Abstract
Background:
Microcirculatory and endothelial dysfunction are signs of cardiovascular engagement in patients with type 2 diabetes. This study tested whether glucose normalisation may reverse this.
Methods:
Thirty-nine T2DM patients (age 61±7 years, 58% females) with signs of mild diastolic dysfunction were randomised to strict glucose control based on insulin (I-group; n=21) or oral agents (O-group; n=18) for four months. Skin microcirculation was studied with Laser Doppler Fluxmetry and endothelial function with brachial artery flow-mediated dilatation.
Results:
Glucose control improved (reduction of HbA1c I-group = -0.5%; O-group -0.7%; p=0.69). Microcirculation improved in the entire group (n=39) determined by foot Laser Doppler Fluxmetry (32.2±13.6 vs. 35.3±13.1 perfusion units; p<0.001) and Laser Doppler Fluxmetry following heating (68.8±34.0 vs. 69.3±25.1 PU; p=0.007). Improvement was more consistent with oral agents than insulin. Endothelial function expressed as flow-mediated dilatation decreased in the I-group (6.0±2.2 to 4.7±3.0%; p=0.037) but remained unchanged in the O-group (4.8±2.3 to 5.0±3.7%; n.s.).
Conclusions:
Glycaemic normalisation improved skin microcirculation but not endothelial function in patients with type 2 diabetes with mild cardiovascular engagement.
Introduction
Hyperglycaemia increases the risk for cardiovascular disease already at levels below those defining type 2 diabetes. 1 Glucose perturbations may alter the structure and function of the vessel wall via disturbed intracellular signal transduction including activation of diacylglycerol, protein kinase C and the receptor for advanced glycation end products due to glucose-induced oxidative stress. 2 This triggers a cascade of molecular events resulting in a limited availability of nitric oxide and an activation of proinflammatory and prothrombotic factors. Impaired microvascular function has indeed been demonstrated already in healthy subjects at an increased risk for developing type 2 diabetes. 3 Furthermore, endothelial dysfunction is a strong and independent predictor of long-term cardiovascular events 4 and an early expression of cardiovascular engagement in patients with diabetes.5,6 With this background the obvious assumption is that a meticulous glycaemic control would preserve vessel wall function, counteracting or at least retarding the development of macro- and microvascular complications, and improve the prognosis in patients with diabetes or its pre-states, an assumption supported by some evidence.7,8 Although not extensively studied, there are indications that improved glycaemic control may improve endothelial function.9,10 Patients in these studies did, however, have a poor glycaemic control with a baseline HbA1c averaging 9.5%, a level that improved without being normalised during treatment. To the best of our knowledge there are no reports on improved microvascular and endothelial function induced by glucose normalisation in patients with type 2 diabetes with only subtle signs of cardiovascular involvement. The importance of such observations increased following presentation of the long-term outcome in the United Kingdom Prospective Diabetes Study (UKPDS). This report suggests that intensive glycaemic control instituted soon after the diagnosis of type 2 diabetes has long-lasting benefits, reducing not only microvascular complications but also myocardial infarction and mortality. It was speculated that this may relate to a sustained, so called legacy effect of the initial intensive glycaemic control. 11
The primary aim of the present investigation was to explore the influence of glucose normalisation on skin microcirculation and endothelial function comparing insulin with oral glucose lowering therapy in patients with type 2 diabetes and only mild cardiovascular engagement.
Patients and methods
This investigation, an originally planned sub-study of the Diastolic Dysfunction And Diabetes (DADD) trial, comprised 39 patients with type 2 diabetes mellitus free from cardiovascular disease but with signs of mild diastolic dysfunction. An extensive description of patient selection and study design has been presented elsewhere. 12 In brief, patients were recruited following a screening process including a thorough case history, physical examination, laboratory specimens, transthoracic Doppler-echocardiography and Tissue Doppler Imaging. Included patients fulfilled the following criteria: type 2 diabetes, diastolic left ventricular dysfunction, age between 40 and 70 years and a fasting plasma glucose (FPG) ≥7.0 mmol/L or HbA1c >5.5% (Mono-S). The main endpoint in DADD was the impact of normalised glucose control on diastolic dysfunction. 12 The most important exclusion criteria were ongoing insulin treatment, systolic left ventricular dysfunction or ischaemic heart disease, symptomatic peripheral vascular disease, poorly controlled hypertension, impaired renal function and left ventricular hypertrophy.
The study was conducted according to the Declaration of Helsinki, and the study protocol was approved by the Regional Ethics Committee. All patients gave their informed written and oral consent to study participation.
Vascular investigations
All participants were asked to refrain from tobacco and caffeine-containing products during the eight hours preceding the investigations. They did, however, receive a light breakfast, sugar-, fat-and caffeine-free, before the tests. These were performed in a quiet, dimly lit room with the temperature kept between 22°C and 24°C with the patients in supine position. All subjects were acclimatised for 15 minutes before the investigations started.
Skin microcirculation
The total skin microcirculation was measured by Laser Doppler Fluxmetry (LDF; Periflux, 4001 Master, Perimed®, Stockholm, Sweden) at the volar side of the forearm and the dorsum of the foot. The LD output signal was continuously recorded on a PC (Perisoft software, Perimed®, Järfälla, Sweden). The LDF signal was recorded during rest for three minutes. The resting LDF value represents the computer integrated mean during this period and is expressed as perfusion units (PUs). Heat LDF was measured at the end of a five minute period of heating of the skin under the LDF probe to 44°C on both the foot (heat foot LDF) and forearm (heat arm LDF) (PeriTemp 4005 with a thermostatic probe PF 457, Perimed®). Post-occlusive LDF in the foot was measured during maximum hyperaemia following a two minute arterial occlusion at the ankle with a cuff pressure of 250 mmHg. The mean intra-individual coefficient variation (CV) for the measurement of hyperaemia during heating and following arterial occlusion in our lab is 7% and 18%. 13
Endothelial function
Flow-mediated dilatation (FMD), studied by a non-invasive examination of the brachial artery of the non-dominant arm, 14 was performed by means of an 8 MHz linear-array transducer connected to a Siemens Sequoia® (Siemens Medical Systems, Mountain View, CA, USA). Baseline images were saved every third second during one minute and a mean value was calculated from these recordings. A blood pressure cuff, positioned below the elbow, was thereafter inflated to 260 mmHg for five minutes. Endothelium-dependent vasodilatation was determined by continuously imaging the artery during three minutes of hyperaemia following cuff release. A mean value was calculated from three recordings at maximum dilatation. Endothelium-independent vasodilatation was determined following sublingual administration of nitroglycerine (spray; 0.4 mg). All images were analysed using proprietary software (Brachial analyser®, Medical Imaging Applications, Iowa City, IA, USA). The maximum diameter, found through beat-to-beat analysis, was measured using an automated contour detection system. Lumen diameter was defined as the distance between the intima of the far and near vessel walls. Dilatation was calculated as maximal lumen diameter after ischaemia or nitroglycerine minus lumen diameter at baseline divided by lumen diameter at baseline. The coefficient of variation for FMD determination on two study occasions is 18%. 15
Study protocol
The study was performed as a Prospective, Randomised and Open study with Blind Evaluation (PROBE). Following the recording of all study variables the patients were randomised into one of two study arms receiving either insulin (I-group; n=21) or oral glucose lowering agents (O-group; n=18) targeting glucose normalisation. The four-months long study period commenced as soon as a patient had reached the glycaemic target of a self-monitored FPG <5.0 mmol/L during at least three consecutive days or after a maximum titration period of eight weeks. Patients in I-group were started on a long lasting insulin analogue, glargine (Lantus®, Aventis Pharma, Stockholm, Sweden) administered once daily in the afternoon (at 17:00). Those not reaching the glycaemic target were in addition prescribed a rapid acting insulin analogue aspart (Novo Rapid®, Novo Nordisk, Copenhagen, Denmark) to meals. Patients in O-group were treated with oral glucose lowering agents, initiated with metformin (Metformin®, Meda, Stockholm, Sweden). If the postprandial values were high after three weeks on metformin, repaglinide (Novonorm®, Novo Nordisk, Copenhagen, Denmark) was added. The treatment regimen was not changed during the four study months apart from minor adjustments related to hypoglycaemic episodes or gastrointestinal side effects. All study variables were obtained by the end of this period.
Laboratory tests
The following tests were obtained at the time of randomisation and the final visit: haemoglobin, white blood cell count, fasting plasma glucose, HbA1c, total serum cholesterol, low density lipoprotein (LDL), high-density lipoprotein (HDL) triglycerides and endothelin-1. Blood samples were drawn after at least eight hours of strict fasting. Almost all analyses were performed at the central Laboratory Department at Karolinska University Hospital. HbA1c was analysed by high performance liquid chromatography presented as Mono-S with a measurement interval of 2.9–17.2%, reference value <5.3%. Swedish HbA1c = 0.989* International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) HbA1c + 0.88%; r2 = 0.996; National Glycohaemoglobin Standardisation Program (NGSP)- HbA1c = 0.915 (IFCC- HbA1c) + 2.15% (r2 = 0.997). 16 Endothelin-1 was analysed by radioimmunoassay.
Statistics
This investigation was a sub-study of the DADD trial. Accordingly power calculation was based on the main study parameter. 12 Continuous variables are reported as mean ± SD or as median and range or quartiles while categorical variables are presented as counts and proportions (%). Group comparisons were made by the Mann–Whitney rank sum test and within-group comparisons by the Wilcoxon matched pair test. A two-sided p<0.05 was considered statistically significant. All statistical analyses were performed in Graph Pad Prism version 4.0 program (Graph Pad Software, San Diego, CA, USA).
Results
Patient characteristics
Pertinent clinical characteristics at baseline are presented in Table 1. Diabetes duration and prescription of glucose lowering treatment were comparable at randomisation. By the end of the study all patients in the I-group were treated with insulin and eight continued metformin. Patients randomised to the O-group were all given metformin and in 12 of them repaglinide had to be added to achieve the glucose target.
Pertinent patient clinical characteristics by group.
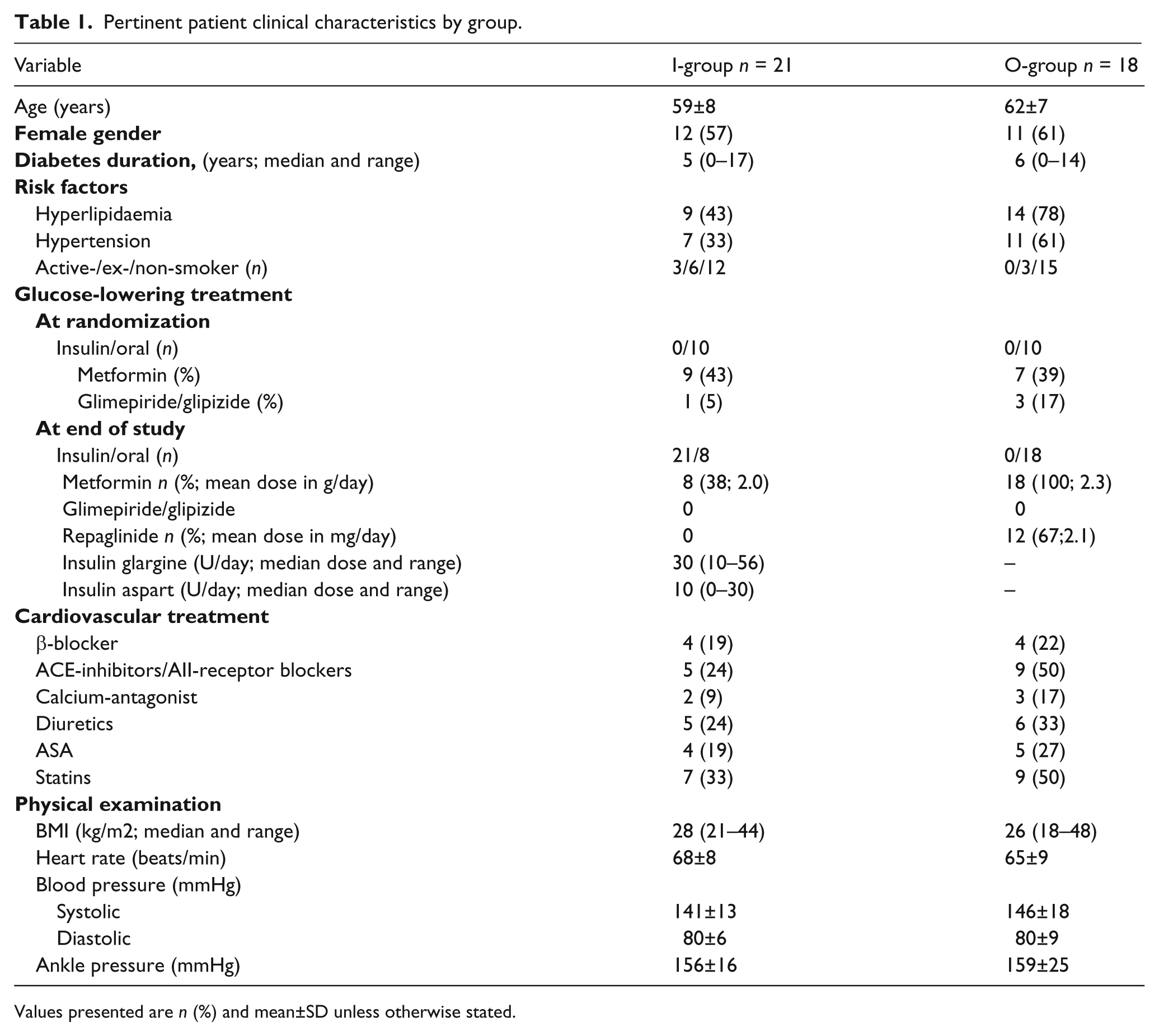
Values presented are n (%) and mean±SD unless otherwise stated.
Biochemical analyses and inflammatory markers
Laboratory data are presented in Table 2. There was a comparable improvement in glycaemic control in both groups as reflected by a decrease in HbA1c by 0.5 and 0.7% in the I- and O-groups respectively. By the end of the study 15 patients (71%) in the I-group and 15 (83%) in the O-group had an HbA1c <5.5%.
Pertinent biochemical analyses at randomisation and end of study by group.
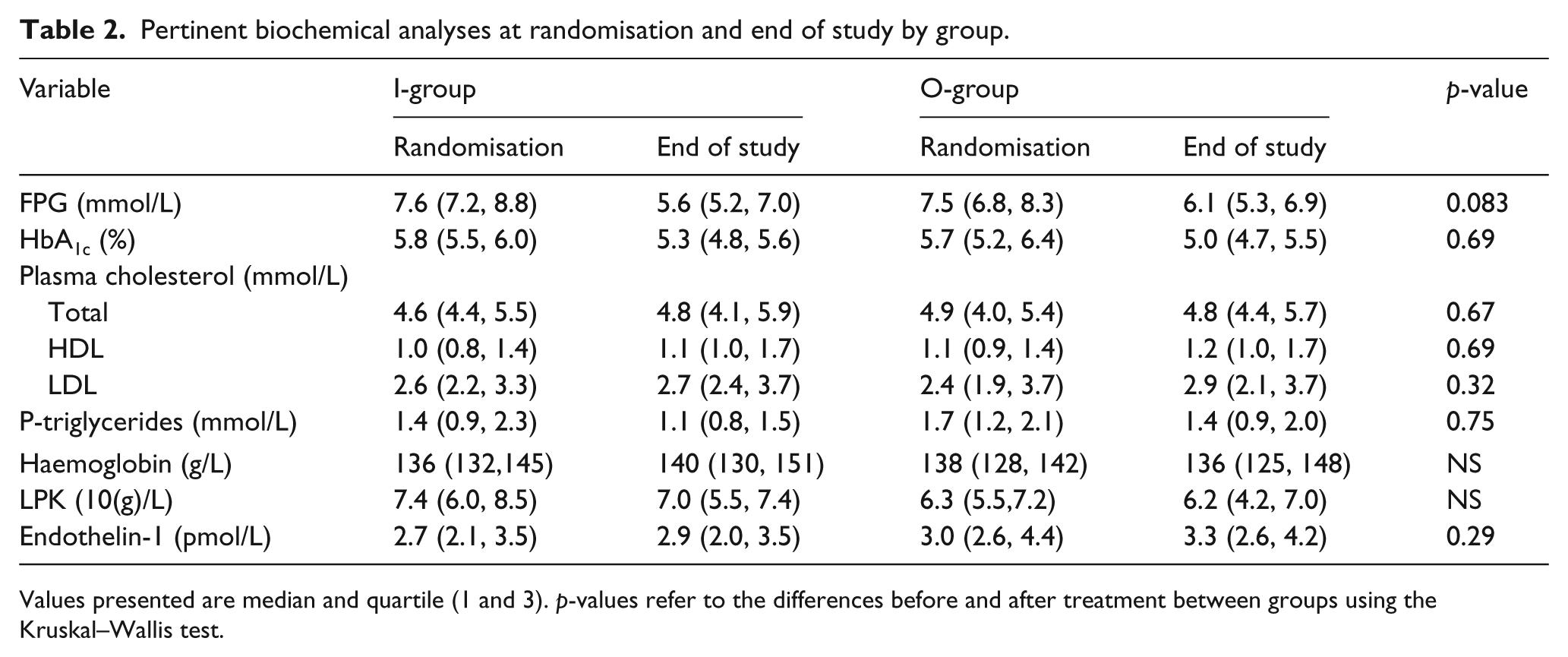
Values presented are median and quartile (1 and 3). p-values refer to the differences before and after treatment between groups using the Kruskal–Wallis test.
Skin microcirculation
Ankle blood pressure (see Table 1) and transcutaneous oxygen tension (data not shown) were comparable in the two groups at randomisation and did not change during the study period. Baseline LDF did not differ between the two groups. Glycaemic normalisation improved microcirculation in the entire group (n = 39) as determined by post occlusive LDF in the foot (baseline vs. follow-up = 32.2 ± 13.6 vs. 35.3 ± 13.1 PU; p < 0.001), heat foot LDF (68.8 ± 34.0 vs. 69.3 ± 25.1 PU; p = 0.007) as well as heat arm LDF (66.1 ± 38.3 vs. 71.5 ± 40.6 PU; p = 0.001). Glucose normalisation induced a significant increase in post occlusive foot LDF in both groups (Figure 1(a)) and increased heat foot as well as heat arm LDF in the O-group (Figure 1(b) and (c)), but there was no significant difference in the I-group (Figure 1(b) and (c)). The change from baseline to follow up in these parameters did, however, not differ significantly between the two groups (Figure 1(d) to (f)).
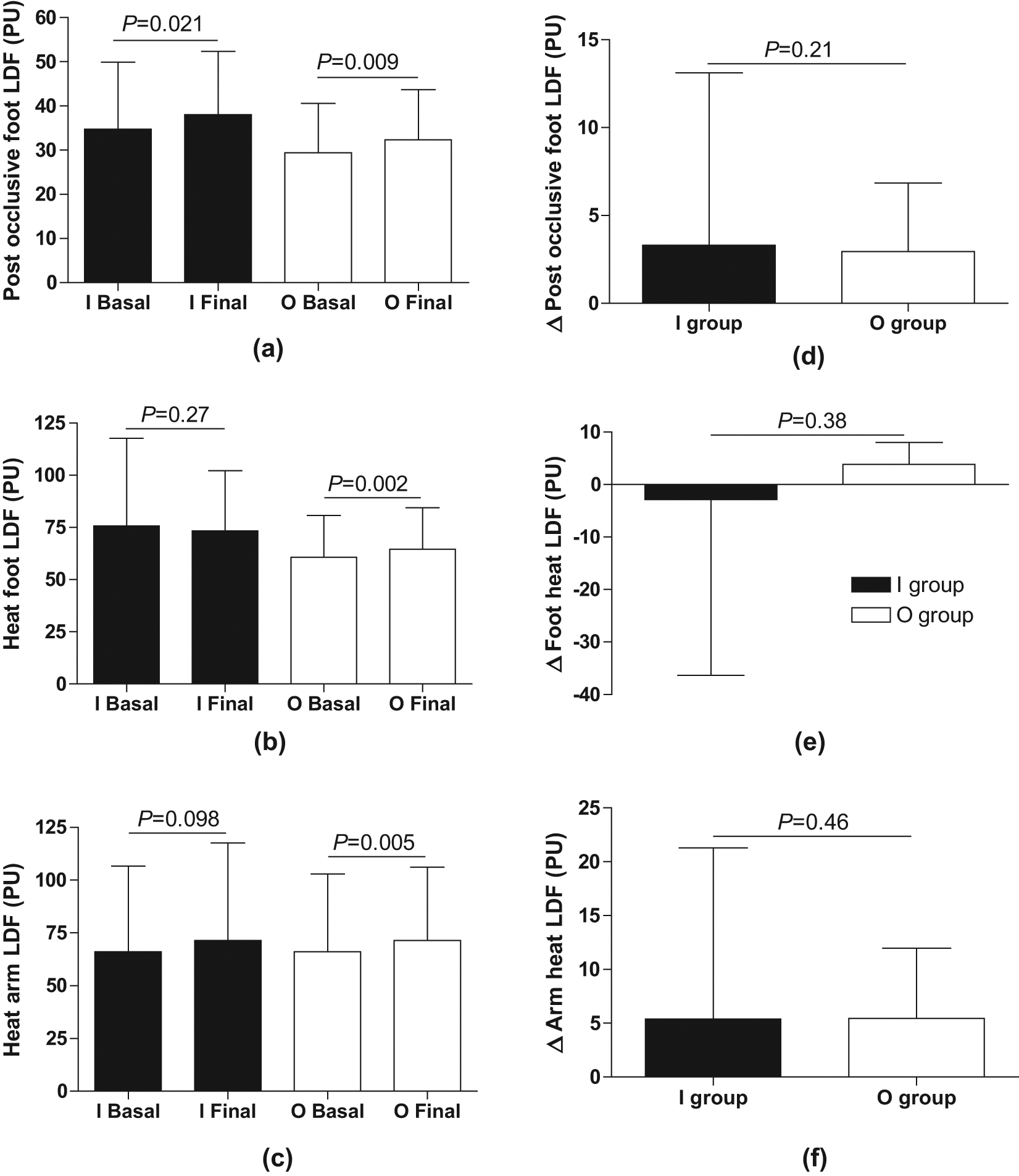
Baseline values (Basal) and effects of glycaemic normalisation (Final) on microcirculation, as determined by Laser Doppler Fluxmetry (LDF) in the foot following a two minute occlusion of the ankle (post occlusive foot LDF; (a), (d)), following five min local heating of the dorsum of the foot to 44°C (heat foot LDF; (b), (e)), and of the volar part of the forearm (heat arm LDF; (c), (f)), expressed as perfusion units (PUs). Insulin group (I; filled bars) and oral group (O; open bars). Δ refers to the in-between group comparison before and after intensified glucose control. Values are presented as means±SD.
Endothelial function
Complete data on flow-mediated dilatation were available in 22 patients (I-group n = 10; O-group n = 12). Brachial artery diameter was similar in the two groups at baseline (I-group 3.3 ± 0.6 mm; O-group 3.3 ± 0.7 mm) and did not change during the study period. Glycaemic normalisation in the entire group did not induce any change in FMD (5.4 ± 2.3% at baseline vs. 4.9 ± 3.3% at follow-up; p = 0.28; n = 22). FMD decreased in the I-group and remained unchanged in the O-group (Figure 2(a)), but the change in FMD following improved glycaemic control did not differ significantly between the two treatment groups (Figure 2(c)). The intensified glycaemic control did not significantly influence endothelium-independent vasodilatation in the entire study group (12.9±4.6% at baseline vs. 16.9±8.9% at follow-up; p=0.14; n=22) or within any of the two groups (Figure 2(b)) and the change from baseline did not differ between the two groups (Figure 2(d)).
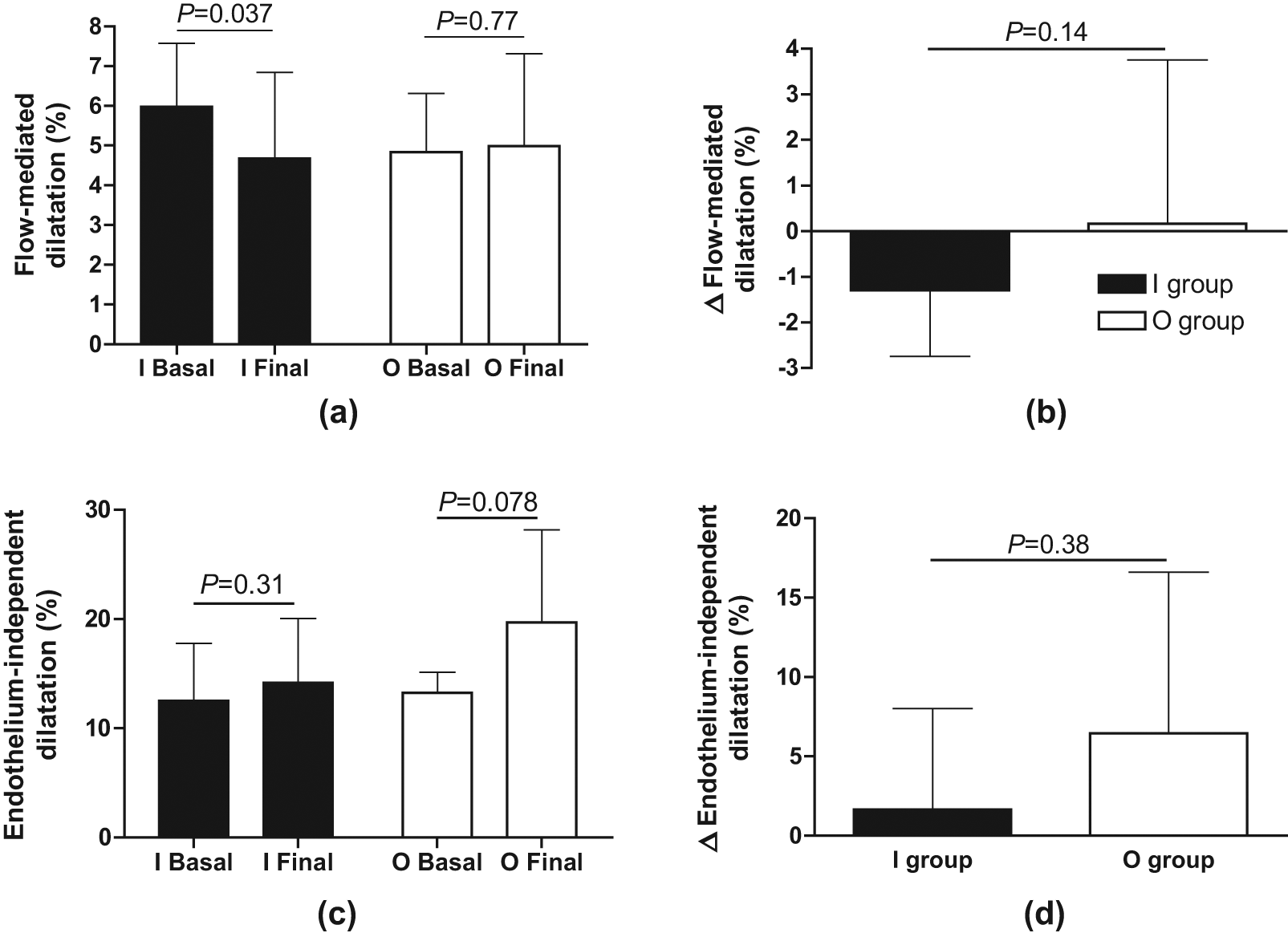
Baseline values (Basal) and effects of glycaemic normalisation (Final) on flow-mediated dilatation in the brachial artery following five minute occlusion of the forearm ((a), (c)), and endothelium-independent vasodilatation following sublingual nitroglycerine (0.4 mg; (b), (d)) expressed as per cent change from baseline. Insulin group (I; filled bars) and oral group (O; open bars). Δ refers to the in-between group comparison before and after intensified glucose control. Values are presented as means±SD.
Discussion
The present study revealed that glycaemic normalisation, especially by means of oral glucose lowering agents, improved skin microvascular function in patients with type 2 diabetes with only minor cardiovascular engagement. Furthermore, brachial artery endothelial function was not affected following glycaemic normalisation in the entire study group although it seemed to deteriorate following insulin treatment.
The present population comprised a group of patients with relatively uncomplicated type 2 diabetes. They were screened to have, at the most, modest signs of myocardial diastolic dysfunction and a glycaemic control above present therapeutic targets but no other cardiovascular disease manifestations. 12 The rationale for the patient selection was to create a possibility to study whether glucose normalisation, by insulin or oral agents, may be motivated from the perspective to improve their vascular function. In this group of patients with type 2 diabetes glucose normalisation did not improve macrovascular function as determined by FMD, either by insulin or oral glucose lowering agents. In contrast microvascular function, as determined by post occlusive, heat foot as well as heat arm LDF, improved in the entire study group.
Microvascular function was studied by LDF, a measure of total skin microcirculation, that is, nutritional capillary as well as non-nutritional sub-papillary blood flow. The maximum microvascular hyperaemic response to local heating and arterial occlusion is likely to involve neurogenic and endothelium-dependent mechanisms induced by structural changes in the vessel wall 17 . An impaired skin maximum hyperaemic response to local heating and arterial occlusion has been observed in patients with diabetes and is associated with macrovascular complications and increased cardiovascular risk.18,19 Moreover, disturbed microvascular function is of great importance for the diabetes-specific complications retinopathy, nephropathy and retarded wound healing. 20
In the present perspective a report showing that healthy middle-aged males with a family history of diabetes exhibited impaired skin microcirculation in the presence of a normal FMD in the brachial artery is of special interest 3 . It supports the assumption that micro- precedes macro-circulatory dysfunction as an early vascular complication in diabetes. This assumption gains support from the present findings that skin microcirculation improved following glycaemic normalisation whereas macrovascular function did not. Furthermore, patients with type 1 diabetes mellitus with good metabolic control (HbA1c 6.3%) had a better post occlusive LDF in the toe compared with patients with bad metabolic control (HbA1c 8.7%). 21 Taken together with the present study this indicates that preservation of normoglycaemia is important for the maintenance of satisfactory skin microcirculation. Collectively these findings may exert a possible mechanistic link as to why intensive glycaemic control instituted soon after the diagnosis of type 2 diabetes has long-lasting benefits, reducing not only microvascular complications but also myocardial infarction and mortality as reported in the long-term follow up of the UKPDS study. 11
In contrast to previous observations glucose normalisation did not improve macrovascular function as determined by FMD in the present study. Thus Vehkavaara and Yki-Jarvinen reported that endothelial function improved following the addition of insulin isophane to ongoing treatment with metformin. 22 Although the patients in these studies were reported to be free from cardiovascular disease their glucose levels were considerably less well controlled at study start and not normalised at follow-up. There are also methodological reasons for the discrepant results. Vehkavaara and co-workers10,22 used an invasive technique with intra-arterial infusions of acetylcholine during measurement of forearm blood flow with venous occlusion plethysmography. This method measures total blood flow reflecting mainly resistance vessel function but also skin circulation, while the presently used non-invasive method reflects only conduit artery function. Of note is that the present FMD values are comparable to those in a lipid- lowering study in our laboratory, which demonstrated improvement in endothelium-dependent vasodilatation. 15 Thus there are reasons to assume that baseline endothelial function in the present study population was compromised and possible to improve by means of pharmacological tools, although not observed in the present setting. Also, other studies have shown an association between different states of insulin resistance and endothelial dysfunction. 23 However, there are studies showing similar endothelial function in patients with and without glucometabolic perturbations. Avogaro et al. 24 were unable to detect any differences in endothelium-dependent vasodilatation as determined by intra-brachial acetylcholine between patients with uncomplicated type 2 diabetes and controls. They concluded that their findings argued against a role of impaired generation of nitric oxide and blood flow regulation as a cause of insulin resistance in such patients. This is supported by the finding that acetylcholine-induced endothelium-dependent forearm vasodilatation was not significantly impaired in a group of clinically healthy subjects with insulin resistance. 25
In the present study there was a significant reduction in brachial artery endothelial function in the I-group following glycaemic normalisation. Furthermore, there was no difference in heat foot LDF or heat arm LDF within the I-group at follow-up. These findings are of particular interest in the light of recent reports on potentially negative consequences of insulin treatment compared with oral glucose-lowering drugs.8,26 Thus it may serve as a hypothesis-generating observation warranting further studies with tight insulin-based glycaemic control to elucidate the effects on vascular function. The mechanisms by which insulin affects endothelial function are very complex. While the activation of the classical PI-3-K pathway increases the endothelial release of nitric oxide, 27 activation of the MAPK pathway results in an increased release of the vasoconstrictive pro-inflammatory peptide endothelin-1 (ET-1). 28 Even though we did not see any significant treatment changes in plasma ET-1 (which is a poor biomarker for tissue ET-1 activity) in the present study it may still be so that these pathways were affected differently in the treatment groups. In line with this insulin resistance may result from impaired PI-3-K-dependent signalling and coexisting hyperinsulinemia results in the activation of genes involved in inflammation and excessively activates the MAPK pathway. 29
Taken together, the present study demonstrates that microvascular function may be improved by glycaemic normalisation before any changes can be detected in conduit artery function or left ventricular diastolic function. 12 This indicates that skin microvascular dysfunction may be an early finding in type 2 diabetes, possible to improve by strict glycaemic control even in patients with relatively good metabolic control.
Study limitations
The lack of an age-matched control group may be seen as a limitation. However, all patients in this study had established diabetes and were on diet. In addition about 50% were already on some type of oral glucose-lowering therapy, which was the reason it was considered unethical to leave them without treatment. Furthermore patients assigned to a placebo would not have reached glycaemic normalisation, an important target in this trial. There were some differences between the two groups as regards concomitant treatment (see Table 1). Some of the prescribed drugs affect endothelial function but given the small differences and that these drugs were not changed over the time of observation it seems unlikely that this was an important confounder. Another limitation is the loss of patients completing the FMD assessments due to temporary illness of the technician responsible for these investigations. In combination with the 18% intra-individual coefficient of variation it is underlined that these results should be interpreted with some caution. However, the reduced FMD following glucose normalisation in the I-group together with the lack of an effect in heat LDF point in the same direction and warrant future more detailed studies of insulin action on vascular function. Another possible limitation could be the presence of coexisting confounding factors such as peripheral neuropathy, medial sclerosis and peripheral vascular disease despite the fact that none of the patients showed any clinical signs of such complications.
Conclusion
Glycaemic normalisation improved skin microcirculation in patients with type 2 diabetes and mild diastolic dysfunction. This improvement was more consistent in patients on oral glucose-lowering agents than in those on insulin. Since endothelial function in the brachial artery was not improved it is possible that glycaemic normalisation may improve microcirculation before any changes in endothelial function may be detected in this type of patient with relatively good metabolic control and only minor cardiovascular engagement.
Footnotes
Acknowledgements
The authors gratefully acknowledge thoughtful comments on the manuscript by Professor John Pernow, Cardiology Unit, Department of Medicine, Karolinska Institutet, and, concerning statistical considerations, by Associate Professor John Öhrvik, Karolinska Institutet. We are grateful to Ann Lindstrom BMA, Marita Wallin BMA and Mattias Lidin RN at our department for skilled handling of patients and data.
Author contributions
All authors contributed to study design, conductance and interpretation of data. CJ took the main responsibility for data interpretation and for writing the manuscript. MK researched data and reviewed/edited the manuscript. FB and LR researched data, contributed to discussion and reviewed/edited the manuscript.
Funding
This work was supported by AFA Insurance and by unconditional research grants from Aventis US. None of these providers of research funds had any role in the design and conduct of the study; collection management, analyses and interpretation of the data; and preparation, review or approval of the manuscript.
Conflict of interest statement
None of the authors has any conflicts of interest to declare in relation to this paper.
