Abstract
This study aimed at investigating the combined effects of albuminuria and reduced estimated glomerular filtration rate (eGFR) on peripheral artery disease (PAD) among elderly patients with diabetes. A total of 236 subjects were cross-classified into four groups according to the presence or absence of albuminuria (urinary albumin creatinine ratio (ACR) ≥ 30 mg/g) and low eGFR (<60 mL/min/1.73 m2). Cardiovascular risk factors and the ankle–brachial index (ABI) were also assessed. After multivariate adjustment using logistic regression analysis, the odds ratios (OR) for prevalent PAD related to albuminuria with preserved eGFR, normoalbuminuria with low eGFR and albuminuria with low eGFR compared to normoalbuminuria with preserved eGFR were 1.10 [95% confidence interval (CI) = 0.43–2.79], 3.14 (95% CI = 1.20–8.22) and 3.87 (95% CI = 1.72–8.72), respectively. In conclusion, in elderly patients with type 2 diabetes, both normoalbuminuria with low eGFR and albuminuria with low eGFR are associated independently with PAD.
Introduction
Peripheral artery disease (PAD) is a diffuse atherosclerotic vascular disease commonly present in patients with diabetes, particularly for the elderly patients aged more than 65 years. 1 In Taiwan, its respective prevalence for the age groups of 65–74 and ≥75 years is 12.9% and 25.3%, respectively. 2 PAD has been linked with cardiovascular disease (CVD) as well as the risk of occurrence of foot-related injuries and amputations.3,4 It has been confirmed that elderly patients with type 2 diabetes after lower extremity amputations are more likely to suffer from a 1.9-fold higher risk of mortality if compared with those of younger patients. 5
Epidemiological studies have identified several risk factors that are associated independently with PAD, among which are older age, smoking habit, higher triglyceride concentration, higher systolic blood pressure (SBP) and pulse pressure, elevated high-sensitive c-reactive peptide (hs-CRP), chronic kidney disease (CKD), presence of albuminuria and low estimated glomerular filtration rate (eGFR).6–8 However, among these factors, the role of both albuminuria and low eGFR in the development of PAD among patients with diabetes has not been well elucidated. Previous studies have indicated that albuminuria is significantly correlated with PAD in elderly patients with diabetes.8,9 In contrast, a recent investigation established that reduced eGFR, independent of albuminuria, is strongly related to PAD in subjects with diabetes. It was proposed that as diabetes progressed, the predictive value of albuminuria gradually diminished after adjustment for renal function. 10
In fact, these investigations only relied on a single criterion to define the risk of PAD, either the presence of albuminuria or a reduction in the eGFR, and their combined effects of both metrics were not evaluated simultaneously.9–11 Since albuminuria and reduced eGFR might be closely related, the correlation between albuminuria and PAD could be confounded by renal function. Consequently, the results of previous research might have underestimated the magnitude of their association. 12 On the basis of these findings, both criteria of CKD should be used simultaneously for the purpose to better characterize the relationship between kidney disease and PAD.
Therefore, this study aimed first to investigate the characteristics of subjects when stratified with PAD and without PAD and second to determine the association between the combined effect of albuminuria and low eGFR with the presence of subclinical PAD in elderly patients with type 2 diabetes mellitus.
Methods
Study population
A total of 236 patients with type 2 diabetes mellitus, aged ≥65 years, were enrolled consecutively from the outpatient department of endocrinology and metabolism at a regional hospital in southern Taiwan. Study patients were mostly recruited into the diabetes shared care programme (DSCP), which provided appropriate care to the population with diabetes. The programme aimed at reducing the incidence of diabetes complications including PAD via a comprehensive strategy of screening and early intervention of the risk factors targeting hyperglycaemic, dyslipidemia and hypertension. Among the recruited patients, diagnosis of type 2 diabetes mellitus was according to the criteria of the American Diabetes Association (ADA), and patients were treated with either oral hypoglycaemic agents or insulin at the time of recruitment. 13 Subjects with acute kidney disease, moderate-to-severe congestive heart failure (New York Heart Association classification of III or IV), fever status and urinary tract infection were excluded. The protocol for this observation study was approved by our Institutional Review Board, and all participants provided informed consent.
Clinical and laboratory measurements
Demographic and medical data, including age, gender, history of smoking, duration of diabetes and comorbid conditions such as CVD and retinopathy, were garnered from medical records. The presence of CVD was diagnosed by the physician as a history of stroke or coronary heart disease (CHD). CHD was defined if they had a history of typical angina, the presence of electrocardiogram (ECG) abnormalities suggestive of CHD, old myocardial infarction or they had undergone coronary artery bypass surgery or angioplasty. Stroke was defined if they had a history of cerebrovascular incidents such as cerebral bleeding or infarction. Retinopathy was assessed by fundoscopy after pupil dilatation. History of smoking was defined as never/past/current.
Central obesity was defined if abdominal circumference was ≥90 cm in men and ≥80 cm in women. Blood pressure was measured by trained observers using an automatic oscillometric monitor while the patients were in the sitting position. Otherwise, information regarding patient medications, including anti-hypertensive agent, statin/fibrate, anti-platelet agent and insulin during the study period, was obtained from medical records.
All laboratory measurements were performed by the Yuan’s General Hospital laboratory using standardized and automated methods. Glycated haemoglobin (HbA1c) was determined by an ion-exchange high-performance liquid chromatography (HLC-723 G8; Tosoh, Tokyo, Japan). Serum creatinine and lipid profiles, including measurements of total cholesterol, triglyceride, high-density lipoprotein (HDL) and low-density lipoprotein (LDL), were measured on an Olympus AU2700 autoanalyzer (Beckman Coulter, Mishima, Japan) using an enzymatic assay. Hs-CRP was measured using an immunonephelometry assay (BN II; Siemens, Deerfield, IL, USA). In addition, urinary creatinine concentration and urinary albumin were measured on an Olympus AU2700 autoanalyzer using the alkaline picrate-kinetic method and immunoturbidimetric assay, respectively.
Measurement of albuminuria and low GFR
The urinary albumin excretion rate was presented as the albumin creatinine ratio (ACR), which was measured from an early morning spot urine sample. Albuminuria was defined as an ACR ≥ 30 mg/g based on the recommendation of ADA. 14 The eGFR was estimated using the Modification of Diet in Renal Disease (MDRD) Study formula as follows: 186 × [serum creatinine (mg/dL)]−1.154 × (age)−0.203 × (0.742 if female). 15 Preserved eGFR was defined as an eGFR of ≥60 and low eGFR or renal insufficiency was defined as an eGFR of <60.
In this study, subjects were then cross-classified into four distinct subgroups determined by albuminuria and low eGFR, defined as follows: normoalbuminuria with preserved eGFR, albuminuria with preserved eGFR, normoalbuminuria with low eGFR and albuminuria with low eGFR.
Ankle–brachial index measurements
In an effort to reduce elderly participant burden, only one measurement was taken for all participants. In brief, a bidirectional portable echo-Doppler of 8 MHz (VasoGuard MicroLite; Nicolet, Madison, WI, USA) and a calibrated mercury sphygmomanometer were used to measure simultaneously bilateral brachial and ankle SBPs. For this measurement, patients were in the supine position in a quiet room after a 10-min rest. The device automatically calculated ankle–brachial index (ABI) by dividing the ankle systolic pressure of either side by the highest of the right and left brachial systolic pressures. Subjects who had an ABI < 0.9 or > 1.4 in either leg were defined as having PAD, which was based on the results of a recent review. 16 If both ABI measures were ≥0.9 and ≤1.4, subjects were defined as having normal ABI.
Statistical analysis
Quantitative variables are presented as mean ± standard deviation or median (interquartile range) if they were normally or non-normally distributed. Qualitative variables are expressed as absolute numbers and percentages. The study groups stratified by prevalent PAD were compared on clinical characteristics using chi-square test for categorical variables, independent t-test for continuous variables and Mann–Whitney test for ACR and hs-CRP.
The odds ratios (OR) for prevalent PAD associated with the cross-classification of albuminuria and low eGFR were calculated using logistic regression models, with subjects having normoalbuminuria with preserved eGFR serving as the reference category. Initially, Model 1 had only minimal adjustment including age and gender. A subsequent fully multivariable-adjusted Model 2 included additional adjustment for those factors that were statistically significant at α < 0.05 in univariate logistic regression analysis (data not shown). Given our sample sizes, our study was powered to detect an estimated OR comparing PAD with non-PAD of 3.09 for albuminuria with eGFR ≥ 60, 3.62 for normoalbuminuria with eGFR < 60 and 2.94 for albuminuria with eGFR < 60, setting at 80% power. The statistical operations were performed using SPSS software (SPSS Version 19; SPSS Inc., Chicago, IL, USA), and the power analysis was carried out with NCSS 2007. A p value of less than 0.05 was considered significant.
Results
This study analysed 236 elderly subjects (47.9% men) with an average age of 72.0 ± 5.0 years. The mean ABI of the overall population was 0.98 ± 0.22; 26.7% of the participants had an ABI < 0.9 or > 1.4, 13.6% had an ABI between 0.7 and 0.89, 8.1% had an ABI between 0.4 and 0.69, 2.1% had an ABI < 0.4 and only 3% had an ABI > 1.4. Regarding the subjects with symptomatic PAD, 12 (5.1%) had intermittent claudication, 4 (1.7%) had foot lesion and 2 (0.9%) had lower extremity amputation.
These study subjects were categorized into PAD and without PAD, and their clinical characteristics were compared (Table 1). Patients with PAD were characterized by more CVD; longer duration of diabetes; higher total cholesterol, triglyceride, LDL-cholesterol, hs-CRP and ACR; and lower HDL-cholesterol and eGFR. Additionally, they had higher prevalence of normoalbuminuria with eGFR < 60 and albuminuria with eGFR < 60, which is twofold higher if compared with subjects without PAD.
Comparison of clinical characteristics according to prevalent peripheral artery disease.
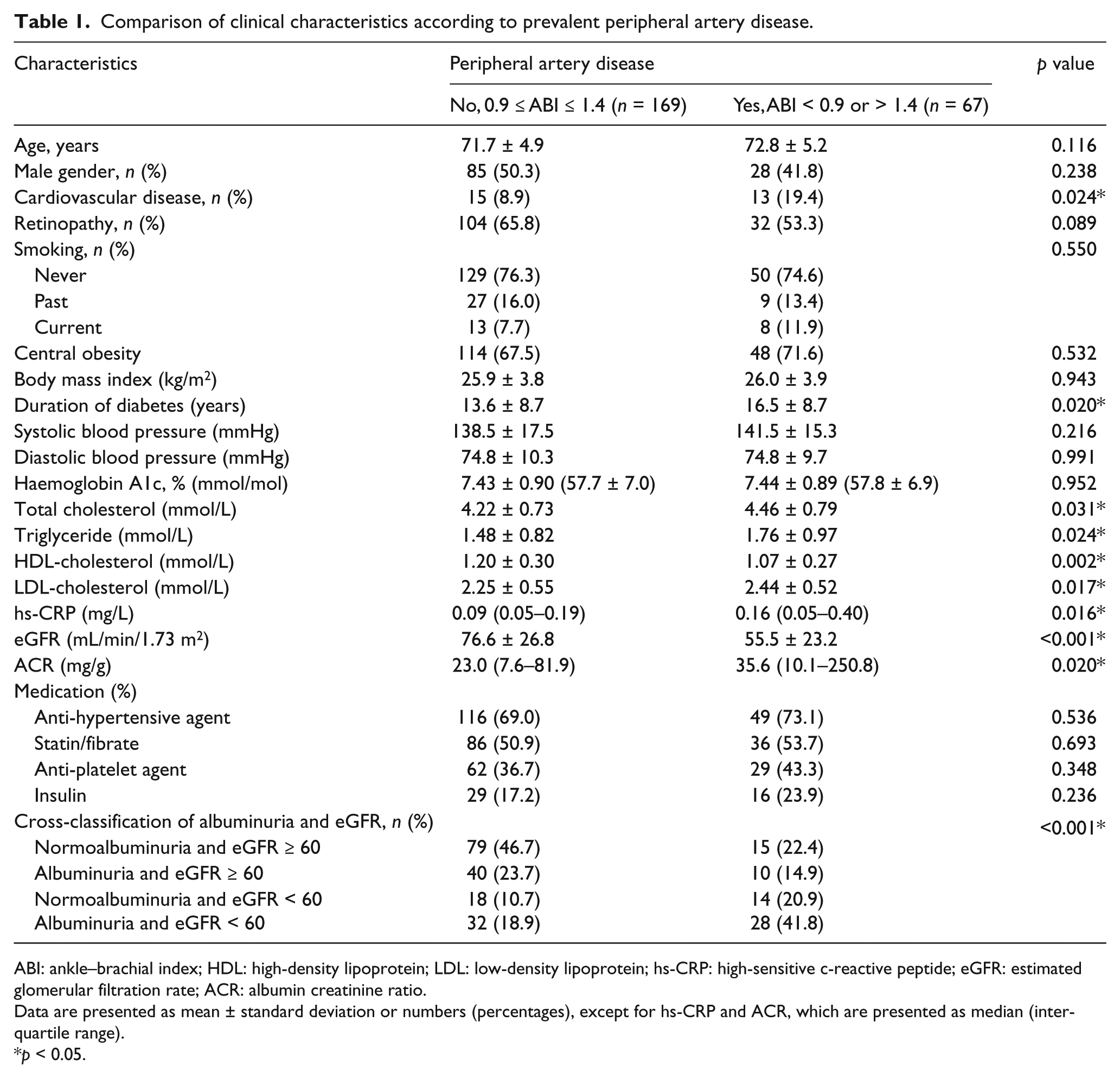
ABI: ankle–brachial index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; hs-CRP: high-sensitive c-reactive peptide; eGFR: estimated glomerular filtration rate; ACR: albumin creatinine ratio.
Data are presented as mean ± standard deviation or numbers (percentages), except for hs-CRP and ACR, which are presented as median (interquartile range).
p < 0.05.
As shown in Figure 1, the prevalence of PAD was greatest in those with albuminuria and low eGFR. Specifically, the prevalence of PAD was 16% among patients with normoalbuminuria and eGFR ≥ 60 and increased up to 20%, 43.8% and 46.7% in patients presenting as albuminuria with eGFR ≥ 60, normoalbuminuria with eGFR < 60 and albuminuria with eGFR < 60, respectively.
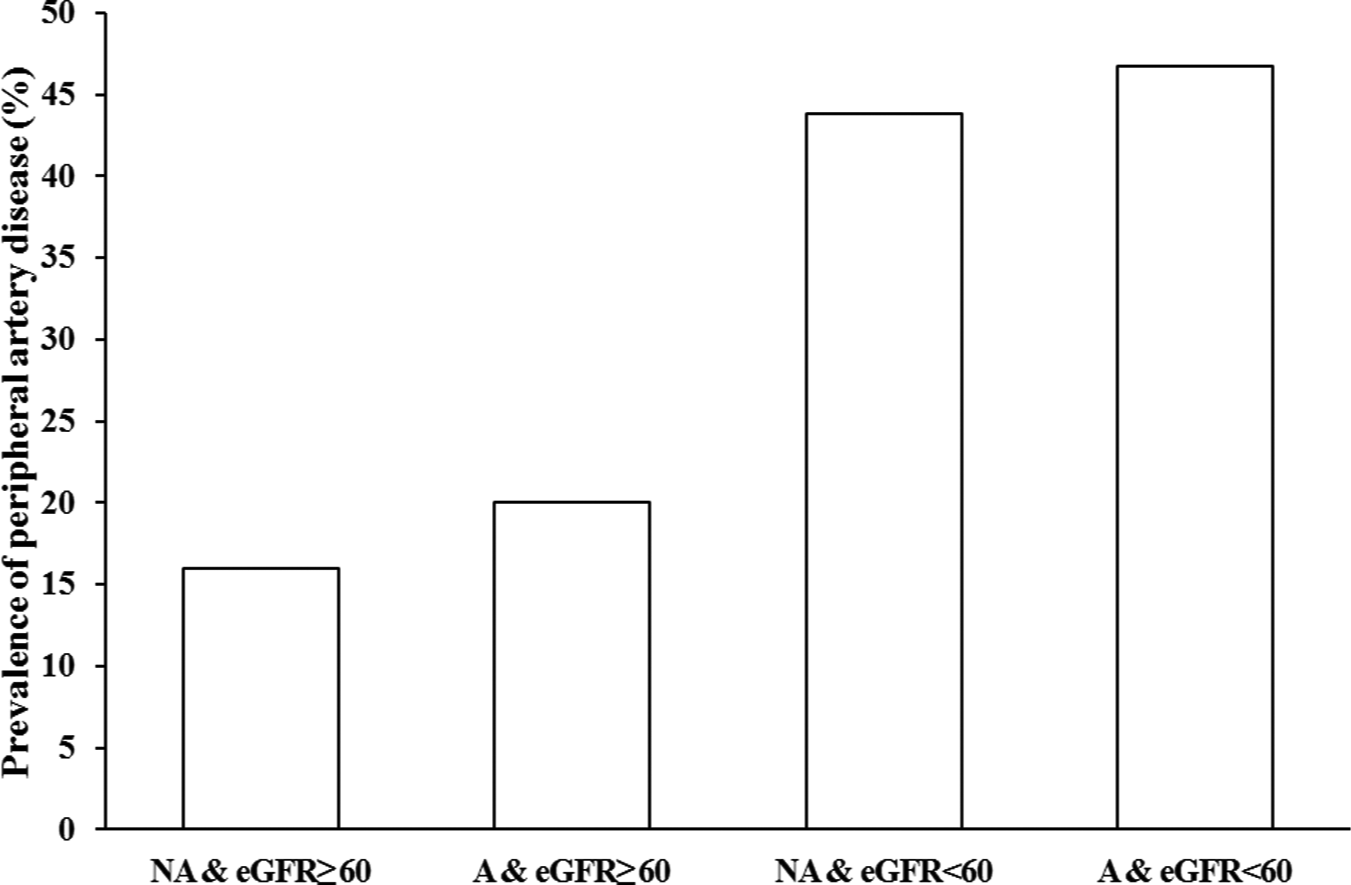
Prevalence of peripheral artery disease after cross-classification of albuminuria and low eGFR.
In Table 2, we performed the multivariate analysis to assess the association between different combined albuminuria and low eGFR categories and PAD. After age and gender adjustment, and compared to participants with normoalbuminuria and eGFR ≥ 60, the ORs [95% confidence interval (CI)] of PAD were 1.28 (0.52–3.14), 3.92 (1.58–9.78) and 4.52 (2.11–9.68) for those with albuminuria and eGFR ≥ 60, normoalbuminuria and eGFR < 60 and albuminuria and eGFR < 60, respectively (Model 1). Finally, with additional adjustment for those factors that were statistically significant at α < 0.05 in univariate logistic analysis (CVD, duration of diabetes, total cholesterol, triglyceride), the OR and 95% CI became 1.10 (0.43–2.79), 3.14 (1.20–8.22) and 3.87 (1.72–8.72), respectively (Model 2). Taken together, adjustment for multiple risk factors for PAD did not result in much attenuation in the odds of PAD among patients presenting as normoalbuminuria with eGFR < 60 and albuminuria with eGFR < 60.
Minimal and multivariable-adjusted ORs (95% confidence interval) for the prevalent peripheral artery disease associated with cross-classification of albuminuria and low eGFR.
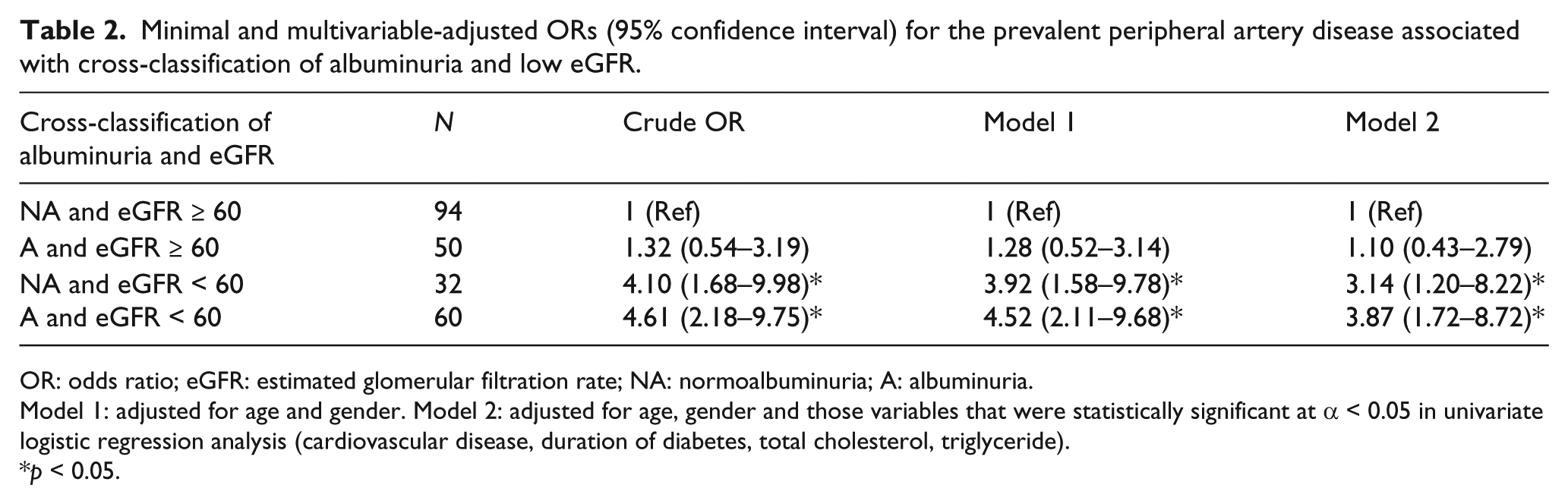
OR: odds ratio; eGFR: estimated glomerular filtration rate; NA: normoalbuminuria; A: albuminuria.
Model 1: adjusted for age and gender. Model 2: adjusted for age, gender and those variables that were statistically significant at α < 0.05 in univariate logistic regression analysis (cardiovascular disease, duration of diabetes, total cholesterol, triglyceride).
p < 0.05.
Discussion
The results of this cross-sectional study involving elderly patients with type 2 diabetes showed that the prevalence of PAD was 26.7% in the overall study population. After cross-classification by the presence of albuminuria and low eGFR, our data demonstrated that normoalbuminuric renal insufficiency and albuminuric renal insufficiency were associated significantly with threefold to fourfold increased likelihood of PAD. We suggest that these two criteria are distinct and complementary, and this might reflect pathophysiological differences among these different cross-classification renal stages.
In this study, we defined PAD as an ABI < 0.9 or > 1.4 based on the findings from several recent investigations instead of only ABI < 0.9.10,16,17 In fact, high ABI values (>1.4) are observed in patients with diabetes, particularly for those concomitant with CKD. 18 According to a subanalysis of the Strong Heart Study, using the 0.90 as the cut-off point may lead to underestimation of the prevalence of PAD because an increased ABI also reflects coronary calcifications and CVD risk. 17 Furthermore, it also led to clinical manifestation of PAD and increased risk of amputation, proposing that elevation of this index could reflect diffuse atheromatous disease.19,20 Since the manifestations or prognosis of high ABI (>1.4) is equivalent with those of low ABI (<0.9), subjects with elevated ABI are thus included in the PAD group for current analysis.
It is well known that most previous analyses have limited their classification to either albuminuria or reduced eGFR to investigate its association with prevalence of PAD.7–10,21 Indeed, the presence of either albuminuria or a reduced eGFR identified different categories of the CKD because only 54.4% of those with albuminuria had a reduced eGFR and only 64.5% of those with a reduced eGFR had albuminuria as shown in our report. This suggested that relying on single criterion alone will miss many cases of pre-existing kidney disease. Prior studies had established that the presence of albuminuria was significantly associated with PAD.9,11 On the contrary, a recent investigation found that reduced eGFR but not albuminuria was independently associated with PAD in patients with diabetes. 10 For the research, which examined the combined effects of albuminuria and reduced eGFR on PAD, the result stated that the prevalence of PAD was significantly elevated accompanied by both abnormalities. 6 A recent population-based study also indicated that the prevalence of PAD was greatest in those with both albuminuria and reduced eGFR, and this association was statistically significant in those with albuminuria alone or reduced eGFR alone. 12
Compared with these previous studies, our research focused on elderly patients with type 2 diabetes. Interestingly, we found that albuminuria with preserved eGFR, which represented the early stage of diabetic nephropathy, was not significantly related to PAD. In contrast, among the patients with late-stage diabetic nephropathy, a significant association between PAD and low eGFR was observed. This finding was in line with the results of a previous report, which revealed that the prevalence of asymptomatic PAD increased with more advanced stage of CKD. 22 To our knowledge, the presence of albuminuria has been recognized as a risk factor of generalized vascular damage. On the other hand, albuminuria may be just a marker of poor glucose control or longer duration of diabetes.23,24 A recent large population-based study demonstrated that albuminuria was not associated with PAD in diabetic subjects. However, in further subgroup analysis, a possible albuminuria and PAD correlation was found in patients with intense glucose control. 10 In summary, the severity of vascular damage cannot be decided simply by the pre-existence of albuminuria, particularly among patients with early-stage diabetic nephropathy.
Although the pathophysiologic mechanisms between PAD and both albuminuria and low eGFR are not fully elucidated, it is believed that the presence of both abnormalities are markers of the generalized burden of atherosclerotic disease. 25 In this study, deterioration of renal function instead of presence of albuminuria may reflect more accurately the severity of generalized atherosclerosis. 26 A prior study confirmed that atherosclerotic change in the renal vessels commonly occurs in the presence of PAD: 56% had either renal artery stenosis or occlusion. 27 Other factors such as hyperhomocysteinemia increase inflammation activity and oxidative stress and could also contribute to the higher prevalence of PAD in patients with renal insufficiency.28,29 In contrast, albuminuria seems related to the generalized endothelial dysfunction, may also be a marker of a prothrombotic state and could possibly lead to the development of lower extremity atherosclerosis. 30 However, this insight mechanism warranted further investigations for confirmation.
The strength of this study is that we studied the combined effects of albuminuria and reduced eGFR, thus making it more likely to find valid associations between CKD and PAD. Several limitations of our study should also be noted. First, the cross-sectional nature limits conclusions about causality and directionality of the association observed in this study. Second, albuminuria measurement was based on a single spot urine sample. However, a prior study indicated that an early morning urinary ACR ≥ 30 mg/g is associated with an albumin excretion rate ≥ 30 mg/min, with high sensitivity and specificity. 31 Third, misclassification due to transient albuminuria could not be completely excluded. Nevertheless, this misclassification would bias the observed results towards the null. Fourth, misclassification of PAD was possible because PAD was diagnosed by a non-invasive method rather than a gold standard technique such as angiography. However, angiography is rarely applied to the elderly patient for the purpose of diagnosis only.
In conclusion, we indicate that both normoalbuminuric and albuminuric renal insufficiencies are independently associated with the presence of PAD among elderly patients with type 2 diabetes mellitus, after adjustment of multiple CVD risk factors. This cross-classification method characterizes a subgroup of patients who have very high risk of PAD as well as CVD, and it is hoped that they would benefit from the early implementation of appropriate therapeutic strategies.
Footnotes
Acknowledgements
The authors thank Mrs Chih-Yun Lin for her excellent statistical work.
Declaration of conflicting interest
The authors report no conflicts of interest.
Funding
This study was supported by a grant from Yuan’s General Hospital (RG10-005), Taiwan.
