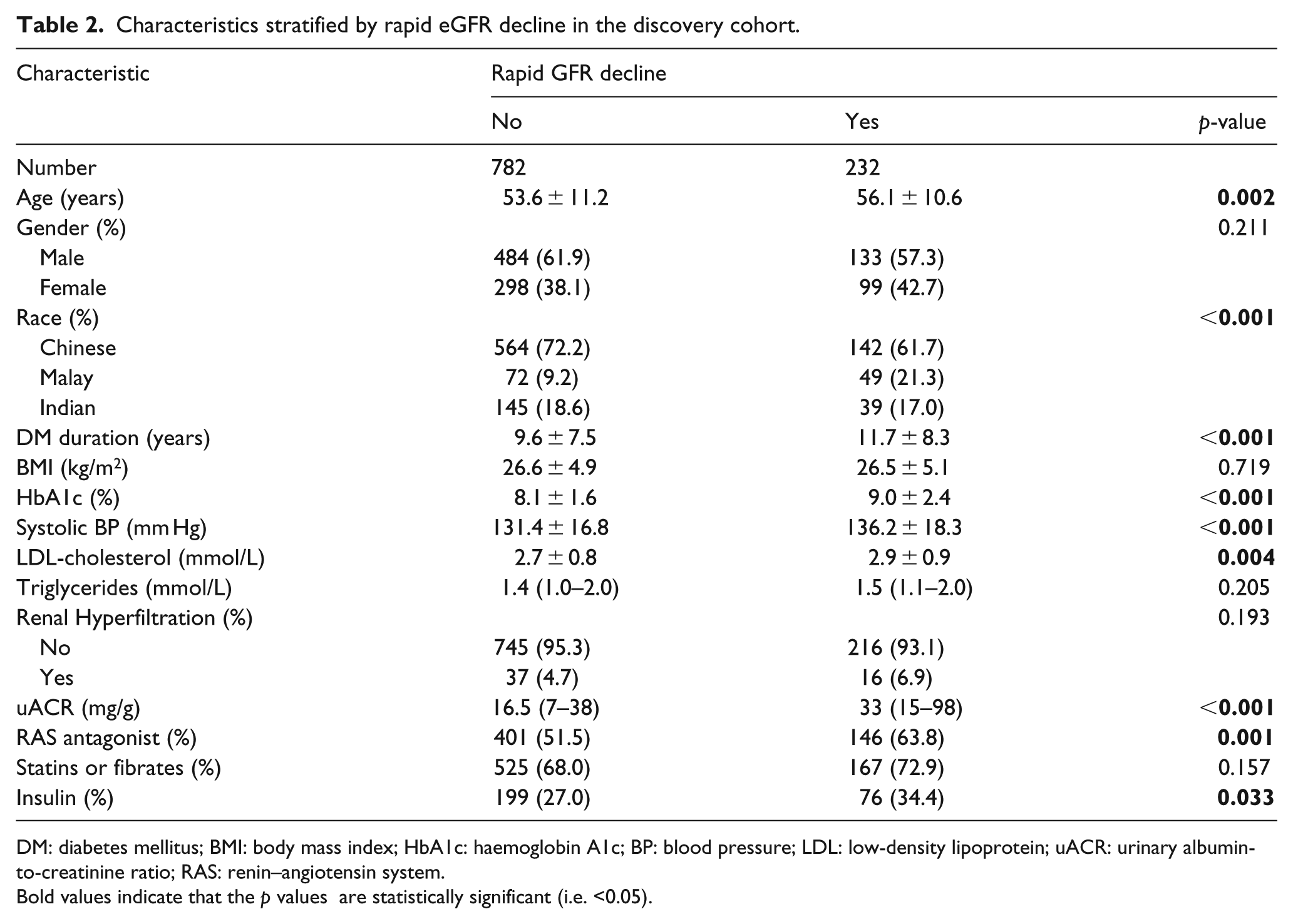
DM: diabetes mellitus; BMI: body mass index; HbA1c: haemoglobin A1c; BP: blood pressure; LDL: low-density lipoprotein; uACR: urinary albumin-to-creatinine ratio; RAS: renin–angiotensin system.
Bold values indicate that the
Baseline hyperfiltration was significantly associated with greater odds of rapid renal decline compared to those without hyperfiltration after adjusting for demographics, DM duration, clinical risk factors and medications with odds ratio (OR) 2.45 [95% confidence interval (CI): 1.15–5.26;
Multivariable model on rapid eGFR decline in the discovery cohort.
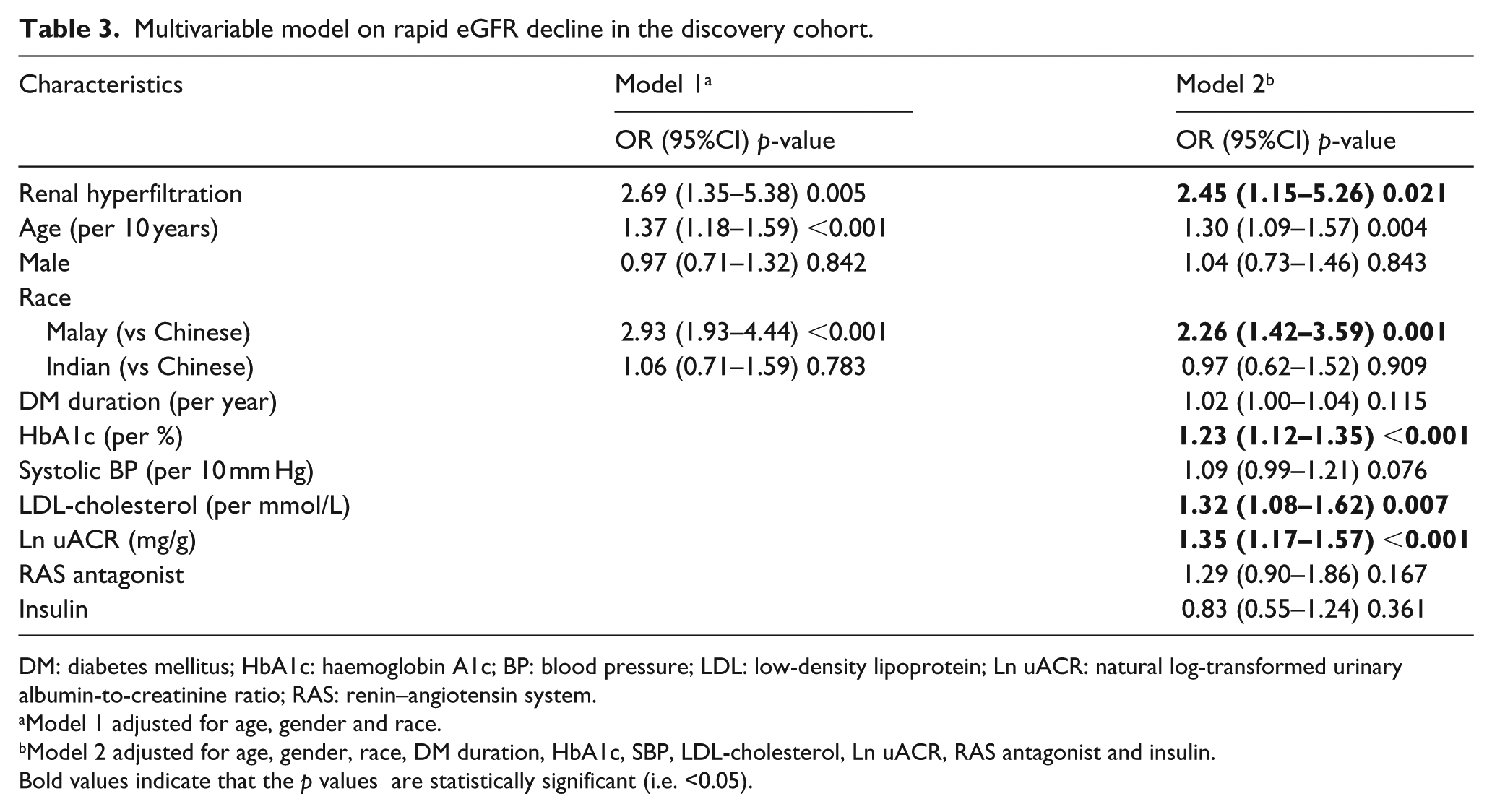
DM: diabetes mellitus; HbA1c: haemoglobin A1c; BP: blood pressure; LDL: low-density lipoprotein; Ln uACR: natural log-transformed urinary albumin-to-creatinine ratio; RAS: renin–angiotensin system.
Model 1 adjusted for age, gender and race.
Model 2 adjusted for age, gender, race, DM duration, HbA1c, SBP, LDL-cholesterol, Ln uACR, RAS antagonist and insulin.
Bold values indicate that the
Multivariable model on rapid eGFR decline in validation cohort.
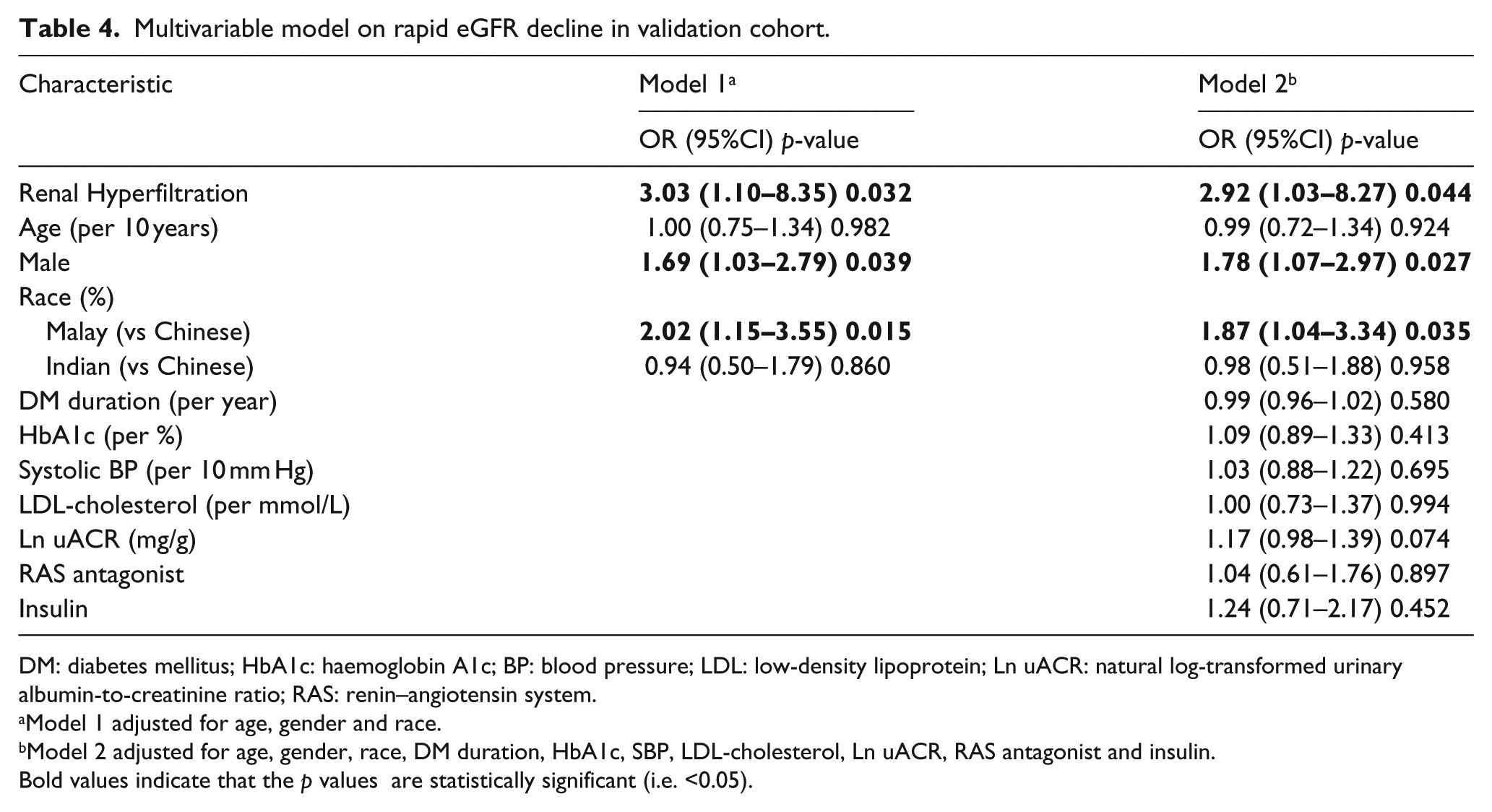
DM: diabetes mellitus; HbA1c: haemoglobin A1c; BP: blood pressure; LDL: low-density lipoprotein; Ln uACR: natural log-transformed urinary albumin-to-creatinine ratio; RAS: renin–angiotensin system.
Model 1 adjusted for age, gender and race.
Model 2 adjusted for age, gender, race, DM duration, HbA1c, SBP, LDL-cholesterol, Ln uACR, RAS antagonist and insulin.
Bold values indicate that the
Hyperfiltration significantly accounted for 35.3% of the association between baseline HbA1c and rapid eGFR decline, having adjusted for age, race, SBP, TG and uACR (
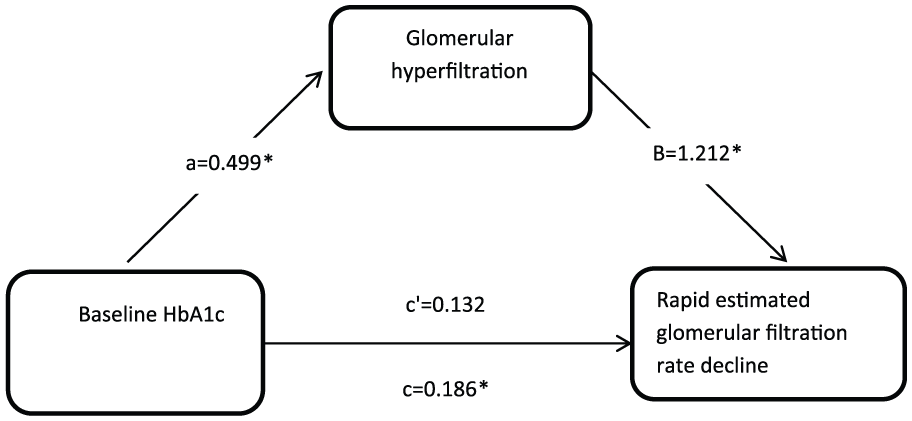
The mediation of hyperfiltration on the association between baseline HbA1c and rapid estimated glomerular filtration rate decline.
Discussion
Our study showed that only a small proportion of patients with T2DM had hyperfiltration (5.2% in discovery cohort and 5.5% in validation cohort). This was lower than the prevalence in most of the earlier studies, which we speculated to be attributable to the long-standing duration of diabetes (~10 years) in our study cohort, since hyperfiltration is an upstream early manifestation of DKD expected to be more prevalent among people with early diabetes.6–8 Nevertheless, the signification association between hyperfiltration and rapid renal decline highlighted the need for closer monitoring and more intensive management of this small group of patients who hyperfiltrate.
The pathogenesis of DKD has been broadly ascribed to two major determinants, namely, metabolic and haemodynamic injuries. 18 In this study, we have demonstrated that glomerular hyperfiltration (a form of haemodynamic insult) was associated with rapid renal decline. This finding was consistent in two different cohorts with T2DM. To date, the pathophysiological mechanism remains unclear. It was suggested that the pathogenesis of hyperfiltration is likely contributed by haemodynamic, vasoactive, tubular and systemic factors.5,7 For example, hyperfiltration was linked to haemodynamic alterations such as vasodilation in afferent arterioles and constriction efferent arterioles. 19 The association between hyperfiltration and raised intra-glomerular pressure also leads to more shear tension, thereby resulting in kidney damage. 20 Furthermore, it was demonstrated that the levels of tubular markers such as urinary neutrophil gelatinase-associated lipocalin (NGAL) and kidney injury molecule-1 (KIM-1) were higher in individuals with hyperfiltration compared to those with no hyperfiltration. This attested to the deleterious effect of hyperfiltration on the kidney. 21
Another notable finding that hyperfiltration mediated the relationship between baseline HbA1c and rapid renal decline. It was postulated that hyperglycaemia leads to increased glucose filtration at the glomerulus. This in turn causes more proximal tubule reabsorption mediated by SGLT2 and less sodium being delivered to the macula dense. The tubuloglomerular feedback is triggered, leading to glomerular hyperfiltration.22–24 A second plausible explanation was that the formation or bioavailability of vasoactive mediators (e.g. nitride oxide system, RAS and endothelin) was raised in the presence of hyperglycaemia. These in turn altered the tone of the afferent and efferent arterioles, resulting in raised glomerular filtration.22,23 Furthermore, sustained elevated HbA1c confers higher risk of microvascular complications in DM.25,26 Poor glycaemic control has also been related to rapid renal decline in earlier research. 27 Our finding shows that the deleterious effect of increasing HbA1c on the kidney was partly contributed by hyperfiltration.
We also observed that ethnic disparity in rapid renal decline where Malays had higher odds of rapid renal decline compared to Chinese and Indians. This is congruent with earlier findings that observed significant association between Malays and ESRD.
28
There are a few possible explanations. First, it was noted in our study that Malays had a more unfavourable clinical profile in terms of higher HbA1c and uACR than Chinese and Indians (
In our study, we used two cohorts to consistently establish the relationship between hyperfiltration and renal decline. We also explored the mediating role of hyperfiltration on the impact of poor glycaemic control on rapid renal decline. Other strengths of our study include availability of rich clinical information, including medication on RAS, which could have altered the course of progression and sampling of the subjects from healthcare setting which provided insights into the ‘real-world’.
There are also limitations in our study. First, our method of ascertaining eGFR by CKD-EPI to assign hyperfiltration status is relatively simple. However, the misclassification of hyperfiltration status is most probably random and hence will not lead to increased bias. Second, we sampled subjects from a diabetes centre in a regional hospital which limits generalizability of results to the general population with diabetes. There could also be residual confounding factors such as socio-economic and behavioural factors which might have influenced the progression of kidney disease.
Nevertheless, our results have a few potential clinical implications. First, hyperfiltration identifies a subset of patients with T2DM who may be more predisposed to rapid renal decline, thereby warranting heightened vigilance and intensive management to prevent renal deterioration. The second implication is that treatments targeted at ameliorating hyperfiltrating glomeruli is now available 31 and may be needed to prevent onset and progression of kidney disease in T2DM. Existing renal-retardation strategies include protein-restricted diet and renin–angiotensin–aldosterone system blockade. It is believed that these interventions work partly through the reduction of intra-glomerular hypertension and associated hyperfiltration. An evolving paradigm is therapeutic agents targeting renal tubules (e.g. SGLT2 inhibitors), which help to regulate intra-glomerular filtration pressure by incompletely understood tubuloglomerular feedback mechanism. 32 This novel strategy may be important in selected individuals experiencing hyperfiltration. 33
In conclusion, glomerular hyperfiltration is an independent predictor of rapid renal decline. Glomerular hyperfiltration mediates the association between increasing HbA1c and rapid renal decline.
Supplemental Material
dvdres-dec-2017-00206-File004 – Supplemental material for Long-term prospective observation suggests that glomerular hyperfiltration is associated with rapid decline in renal filtration function: A multiethnic study
Supplemental material, dvdres-dec-2017-00206-File004 for Long-term prospective observation suggests that glomerular hyperfiltration is associated with rapid decline in renal filtration function: A multiethnic study by Serena Low, Xiao Zhang, Jiexun Wang, Lee Ying Yeoh, Yan Lun Liu, Keven Kue Loong Ang, Wern Ee Tang, Pek Yee Kwan, Subramaniam Tavintharan, Chee Fang Sum and Su Chi Lim in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
S.L., X.Z. and S.C.L. designed the study. S.L. and X.Z. performed statistical analysis. J.W. guided the statistical analysis. S.L. drafted the manuscript. L.Y.Y., Y.L.L., W.E.T., P.Y.K., T.S. and C.F.S. contributed intellectual content in manuscript revision. K.K.L.A. assisted in data collection and data preparation. S.C.L. provided study supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Alexandra Health Grant (AHPL SIGII/11001, SIG/11029 and SIG/12024), National Medical Research Council Grant [NMRC/PPG/AH(KTPH)/2011] and National Medical Research Council Grant (NMRC/CIRG/ 1398/2014).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
