Abstract
Decreased plasma n-3 polyunsaturated fatty acid levels or the n-3/n-6 polyunsaturated fatty acid ratios are associated with a risk of cardiovascular events. In this cross-sectional study, we measured plasma levels of eicosapentaenoic acid, docosahexaenoic acid, and arachidonic acid and investigated the association between the plasma polyunsaturated fatty acid profile and vascular endothelial function in 396 patients with type 2 diabetes. Endothelium-dependent, flow-mediated dilatation of the brachial artery was measured using ultrasonography. Multiple regression analyses, including age, sex, body mass index, and other cardiovascular risk factors, revealed that plasma eicosapentaenoic acid levels (β = 0.140, p = 0.008) and the eicosapentaenoic acid/arachidonic acid ratio (β = 0.127, p = 0.019), but not plasma docosahexaenoic acid levels (β = 0.067, p = 0.220) or the docosahexaenoic acid/arachidonic acid ratio (β = 0.034, p = 0.559), were independently and positively associated with flow-mediated dilatation. In conclusion, plasma eicosapentaenoic acid levels and the eicosapentaenoic acid/arachidonic acid ratio are independently associated with endothelial function in patients with type 2 diabetes. This study indicates a positive association between eicosapentaenoic acid, rather than docosahexaenoic acid, and endothelial function in type 2 diabetes.
Keywords
Introduction
The consumption of n-3 polyunsaturated fatty acids (PUFAs), 1 or the circulating PUFA profile, including eicosapentaenoic acid (EPA) levels, docosahexaenoic acid (DHA) levels, and the n-3 PUFA/arachidonic acid (AA) ratio,2,3 is inversely associated with cardiovascular outcomes. Many clinical trials have also shown that the supplementation of n-3 PUFAs improves vascular endothelial function, a predictor of subclinical cardiovascular outcomes. 4 However, the effect of n-3 PUFA supplementation on endothelial function has been inconsistent in studies on patients with type 2 diabetes,5,6 who evidently have endothelial dysfunction. 7 Moreover, to our knowledge, no study has demonstrated the association between the circulating n-3 PUFA profile and endothelial function in patients with type 2 diabetes. Our hypothesis was that endothelial dysfunction is associated with an abnormal plasma PUFA profile in patients with type 2 diabetes.
Methods
Study design and participants
In this cross-sectional study, we consecutively enrolled 396 patients with type 2 diabetes who were admitted to the Diabetes Centre of the Osaka City University Hospital between January 2009 and June 2013. Patients who were regularly taking drugs containing n-3 PUFAs were excluded from the present study.
This study was performed in accordance with the Declaration of Helsinki (1975, as revised in 2013). The study protocol was approved by the Ethics Committee of Osaka City University Graduate School of Medicine (No. 308). All participants provided written informed consent prior to the study.
Measurements
Frozen plasma samples were shipped to SRL (Tokyo, Japan), and EPA, DHA, and AA concentrations were measured using capillary gas chromatography as previously described.3,8 We measured flow-mediated dilatation (FMD) and endothelium-independent, nitroglycerin-mediated dilatation (NMD) of the brachial artery using an ultrasound system (UNEXEF; Unex Co. Ltd., Nagoya, Japan). The measurements were performed in a quiet, air-conditioned room at 25.0°C for inpatients who had not consumed any foods, caffeine, or tobacco and had not engaged in exercise for at least 12 h before the measurements, according to the International Brachial Artery Reactivity Task Force guideline. 9
Statistical analysis
Correlations were examined using the nonparametric Spearman’s rank correlation test. Multiple regression analyses were used to explore the influence of each of the PUFA levels or the n-3/n-6 PUFA ratio on FMD or NMD. Skewed parameters, such as triglycerides and the plasma PUFA profile, were logarithmically transformed before regression analyses. A p value of < 0.05 was considered significant. Statistical analyses were performed using the JMP 10 software (SAS Institute Inc., Cary, NC, USA).
Results
This study’s participants included 228 men and 168 women, aged 65 years (median). The median duration of diabetes and the body mass index (BMI) were 11 years and 24.9 kg/m2, respectively. The median plasma EPA, DHA, and AA levels were 39.0 μg/mL, 94.3 μg/mL, and 138.7 μg/mL, respectively. The median EPA/AA, DHA/AA, and (EPA + DHA)/AA ratios were 0.29, 0.66, and 0.96, respectively. The median FMD and NMD were 5.9% and 14.5%, respectively.
The FMD was negatively correlated with age (ρ = –0.167, p = 0.001), duration of diabetes (ρ = –0.117, p = 0.020), and systolic blood pressure (ρ = –0.179, p < 0.001), and was positively correlated with estimated glomerular filtration rate (eGFR) (ρ = 0.222, p < 0.001). None of the parameters of the plasma PUFA profile were significantly correlated with FMD in unadjusted analyses. To explore the independent association between plasma PUFA levels or n-3 PUFA/AA ratios and FMD, we performed multiple regression analyses after adjusting for potential confounders. Aside from the traditional risk factors for atherosclerosis, including age, BMI, systolic blood pressure, and glycated haemoglobin (HbA1c) levels, plasma EPA levels and the plasma EPA/AA ratios were found to be independently and positively associated with FMD (Table 1). No significant association was found between FMD and plasma DHA levels, plasma AA levels, the DHA/AA ratio, or the (EPA + DHA)/AA ratio. In contrast, none of the parameters of the plasma PUFA profile were significantly associated with NMD, after adjusting for the same variables as those used in the models for FMD (data not shown).
Multiple regression analysis for the determinants of FMD in all participants with type 2 diabetes.
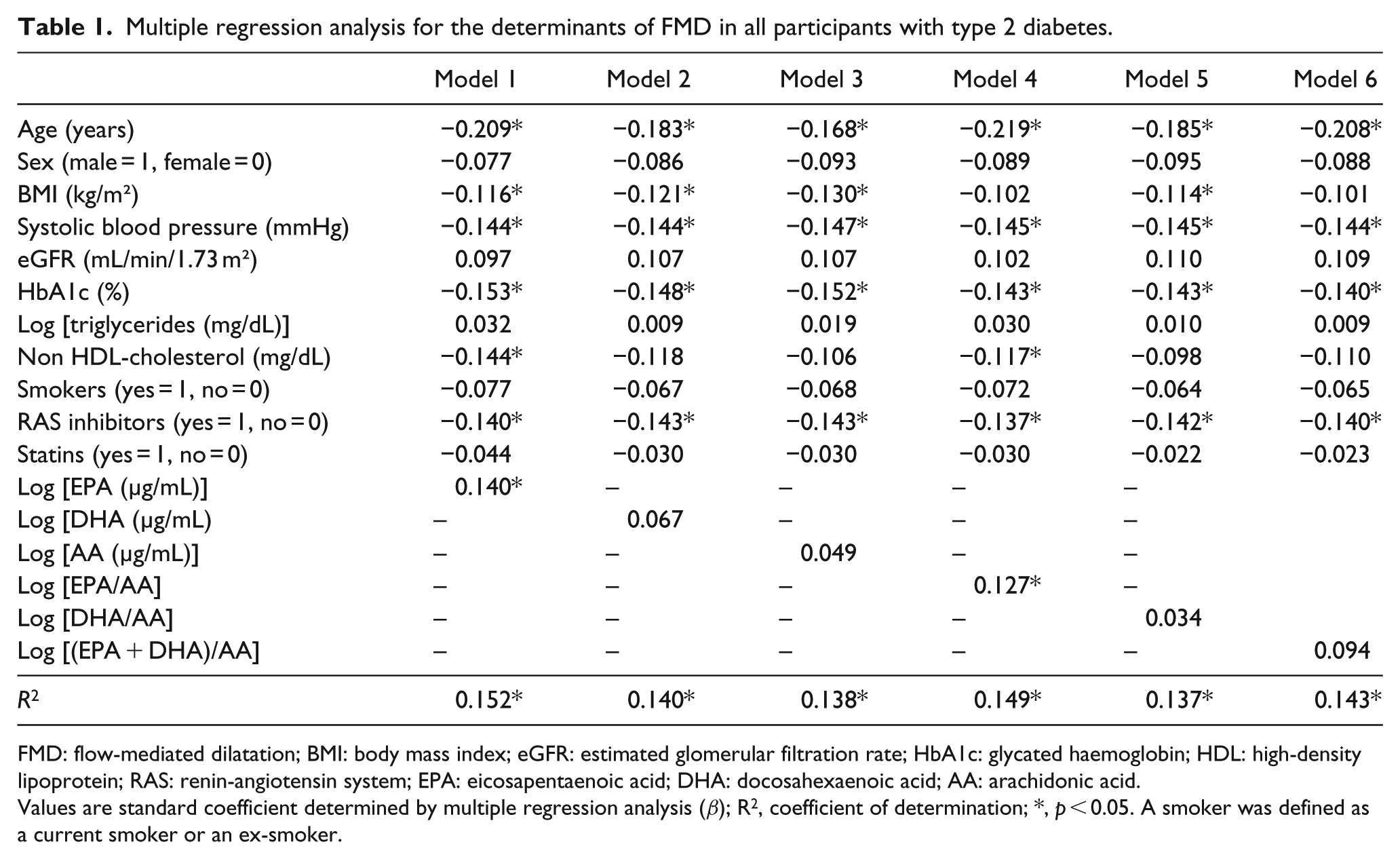
FMD: flow-mediated dilatation; BMI: body mass index; eGFR: estimated glomerular filtration rate; HbA1c: glycated haemoglobin; HDL: high-density lipoprotein; RAS: renin-angiotensin system; EPA: eicosapentaenoic acid; DHA: docosahexaenoic acid; AA: arachidonic acid.
Values are standard coefficient determined by multiple regression analysis (β); R2, coefficient of determination; *, p < 0.05. A smoker was defined as a current smoker or an ex-smoker.
Discussion
The present study demonstrated that plasma EPA levels and the EPA/AA ratio were positively associated with FMD of the brachial artery in patients with type 2 diabetes. Notably, those associations were independent of the traditional cardiovascular risk factors. Accumulating evidence indicates a beneficial effect of n-3 PUFA supplementation on endothelial function in individuals with cardiovascular disease or its risk factors. 4 Because no study has examined the association between circulating levels of n-3 PUFAs and FMD in patients with type 2 diabetes, this is the first study regarding an association between the plasma PUFA profile and endothelial function in those patients.
Contrary to our results, a number of studies performed in patients with diabetes failed to show a beneficial effect of supplementation with n-3 PUFAs on endothelial function.10,11 Impaired endothelial function has been documented in patients with type 2 diabetes. 7 Furthermore, a dysregulated plasma PUFA profile was associated with the presence of type 2 diabetes in our recent study. 8 Taken together, the association between the plasma PUFA profile and endothelial function would be complicated in patients with type 2 diabetes. Therefore, our data may indicate that EPA exerts its vasodilatory effect 12 even in patients with type 2 diabetes. It remains unclear why a significant association between the plasma PUFA profile and FMD was found in our study subjects with type 2 diabetes; however, a relatively large number of participants compared to the number in prior studies10,11 may be one possible reason.
The present study further showed that plasma levels of DHA were not associated with FMD. Results from limited studies indicate that DHA might be more effective than EPA in improving forearm vascular reactivity and that the vasodilatory effects of EPA are endothelial cell-dependent, while those of DHA are endothelial cell-independent. 12 It needs to be mentioned that none of those previous studies were performed in patients with type 2 diabetes. In our results, neither EPA nor DHA was associated with NMD, an endothelium-independent vasodilatation, whereas only EPA was associated with FMD, potentially highlighting the pivotal role of endothelium-dependent, vasodilatory effects of EPA in type 2 diabetes.
This study has several limitations. First, we measured PUFA levels in total plasma lipids, but not in phospholipids from the cellular membrane, which are direct precursors of bioactive eicosanoids. Second, we did not evaluate dietary intake, the use of dietary supplements, or lifestyles of participants, which could have affected vascular function, as well as the plasma PUFA profile. Third, the participants with type 2 diabetes were receiving statin, antihypertensive, and/or anti-diabetic drugs, including insulin, which could affect vascular function and related risk factors. Fourth, a relatively small sample size and a single measurement of plasma PUFA could influence the results of the present study. Finally, we did not include non-diabetic controls. Although previous studies in the non-diabetic population have shown inconsistent results, we could have evaluated whether the presence of diabetes modifies the association between circulating PUFAs and FMD using a non-diabetic control group.
In conclusion, this study demonstrated that plasma EPA levels and the EPA/AA ratio are independently associated with FMD in patients with type 2 diabetes. Our data indicate a positive association between EPA and endothelial function, an established predictor of cardiovascular disease, and further propose the plasma levels of EPA, rather than those of DHA, as a potential biomarker of vascular health, even in patients with type 2 diabetes who have impaired endothelial function.
Footnotes
Acknowledgements
The authors acknowledge the excellent technical assistance of Ms. Setsuko Arita and Ms. Mika Sakaki of the research laboratory in the Department of Metabolism, Endocrinology and Molecular Medicine, Osaka City University Graduate School of Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a Grant-in-Aid for Scientific Research (No. 20591068) from the Japan Society for the Promotion of Science (to M.E. and K. Mor.).
