Abstract
Objective:
Recent studies have indicated the implication of C1q tumour necrosis factor–related protein 9 in vascular pathology of atherosclerosis. This study first investigated the association of C1q tumour necrosis factor–related protein 9 and the severity of lower extremity peripheral arterial disease in type 2 diabetes mellitus patients.
Methods:
A total of 200 patients with type 2 diabetes mellitus had ankle–brachial index examined in this cross-sectional study, 60 patients with ankle–brachial index of ⩽0.9 were diagnosed with peripheral arterial disease and further classified into mild, moderate and severe group with Vivid 7 diagnostic apparatus.
Results:
C1q tumour necrosis factor–related protein 9 and adiponectin were higher in diabetes mellitus with severe peripheral arterial disease than diabetes mellitus with mild and moderate peripheral arterial disease groups. After adjustment of conventional atherosclerosis risk factors, C1q tumour necrosis factor–related protein 9 (r = −0.627, p < 0.001) and adiponectin (r = −0.431, p = 0.003) were negatively correlated with the severity of peripheral arterial disease. Logistic regression analysis showed C1q tumour necrosis factor–related protein 9 was independently associated with the severity of peripheral arterial disease (odds ratio = 0.272, 95% confidence interval = 0.08–0.927, p = 0.037).
Conclusion:
Serum C1q tumour necrosis factor–related protein 9 is negatively and independently associated with the severity of peripheral arterial disease in type 2 diabetes mellitus patients, suggesting the protective potentials of C1q tumour necrosis factor–related protein 9 in the progression of peripheral arterial disease in human type 2 diabetes mellitus.
Keywords
Introduction
Lower extremity peripheral arterial disease (PAD) is widespread, particularly in the elderly and in patients with diabetes mellitus. Over 50% of patients with PAD are asymptomatic and diagnosed through routine screening. PAD identified in this way carries a high mortality and/or vascular event risk. 1 It is important to identify novel sensitive biomarkers to indicate the degree of vascular pathology and monitor the progression of PAD.
C1q tumour necrosis factor–related proteins (CTRPs) are members of a highly conserved family with the same modular organization as adiponectin. Among members of the CTRP superfamily, CTRP9 shares the highest amino acid identity with adiponectin and has effects on glucose metabolism and insulin resistance. 2 Recent research has indicated the involvement of CTRP9 in vascular calcification which is the most important vascular pathology in type 2 diabetes mellitus (T2DM). In aortic vascular rings, CTRP9 causes significant endothelium-dependent vascular relaxation via nitric oxide (NO)-mediated signalling pathway. 3 CTRP9 protein treatment inhibits the proliferative and chemotactic activities of vascular smooth muscle cell (VSMC) induced by growth factors. Adenoviral vectors expressing CTRP9 (Ad-CTRP9) attenuate neointimal formation in mice with a left femoral arteries injury model. 4 CTRP9 overexpression enhances plaque stability in apolipoprotein E knockout mice by reducing pro-inflammatory cytokines in macrophages. 5 CTRP9 prevented inflammatory cytokine-induced nuclear factor kappa B (NF-κB) activation and the expression of adhesion molecules in human aortic endothelial cells (HAECs). 6 Despite these laboratory findings, there is a lack of clinical evidence on the association of CTRP9 with peripheral vascular pathology. The present study investigated whether serum CTRP9 was associated with the severity of PAD in type 2 diabetes patients.
Materials and methods
Subjects
The study was approved by the institutional ethics board (registration no. 2014-03) and complies with the Declaration of Helsinki and Good Clinical Practice. Subjects were recruited from the department of endocrinology at Huai’an Hospital Affiliated to Nanjing University of Traditional Chinese Medicine between January 2015 and December 2016. Diabetes mellitus was diagnosed according to the 1999 WHO T2DM diagnostic criteria. Excluded were subjects with acute or chronic infection, ketoacidosis, hyperosmolar coma, surgery or trauma within the previous 6 months, malignancies, cerebral infarction, heart attack, immunological diseases, disorders of hepatic, renal or haematopoietic systems and subjects receiving systemic glucocorticoid or immunosuppressant. A total of 200 hospitalized T2DM patients had their ankle–brachial index (ABI) examined. Among them, 60 patients were diagnosed as PAD and further classified into mild, moderate and severe according to the degree of vascular pathology.
Laboratory methods
Blood samples were collected in the early morning after an overnight fast. Biochemical parameters were measured by ADVIA1200 Auto biochemical analyser including glycosylated hemoglobin (HbA1c), fasting triglyceride (TG), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C). Serum levels of CTRP9, adiponectin and high-sensitivity C-reactive protein (hs-CRP) were assayed using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Cusabio Biotech Co., Ltd, China).
ABI was determined with DopplexMD200 (Hunlteigh, UK). ABI ⩽ 0.9 was taken as the diagnostic criteria for the presence of PAD. Patients diagnosed as PAD had limb extremity arteries examined by Vivid 7 imaging colour Doppler diagnostic apparatus (GE, USA). The examination sites were the common femoral artery, femoral artery bifurcation, popliteal artery, posterior tibial artery and dorsalis pedis artery. The vascular pathology was evaluated and scored as follows: 7 (1) Artery intima thickness: normal (<1 mm), 0 point; mildly thickened (1–1.2 mm), 1 point; severely thickened (>1.2 mm), 2 points. (2) Hardening: normal, 0 point; mildly hardened (the intima was not thickened, the echo was increased, and without plaque), 1 point; moderately to severely hardened (mildly hardened, associated with plaque or stenosis), 2 points. (3) Plaque: normal (no plaque formation), 0 point; single plaque, 1 points; multiple plaques, 2 points; diffused plaques, 3 points. (4) Stenosis: normal, 0 point; mild stenosis (narrowing by 30%–50%), 1 point; moderate or severe stenosis (narrowing by 50%–75%), 2 points; occlusion (no blood flow), 3 points. The severity of PAD was classified according to the total points: (1) 0 point, normal; (2) <10 mild; (3) 10–20 points, moderate; (4) >20 points, severe.
Statistical analysis
All data were presented as mean ± SD except where otherwise indicated. The Kolmogorov–Smirnov test demonstrated that all measurement data were normally distributed. The chi-squared test was used to compare the differences of enumeration data. One-way analysis of variance and least significance difference (LSD) test were used to analyse clinical and laboratory parameters among the three groups. Pearson’s correlation analysis and partial correlation analysis were used to evaluate the correlation between the scores of vascular pathology and CTRP9, adiponectin and hs-CRP. Logistic regression analysis was used to calculate the odds ratio (OR) for the severity of PAD. Statistical analysis was performed using SPSS 19.0. p-Values of <0.05 were considered significant.
Results
Table 1 shows the clinical and biochemical characteristics of the T2DM patients with mild, moderate and severe PAD. Pearson analysis showed that levels of hs-CRP were positively correlated to the PAD score (r = 0.328, p = 0.011), while levels of CTRP9 (r = −0.584, p < 0.001) and adiponectin (r = −0.499, p < 0.001) were negatively correlated to the PAD score. Partial correlation analysis demonstrated that the association of serum levels of CTRP9 (r = −0.627, p < 0.001), adiponectin (r = −0.431, p = 0.003) and hs-CRP (r = 0.335, p = 0.025) with the PAD score remained after adjustment for atherosclerotic risk factors including gender, age, smoking, body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), HbA1c, TG, LDL-C and HDL-C.
Clinical and biochemical characteristics of type 2 diabetes patients with mild, moderate and severe PAD.
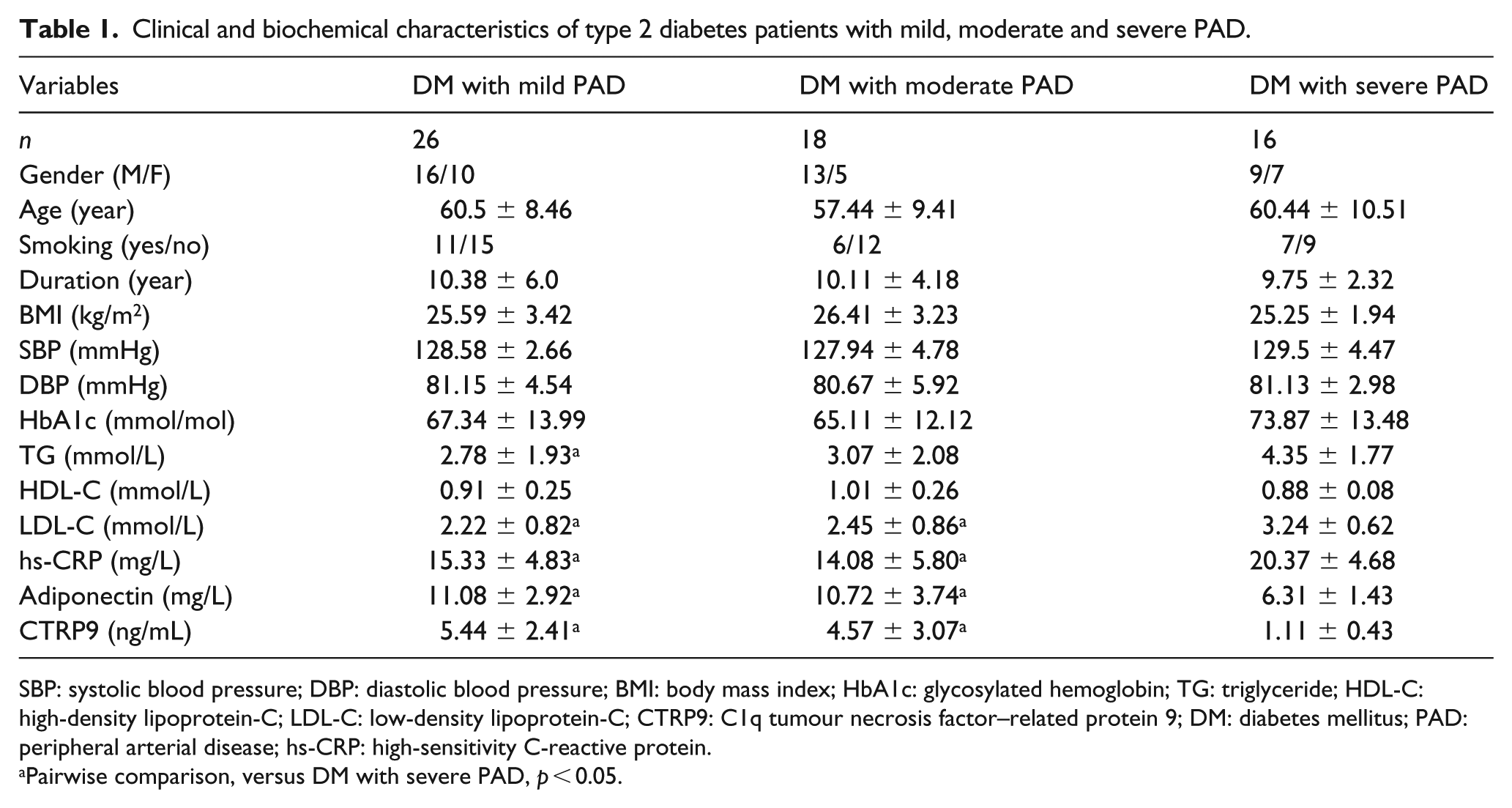
SBP: systolic blood pressure; DBP: diastolic blood pressure; BMI: body mass index; HbA1c: glycosylated hemoglobin; TG: triglyceride; HDL-C: high-density lipoprotein-C; LDL-C: low-density lipoprotein-C; CTRP9: C1q tumour necrosis factor–related protein 9; DM: diabetes mellitus; PAD: peripheral arterial disease; hs-CRP: high-sensitivity C-reactive protein.
Pairwise comparison, versus DM with severe PAD, p < 0.05.
The severity of PAD (0 = mild and moderate PAD, 1 = severe PAD) was taken as dependent variable. Factors including gender, age, duration, smoking, BMI, SBP, DBP, TG, HDL-C, LDL-C, adiponectin, hs-CRP and CTRP9 were taken as independent variables. Logistic regression analysis with forward likelihood ratio method showed that hs-CRP (OR = 1.379, 95% CI = 1.057–1.798, p = 0.018) and LDL-C (OR = 10.780, 95% CI = 1.426–81.489, p = 0.021) were associated with an increased risk for the severity of PAD. Serum CTRP9 (OR = 0.272, 95% CI = 0.08–0.927, p = 0.037) was associated with a decreased risk for the severity of PAD.
Discussion
This study demonstrated that CTRP9 levels were negatively associated with the severity of PAD in T2DM. This association was independent of adiponectin and conventional atherosclerotic risk factors. A low serum level of CTRP9 could provide supplemental information on diabetes patients to predict the possibility of the deterioration of PAD. An interesting finding is that CTRP9 was more strongly associated with PAD scores than adiponectin. Previous studies demonstrated the difference of CTRP9 and adiponectin in cardiac and vascular effects, although they were both identified as independent protective factors for the presence of metabolic syndrome, coronary atherosclerosis and arterial stiffness in several clinical studies.8–10 In aortic vascular rings, the vasoactive potency of CTRP9 surpassed that of adiponectin by threefold. 3 Cardiac expression of CTRP9 exceeded adiponectin by more than 100-fold in mice. 11 Our findings suggest that CTRP9 may play different roles from adiponectin in the vascular pathology of PAD in T2DM.
There are some limitations of this study including the relatively small size of the study population, the cross-sectional design and lack of measures of adiponectin activity. Further prospective studies are warranted to clarify the potential of CTRP9 as a future biomarker for the progression of PAD in T2DM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Science and Technology Bureau of Huai’an (grant no. HAS2014016).
